ABSTRACT
PURPOSE:
To investigate the potential protective effects of enoxaparin against the adverse events of carbon dioxide (CO2) pneumoperitoneum.
METHODS:
Thirty four rats were divided into three groups: Group 1 (sham) underwent insertion of Veress needle into the abdomen and 90 min of anesthesia with no gas insufflation. The animals in control and enoxaparin groups were subjected to 90 min of 14 mmHg CO2 pneumoperitoneum. Enoxaparin (100 u/kg) was administered subcutaneously to the rats in enoxaparin group one hour before the operation. After 90 min of pneumoperitoneum, the rats were allowed for reperfusion through 60 min. Blood and liver samples were obtained for biochemical and histopathological examination.
RESULTS:
Treatment with enoxaparin decreased the histopathological abnormalities when compared with the control group. The highest levels of oxidative stress parameters were found in control group. The use of enoxaparin decreased the levels of all oxidative stress parameters, but the difference between the control and enoxaparin groups was not statistically significant.
CONCLUSION:
Enoxaparin ameliorated the harmful effects of high pressure CO2 pneumoperitoneum on the liver.
Key words:
Laparoscopy; Pneumoperitoneum; Artificial; Carbon Dioxide; Enoxaparin; Liver; Oxidative Stress; Rats
Introduction
Laparoscopic surgery is less damaging to immune functions and has lower rates of systemic inflammation as it is a less invasive method, compared to open abdominal operations. Nonetheless, increased intra-abdominal pressure during pneumoperitoneum and inflation-deflation can lead to splanchnic ischemia-reperfusion and oxidative stress. Hemodynamic changes and the impact of carbon dioxide (CO2) may also exacerbate oxidative stress during pneumoperitoneum11 Arsalani-Zadeh R, Ullah S, Khan S, MacFie J. Oxidative stress in laparoscopic versus open abdominal surgery: a systematic review. J Surg Res. 2011 Jul;169:e59-68. PMID: 21492871.. With the use of high-pressure pneumoperitoneum in laparoscopic surgery, portal venous flow is reduced, which results in physiological changes in hepatic function. These changes in hepatic function during laparoscopic surgery are caused by the duration and level of intra-abdominal pressure22 Hasukic S. Postoperative changes in liver function tests: randomized comparison of low- and high-pressure laparoscopic cholecystectomy. Surg Endosc. 2005;19:1451-5. PMID: 16206003.. With the development of temporary ischemia of the liver during pneumoperitoneum, liver function deteriorates and there is a decreased local cellular immune response. The interlocking mechanisms causing the deleterious effects of pneumoperitoneum on the liver include the changes in hepatic blood flow, changes in monocyte-phagocyte system activity, and decreased immune system function. Abdominal deflation may further aggravate this process, which then creates a reperfusion model in a previously ischemic liver33 Nickkholgh A, Barro-Bejarano M, Liang R, Zorn M, Mehrabi A, Gebhard MM, Büchler MW, Gutt CN, Schemmer P. Signs of reperfusion injury following CO2 pneumoperitoneum: an in vivo microscopy study. Surg Endosc. 2008;22:122-8. PMID: 17483991..
Enoxaparin is one of the most common low molecular weight heparin (LMWH) drugs used to treat many thromboembolic diseases. The safety and efficacy of enoxaparin have been established in a wide range of both arterial and venous thromboembolic conditions. The major anticoagulant effect of the drug is mediated by interaction with antithrombin III, which in turn, inactivates serine protease-like factors IIa (thrombin), IXa and Xa. Thus indirectly, enoxaparin inhibits the conversion of prothrombin to thrombin and the thrombin-mediated conversion of fibrinogen to fibrin is reduced, thereby preventing the formation of clots44 Iqbal Z, Cohen M. Enoxaparin: a pharmacologic and clinical review. Expert Opin Pharmacother. 2011;12:1157-70. PMID: 21470072..
Other than the well-established effects on blood coagulation, studies have evaluated many other activities of LMWH, including anti-inflammatory activity55 Rumelt S, Stolovich C, Segal ZI, Rehany U. Intraoperative enoxaparin minimizes inflammatory reaction after pediatric cataract surgery. Am J Ophthalmol. 2006;141:433-7. PMID: 16490487.
6 Nasiripour S, Gholami K, Mousavi S, Mohagheghi A, Radfar M, Abdollahi M, Khazaeipour Z, Mojtahedzadeh M. Comparison of the effects of enoxaparin and heparin on inflammatory biomarkers in patients with ST-segment elevated myocardial infarction: A prospective open label pilot clinical trial. Iran J Pharm Res. 2014;13:583-90. PMID: 25237354.
7 Baram D, Rashkovsky M, Hershkoviz R, Drucker I, Reshef T, Ben-Shitrit S, Mekori YA. Inhibitory effects of low molecular weight heparin on mediator release by mast cells: preferential inhibition of cytokine production and mast cell-dependent cutaneous inflammation. Clin Exp Immunol. 1997;110:485-91. PMID: 9409655.
8 Shastri MD, Stewart N, Horne J, Zaidi ST, Sohal SS, Peterson GM, Korner H, Gueven N, Patel RP. Non-anticoagulant fractions of enoxaparin suppress inflammatory cytokine release from peripheral blood mononuclear cells of allergic asthmatic individuals. PLoS One. 2015;10(6):e0128803. PMID: 26046354.
9 Shastri MD, Stewart N, Horne J, Peterson GM, Gueven N, Sohal SS, Patel RP. In-vitro suppression of IL-6 and IL-8 release from human pulmonary epithelial cells by non-anticoagulant fraction of enoxaparin. PLoS One. 2015;10:e0126763. PMID: 25961885.
