ABSTRACT
PURPOSE:
To assess the capsules formed by silicone implants coated with polyurethane foam and with a textured surface.
METHODS:
Sixty-four Wistar albinus rats were divided into two groups of 32 each using polyurethane foam and textured surface. The capsules around the implants were analyzed for 30, 50, 70 and 90 days. Were analyzed the following parameters: foreign body reaction, granulation tissue, presence of myofibroblasts, neoangiogenesis, presence of synovial metaplasia, capsular thickness, total area and collagen percentage of type I and III, in capsules formed around silicone implants in both groups.
RESULTS:
The foreign body reaction was only present in the four polyurethane subgroups. The formation of granulation tissue and the presence of myofibroblasts were higher in the four polyurethane subgroups. Regarding to neoangiogenesis and synovial metaplasia, there was no statistical difference between the groups. Polyurethane group presented (all subgroups) a greater capsule thickness, a smaller total area and collagen percentage of type I and a higher percentage area of type III, with statistical difference.
CONCLUSION:
The use of polyurethane-coated implants should be stimulated by the long-term results in a more stable capsule and a lower incidence of capsular contracture, despite developing a more intense and delayed inflammatory reaction in relation to implants with textured surface.
Key words:
Implant Capsular Contracture; Breast Implants; Mammaplasty; Rats
Introduction
Currently, the most performed plastic surgery worldwide is breast augmentation11. Silva MM, Modolin M, Faintuch J, Yamaguchi CM, Zandona CB, Cintra W Jr, Fujiwara H, Curi R, Gemperli R, Ferreira MC. Systemic inflammatory reaction after silicone breast implant. Aesthetic Plast Surg. 2011 Oct;35(5):789-94. doi: 10.1007/s00266-011-9688-x.
https://doi.org/10.1007/s00266-011-9688-...
. Scales et al.22. Wagenführ-Júnior J, Ribas Filho JM, Nascimento MM, Ribas FM, Wanka MV, Godoi Ade L. Histopathological reaction over prosthesis surface covered with silicone and polyurethane foam implanted in rats. Acta Cir Bras. 2012 Dec;27(12):866-73. doi: 10.1590/S0102-86502012001200007.
https://doi.org/10.1590/S0102-8650201200...
defined the properties of an ideal implant: no physical change in tissue; chemically inert; non carcinogenic; non allergic or foreign body reactive; mechanical forces resilience; and made into the desired shape. Unfortunately, there is no such implant, but a lot of progress has been made, since its introduction in 1962 with Cronin and Gerow, to the current generation's implants22. Wagenführ-Júnior J, Ribas Filho JM, Nascimento MM, Ribas FM, Wanka MV, Godoi Ade L. Histopathological reaction over prosthesis surface covered with silicone and polyurethane foam implanted in rats. Acta Cir Bras. 2012 Dec;27(12):866-73. doi: 10.1590/S0102-86502012001200007.
https://doi.org/10.1590/S0102-8650201200...
.
The most commonly used implants are those with textured surface and polyurethane-coated. All material introduced into the human body undergoes a foreign body reaction, which occurs in the formation of a fibrous capsule around itself33. Mendes PR, Bins-Ely J, Lima EA, Vasconcellos ZA, d'Acampora AJ, Neves RD. Histological study on acute inflammatory reaction to polyurethane-coated silicone implants in rats. Acta Cir Bras . 2008 Jan-Feb;23(1):93-101. doi: 10.1590/S0102-86502008000100015.
https://doi.org/10.1590/S0102-8650200800...
.
Unfortunately, there are some patients who undergo to breast augmentation with industrial liquid silicone, which is totally discouraged due to the high index of complications resulting in complex wounds44. Mello DF, Gonçalves KC, Fraga MF, Perin LF, Helene A Jr. Local complications after industrial liquid silicone injection: case series. Rev Col Bras Cir. 2013 Jan-Feb;40(1):37-42. doi: 10.1590/S0100-69912013000100007.
https://doi.org/10.1590/S0100-6991201300...
, often requiring therapy with subatmospheric pressure beyond the usual treatments44. Mello DF, Gonçalves KC, Fraga MF, Perin LF, Helene A Jr. Local complications after industrial liquid silicone injection: case series. Rev Col Bras Cir. 2013 Jan-Feb;40(1):37-42. doi: 10.1590/S0100-69912013000100007.
https://doi.org/10.1590/S0100-6991201300...
,55. Milcheski DA, Ferreira MC, Nakamoto HA, Pereira DD, Batista BN, Tuma P Jr. Subatmospheric pressure therapy in the treatment of traumatic soft tissue injuries. Rev Col Bras Cir . 2013 Sep-Oct;40(5):392-6. doi: 10.1590/S0100-69912013000500008.
https://doi.org/10.1590/S0100-6991201300...
.
In breast augmentation techniques using implants, the more frequent late complication is the capsular contracture. This situation is characterized by breast pain, discomfort, stress, deformations, stiffness and the exchange of implants is often necessary to resolve the problem66. Prantl L, Schreml S, Fichtner-Feigl S, Pöppl N, Eisenmann-Klein M, Schwarze H, Füchtmeier B. Clinical and morphological conditions in capsular contracture formed around silicone breast implants. Plast Reconstr Surg. 2007 Jul;120(1):275-84. doi: 10.1097/01.prs.0000264398.85652.9a.
https://doi.org/10.1097/01.prs.000026439...
