ABSTRACT
PURPOSE:
To investigate the effect of curcumin on visfatin and zinc-α2-glycoprotein (ZAG) expression levels in rats with non-alcoholic fatty liver disease (NAFLD).
METHODS:
Fifty-six male rats were randomly divided into a control group (n=16) and model group (n=40) and were fed on a normal diet or a high-fat diet, respectively. Equal volumes of sodium carboxymethyl cellulose (CMC) were intragastrically administered to the control group for 4 weeks. At the end of the 12th week, visfatin and ZAG protein expression levels were examined by immunohistochemistry. Visfatin mRNA levels were measured by semi-quantitative reverse transcription polymerase chain reaction.
RESULTS:
Compared with the control group, the model group showed significantly increased expression of visfatin in liver tissue (P < 0.01) and significantly decreased expression of ZAG (P < 0.01). These effects were ameliorated by curcumin treatment.
CONCLUSIONS:
Visfatin and zinc-α2-glycoprotein may be involved in the pathogenesis of NAFLD. Treatment of NAFLD in rats by curcumin may be mediated by the decrease of visfatin and the increase of non-alcoholic fatty liver disease.
Key words:
Curcumin; Nicotinamide Phosphoribosyltransferase; Fatty Liver; Rats
Introduction
Non-alcoholic fatty liver disease (NAFLD) is a group of clinical pathological syndromes, and its disease spectrum includes simple fatty liver disease, non-alcoholic steatohepatitis, and related cirrhosis11 Nishioji K, Sumida Y, Kamaguchi M, Mochizuki N, Kobayashi M, Nishimura T, Yamaguchi K, Itoh Y. Prevalence of and risk factors for non-alcoholic fatty liver disease in a non-obese Japanese population, 2011-2012. J Gastroenterol. 2015 Jan;50(1):95-108. doi: 10.1007/s00535-014-0948-9.
https://doi.org/10.1007/s00535-014-0948-...
2 Priore P, Cavallo A, Gnoni A, Damiano F, Gnoni GV, Siculella L. Modulation of hepatic lipid metabolism by olive oil and its phenols in nonalcoholic fatty liver disease. IUBMB Life. 2015 Jan;67(1):9-17. doi: 10.1002/iub.1340.
https://doi.org/10.1002/iub.1340...
3 Machado MV, Michelotti GA, Pereira Tde A, Boursier J, Kruger L, Swiderska-Syn M, Karaca G, Xie G, Guy CD, Bohinc B, Lindblom KR, Johnson E, Kornbluth S, Diehl AM. Reduced lipoapoptosis, hedgehog pathway activation and fibrosis in caspase-2 deficient mice with non-alcoholic steatohepatitis. Gut. 2015 Jul;64(7):1148-57. doi: 10.1136/gutjnl-2014-307362.
https://doi.org/10.1136/gutjnl-2014-3073...
4 Ferreira DM, Castro RE, Machado MV, Evangelista T, Silvestre A, Costa A, Coutinho J, Carepa F, Cortez-Pinto H, Rodrigues CM. Apoptosis and insulin resistance in liver and peripheral tissues of morbidly obese patients is associated with different stages of non-alcoholic fatty liver disease. Diabetologia. 2011 Jul;54(7):1788-98. doi: 10.1007/s00125-011-2130-8.
https://doi.org/10.1007/s00125-011-2130-...
-55 Lam B, Younossi ZM. Treatment options for nomalcoholic fatty liver disease. Therp Adv Gastroenterol. 2010 Mar;3(2):121-37. doi: 10.1177/1756283X09359964.
https://doi.org/10.1177/1756283X09359964...
. The pathogenesis of NAFLD is not yet clear but Day's "two-strike" theory is accepted by most scholars. The role of visceral fat and associated visfatin in the pathogenesis of NAFLD is still controversial, and may cause liver cell steatosis and promote the occurrence of NAFLD. Zinc-α2-glycoprotein (ZAG) can regulate the activity of lipid metabolic enzymes, and hence maintain the balance of lipid metabolism to play a protective role in the pathogenesis of NAFLD66 Russell ST, Tisdale MJ. Role of ß-adrenergic receptors in the anti-obesity and anti-diabetic effects of zinc-a2-glycoprotien (ZAG). Biochim Biophys Acta. 2012 Apr;1821(4):590-9. doi: 10.1016/j.bbalip.2011.12.003.
https://doi.org/10.1016/j.bbalip.2011.12...
7 Zhang L, Zhang HY, Ma CC, Wei W, Jia LH. Increased body weight induced by perinatal exposure to bisphenol A was associated with down-regulation zinc-alpha2-glycoprotein expression in offspring female rats. Mol Cell Toxicol. 2014 Jun;10(2):207-13. doi: 10.1007/s13273-014-0022-y.
https://doi.org/10.1007/s13273-014-0022-...
-88 Gong FY, Deng JY, Zhu HJ, Pan H, Wang LJ, Yang HB. Fatty acid synthase and hormone-sensitive lipase expression in liver are involved in zinc-alpha2-glycoprotein-induced body fat loss in obese mice. Chin Med Sci J. 2010 Sep;25(3):169-75. PMID: 21180279..
So far, there is no satisfactory treatment for NAFLD, and comprehensive therapies are usually performed in clinics. There are efficacy and safety concerns regarding a variety of drugs intended for the treatment of NAFLD. Recent studies have shown that curcumin has many effects such as antioxidant and anti-inflammatory effects, and oxygen free radical-scavenging effect. Curcumin is the main active ingredient in turmeric99 Cheng Y, Ping J, Xu LM. Effects of curcumin on peroxisome proliferator-activated receptor gamma expression and nuclear translocation/redistribution in culture-activated rat hepatic stellate cells. Chin Med J (Engl). 2007 May;120(9):794-801. PMID: 17531121., and research has shown that it has some therapeutic effect on NAFLD1010 Nanji AA, Jokelainen K, Tipoe GL, Rahemtulla A, Thomas P, Dannenberg AJ. Curcumin prevents alcohol-induced liver disease in rats by inhibiting the expression of NF-kappa B-dependent genes. Am J Physiol Gastrointest Liver Physiol. 2003 Feb;284(2):G321-7. PMID: 12388178.,1111 Nair P, Malhotra A, Dhawan DK. Curcumin and quercetin trigger apoptosis during benzo(a)pyrene-induced lung. Mol Cell Biochem. 2015 Feb;400(1-2):51-6. doi: 10.1007/s11010-014-2261-6.
https://doi.org/10.1007/s11010-014-2261-...