10 Grosskopf I, Shaish A, Ray A, Harats D, Kamari Y. Low molecular weight heparin-induced increase in chylomicron-remnants clearance, is associated with decreased plasma TNF-a level and increased hepatic lipase activity. Thromb Res. 2014;133:688-92. PMID: 24525311.-1111 Lean QY, Eri RD, Randall-Demllo S, Sohal SS, Stewart N, Peterson GM, Gueven N, Patel RP. Orally administered enoxaparin ameliorates acute colitis by reducing macrophage-associated inflammatory responses. PLoS One. 2015;10:e0134259. PMID: 26218284.. These studies have demonstrated that enoxaparin could be effective for managing many inflammatory disorders including, inflammatory reaction after pediatric cataract surgery55 Rumelt S, Stolovich C, Segal ZI, Rehany U. Intraoperative enoxaparin minimizes inflammatory reaction after pediatric cataract surgery. Am J Ophthalmol. 2006;141:433-7. PMID: 16490487., ST-segment elevated myocardial infarction66 Nasiripour S, Gholami K, Mousavi S, Mohagheghi A, Radfar M, Abdollahi M, Khazaeipour Z, Mojtahedzadeh M. Comparison of the effects of enoxaparin and heparin on inflammatory biomarkers in patients with ST-segment elevated myocardial infarction: A prospective open label pilot clinical trial. Iran J Pharm Res. 2014;13:583-90. PMID: 25237354., mast cell-mediated allergic inflammation77 Baram D, Rashkovsky M, Hershkoviz R, Drucker I, Reshef T, Ben-Shitrit S, Mekori YA. Inhibitory effects of low molecular weight heparin on mediator release by mast cells: preferential inhibition of cytokine production and mast cell-dependent cutaneous inflammation. Clin Exp Immunol. 1997;110:485-91. PMID: 9409655., allergic asthma88 Shastri MD, Stewart N, Horne J, Zaidi ST, Sohal SS, Peterson GM, Korner H, Gueven N, Patel RP. Non-anticoagulant fractions of enoxaparin suppress inflammatory cytokine release from peripheral blood mononuclear cells of allergic asthmatic individuals. PLoS One. 2015;10(6):e0128803. PMID: 26046354.,99 Shastri MD, Stewart N, Horne J, Peterson GM, Gueven N, Sohal SS, Patel RP. In-vitro suppression of IL-6 and IL-8 release from human pulmonary epithelial cells by non-anticoagulant fraction of enoxaparin. PLoS One. 2015;10:e0126763. PMID: 25961885., and acute colitis1111 Lean QY, Eri RD, Randall-Demllo S, Sohal SS, Stewart N, Peterson GM, Gueven N, Patel RP. Orally administered enoxaparin ameliorates acute colitis by reducing macrophage-associated inflammatory responses. PLoS One. 2015;10:e0134259. PMID: 26218284.. Enoxaparin has also been shown to be effective against fibrosis progression in the rat model of cirrhosis1212 Assy N, Hussein O, Khalil A, Luder A, Szvalb S, Paizi M, Spira G. The beneficial effect of aspirin and enoxaparin on fibrosis progression and regenerative activity in a rat model of cirrhosis. Dig Dis Sci. 2007;52:1187-93. PMID: 17372820., CCL4-induced necrosis and apoptosis of the liver1313 Kukner A, Tore F, Firat T, Terzi EH, Oner H, Balaban YH, Ozogul C. The preventive effect of low molecular weight heparin on CCL4-induced necrosis and apoptosis in rat liver. Ann Hepatol. 2010;9:445-54. PMID: 21057164., dimethylnitrosamine (DMN)-induced liver fibrosis1414 Lee JH, Lee H, Joung YK, Jung KH, Choi JH, Lee DH, Park KD, Hong SS. The use of low molecular weight heparin-pluronic nanogels to impede liver fibrosis by inhibition the TGF-ß/Smad signaling pathway. Biomaterials. 2011;32:1438-45. PMID: 21074845., radiation-induced liver toxicity1515 Seidensticker M, Seidensticker R, Damm R, Mohnike K, Pech M, Sangro B, Hass P, Wust P, Kropf S, Gademann G, Ricke J. Prospective randomized trial of enoxaparin, pentoxifylline and ursodeoxycholic acid for prevention of radiation-induced liver toxicity. PLoS One. 2014;9:e112731. PMID: 25393877., and experimental cholestatic liver injury1616 Abdel-Salam OM, Baiuomy AR, Ameen A, Hassan NS. A study of unfractionated and low molecular weight heparins in a model of cholestatic liver injury in the rat. Pharmacol Res. 2005;51:59-67. PMID: 15519536.. In addition, the protective effects of heparin have been shown in an experimental hepatic ischemia-reperfusion model1717 Taha MO, Gonçalves PF, Vidigal RO, Sacchi GP, Pinheiro FG, Brandão LS, Feguri S, Moraes-Filho AM, Ghadie MM, Magalhães KG, Simões MJ, Tersariol IL, Monteiro HP, Oliveira-Júnior IS, Jurkiewicz A, Caricati-Neto A. Protective effects of heparin on hepatic ischemia and reperfusion lesions in rabbits. Transplant Proc. 2009;41:812-5. PMID: 19376359..
Based on these effects of enoxaparin, the present study was conducted to investigate the potential protective effects of enoxaparin against the adverse events of high pressure CO2 pneumoperitoneum. The main outcome was to evaluate the effects of enoxaparin on histopathological changes in liver tissue caused by pneumoperitoneum. The possible effects on biochemical parameters and oxidative status were also assessed.
Methods
This study was conducted in the Husnu Sakal Experimental and Clinical Practice Center, Ankara, Turkey. The procedures in this experimental study were performed in accordance with the National Guidelines for the Use and Care of Laboratory Animals and the Animal Ethics Committee of Ankara Education and Research Hospital (Ankara, Turkey) granted approval for the study.
The study was carried out on 34 Wistar-Albino female rats, each weighing 250±25 g. The rats were allowed to adapt to laboratory conditions for 1 week before experimental use. The animals were housed under standard laboratory conditions at a constant temperature (21±2 ̊C) and relative humidity of 65-70% with a 12-hour light-dark cycle. The animals were fed with standard laboratory chow and water ad libitum. No enteral or parenteral antibiotics were administered during the study.
All animals were anesthetized with an intramuscular injection of 50 mg/kg ketamine hydrochloride (Ketalar(r), Parke-Davis, Istanbul) and 5 mg/kg xylazine (Rompun(r), Bayer, Istanbul). The animals were divided into three groups: Group 1 (sham, n=8) underwent insertion of 7 Gauge Veress needle (Wollex(r), Greatcare Medical GmbH, Gildenweg, Germany) into the abdomen and 90 min of anesthesia with no gas insufflation, maintaining intra-abdominal pressure at 0 mmHg. The animals in Group 2 (control, n=12) and Group 3 (enoxaparin group, n=14) were subjected to 90 min of 14 mmHg CO2 pneumoperitoneum. Enoxaparin (100 u/kg) was administered subcutaneously to the rats in Group 3 1 hour before the operation. The pneumoperitoneum was created using an electronic insufflator (Ackermann Instrumente, 15-1711, high performance insufflation system, Germany) through a 7-gauge Veress needle maintaining intra-abdominal pressure at 14 mmHg. After 90 min of pneumoperitoneum, the Veress needles were withdrawn and the abdomens of the rats were desufflated. The rats were then allowed free movement for reperfusion through 60 min. At the end of this period, blood and liver samples were obtained for biochemical and histopathological examination.