,77. Araco A, Caruso R, Araco F, Overton J, Gravante G. Capsular contractures: a systematic review. Plast Reconstr Surg . 2009 Dec;124(6):1808-19. doi: 10.1097/PRS.0b013e3181bf7f26.
https://doi.org/10.1097/PRS.0b013e3181bf...
.
The incidence of contracture varies depending on the coating of the silicone implant, being the polyurethane-coated at about 1%; the textured surface around 10%; and with smooth surface at rates higher than 50%. So, the use of smooth implants is being phased out22. Wagenführ-Júnior J, Ribas Filho JM, Nascimento MM, Ribas FM, Wanka MV, Godoi Ade L. Histopathological reaction over prosthesis surface covered with silicone and polyurethane foam implanted in rats. Acta Cir Bras. 2012 Dec;27(12):866-73. doi: 10.1590/S0102-86502012001200007.
https://doi.org/10.1590/S0102-8650201200...
,88. Barnsley GP, Sigurdson LJ, Barnsley SE. Textured surface breast implants in the prevention of capsular contracture among breast augmentation patients: a meta-analysis of randomized controlled trials. Plast Reconstr Surg . 2006 Jun;117(7):2182-90. doi: 10.1097/01.prs.0000218184.47372.d5.
https://doi.org/10.1097/01.prs.000021818...
9. Wong CH, Samuel M, Tan BK, Song C. Capsular contracture in subglandular breast augmentation with textured versus smooth breast implants: a systematic review. doi: 10.1097/01.prs.0000237013.50283.d2.
https://doi.org/10.1097/01.prs.000023701...
-1010. Balderrama CM, Ribas-Filho JM, Malafaia O, Czeczko NG, Dietz UA, Sakamoto DG, Bittencourt LP. Healing reaction to mammary prostheses covered by textured silicone and silicone foam in rats. Acta Cir Bras . 2009 Sep-Oct;24(5):367-76. doi: 10.1590/S0102-86502009000500006.
https://doi.org/10.1590/S0102-8650200900...
.
The present study aims to assess the capsules formed by silicone implants coated with polyurethane foam and with a textured surface.
Methods
This study was performed in the vivarium , Universidade Estadual de Ponta Grossa, after was approved by the Ethics Committee on the Use of Animals of this university.
Sixty-four female Wistar rats were used (Rattus norvegicus albinus , Roentia mammalia), weighing 190-250 g, 30 to 90 days of life, distributed in acrylic boxes with 500 cm3 containing four rats in each with free access to water and specific diet for the species, room temperature and alternating 12 h cycles of light and dark. They were distributed into two groups of 32 and subdivided into four subgroups according to the time of death (30, 50, 70 and 90 days).
Experimental procedure
The rats were anesthetized by intramuscular injection consisting of a combination of 1% ketamine (40 mg/kg) and 2% xylazine (8 mg/kg)1010. Balderrama CM, Ribas-Filho JM, Malafaia O, Czeczko NG, Dietz UA, Sakamoto DG, Bittencourt LP. Healing reaction to mammary prostheses covered by textured silicone and silicone foam in rats. Acta Cir Bras . 2009 Sep-Oct;24(5):367-76. doi: 10.1590/S0102-86502009000500006.
https://doi.org/10.1590/S0102-8650200900...
. The effectiveness of the anesthesia was assessed by the lack of movement, corneal reflex and motor reaction after holding a clamp on the adipose cushion of one of the hind legs, and a good ventilatory pattern33. Mendes PR, Bins-Ely J, Lima EA, Vasconcellos ZA, d'Acampora AJ, Neves RD. Histological study on acute inflammatory reaction to polyurethane-coated silicone implants in rats. Acta Cir Bras . 2008 Jan-Feb;23(1):93-101. doi: 10.1590/S0102-86502008000100015.
https://doi.org/10.1590/S0102-8650200800...
.
The rats were placed in the prone position with shaving performed in the dorsal region followed by antisepsis.
For delimiting the incision, the crossing point of a horizontal line following the posterior inferior costal margin to the sagittal midline was defined. With a number 3 scalpel, coupled with a number 15 blade, a horizontal incision was made with a horizontal extension of 1 cm at the intersection of these virtual lines.
The pocket, which housed the implants, was made on the retromuscular plane with scissors in a cranial direction, starting 5 mm from the incision and square shape. After being made, the implant was inserted (Figure 1) and placed 5 mm from the incision (Figure 2), and skin closure was performed with three simple knots using mononylon 5-0.
The wound was kept exposed and postoperative analgesia was applied with a single, intramuscular injection of dipyrone (20 mg/kg) on the outside of the hind limb1111. Mendes FH, Viterbo F, DeLucca L. The influence of external ultrasound on the histologic architecture of the organic capsule around smooth silicone implants: experimental study in rats. Aesthetic Plast Surg . 2008 May;32(3):442-50. doi: 10.1007/s00266-007-9063-0.
https://doi.org/10.1007/s00266-007-9063-...
. There have been no postoperative dressings or stitches withdrawal.
The rats were killed according to their subgroups with an application of ketamine and xylazine, corresponding to four times the therapeutic dose and subsequent cervical dislocation. Because of implant extrusion in seven animals (four from the polyurethane group and three from the textured group), they were excluded from the study.