. However, the specific mechanism is unclear.
Methods
Animals and establishment of NAFLD model
This study was carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health. The animal use protocol has been reviewed and approved by the Institutional Animal Care and Use Committee (IACUC) of the First Affiliated Hospital of Sichuan Medical University.
Fifty-six healthy male Sprague Dawley rats, weighing 210 ± 30 g, were provided by the experimental animal center of Luzhou Medical College. They were raised, 8 rats per cage, in the laboratory animal center of Affiliated Hospital of Luzhou Medical College. Temperature was maintained at 15-25°C with 12 h of light and dark, and all the rats were free to eat and drink. They were randomly divided into the control group (16 rats) and the model group (40 rats), and were fed with a normal diet or high-fat diet (82.5% normal diet + 10% lard + 2% cholesterol (Lanji Technology Development Co., Ltd., Shanghai, China) + 0.5% sodium cholate (Lanji Technology Development Co., Ltd., Shanghai, China) + 5% sucrose), respectively. Rats in both groups were sacrificed at the end of the 8th week, and their livers were taken for pathological examination, in order to confirm that the NAFLD rat model was successfully established.
Drug intervention
At the end of the 8th week, rats in the model group were randomly divided into groups that received low (50 mg/kg/d), medium (100 mg/kg/d), and high (200 mg/kg/d) dose of curcumin (98%; Mann Stewart Biological Technology Co. Ltd., Chengdu, China). The control group received equivoluminal sodium carboxymethyl cellulose (CMC) intragastrically for 4 weeks.
Test of liver function and HE staining of liver tissue
Rats were sacrificed at 8 and 12 weeks (8 rats per group). Serum alanine aminotransferase (ALT), aspartate transaminase (AST), triglyceride (TG), total cholesterol (TC), fasting blood-glucose (FBG), and fasting insulin (FINS) levels, and the insulin resistance index (HOMA-IR) were assessed by a steady state model. HOMA-IR was calculated with the formula: HOMA-IR = (FBG × FINS)/22.5. Liver tissue (1 cm × 1 cm × 0.5 cm) was placed in 4% multi-formaldehyde. After fixing for 24 h, liver tissue was rinsed with water, dehydrated, made transparent, dipped in wax, embedded, and cut into 5 μm sections, which were subjected to HE staining. Histopathologic changes were observed under a microscope, the steatosis and fibrosis of liver fat evaluated and inflammation activity scores were computed1212 Song YM, , Lee YH, Kim JW, Ham DS, Kang ES, Cha BS, Lee HC, Lee BW. Metformin alleviates hepatosteatosis by restoring SIRT1-mediated autophagy induction via an AMP-activated protein kinase-independent pathway. Autophagy. 2015;11(1):46-59. doi: 10.4161/15548627.2014.984271.
https://doi.org/10.4161/15548627.2014.98...
,1313 Hecht L, Weiss R. Nonalcoholic fatty liver disease and type 2 diabetes in obese children. Curr Diab Rep. 2014 Jan;14(1):448. doi: 10.1007/s11892-013-0448-y.
https://doi.org/10.1007/s11892-013-0448-...
.
Immunohistochemistry
Sections were deparaffinized with water and citric acid buffer (pH = 6.0) for high-pressure antigen repair, and soaked for 10 min (in darkness) with 3% methanol and hydrogen peroxide to eliminate the activity of endogenous catalase. Visfatin rabbit-anti-rat polyclonal antibody (Proteintech Bio Technology Co. Ltd., Shanghai, China) and ZAG rabbit-anti-rat polyclonal antibody (1:100; Bioworld Bio Technology Co. Ltd., Guangzhou, China) were added and incubated at 27°C for 60 min. Next, secondary antibody was added and incubated at 27°C for 30 min. 3,3′-Diaminobenzidine (DAB) staining of the sections for 5-10 min was monitored under a light microscope, the sections washed under running water and counterstained with hematoxylin for 2 min. This was then followed by 0.1% dilute hydrochloric acid for differentiation and saturated with lithium carbonate, dehydrated, made transparent, and mounted. Integrated optical density (IOD) of visfatin and ZAG protein via immunohistochemistry was calculated with the software Image-Pro Plus.
Semi-quantitative reverse transcription polymerase chain reaction (RT-PCR) for visfatin mRNA
Liver tissue was placed in liquid nitrogen and crushed. Next, to about 100 mg of crushed tissue were added 1 ml of Trizol and 0.2 ml of chloroform, mixed well, and centrifuged at 12,000 rpm (4°C, 15 min). The upper phase was moved to a clean EP tube, equal volume of isopropanol (0.5 ml) was added to it, and centrifuged at 12,000 rpm (4°C, 10 min). The supernatant was removed, and the pellet was washed with 1 ml of 0.1% diethylpyrocarbonate (DEPC) water solution, mixed well and centrifuged at 8,000 rpm (4°C, 5 min). The supernatant was removed, and the tube dried at room temperature, then 25 μl of 0.1% DEPC water solution was added and mixed well. Added 2 μl RNA extract to a clean EP tube and added 98 μl 0.1% DEPC water, mixed well. Next, 5 μl RNA samples were added to 6× loading buffer for electrophoresis (30 min, 100 V). RNA was extracted by using chloroform-ethanol-75% alcohol method, and 20 μl reverse transcription reaction system was prepared by using the reverse transcription kit (Borik Biotech. Co., Chengdu, China). The PCR mix consisted of 1 μl of cDNA products, 1 µl of upstream and downstream primers (10 µmol/L), 12.5 µl of 2×MasterMix, and 9.5 µl of ddH2O to a total volume of 25 µl. After the reaction, 5 µl of PCR products were used for 1.5% agarose gel electrophoresis (100 V, 15-30 minutes), images collected, gray-scale scanning done and the visfatin/GAPDH ratio calculated. Primer sequences were shown in Table 1.
Statistical analysis
SPSS19.0 software was used to analyze data, and the experimental data were expressed by ±s. One-way ANOVA was used in the comparison between groups, and P < 0.05 was considered statistically significant.
Results
General observations
The rats in the control group were in good mental state, agile, and active. Their fur was bright, and their weight gradually increased on a normal diet. However, in the model group, rats were lazy to move and inactive, their fur was less shiny, and early weight gained on their diet decreased slightly. The state of the intervention group was between that of the control and model group.