The histopathological analyses were performed in the Pathology Department of Ankara Education and Research Hospital. For light microscopic analyses, the samples were obtained from the liver and fixed in 10% neutral buffered formalin solution for 2 days. The tissues were washed in running water and were dehydrated with increasing concentrations of ethanol (50%, 75%, 96%, 100%). After dehydration, the specimens were put into xylene to obtain transparency and were then infiltrated with and embedded in paraffin. The embedded tissues were cut into 4-μm thick sections using a Leica RM 2125 RT microtome and stained with hematoxylin & eosin. Histopathological examinations were performed with a light microscope (Olympus BX51TF, Tokyo, Japan) by a pathologist blinded to the study design. Histopathological parameters including sinusoidal congestion, hepatocyte necrosis and liver cell vacuolization were evaluated semi-quantitatively according to the modified Suzuki scoring system1818 Suzuki S, Toledo-Peneyra LH, Rodriquez FJ, Cejalvo D. Neutrophil infiltration as an important factor in liver ischemia and reperfusion injury. Modulating effects of FK506 and cylosporine. Transplantation. 1993;55:1265-72. PMID: 7685932.. The scoring system of these parameters is given in Table 1.
Serum alanine aminotransferase (ALT), aspartate aminotransferase (AST), and alkaline phosphatase (ALP) measurements were performed with a clinical chemistry analyzer Beckman Coulter 5800 using Beckman Coulter commercial kits.
The evaluation of oxidative stress parameters was performed in the Biochemistry Department of Ankara Education and Research Hospital. Tissues were stored at -80°C until the assays. Tissue malondialdehyde (MDA), advanced oxidation protein products (AOPP) levels and tissue fluorescent oxidation products (TFOP) were measured.
MDA levels were calculated using the fluorometric method, as described by Wasowicz et al.1919 Wasowicz W, Nève J, Peretz A. Optimized steps in fluorometric determination of thiobarbituric acid-reactive substances in serum: importance of extraction pH and influence of sample preservation and storage. Clin Chem. 1993;39:2522-6. PMID: 8252725. After the reaction of thiobarbituric acid (TBA) with MDA, the reaction product was extracted in butanol and was measured spectrofluorometrically at wavelengths of 525 nm for excitation and 547 nm for emission. 0-5 μmol/L 1,1',3,3'-tetraethoxypropane solution was used as standard. For the measurement of tissue MDA levels; 50 μL of homogenate was added and introduced into 10 mL glass tubes containing 1 mL of distilled water. After the addition of 1 mL of the solution containing 29 mmol/L TBA in acetic acid and mixing, the samples were placed in a water bath and heated for 1 h at 95-100°C. The samples were then cooled, 25 μL of 5 mol/L hydrochloric acid (HCL) was added and the reaction mixture was extracted by agitation for 5 min with 3.5 mL n-butanol. After separation of the butanol phase by centrifugation at 1500 x g for 10 min, the fluorescence of the butanol extract was measured with a fluorometer (HITACHI F-2500) at wave-lengths of 525 nm for excitation and 547 nm for emission. 0-5 μmol/L 1,1',3,3'-tetraethoxypropane solutions were used as standard. MDA levels were presented as μmol/g wet tissue.
AOPP were determined according to method described by Witko-Sarsat et al.2020 Witko-Sarsat V, Friedlander M, Nguyen Khoa T, Capeillère-Blandin C, Nguyen AT, Canteloup S, Dayer JM, Jungers P, Drüeke T, Descamps-Latscha B. Advanced oxidation protein and products as novel mediators of inflammation and monocyte activation in chronic renal failure. J Immunol. 1998;161:2524-32. PMID: 9725252. AOPP were measured by spectrophotometrically and were calibrated with Cloramin-T solutions that in the presence of potassium iodide absorb at 340 nm. In standard tubes, 50 µL of 1.16 M potassium iodide (KI) was added to 1000 µL of chloramine-T solution (0-100 mmol/liter) followed by 100 µL of acetic acid. In test tube, 1000 µL of tissue homogenate diluted 1/5 in PBS, 50 µL 1.16 M KI and 100 µL of acetic acid were mixed and the absorbance of the reaction mixture is immediately read at 340 nm on the spectrophotometer against blank containing 1000 µL PBS, 100 µL acetic acid and 50 µL 1.16 M KI.
Plasma was extracted with ethanol-ether (3/1, v/v) and was measured by spectrofluorometer at wavelength 360/430 (excitation/emission wavelength). Fluorescence was determined as relative fluorescent intensity (FI) units per milliliter (FI/ml) of plasma2121 Wu T, Willett WC, Rifai N, Rimm EB. Plasma fluorescent oxidation products as potential markers of oxidative stress for epidemiologic studies. Am J Epidemiol. 2007;166:552-60. PMID: 17615091..
Data analysis was performed using the Statistical Package for Social Sciences (SPSS) version 15.0 for Windows (SPSS Inc, Chicago, IL). All variables were normally distributed about the mean. Data were presented as mean±SD. Differences between the groups were evaluated by One-Way analysis of variance (ANOVA) or Kruskal-Wallis variance analysis, whichever was appropriate. When the p values from the variance analysis were statistically significant, the Tukey honestly significant difference (HSD) or Mann-Whitney U multiple comparison test was used to determine which group was different from the others. A value of p<0.05 was considered to be statistically significant.
Results
The mean histopathological scores of the groups are given in Table 2. When statistical analyses of the histopathological scores were performed, it was found that congestion and necrosis scores in the enoxaparin-treated group were significantly lower than the scores in the control group (p<0.05 for both parameters). The congestion and necrosis scores of the sham group were also significantly lower than those of the control group (p<0.05 for both scores). There was no significant difference between the necrosis scores of the sham and enoxaparin groups (p>0.05), but the congestion scores of the enoxaparin group were higher than those of the sham group (p<0.05). No vacuolization was detected in any group (mean vacuolization score was 0.00 for all groups).
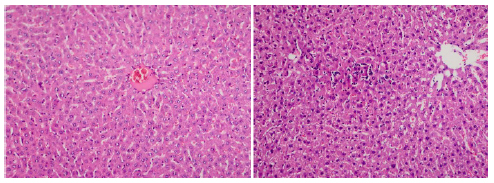
The histopathological findings are presented in Figures 1-3. Liver tissue sections from the sham group exhibited almost normal morphology. The portal areas and lobuli were regular and there was minimal inflammation or sinusoidal congestion. Pneumoperitoneum caused marked inflammatory cell infiltration, hepatocyte necrosis and sinusoidal congestion. Treatment with enoxaparin decreased the histopathological abnormalities when compared with the control group. Sparse mononuclear inflammatory cell infiltration and mild sinusoidal congestion were found in the liver specimens of the enoxaparin group.