Histological evaluation
Hematoxylin-eosin staining
With this staining the following histological variables were evaluated: foreign body reaction, granulation tissue, myofibroblasts, neoangiogenesis and synovial metaplasia1213. Suvik A, Effendy AWM. The use of modified Masson's trichrome staining in collagen evaluation in wound healing study. Malaysian J Vet Res. 2012;3(1)39-47.. The analyses of these variables were measured in four levels: absent, and discrete, moderate and intense presence.
Masson's trichrome acid staining
Through this staining, the measurement of the thickness of the capsule showing the muscle, collagen fibers, fibrin and erythrocytes were evaluated1213. Suvik A, Effendy AWM. The use of modified Masson's trichrome staining in collagen evaluation in wound healing study. Malaysian J Vet Res. 2012;3(1)39-47..
Picrosirius staining
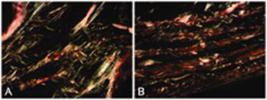
Through this staining, collagens type I and III were quantified. The study of the microscope slides was a closed-type, double-blind and read by polarized light (Figure 3) for further analysis by the software Image Pro-Plus (Figure 4)1314. Montes GS, Junqueira LC. The use of the Picrosirius-polarization method for the study of the biopathology of collagen. Mem Inst Oswaldo Cruz. 1991;86 Suppl 3:1-11. doi: 10.1590/S0074-02761991000700002.
https://doi.org/10.1590/S0074-0276199100...
,1415. Montes GS. Structural biology of the fibres of the collagenous and elastic systems. Cell Biol Int. 1996 Jan;20(1):15-27. doi: 10.1006/cbir.1996.0004.
https://doi.org/10.1006/cbir.1996.0004....
.
Photo with six different photomicrographs (increase of x400) showing collagen fibers stained by Picrosirius and visualized in the microscope with polarized light.
The same six previous photomicrographs displayed on the computer after application of the mask by software Image Pro-Plus.
Statistical analysis
For the analysis of quantitative variables the nonparametric Mann-Whitney and Kruskal-Wallis were used. Qualitative variables were analyzed using Fisher's exact test. The significance level was 0.05, adjusted by Bonferroni for multiple comparisons of the groups (p<0.008). Data was analyzed with the computer program IBM SPSS v.20.0.
RESULTS
Foreign body reaction
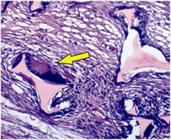
Most animals in polyurethane group had moderate or severe presence of a foreign body reaction (Figure 5), whereas in the textured group was not observed this variable.
Photomicrography of polyurethane foam showing the foreign body reaction, the arrow showing the foreign body reaction.
The foreign body reaction only was present in the polyurethane group within the four evaluated subgroups (30 days p<0.0001/ 50 days p<0.002/ 70 days p<0.001/ 90 days p<0.0002, Figure 6).
Granulation tissue
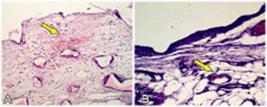
While every rat of the polyurethane group had moderate or intense presence of the formation of granulation tissue, in the textured group absent or presence of this discrete variable was observed (Figure 7).
Photomicrography of polyurethane foam (A) and textured surface (B) showing the granulation tissue with arrow showing fibroplasia and reactional neoangiogenesis.
Granulation tissue formation was statistically significantly greater in the polyurethane group within the four evaluated subgroups (30 days p<0.0001/ 50 days p<0.002/ 70 days p<0.0002/ 90 days p<0.0002, Figure 6).
Myofibroblasts
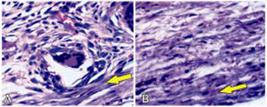
In the polyurethane group most rats had moderate or severe presence of myofibroblasts, whereas in the textured group all rats had absent or discrete presence of the variable (Figure 8).
Photomicrography of polyurethane foam (A) and textured surface (B) showing, in arrows, the myofibroblasts.
The presence of myofibroblasts was statistically significantly greater in the group polyurethane within the four evaluated in subgroups (30 days p<0.0001/ 50 days p<0.015/ 70 days p<0.007/ 90 days p<0.0002, Figure 6).
Neoangiogenesis
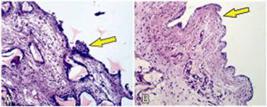
Only 16.7% of the animals in the 50 days subgroup of the polyurethane group had moderate or severe presence of neoangiogenesis, in the rest of the rats this variable was absent or had discrete presence (Figure 9).
Photomicrography of polyurethane foam (A) and textured surface (B) showing the neoangiogenesis and, in arrows, blood vessels.
No statistically significant differences were found between the two groups in the several subgroups (Figure 6).
Synovial metaplasia
Only 12.5% of the rats in the 70 days subgroup of the textured group had moderate or severe presence of synovial metaplasia, the other rats had absent or discrete presence of this variable (Figure 10).
Photomicrography of polyurethane foam (A) and textured surface (B) showing, in arrows, the synovial metaplasia.
There were no statistically significant differences being the two groups in all subgroups (Figure 6).
Measurement of the thickness of the capsule
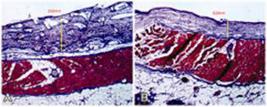
The capsule in the polyurethane group was thicker compared to textured group (Figure 11), with statistical significance in all evaluated subgroups (30 days p=0.001/ 50 days p=0.006/ 70 days p=0.001/ 90 days p=0.001, Table 1).
Photomicrography of polyurethane foam (A) and textured surface (B) showing the capsule with 0.53 mm in A and 0.24 mm in B.
The comparison of the measurement of the capsule in each subgroup (30, 50, 70 and 90 days) of the polyurethane group was statistically significant at p=0.008, while in the textured group there was no statistical significance (Table 2).