Liver index of the rats
The liver index of rats in the model group was significantly increased compared with the control group (P < 0.01), and the liver index increased (P < 0.05) with increase in time. Curcumin administered at low, medium, and high dose could reduce the liver index in rats (treated groups compared to the model group, P < 0.01). The liver index decreased with increasing concentration of curcumin, but there was no significant difference between the groups with high dose (P > 0.05), and the liver index was not reduced to normal with curcumin intervention (P < 0.05).
Liver function of the rats
The serum ALT and AST of the model group increased as compared to the control group (P < 0.01), and continued to increase as time passed (P < 0.05). Curcumin could decrease serum ALT and AST levels in the rats (compared with the model group, P < 0.01). Furthermore, AST and ALT levels decreased with increasing concentration of curcumin and were significantly different between the groups with low, medium, and high dose (P < 0.05). However, the serum levels of ALT and AST did not decrease to normal with curcumin intervention (P < 0.05).
Blood lipid of the rats
The levels of TC and TG in the model group were significantly higher than the control group (P < 0.01) and they increased (P < 0.05) with passing time. Curcumin could decrease serum TC and TG levels in treated rats (compared with the model group, P < 0.05), and the effect was dose dependent. Serum TC levels was significantly different between treated groups (P < 0.01), but there was no significant difference in serum TG (P > 0.05). The serum TC and TG levels were not reduced to normal with curcumin intervention (P < 0.05).
Fasting blood-glucose (FBG), fasting insulin (FINS), and the insulin resistance index (HOMA-IR) of the rats
The FBG, FINS, and HOMA-IR of rats in the model group were increased compared to the control group (P < 0.01) during the same period. The levels of serum FBG, FINS, and HOMA-IR increased with increase in time (P < 0.05). Curcumin could reduce HOMA-IR, FINS, and FBG serum levels in treated rats (P < 0.05), and this effect was dose dependent. HOMA-IR was significantly different between groups with low, medium, and high dose (P < 0.05). However, there was no significant difference between groups with low and medium dose for FBG (P > 0.05) and between groups with medium and high dose for FINS (P > 0.05). The levels of FBG, FINS, and HOMA-IR in the serum were not restored to normal levels with the curcumin intervention (P < 0.05).
Liver pathology of the rats
The examination of the control rats' livers showed that structure of hepatic lobule was intact, hepatic cord was radial, and central vein and portal tracts were clear. Furthermore, there was neither steatosis of the liver cell, ballooning degeneration, nor infiltration by inflammatory cells.
Vacuoles were visible in liver cells of rats in the model group after 8 weeks, showing severe macrovesicular fatty change, obvious ballooning degeneration with infiltration of a small number of inflammatory cells. In the model group, the structural disorder of the liver was observed at 12-weeks. The liver cells were swollen with a large number of lipid droplets. Varying degrees of inflammation with punctiform or piecemeal necrosis could be seen. More necrosis foci than at 8-weeks with obvious ballooning degeneration were observed.
The curcumin-treated groups showed disorder of hepatic lobule structure, and varying degrees of inflammation in lobular and portal tracts, which was slightly lighter than that of the model group at 12-weeks. Visible lipid droplets could be seen in liver cells, with punctiform or piecemeal necrosis. However, the necrosis foci were fewer than that of the model group at 12-weeks with obvious ballooning degeneration. The control group was similar to the model group (Figure 1).
HE staining of liver tissues (×200). A: The control group; B: The model group at 8th week; C: The model group at 12thweek; D: The low dose group at 12th week; E: The medium dose group at 12th week; F: The high dose group at 12th week.
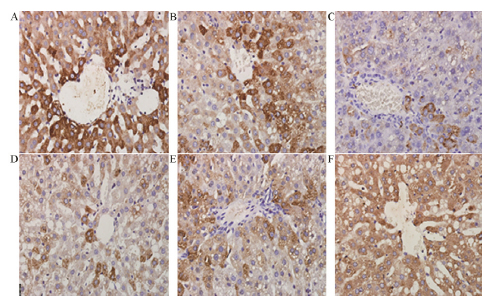
Visfatin and ZAG protein expression in rat livers by immunohistochemistry
Compared with the control group, visfatin level increased (P < 0.01) but ZAG level decreased in the model group (P < 0.01). This trend was maintained with increasing time (P < 0.01). Curcumin could reduce visfatin levels and increase ZAG levels in liver tissue of rats (compared with the model group, P < 0.01), and the effect was dose dependent. Visfatin and ZAG levels in liver tissue were significantly different between groups treated with low, medium, and high dose (P < 0.01). Curcumin intervention did not restore either visfatin or ZAG levels to normal (P < 0.05). The control group was similar to the model group (Table 2, Figures 2 and 3).
Expression of visfatin of liver tissues (×400). A: The control group; B: The model group at 8th week; C: The model group at 12thweek; D: The low dose group at 12th week; E: The medium dose group at 12th week; F: The high dose group at 12th week.
Expression of ZAG of liver tissues (×400). A: The control group; B: The model group at 8th week; C: The model group at 12thweek; D: The low dose group at 12th week; E: The medium dose group at 12th week; F: The high dose group at 12th week.
Visfatin mRNA expression in rat livers by semi quantitative RT-PCR
In the model group, the level of visfatin mRNA was significantly higher than the control group at the same time (P < 0.01). Visfatin mRNA level increased with time while establishing the model (P < 0.01). Curcumin could reduce visfatin mRNA levels in liver tissue of rats (P < 0.01), and the effect was dose dependent. There was significant difference between groups treated with low, medium, and high dose (P < 0.05), but the visfatin mRNA level in liver tissues was not restored to normal with the curcumin intervention (P < 0.05). The control group was similar to the model group (Table 3, Figure 4).
Expression of visfatin mRNA in liver tissues of rats by RT-PCR. 1: The control group at 8th week; 2: The model group at 8th week; 3: The controlgroup at 12th week; 4: The model group at 12th week; 5: The low dose group at 12th week; 6: The medium dose group at 12th week; 7: The high dose group at 12th week.