Sham group- Portal areas and lobuli are regular in form, there is no inflammation or sinusoidal congestion (x100, H&E).
Control group (only pneumoperitoneum with no treatment-group 2)-Mononuclear cell infiltration, necrosis and sinusoidal congestion in the liver tissue around the central vein were more intensive than in the group treated with enoxaparin (Group 3) (x400, x200, H&E).
Sparse mononuclear cell infiltration and mild sinusoidal congestion in the enoxaparin group (x200, H&E).
The mean levels of the liver function tests (AST, ALT and ALP) are summarized in Table 3. There was no significant difference in ALP levels between the groups (p>0.05). The highest tissue ALP levels were detected in the control group. The levels in the enoxaparin group were lower than those of the control group, but this difference was not statistically significant (p>0.05).
The differences between the sham and other groups were significant for ALT and AST levels (p<0.05). The highest AST and ALT values were found in the control group. The use of enoxaparin decreased these levels, but the difference between the control and enoxaparin groups was not statistically significant (p>0.05).
The mean levels of the oxidative stress parameters for the liver (MDA, AOPP and TFOP) are summarized in Table 4. For MDA, AOPP, and TFOP levels, the differences between the sham and other groups were significant (p<0.05 for all parameters). The highest MDA, AOPP, and TFOP values were found in the control group. The use of enoxaparin decreased the levels of all oxidative stress parameters, but the difference between the control and enoxaparin groups was not statistically significant (p>0.05 for all parameters).
Discussion
The impact of any surgical procedure on the human body can be described as surgical stress. As a result of surgical procedures, there may be a complex physiological stress response including the activation of inflammatory, metabolic, endocrine, and immunological mediators. In recent years, there has been increasing use of laparoscopic surgery for diagnostic and therapeutic purposes. A reduced requirement for analgesia, shorter hospital stay, decreased blood loss, less operative morbidity, and better cosmesis are just some of the patient-related benefits provided by this type of surgery. The smaller abdominal incisions and less tissue manipulation required by minimally invasive surgery result in less trauma and therefore, a lower stress response is expected. In the application of laparoscopic surgery, pneumoperitoneum is necessary for adequate visualization and operative manipulation. Several systems are affected by laparoscopic surgery and insufflation of CO2 into the peritoneal cavity, leading to changes in blood gases, acid-base balance, bacterial translocation, CO2 embolism, ischemia of various splanchnic organs, and cardiovascular, hepatic, renal, and respiratory physiology. Following insufflation and deflation of CO2, ischemia-reperfusion injury develops, thereby inducing oxidative stress2222 Komori Y, Iwashita Y, Ohta M, Kawano Y, Inomata M, Kitano S. Effects of different pressure levels of CO2 pneumoperitoneum on liver regeneration after liver resection in a rat model. Surg Endosc. 2014;28:2466-73. PMID: 24619333.,2323 Yiannakopoulou E, Nikiteas N, Perrea D, Tsigris C. Minimally invasive surgery and oxidative stress response: what have we learned from animal studies? Surg Laparosc Endosc Percutan Tech. 2013;23:25-8.PMID: 23386145..
The level of intra-abdominal pressure may have an effect on the visceral organ injury. In a study by Komori et al.2222 Komori Y, Iwashita Y, Ohta M, Kawano Y, Inomata M, Kitano S. Effects of different pressure levels of CO2 pneumoperitoneum on liver regeneration after liver resection in a rat model. Surg Endosc. 2014;28:2466-73. PMID: 24619333., it was demonstrated that pneumoperitoneum at high pressure had a negative effect on postoperative liver regeneration but when the pressure was maintained at a low level, no adverse effects were observed. In all the parameters, there was no significant difference between the control and low-pressure groups. In the high pressure group, a significant decrease was determined in the rate of liver regeneration and mitotic count. In addition, hepatocellular damage, serum MDA and serum IL-6 were determined to be significantly higher in the high-pressure group compared with the control and/or low-pressure groups. It was concluded that the application of high-pressure pneumoperitoneum before 70% liver resection results in impaired postoperative liver regeneration.
Compared to unfractionated heparin (UFH), low-molecular-weight heparin (LMWH) enoxaparin provides more stable anticoagulation, without any requirement for therapeutic monitoring. In the same way as UFH, Enoxaparin binds to and potentiates antithrombin action, as an endogenous inhibitor of coagulation factors XIa, IXa, Xa and IIa. Through the inhibition of these serine proteases, the coagulation cascade is inhibited, thereby preventing blood clot formation2424 Meneveau N. Safety evaluation of enoxaparin in currently approved indications. Expert Opin Drug Saf. 2009;8:745-54. PMID: 19860536..
Beyond the anticoagulative effects, the anti-inflammatory activities of LMWHs have been shown in several studies55 Rumelt S, Stolovich C, Segal ZI, Rehany U. Intraoperative enoxaparin minimizes inflammatory reaction after pediatric cataract surgery. Am J Ophthalmol. 2006;141:433-7. PMID: 16490487.
6 Nasiripour S, Gholami K, Mousavi S, Mohagheghi A, Radfar M, Abdollahi M, Khazaeipour Z, Mojtahedzadeh M. Comparison of the effects of enoxaparin and heparin on inflammatory biomarkers in patients with ST-segment elevated myocardial infarction: A prospective open label pilot clinical trial. Iran J Pharm Res. 2014;13:583-90. PMID: 25237354.
7 Baram D, Rashkovsky M, Hershkoviz R, Drucker I, Reshef T, Ben-Shitrit S, Mekori YA. Inhibitory effects of low molecular weight heparin on mediator release by mast cells: preferential inhibition of cytokine production and mast cell-dependent cutaneous inflammation. Clin Exp Immunol. 1997;110:485-91. PMID: 9409655.
8 Shastri MD, Stewart N, Horne J, Zaidi ST, Sohal SS, Peterson GM, Korner H, Gueven N, Patel RP. Non-anticoagulant fractions of enoxaparin suppress inflammatory cytokine release from peripheral blood mononuclear cells of allergic asthmatic individuals. PLoS One. 2015;10(6):e0128803. PMID: 26046354.
9 Shastri MD, Stewart N, Horne J, Peterson GM, Gueven N, Sohal SS, Patel RP. In-vitro suppression of IL-6 and IL-8 release from human pulmonary epithelial cells by non-anticoagulant fraction of enoxaparin. PLoS One. 2015;10:e0126763. PMID: 25961885.