Due to the rejection of the hypothesis of equality to all subgroups for the polyurethane group, the subgroups were compared in pairs.
In the polyurethane group by comparing the different subgroups to each other. Statistical significance between 30 and 50 days in the polyurethane group was obtained (p=0.009), 30 and 70 days (p=0.009), and 30 and 90 days (p<0.001).
Measurement of the total and percentage areas of collagen type I and III
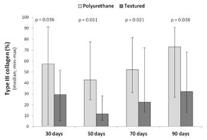
The total area of type I collagen was larger in the textured group (30 days p=0.046; 50 days, p=0.045; 70 days, p=0.021; 90 days, p= 0.037). The area percentage of type I collagen was bigger in the textured group (30 days, p=0.036; 50 days, p=0.011; 70 days, p=0.021; 90 days, p=0.028). The total area of type III collagen was higher in the polyurethane group (50 days, p=0.028). The percentage area of type III collagen was larger in the polyurethane group (30 days, p=0.036; 50 days, p=0.011; 70 days, p=0.021; 90 days, p=0.028, Figures 12 and 13).
Photomicrography of polyurethane foam (A) and textured surface (B) showing collagen fibers: in red type I and in green type III.
Discussion
Histological aspects
Since its discovery to the present day silicone implants have been defined as a biocompatible material. However, for Ratner1516. Ratner BD. The biocompatibility manifesto: biocompatibility for the twenty-first century. J Cardiovasc Transl Res. 2011 Oct;4(5):523-7. doi: 10.1007/s12265-011-9287-x.
https://doi.org/10.1007/s12265-011-9287-...
, the capsule formed around the implants is an attempt by the body to isolate it, which is a matter of incompatibility. According to this same author, the best concept that could be applied to implants in general, including silicone, is biotolerability, which is the ability of a material to reside in the body for long periods of time with only low-grade inflammation.
Evaluation of foreign body reaction
According to Ratner1516. Ratner BD. The biocompatibility manifesto: biocompatibility for the twenty-first century. J Cardiovasc Transl Res. 2011 Oct;4(5):523-7. doi: 10.1007/s12265-011-9287-x.
https://doi.org/10.1007/s12265-011-9287-...
, a foreign body reaction is more evident when the pores of the implant are either larger or smaller than 30-40 micrometers (μm). Disagreeing with the author, in the textured group in which the pore size exceeded this range at various times (200-500 μm), the foreign body reaction was absent in all subgroups analyzed in this research.
In accordance to Wagenführ22. Wagenführ-Júnior J, Ribas Filho JM, Nascimento MM, Ribas FM, Wanka MV, Godoi Ade L. Histopathological reaction over prosthesis surface covered with silicone and polyurethane foam implanted in rats. Acta Cir Bras. 2012 Dec;27(12):866-73. doi: 10.1590/S0102-86502012001200007.
https://doi.org/10.1590/S0102-8650201200...
, most of the rats in the polyurethane group had moderate or intense presence of a foreign body reaction in all subgroups evaluated. Although the same author obtained statistical significance only at 60 days subgroup, in this study, the results were statistically significant in the subgroups 30, 50, 70 and 90 days. This fact can be explained because Wagenführ22. Wagenführ-Júnior J, Ribas Filho JM, Nascimento MM, Ribas FM, Wanka MV, Godoi Ade L. Histopathological reaction over prosthesis surface covered with silicone and polyurethane foam implanted in rats. Acta Cir Bras. 2012 Dec;27(12):866-73. doi: 10.1590/S0102-86502012001200007.
https://doi.org/10.1590/S0102-8650201200...
compared implants coated with polyurethane foam and with silicone foam. In this experiment the comparison was made with implants with textured surfaces.
Haddad Filho et al.1617. Haddad Filho D, Zveibel DK, Alonso N, Gemperli R. Comparison between textured silicone implants and those bonded with expanded polytetrafluoroethylene in rats. Acta Cir Bras . 2007 May-Jun;22(3):187-94. doi: 10.1590/S0102-86502007000300006.
https://doi.org/10.1590/S0102-8650200700...
after comparing silicone implants with a textured surface and coated PTFE-E, it was concluded that both groups induced a weak foreign body reaction, which was higher in textured group. Disagreeing with this author, in this study the foreign body reaction was absent in the textured group at all subgroups.
Corroborating with Balderrama et al.1010. Balderrama CM, Ribas-Filho JM, Malafaia O, Czeczko NG, Dietz UA, Sakamoto DG, Bittencourt LP. Healing reaction to mammary prostheses covered by textured silicone and silicone foam in rats. Acta Cir Bras . 2009 Sep-Oct;24(5):367-76. doi: 10.1590/S0102-86502009000500006.
https://doi.org/10.1590/S0102-8650200900...
findings, it was confirmed with significant difference that the foreign body reaction was lower in textured group at the time of 60 days when compared to the foam silicone group. In this research lower foreign body reaction in textured group in all subgroups was also obtained, although the comparison was made with implants coated with polyurethane foam.
In according to Bassetto et al.1718. Bassetto F, Scarpa C, Caccialanza E, Montesco MC, Magnani P. Histological features of periprosthetic mammary capsules: silicone vs. polyurethane. Aesthetic Plast Surg . 2010 Aug;34(4):481-5. doi: 10.1007/s00266-010-9483-0.
https://doi.org/10.1007/s00266-010-9483-...