Discussion
Fukuhara et al.1414 Fukuhara A, Matsuda M, Nishizawa M, Segawa K, Tanaka M, Kishimoto K, Matsuki Y, Murakami M, Ichisaka T, Murakami H, Watanabe E, Takagi T, Akiyoshi M, Ohtsubo T, Kihara S, Yamashita S, Makishima M, Funahashi T, Yamanaka S, Hiramatsu R, Matsuzawa Y, Shimomura I. Visfatin: a protein secreted by visceral fat that mimics the effects of insulin. Science. 2005;307(5708):426-30. PMID: 15604343. found a new type of fat factor visfatin, which was expressed in visceral fat. Visfatin can promote the accumulation of triglyceride in front of fat cells, accelerate synthesis of glucose, and induce differentiation of fat cells. Treatment with visfatin can induce expression of adiponectin and fatty acid synthetase (FAS). In addition, circulating visfatin promotes nicotinamide adenine dinucleotide (NAD) biosynthesis activity1515 Revollo JR, Körner A, Mills KF, Satoh A, Wang T, Garten A, Dasgupta B, Sasaki Y, Wolberger C, Townsend RR, Milbrandt J, Kiess W, Imai S. Nampt/PBEF/Visfatin regulates insulin secretion in beta cells as a systemi NAD biosynthetic enzyme. Cell Metab. 2007 Nov;6(5):363-75. PMID: 17983582.. It can regulate the function of islet β cell by maintaining NM levels1616 Revollo JR, Grimm AA, Imai S. The regulation of nicotinamide adenine dinucleotide biosynthesis by Nampt/PBEF/visfatin in mammals. Curr Opin Gastroenterol. 2007 Mar;23(2):164-70. PMID: 17268245.. Meanwhile, visfatin is an inflammatory mediator and recombinant visfatin can induce tumor necrosis factor (TNF)-ɑ and interleukin (IL)-6. The expression of IL-6 mRNA in the small intestine of rats and the level of circulating IL-6 both increased by intraperitoneal injection of visfatin1717 Moschen AR, Kaser A, Enrich B, Mosheimer B, Theurl M, Niederegger H, Tilg H. Visfatin, an adipocytokine with proinflammatory and immunomodulating properties. J Immunol. 2007 Feb;178(3):1748-58. PMID: 17237424.. Visfatin can also promote the production of monocyte chemotactic protein 1 (MCP-1)1818 Liu SW, Qiao SB, Yuan JS, Liu DQ. Visfatin stimulates production of monocyte chemotactic protein-1 and interleukin-6 in human vein umbilical endothelial cells. Horm Metab Res. 2009 Apr;41(4):281-6. doi: 10.1055/s-0028-1102914.
https://doi.org/10.1055/s-0028-1102914...
.
The concentration of serum visfatin in NAFLD group was significantly higher than the control group1919 Auguet T, Terra X, Porras JA, Orellana-Gavaldà JM, Martinez S, Aguilar C, Lucas A, Pellitero S, Hernández M, Del Castillo D, Richart C. Plasma visfatin levels and gene expression in morbidly obese women with associated fatty liver disease. Clin Biochem. 2013 Feb;46(3):202-8. doi: 10.1016/j.clinbiochem.2012.
https://doi.org/10.1016/j.clinbiochem.20...
, which may be due to positive-negative feedback loop with the visfatin level and IL-6 interacting with each other in NAFLD patients2020 Jarrar MH, Baranova A, Collantes R, Ranard B, Stepanova M, Bennett C, Fang Y, Elariny H, Goodman Z, Chandhoke V, Younossi ZM. Adipokines and cytokines in non-alcoholic fatty liver disease. Aliment Pharmacol Ther. 2008 Mar;27(5):412-21. PMID: 18081738.. Visfatin can promote the secretion of IL-6, and IL-6 has a negative regulatory effect on the expression of visfatin2121 Teoh N, Field J, Farrell G. Interleukin-6 is a key mediator of the hepatoprotective and pro-proliferative effects of ischaemic preconditioning in mice. J Hepatol. 2006 Jul;45(1):20-7. PMID: 16600417.. Interaction with each other can promote the differentiation of the former fat cells and increase insulin resistance (IR). On the contrary, IR with high serum insulin may lead to an increase in IL-6 and visfatin, and promote hepatic steatosis, resulting in the occurrence of NAFLD. Visfatin can also promote the production of ROS2222 Oita RC, Ferdinando D, Wilson S, Bunce C, Mazzatti DJ. Visfatin induces oxidative stress in differentiated C2C12 myotubes in an Akt- and MAPK-independent, NFkB-dependent manner. Pflugers Arch. 2010 Mar;459(4):619-30. doi: 10.1007/s00424-009-0752-1.
https://doi.org/10.1007/s00424-009-0752-...
, causing further inflammation, necrosis, and fibrosis of the fatty liver. The expression of NAFLD in liver of patients with morbid obesity increased, and its expression was positively correlated with stage of liver fibrosis2323 Kukla M, Ciupinska-Kajor M, Kajor M, Wylezol M, Zwirska-Korczala K, Hartleb M, Berdowska A, Mazur W. Liver visfatin expression in morbidly obese patients with nonalcoholic fatty liver disease undergoing bariatric surgery. Pol J Pathol. 2010;61(3):147-53. PMID: 21225497..
Our results showed that expression of visfatin protein could be detected in the control group, while in the model group in the same period levels of serum TC and TG increased, which promotes liver steatosis, and expression of visfatin in liver tissue increased (compared with control group). It can be inferred that the expression of visfatin can be induced by steatosis of liver cells. Compared with the control group, serum FBG, FINS, and HOMA-IR of the model group increased, indicating obvious IR. With the increase in duration of high-fat feeding, the degree of fatty degeneration of liver tissue in rats was aggravated, and the expression of visfatin protein in liver tissue gradually increased. Furthermore, the hyperinsulinemia and pathological changes to liver tissue in the model group further deteriorated. This shows that when fat in liver cells degenerated, the compensatory expression of visfatin protein increased and the function of islet β cell was regulated. However, overexpression of visfatin protein aggravated IR, which causes the production of IL-6, reactive oxygen species (ROS), and so on, and further damages liver function.
ZAG is secreted by fat cells and epithelial cells2424 Bing C, Mracek T, Gao D, Trayhurn P. Zinc-a2-glycoprotein: an adipokine modulator of body fat mass? Int J Obes (Lond). 2010 Nov;34(11):1559-65. doi: 10.1038/ijo.2010.105.
https://doi.org/10.1038/ijo.2010.105...