10 Grosskopf I, Shaish A, Ray A, Harats D, Kamari Y. Low molecular weight heparin-induced increase in chylomicron-remnants clearance, is associated with decreased plasma TNF-a level and increased hepatic lipase activity. Thromb Res. 2014;133:688-92. PMID: 24525311.-1111 Lean QY, Eri RD, Randall-Demllo S, Sohal SS, Stewart N, Peterson GM, Gueven N, Patel RP. Orally administered enoxaparin ameliorates acute colitis by reducing macrophage-associated inflammatory responses. PLoS One. 2015;10:e0134259. PMID: 26218284.. These studies have demonstrated that enoxaparin could be effective for managing many inflammatory disorders including inflammatory reaction after pediatric cataract surgery55 Rumelt S, Stolovich C, Segal ZI, Rehany U. Intraoperative enoxaparin minimizes inflammatory reaction after pediatric cataract surgery. Am J Ophthalmol. 2006;141:433-7. PMID: 16490487., ST-segment elevated myocardial infarction66 Nasiripour S, Gholami K, Mousavi S, Mohagheghi A, Radfar M, Abdollahi M, Khazaeipour Z, Mojtahedzadeh M. Comparison of the effects of enoxaparin and heparin on inflammatory biomarkers in patients with ST-segment elevated myocardial infarction: A prospective open label pilot clinical trial. Iran J Pharm Res. 2014;13:583-90. PMID: 25237354., mast cell-mediated allergic inflammation77 Baram D, Rashkovsky M, Hershkoviz R, Drucker I, Reshef T, Ben-Shitrit S, Mekori YA. Inhibitory effects of low molecular weight heparin on mediator release by mast cells: preferential inhibition of cytokine production and mast cell-dependent cutaneous inflammation. Clin Exp Immunol. 1997;110:485-91. PMID: 9409655., allergic asthma88 Shastri MD, Stewart N, Horne J, Zaidi ST, Sohal SS, Peterson GM, Korner H, Gueven N, Patel RP. Non-anticoagulant fractions of enoxaparin suppress inflammatory cytokine release from peripheral blood mononuclear cells of allergic asthmatic individuals. PLoS One. 2015;10(6):e0128803. PMID: 26046354.,99 Shastri MD, Stewart N, Horne J, Peterson GM, Gueven N, Sohal SS, Patel RP. In-vitro suppression of IL-6 and IL-8 release from human pulmonary epithelial cells by non-anticoagulant fraction of enoxaparin. PLoS One. 2015;10:e0126763. PMID: 25961885., and acute colitis1111 Lean QY, Eri RD, Randall-Demllo S, Sohal SS, Stewart N, Peterson GM, Gueven N, Patel RP. Orally administered enoxaparin ameliorates acute colitis by reducing macrophage-associated inflammatory responses. PLoS One. 2015;10:e0134259. PMID: 26218284.. Enoxaparin has also been shown to be effective against many hepatic disorders and has a well-established hepatoprotective effect1212 Assy N, Hussein O, Khalil A, Luder A, Szvalb S, Paizi M, Spira G. The beneficial effect of aspirin and enoxaparin on fibrosis progression and regenerative activity in a rat model of cirrhosis. Dig Dis Sci. 2007;52:1187-93. PMID: 17372820.
13 Kukner A, Tore F, Firat T, Terzi EH, Oner H, Balaban YH, Ozogul C. The preventive effect of low molecular weight heparin on CCL4-induced necrosis and apoptosis in rat liver. Ann Hepatol. 2010;9:445-54. PMID: 21057164.
14 Lee JH, Lee H, Joung YK, Jung KH, Choi JH, Lee DH, Park KD, Hong SS. The use of low molecular weight heparin-pluronic nanogels to impede liver fibrosis by inhibition the TGF-ß/Smad signaling pathway. Biomaterials. 2011;32:1438-45. PMID: 21074845.
15 Seidensticker M, Seidensticker R, Damm R, Mohnike K, Pech M, Sangro B, Hass P, Wust P, Kropf S, Gademann G, Ricke J. Prospective randomized trial of enoxaparin, pentoxifylline and ursodeoxycholic acid for prevention of radiation-induced liver toxicity. PLoS One. 2014;9:e112731. PMID: 25393877.
16 Abdel-Salam OM, Baiuomy AR, Ameen A, Hassan NS. A study of unfractionated and low molecular weight heparins in a model of cholestatic liver injury in the rat. Pharmacol Res. 2005;51:59-67. PMID: 15519536.-1717 Taha MO, Gonçalves PF, Vidigal RO, Sacchi GP, Pinheiro FG, Brandão LS, Feguri S, Moraes-Filho AM, Ghadie MM, Magalhães KG, Simões MJ, Tersariol IL, Monteiro HP, Oliveira-Júnior IS, Jurkiewicz A, Caricati-Neto A. Protective effects of heparin on hepatic ischemia and reperfusion lesions in rabbits. Transplant Proc. 2009;41:812-5. PMID: 19376359..
In a study by Kukner et al.1313 Kukner A, Tore F, Firat T, Terzi EH, Oner H, Balaban YH, Ozogul C. The preventive effect of low molecular weight heparin on CCL4-induced necrosis and apoptosis in rat liver. Ann Hepatol. 2010;9:445-54. PMID: 21057164. the beneficial effects of enoxaparin against necrosis and apoptosis were investigated in the early stage of CCL4-induced liver damage. Enoxaparin was determined to reduce necrosis, apoptosis, and the number of mast cells but no effect was observed on lipid droplets in hepatocytes.
Enoxaparin has been shown to have beneficial effects on fibrosis progression and regenerative activity in the cirrhosis model1212 Assy N, Hussein O, Khalil A, Luder A, Szvalb S, Paizi M, Spira G. The beneficial effect of aspirin and enoxaparin on fibrosis progression and regenerative activity in a rat model of cirrhosis. Dig Dis Sci. 2007;52:1187-93. PMID: 17372820.. In a study by Lee et al.1414 Lee JH, Lee H, Joung YK, Jung KH, Choi JH, Lee DH, Park KD, Hong SS. The use of low molecular weight heparin-pluronic nanogels to impede liver fibrosis by inhibition the TGF-ß/Smad signaling pathway. Biomaterials. 2011;32:1438-45. PMID: 21074845. LMWH was seen to exert anti-fibrotic effects via the inhibition of the TGF-b/Smad pathway in addition to the elimination of the extracellular matrix. Enoxaparin has also shown positive effects on radiation-induced liver toxicity1515 Seidensticker M, Seidensticker R, Damm R, Mohnike K, Pech M, Sangro B, Hass P, Wust P, Kropf S, Gademann G, Ricke J. Prospective randomized trial of enoxaparin, pentoxifylline and ursodeoxycholic acid for prevention of radiation-induced liver toxicity. PLoS One. 2014;9:e112731. PMID: 25393877., and experimental cholestatic liver injury1616 Abdel-Salam OM, Baiuomy AR, Ameen A, Hassan NS. A study of unfractionated and low molecular weight heparins in a model of cholestatic liver injury in the rat. Pharmacol Res. 2005;51:59-67. PMID: 15519536..