, the foreign body reaction was bigger in the polyurethane group. In the textured group, this reaction was present in all members in a milder way, differing from the present study in which the foreign body reaction was absent in the textured group, although this study has been carried out in rats and the other in humans.
Evaluation of granulation tissue
Corroborating Vieira1819. Vieira VJ, d'Acampora AJ, Marcos AB, Di Giunta G, de Vasconcellos ZA, Bins-Ely J, d'Eça Neves R, Figueiredo CP. Vascular endothelial growth factor overexpression positively modulates the characteristics of periprosthetic tissue of polyurethane-coated silicone breast implant in rats. Plast Reconstr Surg . 2010 Dec;126(6):1899-910. doi: 10.1097/PRS.0b013e3181f446d5.
https://doi.org/10.1097/PRS.0b013e3181f4...
, who found that the polyurethane group had increased formation of granulation tissue in relation to the textured group in subgroups of 30 and 90 days with statistical significance, in this study in all subgroups, this variable was more intense in the polyurethane group, whereas in the textured group it was absent or showed discrete presence, also with significant difference.
Evaluation of myofibroblasts
Disagreeing with Hwang et al .1920. Hwang K, Sim HB, Huan F, Kim DJ. Myofibroblasts and capsular tissue tension in breast capsular contracture. Aesthetic Plast Surg . 2010 Dec;34(6):716-21. doi: 10.1007/s00266-010-9532-8
https://doi.org/10.1007/s00266-010-9532-...
when saying that the preferential localization of myofibroblasts is in the outer layer of the capsule, in this study, like in Minami et al.2021. Minami E, Koh IH, Ferreira JC, Waitzberg AF, Chifferi V, Rosewick TF, Pereira MD, Saldiva PH, de Figueiredo LF. The composition and behavior of capsules around smooth and textured breast implants in pigs. Plast Reconstr Surg . 2006 Sep 15;118(4):874-84. doi: 10.1097/01.prs.0000240878.24213.b7.
https://doi.org/10.1097/01.prs.000024087...
and Prantl et al.66. Prantl L, Schreml S, Fichtner-Feigl S, Pöppl N, Eisenmann-Klein M, Schwarze H, Füchtmeier B. Clinical and morphological conditions in capsular contracture formed around silicone breast implants. Plast Reconstr Surg. 2007 Jul;120(1):275-84. doi: 10.1097/01.prs.0000264398.85652.9a.
https://doi.org/10.1097/01.prs.000026439...
, it was found in the inner layer.
According to Bui et al.2122. Bui JM, Perry T, Ren CD, Nofrey B, Teitelbaum S, Van Epps DE. Histological characterization of human breast implant capsules. Aesthetic Plast Surg . 2015 Jun;39(3):306-15. doi: 10.1007/s00266-014-0439-7
https://doi.org/10.1007/s00266-014-0439-...
, in the current study all rats of the textured group had absent or mild presence of myofibroblasts, while in the polyurethane group most animals had moderate or severe presence of myofibroblasts, thereby strengthening the interdependence of the type of implant coating and the amount of myofibroblasts. An important observation was that these authors have conducted the study in humans.
Evaluation of neoangiogenesis
Disagreeing with Haddad Filho et al.1617. Haddad Filho D, Zveibel DK, Alonso N, Gemperli R. Comparison between textured silicone implants and those bonded with expanded polytetrafluoroethylene in rats. Acta Cir Bras . 2007 May-Jun;22(3):187-94. doi: 10.1590/S0102-86502007000300006.
https://doi.org/10.1590/S0102-8650200700...
that found greater number of capillaries in the textured and PTFE-E groups in relation to the control group with statistical significance in various subgroups, in this study all rats that took part in the textured group had absence or discrete presence of this variable with no significant difference.
Disagreeing with Wagenführ22. Wagenführ-Júnior J, Ribas Filho JM, Nascimento MM, Ribas FM, Wanka MV, Godoi Ade L. Histopathological reaction over prosthesis surface covered with silicone and polyurethane foam implanted in rats. Acta Cir Bras. 2012 Dec;27(12):866-73. doi: 10.1590/S0102-86502012001200007.
https://doi.org/10.1590/S0102-8650201200...
and Vieira1819. Vieira VJ, d'Acampora AJ, Marcos AB, Di Giunta G, de Vasconcellos ZA, Bins-Ely J, d'Eça Neves R, Figueiredo CP. Vascular endothelial growth factor overexpression positively modulates the characteristics of periprosthetic tissue of polyurethane-coated silicone breast implant in rats. Plast Reconstr Surg . 2010 Dec;126(6):1899-910. doi: 10.1097/PRS.0b013e3181f446d5.
https://doi.org/10.1097/PRS.0b013e3181f4...
that found in the polyurethane group larger amount of vessels statistically significant, in this study the majority of the rats showed absence of neoangiogenesis or discrete presence, but without significant difference.
Evaluation of synovial metaplasia
In accordance with Prantl et al.66. Prantl L, Schreml S, Fichtner-Feigl S, Pöppl N, Eisenmann-Klein M, Schwarze H, Füchtmeier B. Clinical and morphological conditions in capsular contracture formed around silicone breast implants. Plast Reconstr Surg. 2007 Jul;120(1):275-84. doi: 10.1097/01.prs.0000264398.85652.9a.
https://doi.org/10.1097/01.prs.000026439...