. Studies have shown that ZAG could reduce fat in mice, increase serum FFA level, and stimulate fat decomposition of fat cells. Lipolysis is carried out by upregulation of cAMP level via adrenergic β3 receptor2525 Russell ST, Hirai K, Tisdale MJ. Role of beta3-adrenergic receptors in the action of a tumour lipid mobilizing factor. Br J Cancer. 2002 Feb;86(3):424-8. PMID: 11875710.. At the same time, by regulating FAS activity, ZAG can maintain the balance of lipid metabolism in the liver2626 Liu W, Baker SS, Baker RD, Zhu L. Antioxidant mechanisms in nonalcoholic fatty liver disease. Curr Drug Targ. 2015;16(12):1301-14. PMID: 25915484.. Moreover, ZAG can also inhibit the differentiation of mouse cells and stimulate cell proliferation, the former may be due to the declined expression of CCAAT-enhancer-binding protein α (C/EBPα), peroxisome proliferator-activated receptor γ (PPARγ), and FAS2727 Zhu HJ, Ding HH, Deng JY, Pan H, Wang LJ, Li NS, Wang XQ, Shi YF, Gong FY. Inhibition of preadipocyte differentiation and adipogenesis by zinc-a2-glycoprotein treatment in 3T3-L1 cells. J Diabetes Investig. 2013 May;4(3):252-60. doi: 10.1111/jdi.12046.
https://doi.org/10.1111/jdi.12046...
.
The expression level of ZAG was low in obese mice, inducing increased expression of ZAG. Consequently, the weight of mice reduced and fat tissue was decreased2828 Imajo K, Yoneda M, Ogawa Y, Wada K, Nakajima A. Microbiota and nonalcoholic steatohepatitis. Semin Immunopathol. 2014 Jan;36(1):115-32. doi: 10.1007/s00281-013-0404-6.
https://doi.org/10.1007/s00281-013-0404-...
. Mracek et al.2929 Mracek T, Gao D, Tzanavari T, Bao Y, Xiao X, Stocker C, Trayhurn P, Bing C. Downregulation of zinc-{alpha}2-glycoprotein in adipose tissue and liver of obese ob/ob mice and by tumour necrosis factor-alpha in adipocytes. J Endocrinol. 2010 Feb;204(2):165-72. doi: 10.1677/JOE-09-0299.
https://doi.org/10.1677/JOE-09-0299...
found that the expression level of ZAG mRNA in normal mice was higher than that of the NAFLD model mice, which may due to the increased expression of TNF-α2121 Teoh N, Field J, Farrell G. Interleukin-6 is a key mediator of the hepatoprotective and pro-proliferative effects of ischaemic preconditioning in mice. J Hepatol. 2006 Jul;45(1):20-7. PMID: 16600417.. The expression of ZAG in the liver decreased, activity of ACC2 and CPT1 oxidated by fatty acid β declined in the lipid decomposition pathway, and the activity of ACC1 and ME enhanced in the lipid synthesis pathway3030 Choi JW, Liu H, Mukherjee R, Yun JW. Downregulation of fetuin-B and zinc-a2-glycoprotein is linked to impaired fatty acid metabolism in liver cells. Cell Physiol Biochem. 2012;30(2):295-306. doi: 10.1159/000339065.
https://doi.org/10.1159/000339065...
, these changes led to disorders of lipid metabolism, accumulation of liver fat and liver steatosis, ultimately forming NAFLD.
Our results showed that compared with the control group, the expression of ZAG in the model group was significantly decreased, and that with increasing duration of high-fat feeding, the expression further decreased. The high level of insulin, hyperlipidemia, hyperglycemia, and aggravated pathological changes of liver tissue, proving that ZAG of the liver tissue played a protective role in the development of NAFLD. The decline in ZAG expression of liver tissue could lead to disorder of hepatic lipid metabolism, thus forming NAFLD.
The severity of IR and lipid metabolism disorders in NAFLD patients was positively correlated with the progression of NAFLD3131 Ying X, Jiang Y, Qian Y, Jiang Z, Song Z, Zhao C. Association between insulin resistance, metabolic syndrome and nonalcoholic fatty liver disease in chinese adults. Iran J Public Health. 2012;41(1):45-9. PMID: 23113121.. Therefore, improving IR and correcting disorders of lipid metabolism are one of the methods for the treatment of NAFLD. Studies have found that curcumin can regulate the activity of hepatic stellate cells, thus improving liver cell function3232 Subudhi U, Das K, Paital B. Supplementation of curcumin and vitamin E enhances oxidative stress, but restores hepatic histoarchitecture inhypothyroid rats. Life Sci. 2009;84(11-12):372-9. doi: 10.1016/j.lfs.2008.12.024.
https://doi.org/10.1016/j.lfs.2008.12.02...
. By inhibiting the biosynthesis of endogenous cholesterol, curcumin can reduce the free cholesterol in fatty liver cells3333 Arafa HM. Curcumin attenuates diet-induced hypercholesterolemia in rats. Med Sci Monit. 2005 Jul;11(7):BR228-34. PMID: 15990684.. Studies showed that the protective effect of curcumin on liver may be related to the decrease of MCP-1, CD11b, and TIMP-13434 Vizzutti F, Provenzano A, Galastri S, Milani S, Delogu W, Novo E, Caligiuri A, Zamara E, Arena U, Laffi G, Parola M, Pinzani M, Marra F. Curcumin limits the fibrogenic evolution of experimental steatohepatitis. Lab Invest. 2010 Jan;90(1):104-15. doi: 10.1038/labinvest.2009.112.
https://doi.org/10.1038/labinvest.2009.1...
, and influence the expression of TNF-α and TGF-β13535 Xue X, Yu JL, Sun DQ, Zou W, Kong F, Wu J, Liu HP, Qu XJ, Wang RM. Curcumin as a multidrug resistance modulator: a quick review. Biomed Prevent Nutr. 2013 Apr;3(2):173-6. doi: 10.1016/j.bionut.2012.12.001.
https://doi.org/10.1016/j.bionut.2012.12...
,3636 Jeon Y, Kim CE, Jung D, Kwak K, Park S, Lim D, Kim S, Baek W. Curcumin could prevent the development of chronic neuropathic pain in rats with peripheral nerve injury. Curr Ther Res Clin Rep. 2013 Jun;74:1-4. doi: 10.1016/j.curtheres.2012.10.001.
https://doi.org/10.1016/j.curtheres.2012...