Taha et al.1717 Taha MO, Gonçalves PF, Vidigal RO, Sacchi GP, Pinheiro FG, Brandão LS, Feguri S, Moraes-Filho AM, Ghadie MM, Magalhães KG, Simões MJ, Tersariol IL, Monteiro HP, Oliveira-Júnior IS, Jurkiewicz A, Caricati-Neto A. Protective effects of heparin on hepatic ischemia and reperfusion lesions in rabbits. Transplant Proc. 2009;41:812-5. PMID: 19376359. researched the role of heparin in hepatic ischemia/reperfusion (I/R) injury in rabbits treated with 100 U/kg heparin or saline solution. Lower increases were seen in the levels of AST, ALT, and LDH in the rabbits treated with heparin. In the histological examination of the liver tissue of rabbits undergoing I/R, evident lesions were observed in the central lobule with significant inflammatory infiltration. Treatment with heparin led to a significant reduction in the lesions caused by I/R.
In a previous study by the current authors2525 Cavusoglu T, Ozcan N, Ogus E, Ozcan A, Kaymak C, Sahin S, Yilmaz FM, Tezel S. Enoxaparin effect on malondialdehyde levels in laparoscopic cholecystectomy. J Invest Surg. 2011;24:211-6. PMID: 21867390., an evaluation was made of the effects of low dose enoxaparin on plasma MDA levels and liver function tests during laparoscopic cholecystectomy. At 20 mins after deflation, MDA levels were seen to have significantly increased in the control group compared with the enoxaparin group. A significant increase in plasma MDA concentrations compared to the initial levels was determined in the control group. It was concluded that a mini dose of enoxaparin prevented the MDA increase after laparoscopic cholecystectomy without causing any bleeding disorder while having no effect on serum ALT and AST elevations. In the current study, significant differences were seen between the sham and other groups in respect of the ALT and AST levels. The highest AST and ALT values were determined in the control group. While enoxaparin was determined to have decreased these levels, the difference between the control and enoxaparin groups was not statistically significant. No significant difference was found in the ALP levels between the groups. The highest tissue ALP levels were in the control group and although the levels in the enoxaparin group were lower than those of the control group, the difference was not statistically significant. The results obtained in the current study were similar to the results of our previous study2525 Cavusoglu T, Ozcan N, Ogus E, Ozcan A, Kaymak C, Sahin S, Yilmaz FM, Tezel S. Enoxaparin effect on malondialdehyde levels in laparoscopic cholecystectomy. J Invest Surg. 2011;24:211-6. PMID: 21867390. except for the MDA levels. Although the use of enoxaparin decreased the MDA values, the differences between the control and enoxaparin groups were not statistically significant in the present study. This might be due to various factors such as the enoxaparin doses used in the studies, or the different responses of human and rat organisms. Thus, there is need for further detailed studies of the antioxidant effects of enoxaparin.
Since our previous study was conducted on humans, biopsies for determination of pathological changes and the effects of enoxaparin on these changes were not taken and only biochemical changes were evaluated. The current study was planned primarily for the detection of the histopathological injury to the liver tissue and the possible effects of enoxaparin on this injury. Liver tissue sections from the sham group were found to have almost normal morphology. The portal areas and lobuli were regular and there was minimal inflammation or sinusoidal congestion in this group. Pneumoperitoneum resulted in marked inflammatory cell infiltration, hepatocyte necrosis and sinusoidal congestion. Treatment with enoxaparin decreased the histopathological abnormalities caused by pneumoperitoneum. Sparse mononuclear inflammatory cell infiltration and mild sinusoidal congestion were found in the liver specimens of the enoxaparin group and the histopathological scores were significantly lower in the enoxaparin group when compared with the control group (pneumoperitoneum with no treatment group). No vacuolization was detected in any of the groups. This might be because of the short duration of the experimental process. These results indicated that enoxaparin reduced the harmful effects of pneumoperitoneum on the liver and this effect might be primarily due to its anti-inflammatory activity. The results of the current study were also in accordance with previous studies that have shown the beneficial hepatoprotective effects of enoxaparin on different hepatic disorders1212 Assy N, Hussein O, Khalil A, Luder A, Szvalb S, Paizi M, Spira G. The beneficial effect of aspirin and enoxaparin on fibrosis progression and regenerative activity in a rat model of cirrhosis. Dig Dis Sci. 2007;52:1187-93. PMID: 17372820.
13 Kukner A, Tore F, Firat T, Terzi EH, Oner H, Balaban YH, Ozogul C. The preventive effect of low molecular weight heparin on CCL4-induced necrosis and apoptosis in rat liver. Ann Hepatol. 2010;9:445-54. PMID: 21057164.
14 Lee JH, Lee H, Joung YK, Jung KH, Choi JH, Lee DH, Park KD, Hong SS. The use of low molecular weight heparin-pluronic nanogels to impede liver fibrosis by inhibition the TGF-ß/Smad signaling pathway. Biomaterials. 2011;32:1438-45. PMID: 21074845.
15 Seidensticker M, Seidensticker R, Damm R, Mohnike K, Pech M, Sangro B, Hass P, Wust P, Kropf S, Gademann G, Ricke J. Prospective randomized trial of enoxaparin, pentoxifylline and ursodeoxycholic acid for prevention of radiation-induced liver toxicity. PLoS One. 2014;9:e112731. PMID: 25393877.
16 Abdel-Salam OM, Baiuomy AR, Ameen A, Hassan NS. A study of unfractionated and low molecular weight heparins in a model of cholestatic liver injury in the rat. Pharmacol Res. 2005;51:59-67. PMID: 15519536.-1717 Taha MO, Gonçalves PF, Vidigal RO, Sacchi GP, Pinheiro FG, Brandão LS, Feguri S, Moraes-Filho AM, Ghadie MM, Magalhães KG, Simões MJ, Tersariol IL, Monteiro HP, Oliveira-Júnior IS, Jurkiewicz A, Caricati-Neto A. Protective effects of heparin on hepatic ischemia and reperfusion lesions in rabbits. Transplant Proc. 2009;41:812-5. PMID: 19376359..