,2223. Prantl L, Pöppl N, Horvat N, Heine N, Eisenmann-Klein M. Serologic and histologic findings in patients with capsular contracture after breast augmentation with smooth silicone gel implants: is serum hyaluronan a potential predictor? Aesthetic Plast Surg . 2005 Nov-Dec;29(6):510-8. doi: 10.1007/s00266-005-5049-y.
https://doi.org/10.1007/s00266-005-5049-...
, that found an inner layer with synovial metaplasia in 62.5% of cases, in this study mild synovial metaplasia was found in 100% of the rats, however, there was no significant difference between groups.
Partially agreeing with Bassetto et al.1718. Bassetto F, Scarpa C, Caccialanza E, Montesco MC, Magnani P. Histological features of periprosthetic mammary capsules: silicone vs. polyurethane. Aesthetic Plast Surg . 2010 Aug;34(4):481-5. doi: 10.1007/s00266-010-9483-0.
https://doi.org/10.1007/s00266-010-9483-...
, who obtained the presence of synovial metaplasia in the polyurethane and textured groups, which was bigger in the polyurethane group, in this study was found this variable, however, there was no difference between the groups. The same authors concluded that synovial metaplasia is the most important protective factor against capsular contracture. This becomes clear when Bui et al.2122. Bui JM, Perry T, Ren CD, Nofrey B, Teitelbaum S, Van Epps DE. Histological characterization of human breast implant capsules. Aesthetic Plast Surg . 2015 Jun;39(3):306-15. doi: 10.1007/s00266-014-0439-7
https://doi.org/10.1007/s00266-014-0439-...
stated synovial metaplasia to be the most common in capsules Baker I/II, compared to Baker III/IV capsules.
Evaluation of the thickness of the capsule
Corroborating Minami et al.2021. Minami E, Koh IH, Ferreira JC, Waitzberg AF, Chifferi V, Rosewick TF, Pereira MD, Saldiva PH, de Figueiredo LF. The composition and behavior of capsules around smooth and textured breast implants in pigs. Plast Reconstr Surg . 2006 Sep 15;118(4):874-84. doi: 10.1097/01.prs.0000240878.24213.b7.
https://doi.org/10.1097/01.prs.000024087...
and Vieira1819. Vieira VJ, d'Acampora AJ, Marcos AB, Di Giunta G, de Vasconcellos ZA, Bins-Ely J, d'Eça Neves R, Figueiredo CP. Vascular endothelial growth factor overexpression positively modulates the characteristics of periprosthetic tissue of polyurethane-coated silicone breast implant in rats. Plast Reconstr Surg . 2010 Dec;126(6):1899-910. doi: 10.1097/PRS.0b013e3181f446d5.
https://doi.org/10.1097/PRS.0b013e3181f4...
that observed variation in the thickness of the capsule due to increase in the non-collagenous layers (intermediate and internal), in this study this increase also occurred in these layers.
In accordance to Balderrama et al.1010. Balderrama CM, Ribas-Filho JM, Malafaia O, Czeczko NG, Dietz UA, Sakamoto DG, Bittencourt LP. Healing reaction to mammary prostheses covered by textured silicone and silicone foam in rats. Acta Cir Bras . 2009 Sep-Oct;24(5):367-76. doi: 10.1590/S0102-86502009000500006.
https://doi.org/10.1590/S0102-8650200900...
that found thinner capsule in the textured group in the subgroups of 7 and 60 days compared to silicone foam group with statistical significance, all subgroups evaluated in this study (30, 50, 70 and 90 days) the capsule was thinner in the textured group with a significant difference, although the comparison has been made with polyurethane-coated implants.
Agreeing with Vieira1819. Vieira VJ, d'Acampora AJ, Marcos AB, Di Giunta G, de Vasconcellos ZA, Bins-Ely J, d'Eça Neves R, Figueiredo CP. Vascular endothelial growth factor overexpression positively modulates the characteristics of periprosthetic tissue of polyurethane-coated silicone breast implant in rats. Plast Reconstr Surg . 2010 Dec;126(6):1899-910. doi: 10.1097/PRS.0b013e3181f446d5.
https://doi.org/10.1097/PRS.0b013e3181f4...
, who obtained ,statistically significant thinner capsule in the textured group in the subgroups of 30 and 90 days compared to polyurethane group, in all evaluated subgroups in this study the capsule was thinner in the textured group with statistical significance. Disagreeing with this author who found thinner layer of collagen in the polyurethane group compared to the textured group, in the current study were evaluated all subgroups and was found an opposite result.
Evaluation of the total and percentage areas of collagen type I and III
Disagreeing with Ratner1516. Ratner BD. The biocompatibility manifesto: biocompatibility for the twenty-first century. J Cardiovasc Transl Res. 2011 Oct;4(5):523-7. doi: 10.1007/s12265-011-9287-x.
https://doi.org/10.1007/s12265-011-9287-...
that studied the coating of implants and concluded that a greater angiogenesis occurs when the pores on the surface of the implants are between 30-40 μm with less collagen deposition, in this study the pores measured 50-250 μm in the textured group and 250-350 μm in polyurethane group, and was obtained in both groups and in all subgroups, discrete angiogenesis with a significant deposition of collagen which was larger in the polyurethane group.