. Curcumin can activate AMPK and reduce the expression of SREBP-1 and Fas, finally inhibiting liver fat and improving the antioxidant capacity of liver3737 Kang OH, Kim SB, Seo YS, Joung DK, Mun SH, Choi JG, Lee YM, Kang DG, Lee HS, Kwon DY. Curcumin decreases oleic acid-induced lipid accumulation via AMPK phosphorylation in hepatocarcinoma cells. Eur Rev Med Pharmacol Sci. 2013 Oct;17(19):2578-86. PMID: 24142602.. Vera-Ramirez et al.3838 Vera-Ramirez L, Pérez-Lopez P, Varela-Lopez A, Ramirez-Tortosa M, Battino M, Quiles JL. Curcumin and liver disease. Biofactors. 2013 Jan-Feb;39(1):88-100. doi: 10.1002/biof.1057.
https://doi.org/10.1002/biof.1057...
found that curcumin had a vital effect on NF-κβ in the treatment of NAFLD. Nabavi et al.3939 Nabavi SF, Thiagarajan R, Rastrelli L, Daglia M, Sobarzo-Sánchez E, Alinezhad H, Nabavi SM. Curcumin: a natural product for diabetes and its complications. Curr Top Med Chem. 2015;15(23):2445-55. PMID: 26088351. suggested that curcumin could significantly decrease the serum parameters of non-alcoholic fatty liver, and its mechanism was related to the regulation of mitochondrial function and the maintenance of normal structure of liver cells.
In this study, we showed that the expression of visfatin protein increased and that of ZAG decreased in NAFLD rats. The change in expression of ZAG and visfatin in liver tissue of the rats induced NAFLD. Therefore, is the expression of visfatin and ZAG protein a new mechanism of NAFLD, and can curcumin inhibit visfatin expression and induce ZAG expression in liver tissue for the treatment of NAFLD? Although the effect of curcumin as an anti-inflammatory and anti-oxidant has been proved, further studies on the prevention and treatment of NASH were still needed. In this study, curcumin treatment was used for NAFLD rats fed on a high fat diet for 4 weeks, and it was found that it could significantly improve liver function, relieve hepatic steatosis and inflammation, thereby proving the therapeutic effect of curcumin on NAFLD. Palma et al.4040 Palma HE, Wolkmer P, Gallio M, Corrêa MM, Schmatz R, Thomé GR, Pereira LB, Castro VS, Pereira AB, Bueno A, de Oliveira LS, Rosolen D, Mann TR, de Cecco BS, Graça DL, Lopes ST, Mazzanti CM. Oxidative stress parameters in blood, liver, and kidney of diabetic rats treated with curcumin and/or insulin. Mol Cell Biochem. 2014 Jan;386(1-2):199-210. doi: 10.1007/s11010-013-1858-5.
https://doi.org/10.1007/s11010-013-1858-...
found that curcumin can promote differentiation of preadipocytes and reduce visfatin and resistin mRNA expression. This study showed that curcumin could improve IR, and significantly reduce levels of serum TG, TC. It could also increase ZAG expression and reduce visfatin activity as observed by the significant decrease in levels of serum ALT and AST, and improvement of liver histopathology.
In this study, liver fat changes were observed in the 8th and 12th week, and the partial infiltration of inflammatory cells and necrosis of liver cells were observed. AST and ALT levels were significantly increased, indicating that there were varying degrees of liver damage after high-fat feeding. Four weeks after curcumin treatment, liver index of rats decreased (compared with the model group). Furthermore, levels of serum ALT and AST were significantly decreased (compared with the model group), as were levels of serum TC and TG (compared with the model group). These changes were accompanied by decreased expression of visfatin mRNA and protein (compared with the model group). Expression of ZAG in liver tissue increased (compared with the model group). Meanwhile, FBG, FINS, and HOMA-IR serum levels decreased (compared with the model group). These changes would ultimately alleviate hepatic steatosis, inflammatory response, and liver damage. This study shows that curcumin has an effect on regulation of lipid, improving IR, and is anti-inflammatory. It can also improve IR and reduce production of ROS by downregulating the expression of visfatin in liver tissue for the treatment of NAFLD. Furthermore, curcumin can correct the disorders of lipid metabolism by upregulating the expression of ZAG in liver tissue.
In this study, we did not measure TNF-α and IL-6 levels, which was a limitation of the study.
Conclusion
Visfatin and zinc-α2-glycoprotein may be involved in the pathogenesis of NAFLD. Treatment of NAFLD in rats by curcumin may be mediated by the decrease of visfatin and the increase of non-alcoholic fatty liver disease.
References
-
1Nishioji K, Sumida Y, Kamaguchi M, Mochizuki N, Kobayashi M, Nishimura T, Yamaguchi K, Itoh Y. Prevalence of and risk factors for non-alcoholic fatty liver disease in a non-obese Japanese population, 2011-2012. J Gastroenterol. 2015 Jan;50(1):95-108. doi: 10.1007/s00535-014-0948-9.
» https://doi.org/10.1007/s00535-014-0948-9 -
2Priore P, Cavallo A, Gnoni A, Damiano F, Gnoni GV, Siculella L. Modulation of hepatic lipid metabolism by olive oil and its phenols in nonalcoholic fatty liver disease. IUBMB Life. 2015 Jan;67(1):9-17. doi: 10.1002/iub.1340.
» https://doi.org/10.1002/iub.1340 -
3Machado MV, Michelotti GA, Pereira Tde A, Boursier J, Kruger L, Swiderska-Syn M, Karaca G, Xie G, Guy CD, Bohinc B, Lindblom KR, Johnson E, Kornbluth S, Diehl AM. Reduced lipoapoptosis, hedgehog pathway activation and fibrosis in caspase-2 deficient mice with non-alcoholic steatohepatitis. Gut. 2015 Jul;64(7):1148-57. doi: 10.1136/gutjnl-2014-307362.
» https://doi.org/10.1136/gutjnl-2014-307362 -
4Ferreira DM, Castro RE, Machado MV, Evangelista T, Silvestre A, Costa A, Coutinho J, Carepa F, Cortez-Pinto H, Rodrigues CM. Apoptosis and insulin resistance in liver and peripheral tissues of morbidly obese patients is associated with different stages of non-alcoholic fatty liver disease. Diabetologia. 2011 Jul;54(7):1788-98. doi: 10.1007/s00125-011-2130-8.
» https://doi.org/10.1007/s00125-011-2130-8 -
5Lam B, Younossi ZM. Treatment options for nomalcoholic fatty liver disease. Therp Adv Gastroenterol. 2010 Mar;3(2):121-37. doi: 10.1177/1756283X09359964.