There have been various studies that have investigated the biochemical alterations in laparoscopic operations and experimental pneumoperitoneum models. Changes in serum levels of ALT, AST, gamma-glutamyl-transferase (GGT), ALP, and lactate dehydrogenase (LDH) were evaluated by Guven et al.2626 Guven HE, Oral S. Liver enzyme alterations after laparoscopic cholecystectomy. J Gastrointestin Liver Dis. 2007;16:391-4. PMID: 18193120. in patients that had undergone laparoscopic cholecystectomy compared to open cholecystectomy. In a comparison of pre and postoperative biochemical tests from blood samples taken preoperatively and at 24 hours postoperatively, liver enzyme levels were found to be significantly increased in the laparoscopic cholecystectomy group. Significant differences were also determined between the laparoscopic cholecystectomy and open cholecystectomy groups. The conclusion of that study was that these elevated enzyme levels could be attributed in the main to the negative effects of the pneumoperitoneum on hepatic blood flow.
In another study, using indocyanine green elimination tests (ICG-PDR), Eryilmaz et al.2727 Eryilmaz HB, Memis D, Sezer A, Inal MT. The effects of different insufflation pressures on liver functions assessed with LiMON on patients undergoing laparoscopic cholecystectomy. Scientific World J. 2012;2012:172575. PMID: 22619616. examined the effects on liver functions of insufflation at different pressures. Measurements of serum AST, ALT, and total bilirubin levels were taken preoperatively, and at postoperative 1 hour and 24 hours. The results showed that 14mmHg pressure pneumoperitoneum decreased blood flow to the liver and increased postoperative 1-hour serum AST and ALT levels. It was therefore suggested that 10mmHg pressure pneumoperitoneum is superior to 14mmHg in laparoscopic cholecystectomy.
In the present study, the differences between the sham and other groups were significant for ALT and AST levels (p<0.05). The highest AST and ALT values were found in the control group. The use of enoxaparin decreased these levels, but the difference between the control and enoxaparin groups was not statistically significant (p>0.05). Although the minimal changes of liver enzymes during laparoscopic procedures do not seem to be clinically important, care should be taken before deciding to perform laparoscopic operations in patients with liver diseases.
Free radicals are highly reactive molecules with unpaired electrons, and undertake significant biological functions, including redox signaling and antibacterial defence. In laparoscopic surgery, oxidative stress may develop with the effect of several factors including the level of intra-abdominal pressure, and the type, temperature and humidity of the gas11 Arsalani-Zadeh R, Ullah S, Khan S, MacFie J. Oxidative stress in laparoscopic versus open abdominal surgery: a systematic review. J Surg Res. 2011 Jul;169:e59-68. PMID: 21492871.. Free radicals can react with lipids in the cell and mitochondrial membranes and consequently initiate lipid peroxidation. MDA, which is the end product of lipid peroxidation, is commonly used as a marker of oxidative activity2828 Cighetti G1, Duca L, Bortone L, Sala S, Nava I, Fiorelli G, Cappellini MD. Oxidative status and malondialdehyde in beta- thalassemia patients. Eur J Clin Invest. 2002;32(Suppl 1):55-60. PMID: 11886433. Neutrophil-derived oxidants can change protein structure, resulting in denaturation, protein fragmentation, and at very high levels, protein cross-linking. AOPP measure highly oxidized protein levels and may be used as an in vivo marker of oxidant-mediated protein damage2020 Witko-Sarsat V, Friedlander M, Nguyen Khoa T, Capeillère-Blandin C, Nguyen AT, Canteloup S, Dayer JM, Jungers P, Drüeke T, Descamps-Latscha B. Advanced oxidation protein and products as novel mediators of inflammation and monocyte activation in chronic renal failure. J Immunol. 1998;161:2524-32. PMID: 9725252.. A sensitive measurement method of oxidation is the measurement of fluorescent oxidation products (FOP), as it has been reported to be 10-100 times more sensitive than the colorimetric thiobarbituric acid-reactive substances assay. Different conditions associated with systemic oxidative stress are known to significantly increase the levels of fluorescent oxidation products2121 Wu T, Willett WC, Rifai N, Rimm EB. Plasma fluorescent oxidation products as potential markers of oxidative stress for epidemiologic studies. Am J Epidemiol. 2007;166:552-60. PMID: 17615091..
In the current study, the levels of MDA, AOPP, and TFOP were measured for the evaluation of oxidative status and the effects of enoxaparin on oxidative stress during high-pressure pneumoperitoneum. The differences between the sham and other groups were significant for all parameters (p<0.05). The highest levels of oxidative stress parameters were found in the control group, in other words, high-pressure pneumoperitoneum increased the oxidative stress. Although the use of enoxaparin decreased these values, the differences between the control and enoxaparin groups were not statistically significant (p>0.05 for all parameters). In the light of these results, it was concluded that enoxaparin had a partial antioxidant effect.
The beneficial effects of enoxaparin was thought to be primarily due to its anti-inflammatory effect, together with a partial antioxidant effect. Enoxaparin has been used widely preoperatively for the prevention of thromboembolism in surgical clinics all over the world.
Conclusions
Enoxaparin ameliorated the harmful effects of high pressure CO2 pneumoperitoneum on the liver. The use of enoxaparin in laparoscopic operations for its hepatoprotective effect might be recommended after prospective randomized clinical trials.
References
-
1Arsalani-Zadeh R, Ullah S, Khan S, MacFie J. Oxidative stress in laparoscopic versus open abdominal surgery: a systematic review. J Surg Res. 2011 Jul;169:e59-68. PMID: 21492871.
-
2Hasukic S. Postoperative changes in liver function tests: randomized comparison of low- and high-pressure laparoscopic cholecystectomy. Surg Endosc. 2005;19:1451-5. PMID: 16206003.
-
3Nickkholgh A, Barro-Bejarano M, Liang R, Zorn M, Mehrabi A, Gebhard MM, Büchler MW, Gutt CN, Schemmer P. Signs of reperfusion injury following CO2 pneumoperitoneum: an in vivo microscopy study. Surg Endosc. 2008;22:122-8. PMID: 17483991.
-
4Iqbal Z, Cohen M. Enoxaparin: a pharmacologic and clinical review. Expert Opin Pharmacother. 2011;12:1157-70. PMID: 21470072.
-
5Rumelt S, Stolovich C, Segal ZI, Rehany U. Intraoperative enoxaparin minimizes inflammatory reaction after pediatric cataract surgery. Am J Ophthalmol. 2006;141:433-7. PMID: 16490487.
-
6Nasiripour S, Gholami K, Mousavi S, Mohagheghi A, Radfar M, Abdollahi M, Khazaeipour Z, Mojtahedzadeh M. Comparison of the effects of enoxaparin and heparin on inflammatory biomarkers in patients with ST-segment elevated myocardial infarction: A prospective open label pilot clinical trial. Iran J Pharm Res. 2014;13:583-90. PMID: 25237354.
-
7Baram D, Rashkovsky M, Hershkoviz R, Drucker I, Reshef T, Ben-Shitrit S, Mekori YA. Inhibitory effects of low molecular weight heparin on mediator release by mast cells: preferential inhibition of cytokine production and mast cell-dependent cutaneous inflammation. Clin Exp Immunol. 1997;110:485-91. PMID: 9409655.