Regarding the characteristics of the collagen fibers according to Minami et al.2021. Minami E, Koh IH, Ferreira JC, Waitzberg AF, Chifferi V, Rosewick TF, Pereira MD, Saldiva PH, de Figueiredo LF. The composition and behavior of capsules around smooth and textured breast implants in pigs. Plast Reconstr Surg . 2006 Sep 15;118(4):874-84. doi: 10.1097/01.prs.0000240878.24213.b7.
https://doi.org/10.1097/01.prs.000024087...
and Balderrama et al.1010. Balderrama CM, Ribas-Filho JM, Malafaia O, Czeczko NG, Dietz UA, Sakamoto DG, Bittencourt LP. Healing reaction to mammary prostheses covered by textured silicone and silicone foam in rats. Acta Cir Bras . 2009 Sep-Oct;24(5):367-76. doi: 10.1590/S0102-86502009000500006.
https://doi.org/10.1590/S0102-8650200900...
that found a decline in the amount of type III collagen fibers with increased type I collagen fibers with significant differences, the same result was found in this study from 50 days on.
Disagreeing with Wagenführ22. Wagenführ-Júnior J, Ribas Filho JM, Nascimento MM, Ribas FM, Wanka MV, Godoi Ade L. Histopathological reaction over prosthesis surface covered with silicone and polyurethane foam implanted in rats. Acta Cir Bras. 2012 Dec;27(12):866-73. doi: 10.1590/S0102-86502012001200007.
https://doi.org/10.1590/S0102-8650201200...
that obtained a greater amount of collagen in the subgroups of 28 and 60 days of the polyurethane group than in the silicone foam group with statistical significance, in this study all the textured subgroups evaluated had a higher amount of collagen, statistically significant, than the polyurethane group.
When analyzed the same fraction area, Minami et al .2021. Minami E, Koh IH, Ferreira JC, Waitzberg AF, Chifferi V, Rosewick TF, Pereira MD, Saldiva PH, de Figueiredo LF. The composition and behavior of capsules around smooth and textured breast implants in pigs. Plast Reconstr Surg . 2006 Sep 15;118(4):874-84. doi: 10.1097/01.prs.0000240878.24213.b7.
https://doi.org/10.1097/01.prs.000024087...
found a greater increase of type I collagen fibers in the smooth implants for textured implants with a significant difference. In this study the textured group had a greater increase of type I collagen fibers in relation to the polyurethane group with statistical significance.
Conclusion
The use of polyurethane-coated implants should be stimulated by the long-term results in a more stable capsule and a lower incidence of capsular contracture, despite developing a more intense and delayed inflammatory reaction in relation to implants with textured surface.
References
-
1Silva MM, Modolin M, Faintuch J, Yamaguchi CM, Zandona CB, Cintra W Jr, Fujiwara H, Curi R, Gemperli R, Ferreira MC. Systemic inflammatory reaction after silicone breast implant. Aesthetic Plast Surg. 2011 Oct;35(5):789-94. doi: 10.1007/s00266-011-9688-x.
» https://doi.org/10.1007/s00266-011-9688-x -
2Wagenführ-Júnior J, Ribas Filho JM, Nascimento MM, Ribas FM, Wanka MV, Godoi Ade L. Histopathological reaction over prosthesis surface covered with silicone and polyurethane foam implanted in rats. Acta Cir Bras. 2012 Dec;27(12):866-73. doi: 10.1590/S0102-86502012001200007.
» https://doi.org/10.1590/S0102-86502012001200007 -
3Mendes PR, Bins-Ely J, Lima EA, Vasconcellos ZA, d'Acampora AJ, Neves RD. Histological study on acute inflammatory reaction to polyurethane-coated silicone implants in rats. Acta Cir Bras . 2008 Jan-Feb;23(1):93-101. doi: 10.1590/S0102-86502008000100015.
» https://doi.org/10.1590/S0102-86502008000100015 -
4Mello DF, Gonçalves KC, Fraga MF, Perin LF, Helene A Jr. Local complications after industrial liquid silicone injection: case series. Rev Col Bras Cir. 2013 Jan-Feb;40(1):37-42. doi: 10.1590/S0100-69912013000100007.
» https://doi.org/10.1590/S0100-69912013000100007 -
5Milcheski DA, Ferreira MC, Nakamoto HA, Pereira DD, Batista BN, Tuma P Jr. Subatmospheric pressure therapy in the treatment of traumatic soft tissue injuries. Rev Col Bras Cir . 2013 Sep-Oct;40(5):392-6. doi: 10.1590/S0100-69912013000500008.
» https://doi.org/10.1590/S0100-69912013000500008. -
6Prantl L, Schreml S, Fichtner-Feigl S, Pöppl N, Eisenmann-Klein M, Schwarze H, Füchtmeier B. Clinical and morphological conditions in capsular contracture formed around silicone breast implants. Plast Reconstr Surg. 2007 Jul;120(1):275-84. doi: 10.1097/01.prs.0000264398.85652.9a.
» https://doi.org/10.1097/01.prs.0000264398.85652.9a. -
7Araco A, Caruso R, Araco F, Overton J, Gravante G. Capsular contractures: a systematic review. Plast Reconstr Surg . 2009 Dec;124(6):1808-19. doi: 10.1097/PRS.0b013e3181bf7f26.
» https://doi.org/10.1097/PRS.0b013e3181bf7f26. -
8Barnsley GP, Sigurdson LJ, Barnsley SE. Textured surface breast implants in the prevention of capsular contracture among breast augmentation patients: a meta-analysis of randomized controlled trials. Plast Reconstr Surg . 2006 Jun;117(7):2182-90. doi: 10.1097/01.prs.0000218184.47372.d5.