» https://doi.org/10.1177/1756283X09359964 -
6Russell ST, Tisdale MJ. Role of ß-adrenergic receptors in the anti-obesity and anti-diabetic effects of zinc-a2-glycoprotien (ZAG). Biochim Biophys Acta. 2012 Apr;1821(4):590-9. doi: 10.1016/j.bbalip.2011.12.003.
» https://doi.org/10.1016/j.bbalip.2011.12.003 -
7Zhang L, Zhang HY, Ma CC, Wei W, Jia LH. Increased body weight induced by perinatal exposure to bisphenol A was associated with down-regulation zinc-alpha2-glycoprotein expression in offspring female rats. Mol Cell Toxicol. 2014 Jun;10(2):207-13. doi: 10.1007/s13273-014-0022-y.
» https://doi.org/10.1007/s13273-014-0022-y -
8Gong FY, Deng JY, Zhu HJ, Pan H, Wang LJ, Yang HB. Fatty acid synthase and hormone-sensitive lipase expression in liver are involved in zinc-alpha2-glycoprotein-induced body fat loss in obese mice. Chin Med Sci J. 2010 Sep;25(3):169-75. PMID: 21180279.
-
9Cheng Y, Ping J, Xu LM. Effects of curcumin on peroxisome proliferator-activated receptor gamma expression and nuclear translocation/redistribution in culture-activated rat hepatic stellate cells. Chin Med J (Engl). 2007 May;120(9):794-801. PMID: 17531121.
-
10Nanji AA, Jokelainen K, Tipoe GL, Rahemtulla A, Thomas P, Dannenberg AJ. Curcumin prevents alcohol-induced liver disease in rats by inhibiting the expression of NF-kappa B-dependent genes. Am J Physiol Gastrointest Liver Physiol. 2003 Feb;284(2):G321-7. PMID: 12388178.
-
11Nair P, Malhotra A, Dhawan DK. Curcumin and quercetin trigger apoptosis during benzo(a)pyrene-induced lung. Mol Cell Biochem. 2015 Feb;400(1-2):51-6. doi: 10.1007/s11010-014-2261-6.
» https://doi.org/10.1007/s11010-014-2261-6 -
12Song YM, , Lee YH, Kim JW, Ham DS, Kang ES, Cha BS, Lee HC, Lee BW. Metformin alleviates hepatosteatosis by restoring SIRT1-mediated autophagy induction via an AMP-activated protein kinase-independent pathway. Autophagy. 2015;11(1):46-59. doi: 10.4161/15548627.2014.984271.
» https://doi.org/10.4161/15548627.2014.984271 -
13Hecht L, Weiss R. Nonalcoholic fatty liver disease and type 2 diabetes in obese children. Curr Diab Rep. 2014 Jan;14(1):448. doi: 10.1007/s11892-013-0448-y.
» https://doi.org/10.1007/s11892-013-0448-y -
14Fukuhara A, Matsuda M, Nishizawa M, Segawa K, Tanaka M, Kishimoto K, Matsuki Y, Murakami M, Ichisaka T, Murakami H, Watanabe E, Takagi T, Akiyoshi M, Ohtsubo T, Kihara S, Yamashita S, Makishima M, Funahashi T, Yamanaka S, Hiramatsu R, Matsuzawa Y, Shimomura I. Visfatin: a protein secreted by visceral fat that mimics the effects of insulin. Science. 2005;307(5708):426-30. PMID: 15604343.
-
15Revollo JR, Körner A, Mills KF, Satoh A, Wang T, Garten A, Dasgupta B, Sasaki Y, Wolberger C, Townsend RR, Milbrandt J, Kiess W, Imai S. Nampt/PBEF/Visfatin regulates insulin secretion in beta cells as a systemi NAD biosynthetic enzyme. Cell Metab. 2007 Nov;6(5):363-75. PMID: 17983582.
-
16Revollo JR, Grimm AA, Imai S. The regulation of nicotinamide adenine dinucleotide biosynthesis by Nampt/PBEF/visfatin in mammals. Curr Opin Gastroenterol. 2007 Mar;23(2):164-70. PMID: 17268245.
-
17Moschen AR, Kaser A, Enrich B, Mosheimer B, Theurl M, Niederegger H, Tilg H. Visfatin, an adipocytokine with proinflammatory and immunomodulating properties. J Immunol. 2007 Feb;178(3):1748-58. PMID: 17237424.
-
18Liu SW, Qiao SB, Yuan JS, Liu DQ. Visfatin stimulates production of monocyte chemotactic protein-1 and interleukin-6 in human vein umbilical endothelial cells. Horm Metab Res. 2009 Apr;41(4):281-6. doi: 10.1055/s-0028-1102914.
» https://doi.org/10.1055/s-0028-1102914 -
19Auguet T, Terra X, Porras JA, Orellana-Gavaldà JM, Martinez S, Aguilar C, Lucas A, Pellitero S, Hernández M, Del Castillo D, Richart C. Plasma visfatin levels and gene expression in morbidly obese women with associated fatty liver disease. Clin Biochem. 2013 Feb;46(3):202-8. doi: 10.1016/j.clinbiochem.2012.
» https://doi.org/10.1016/j.clinbiochem.2012 -
20Jarrar MH, Baranova A, Collantes R, Ranard B, Stepanova M, Bennett C, Fang Y, Elariny H, Goodman Z, Chandhoke V, Younossi ZM. Adipokines and cytokines in non-alcoholic fatty liver disease. Aliment Pharmacol Ther. 2008 Mar;27(5):412-21. PMID: 18081738.
-
21Teoh N, Field J, Farrell G. Interleukin-6 is a key mediator of the hepatoprotective and pro-proliferative effects of ischaemic preconditioning in mice. J Hepatol. 2006 Jul;45(1):20-7. PMID: 16600417.
-
22Oita RC, Ferdinando D, Wilson S, Bunce C, Mazzatti DJ. Visfatin induces oxidative stress in differentiated C2C12 myotubes in an Akt- and MAPK-independent, NFkB-dependent manner. Pflugers Arch. 2010 Mar;459(4):619-30. doi: 10.1007/s00424-009-0752-1.
» https://doi.org/10.1007/s00424-009-0752-1 -
23Kukla M, Ciupinska-Kajor M, Kajor M, Wylezol M, Zwirska-Korczala K, Hartleb M, Berdowska A, Mazur W. Liver visfatin expression in morbidly obese patients with nonalcoholic fatty liver disease undergoing bariatric surgery. Pol J Pathol. 2010;61(3):147-53. PMID: 21225497.