-
8Shastri MD, Stewart N, Horne J, Zaidi ST, Sohal SS, Peterson GM, Korner H, Gueven N, Patel RP. Non-anticoagulant fractions of enoxaparin suppress inflammatory cytokine release from peripheral blood mononuclear cells of allergic asthmatic individuals. PLoS One. 2015;10(6):e0128803. PMID: 26046354.
-
9Shastri MD, Stewart N, Horne J, Peterson GM, Gueven N, Sohal SS, Patel RP. In-vitro suppression of IL-6 and IL-8 release from human pulmonary epithelial cells by non-anticoagulant fraction of enoxaparin. PLoS One. 2015;10:e0126763. PMID: 25961885.
-
10Grosskopf I, Shaish A, Ray A, Harats D, Kamari Y. Low molecular weight heparin-induced increase in chylomicron-remnants clearance, is associated with decreased plasma TNF-a level and increased hepatic lipase activity. Thromb Res. 2014;133:688-92. PMID: 24525311.
-
11Lean QY, Eri RD, Randall-Demllo S, Sohal SS, Stewart N, Peterson GM, Gueven N, Patel RP. Orally administered enoxaparin ameliorates acute colitis by reducing macrophage-associated inflammatory responses. PLoS One. 2015;10:e0134259. PMID: 26218284.
-
12Assy N, Hussein O, Khalil A, Luder A, Szvalb S, Paizi M, Spira G. The beneficial effect of aspirin and enoxaparin on fibrosis progression and regenerative activity in a rat model of cirrhosis. Dig Dis Sci. 2007;52:1187-93. PMID: 17372820.
-
13Kukner A, Tore F, Firat T, Terzi EH, Oner H, Balaban YH, Ozogul C. The preventive effect of low molecular weight heparin on CCL4-induced necrosis and apoptosis in rat liver. Ann Hepatol. 2010;9:445-54. PMID: 21057164.
-
14Lee JH, Lee H, Joung YK, Jung KH, Choi JH, Lee DH, Park KD, Hong SS. The use of low molecular weight heparin-pluronic nanogels to impede liver fibrosis by inhibition the TGF-ß/Smad signaling pathway. Biomaterials. 2011;32:1438-45. PMID: 21074845.
-
15Seidensticker M, Seidensticker R, Damm R, Mohnike K, Pech M, Sangro B, Hass P, Wust P, Kropf S, Gademann G, Ricke J. Prospective randomized trial of enoxaparin, pentoxifylline and ursodeoxycholic acid for prevention of radiation-induced liver toxicity. PLoS One. 2014;9:e112731. PMID: 25393877.
-
16Abdel-Salam OM, Baiuomy AR, Ameen A, Hassan NS. A study of unfractionated and low molecular weight heparins in a model of cholestatic liver injury in the rat. Pharmacol Res. 2005;51:59-67. PMID: 15519536.
-
17Taha MO, Gonçalves PF, Vidigal RO, Sacchi GP, Pinheiro FG, Brandão LS, Feguri S, Moraes-Filho AM, Ghadie MM, Magalhães KG, Simões MJ, Tersariol IL, Monteiro HP, Oliveira-Júnior IS, Jurkiewicz A, Caricati-Neto A. Protective effects of heparin on hepatic ischemia and reperfusion lesions in rabbits. Transplant Proc. 2009;41:812-5. PMID: 19376359.
-
18Suzuki S, Toledo-Peneyra LH, Rodriquez FJ, Cejalvo D. Neutrophil infiltration as an important factor in liver ischemia and reperfusion injury. Modulating effects of FK506 and cylosporine. Transplantation. 1993;55:1265-72. PMID: 7685932.
-
19Wasowicz W, Nève J, Peretz A. Optimized steps in fluorometric determination of thiobarbituric acid-reactive substances in serum: importance of extraction pH and influence of sample preservation and storage. Clin Chem. 1993;39:2522-6. PMID: 8252725.
-
20Witko-Sarsat V, Friedlander M, Nguyen Khoa T, Capeillère-Blandin C, Nguyen AT, Canteloup S, Dayer JM, Jungers P, Drüeke T, Descamps-Latscha B. Advanced oxidation protein and products as novel mediators of inflammation and monocyte activation in chronic renal failure. J Immunol. 1998;161:2524-32. PMID: 9725252.
-
21Wu T, Willett WC, Rifai N, Rimm EB. Plasma fluorescent oxidation products as potential markers of oxidative stress for epidemiologic studies. Am J Epidemiol. 2007;166:552-60. PMID: 17615091.
-
22Komori Y, Iwashita Y, Ohta M, Kawano Y, Inomata M, Kitano S. Effects of different pressure levels of CO2 pneumoperitoneum on liver regeneration after liver resection in a rat model. Surg Endosc. 2014;28:2466-73. PMID: 24619333.
-
23Yiannakopoulou E, Nikiteas N, Perrea D, Tsigris C. Minimally invasive surgery and oxidative stress response: what have we learned from animal studies? Surg Laparosc Endosc Percutan Tech. 2013;23:25-8.PMID: 23386145.
-
24Meneveau N. Safety evaluation of enoxaparin in currently approved indications. Expert Opin Drug Saf. 2009;8:745-54. PMID: 19860536.
-
25Cavusoglu T, Ozcan N, Ogus E, Ozcan A, Kaymak C, Sahin S, Yilmaz FM, Tezel S. Enoxaparin effect on malondialdehyde levels in laparoscopic cholecystectomy. J Invest Surg. 2011;24:211-6. PMID: 21867390.
-
26Guven HE, Oral S. Liver enzyme alterations after laparoscopic cholecystectomy. J Gastrointestin Liver Dis. 2007;16:391-4. PMID: 18193120.
-
27Eryilmaz HB, Memis D, Sezer A, Inal MT. The effects of different insufflation pressures on liver functions assessed with LiMON on patients undergoing laparoscopic cholecystectomy. Scientific World J. 2012;2012:172575. PMID: 22619616.
-
28Cighetti G1, Duca L, Bortone L, Sala S, Nava I, Fiorelli G, Cappellini MD. Oxidative status and malondialdehyde in beta- thalassemia patients. Eur J Clin Invest. 2002;32(Suppl 1):55-60. PMID: 11886433
-
Financial source:
none -
1
Research performed at Department of General Surgery, Ankara Education and Research Hospital, Turkey.
Publication Dates
-
Publication in this collection
Nov 2016
History
-
Received
25 July 2016 -
Reviewed
26 Sept 2016 -
Accepted
24 Oct 2016