» https://doi.org/10.1097/01.prs.0000218184.47372.d5. -
9Wong CH, Samuel M, Tan BK, Song C. Capsular contracture in subglandular breast augmentation with textured versus smooth breast implants: a systematic review. doi: 10.1097/01.prs.0000237013.50283.d2.
» https://doi.org/10.1097/01.prs.0000237013.50283.d2 -
10Balderrama CM, Ribas-Filho JM, Malafaia O, Czeczko NG, Dietz UA, Sakamoto DG, Bittencourt LP. Healing reaction to mammary prostheses covered by textured silicone and silicone foam in rats. Acta Cir Bras . 2009 Sep-Oct;24(5):367-76. doi: 10.1590/S0102-86502009000500006.
» https://doi.org/10.1590/S0102-86502009000500006. -
11Mendes FH, Viterbo F, DeLucca L. The influence of external ultrasound on the histologic architecture of the organic capsule around smooth silicone implants: experimental study in rats. Aesthetic Plast Surg . 2008 May;32(3):442-50. doi: 10.1007/s00266-007-9063-0.
» https://doi.org/10.1007/s00266-007-9063-0. -
13Suvik A, Effendy AWM. The use of modified Masson's trichrome staining in collagen evaluation in wound healing study. Malaysian J Vet Res. 2012;3(1)39-47.
-
14Montes GS, Junqueira LC. The use of the Picrosirius-polarization method for the study of the biopathology of collagen. Mem Inst Oswaldo Cruz. 1991;86 Suppl 3:1-11. doi: 10.1590/S0074-02761991000700002.
» https://doi.org/10.1590/S0074-02761991000700002. -
15Montes GS. Structural biology of the fibres of the collagenous and elastic systems. Cell Biol Int. 1996 Jan;20(1):15-27. doi: 10.1006/cbir.1996.0004.
» https://doi.org/10.1006/cbir.1996.0004. -
16Ratner BD. The biocompatibility manifesto: biocompatibility for the twenty-first century. J Cardiovasc Transl Res. 2011 Oct;4(5):523-7. doi: 10.1007/s12265-011-9287-x.
» https://doi.org/10.1007/s12265-011-9287-x. -
17Haddad Filho D, Zveibel DK, Alonso N, Gemperli R. Comparison between textured silicone implants and those bonded with expanded polytetrafluoroethylene in rats. Acta Cir Bras . 2007 May-Jun;22(3):187-94. doi: 10.1590/S0102-86502007000300006.
» https://doi.org/10.1590/S0102-86502007000300006. -
18Bassetto F, Scarpa C, Caccialanza E, Montesco MC, Magnani P. Histological features of periprosthetic mammary capsules: silicone vs. polyurethane. Aesthetic Plast Surg . 2010 Aug;34(4):481-5. doi: 10.1007/s00266-010-9483-0.
» https://doi.org/10.1007/s00266-010-9483-0. -
19Vieira VJ, d'Acampora AJ, Marcos AB, Di Giunta G, de Vasconcellos ZA, Bins-Ely J, d'Eça Neves R, Figueiredo CP. Vascular endothelial growth factor overexpression positively modulates the characteristics of periprosthetic tissue of polyurethane-coated silicone breast implant in rats. Plast Reconstr Surg . 2010 Dec;126(6):1899-910. doi: 10.1097/PRS.0b013e3181f446d5.
» https://doi.org/10.1097/PRS.0b013e3181f446d5. -
20Hwang K, Sim HB, Huan F, Kim DJ. Myofibroblasts and capsular tissue tension in breast capsular contracture. Aesthetic Plast Surg . 2010 Dec;34(6):716-21. doi: 10.1007/s00266-010-9532-8
» https://doi.org/10.1007/s00266-010-9532-8 -
21Minami E, Koh IH, Ferreira JC, Waitzberg AF, Chifferi V, Rosewick TF, Pereira MD, Saldiva PH, de Figueiredo LF. The composition and behavior of capsules around smooth and textured breast implants in pigs. Plast Reconstr Surg . 2006 Sep 15;118(4):874-84. doi: 10.1097/01.prs.0000240878.24213.b7.
» https://doi.org/10.1097/01.prs.0000240878.24213.b7. -
22Bui JM, Perry T, Ren CD, Nofrey B, Teitelbaum S, Van Epps DE. Histological characterization of human breast implant capsules. Aesthetic Plast Surg . 2015 Jun;39(3):306-15. doi: 10.1007/s00266-014-0439-7
» https://doi.org/10.1007/s00266-014-0439-7 -
23Prantl L, Pöppl N, Horvat N, Heine N, Eisenmann-Klein M. Serologic and histologic findings in patients with capsular contracture after breast augmentation with smooth silicone gel implants: is serum hyaluronan a potential predictor? Aesthetic Plast Surg . 2005 Nov-Dec;29(6):510-8. doi: 10.1007/s00266-005-5049-y.
» https://doi.org/10.1007/s00266-005-5049-y
-
Financial source: none
-
1
Research performed at Postgraduate Program in Principles of Surgery, Faculdade Evangélica do Paraná (FEPAR), Evangelic University Hospital (HUEC) and Institute for Medical Research (IPEM), Curitiba-PR, Brazil
Publication Dates
-
Publication in this collection
Dec 2016
History
-
Received
12 Aug 2016 -
Reviewed
14 Oct 2016 -
Accepted
14 Nov 2016