-
24Bing C, Mracek T, Gao D, Trayhurn P. Zinc-a2-glycoprotein: an adipokine modulator of body fat mass? Int J Obes (Lond). 2010 Nov;34(11):1559-65. doi: 10.1038/ijo.2010.105.
» https://doi.org/10.1038/ijo.2010.105 -
25Russell ST, Hirai K, Tisdale MJ. Role of beta3-adrenergic receptors in the action of a tumour lipid mobilizing factor. Br J Cancer. 2002 Feb;86(3):424-8. PMID: 11875710.
-
26Liu W, Baker SS, Baker RD, Zhu L. Antioxidant mechanisms in nonalcoholic fatty liver disease. Curr Drug Targ. 2015;16(12):1301-14. PMID: 25915484.
-
27Zhu HJ, Ding HH, Deng JY, Pan H, Wang LJ, Li NS, Wang XQ, Shi YF, Gong FY. Inhibition of preadipocyte differentiation and adipogenesis by zinc-a2-glycoprotein treatment in 3T3-L1 cells. J Diabetes Investig. 2013 May;4(3):252-60. doi: 10.1111/jdi.12046.
» https://doi.org/10.1111/jdi.12046 -
28Imajo K, Yoneda M, Ogawa Y, Wada K, Nakajima A. Microbiota and nonalcoholic steatohepatitis. Semin Immunopathol. 2014 Jan;36(1):115-32. doi: 10.1007/s00281-013-0404-6.
» https://doi.org/10.1007/s00281-013-0404-6 -
29Mracek T, Gao D, Tzanavari T, Bao Y, Xiao X, Stocker C, Trayhurn P, Bing C. Downregulation of zinc-{alpha}2-glycoprotein in adipose tissue and liver of obese ob/ob mice and by tumour necrosis factor-alpha in adipocytes. J Endocrinol. 2010 Feb;204(2):165-72. doi: 10.1677/JOE-09-0299.
» https://doi.org/10.1677/JOE-09-0299 -
30Choi JW, Liu H, Mukherjee R, Yun JW. Downregulation of fetuin-B and zinc-a2-glycoprotein is linked to impaired fatty acid metabolism in liver cells. Cell Physiol Biochem. 2012;30(2):295-306. doi: 10.1159/000339065.
» https://doi.org/10.1159/000339065 -
31Ying X, Jiang Y, Qian Y, Jiang Z, Song Z, Zhao C. Association between insulin resistance, metabolic syndrome and nonalcoholic fatty liver disease in chinese adults. Iran J Public Health. 2012;41(1):45-9. PMID: 23113121.
-
32Subudhi U, Das K, Paital B. Supplementation of curcumin and vitamin E enhances oxidative stress, but restores hepatic histoarchitecture inhypothyroid rats. Life Sci. 2009;84(11-12):372-9. doi: 10.1016/j.lfs.2008.12.024.
» https://doi.org/10.1016/j.lfs.2008.12.024 -
33Arafa HM. Curcumin attenuates diet-induced hypercholesterolemia in rats. Med Sci Monit. 2005 Jul;11(7):BR228-34. PMID: 15990684.
-
34Vizzutti F, Provenzano A, Galastri S, Milani S, Delogu W, Novo E, Caligiuri A, Zamara E, Arena U, Laffi G, Parola M, Pinzani M, Marra F. Curcumin limits the fibrogenic evolution of experimental steatohepatitis. Lab Invest. 2010 Jan;90(1):104-15. doi: 10.1038/labinvest.2009.112.
» https://doi.org/10.1038/labinvest.2009.112 -
35Xue X, Yu JL, Sun DQ, Zou W, Kong F, Wu J, Liu HP, Qu XJ, Wang RM. Curcumin as a multidrug resistance modulator: a quick review. Biomed Prevent Nutr. 2013 Apr;3(2):173-6. doi: 10.1016/j.bionut.2012.12.001.
» https://doi.org/10.1016/j.bionut.2012.12.001 -
36Jeon Y, Kim CE, Jung D, Kwak K, Park S, Lim D, Kim S, Baek W. Curcumin could prevent the development of chronic neuropathic pain in rats with peripheral nerve injury. Curr Ther Res Clin Rep. 2013 Jun;74:1-4. doi: 10.1016/j.curtheres.2012.10.001.
» https://doi.org/10.1016/j.curtheres.2012.10.001 -
37Kang OH, Kim SB, Seo YS, Joung DK, Mun SH, Choi JG, Lee YM, Kang DG, Lee HS, Kwon DY. Curcumin decreases oleic acid-induced lipid accumulation via AMPK phosphorylation in hepatocarcinoma cells. Eur Rev Med Pharmacol Sci. 2013 Oct;17(19):2578-86. PMID: 24142602.
-
38Vera-Ramirez L, Pérez-Lopez P, Varela-Lopez A, Ramirez-Tortosa M, Battino M, Quiles JL. Curcumin and liver disease. Biofactors. 2013 Jan-Feb;39(1):88-100. doi: 10.1002/biof.1057.
» https://doi.org/10.1002/biof.1057 -
39Nabavi SF, Thiagarajan R, Rastrelli L, Daglia M, Sobarzo-Sánchez E, Alinezhad H, Nabavi SM. Curcumin: a natural product for diabetes and its complications. Curr Top Med Chem. 2015;15(23):2445-55. PMID: 26088351.
-
40Palma HE, Wolkmer P, Gallio M, Corrêa MM, Schmatz R, Thomé GR, Pereira LB, Castro VS, Pereira AB, Bueno A, de Oliveira LS, Rosolen D, Mann TR, de Cecco BS, Graça DL, Lopes ST, Mazzanti CM. Oxidative stress parameters in blood, liver, and kidney of diabetic rats treated with curcumin and/or insulin. Mol Cell Biochem. 2014 Jan;386(1-2):199-210. doi: 10.1007/s11010-013-1858-5.
» https://doi.org/10.1007/s11010-013-1858-5
-
Financial sources:
Science and Technology Department and Technology Bureau, Joint Project of Luzhou Medical College (No. 14JC0087); Medical Association of Sichuan province (No. IJ2015026) -
1
Research performed at Department of Gastroenterology, Affiliated Hospital, Southwest Medical University, Luzhou, China.
Publication Dates
-
Publication in this collection
Nov 2016
History
-
Received
14 July 2016 -
Reviewed
15 Sept 2016 -
Accepted
16 Oct 2016