Abstract
Purpose:
To evaluate the changes of caveolin-1 in lung fibroblasts in newborn Wistar rats when exposed to hyperoxic conditions, as well as lung fibroblasts cell cycle.
Methods:
One hundred newborn Wistar rats were randomly divided (50 rats/group) into experimental and control groups, exposed to hyperoxic conditions or normal air, respectively. The fraction of inspired oxygen (FiO2) in the experimental group was 90%, whereas this value was 21% in the control group. Lung fibroblasts were collected on days 3, 7, and 14 of the experiment. Caveolin-1 expression dynamics in lung fibroblasts was assayed in each group by immunofluorescence and Western blot analyses. Flow cytometry (FCM) was used to assess the proportions of lung fibroblasts at different stages of the cell cycle.
Results:
On day 3, no significant difference in caveolin-1 expression was observed between the hyperoxic and control groups; however, on days 7 and 14, caveolin-1 expression was significantly lower in the hyperoxic group than in the control (P<0.05). No apparent differences were observed in caveolin-1 expression in the control group at the different time points. Using FCM analysis, we showed that the proportion of lung fibroblasts in G0/G1 phase in the hyperoxic group decreased compared to that of the control group on day 7, while the proportion of S-phase cells increased (P<0.05). These differences were more significant when the groups were compared on day 14 (P<0.01).
Conclusion:
After seven days the exposure to hyperoxic conditions, lung fibroblasts proliferated and caveolin-1 expression decreased.
Key words:
Lung; Fibroblasts; Pulmonary Fibrosis; Caveolin 1; Cell Cycle; Rats
Introduction
Although the pathogenesis of chronic lung disease (CLD) in preterm children is not well understood, its delayed pathological outcome, pulmonary fibrosis, is unquestionable11 Jensen EA, Schmidt B. Epidemiology of bronchopulmonary dysplasia. Birth Defects Res A Clin Mol Teratol. 2014 Mar;100(3):145-57. doi: 10.1002/bdra.23235.
https://doi.org/10.1002/bdra.23235...
. If we find what is the main cause of the pulmonary fibrosis, the treatment would be more effect and lead to better prognosis.
The prevention and treatment of pulmonary fibrosis has posed a significant challenge in the treatment of CLD with non-interruption study22 Ambalavanan N, Mourani P. Pulmonary hypertension in bronchopulmonary dysplasia. Birth Defects Res A Clin Mol Teratol. 2014 Mar;100(3):240-6. doi: 10.1002/bdra.23241.
https://doi.org/10.1002/bdra.23241...
. Lung fibroblasts (LFs) are recognized as effector cells of pulmonary fibrosis33 Chelstowska S, Wajs J, Rzepecki P, Plusa T. The role of fibrocytes in pulmonary fibrosis. Pol Merkur Lekarski. 2014 Jan;36(211):39-41. PMID: 24645577.. Pulmonary fibrosis is associated with excessively proliferating LFs that deposit in the interstitial lung or replace the normal lung epithelium44 Ansarie M, Naseem A, Kasmani A, Ahmed R, Azeemuddin M. Profile of interstitial lung diseases in pakistan, karachi pulmonology clinics registry data: jan 2012 - aug 2013. Chest. 2014;145(3 Suppl):241A.. Directly culturing LFs in vitro at high concentrations of oxygen can inhibit cell proliferation55 Wang XQ, Mao LJ, Fang QH, Kobayashi T, Kim HJ, Sugiura H, Kawasaki S, Togo S, Kamio K, Liu X, Rennard SI. Sphingosylphosphorylcholine induces a-smooth muscle actin expression in human lung fibroblasts and fibroblasts mediated gel contraction via S1P2 receptor and Rho/Rho-Kinase pathway. Prostaglandins Other Lipid Mediat. 2014 Jan;108:23-30. doi: 10.1016/j.prostaglandins.2014.02.002.
https://doi.org/10.1016/j.prostaglandins...
, while the growth of LFs in human or animal lung tissues that have inhaled high concentrations of oxygen show the opposite effect. The primary culture of LFs from CLD newborn rats is the only feasible method to study the mechanism controlling this excessive proliferation at the cellular level. In the present study, we established a reliable primary cell culture method for LFs derived from CLD newborn rats to create a platform to study the mechanism of CLD stromal cell fibrosis in vitro.
Caveolin-1, which is highly expressed in type I alveolar epithelial cells, endothelial cells, and fibroblasts, is associated with cell proliferation66 Cai L, Yi F, Dai Z, Huang X, Zhao YD, Mirza MK, Xu J, Vogel SM, Zhao YY. Loss of caveolin-1 and adiponectin induces severe inflammatory lung injury following LPS challenge through excessive oxidative/nitrative stress. Am J Physiol Lung Cell Mol Physiol. 2014 Mar 15;306(6):L566-73. doi: 10.1152/ajplung.00182.2013.
https://doi.org/10.1152/ajplung.00182.20...
. Thus, we questioned whether abnormal expression levels of caveolin-1 correlated with the pulmonary LF hyperproliferative process that occurs during CLD stromal cell fibrosis77 Reese C, Perry B, Heywood J, Bonner M, Visconti RP, Lee R, Hatfield CM, Silver RM, Hoffman S, Tourkina E. Caveolin-1 deficiency predisposes African-Americans to SSc-ILD. Arthritis Rheumatol. 2014 Jul;66(7):1909-19. doi: 10.1002/art.38572.
https://doi.org/10.1002/art.38572...
. In this study, the expression levels of caveolin-1 in LFs at different stages of hyperoxia-induced CLD development in rats were examined to investigate its role in lung fibroblast proliferation and in the incidence and development of pulmonary fibrosis. This study was to find the changes of caveolin-1 in lung fibroblasts in newborn Wistar rats when exposed to hyperoxic conditions, as well as lung fibroblasts cell cycle to find if more fibroblasts conversed into myofibroblasts.
Methods
The animal use protocol has been reviewed and approved by the Institutional Animal Care and Use Committee (IACUC) of China Medical University (Permit Number: 20060828001). This study was carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health.
One hundred newborn Wistar rats were provided by the Department of Animal Laboratory. All surgery on rats was performed under pentobarbital sodium (3% pentobarbital sodium 1 ml/100 g mouse, intraperitoneal injection) anesthesia, and all efforts were made to minimize suffering.
Healthy neonatal Wistar rats were randomly divided into two groups (50 rats/group), the experimental and control groups that were exposed to hyperoxic condition and normal air, respectively. The newborn Wistar rats in the experimental group, together with female rats, were placed in an oxygen tank after birth. The tank had a continuous input of oxygen and the fraction of inspired oxygen (FiO2) was maintained at 90% (U.S. OM-25ME-type oxygen analyzer, Maxtec, Salt Lake City, Utah, United States of America). CO2 was absorbed using soda lime to maintain the concentration below 0.5% (Dapex gas analyzer). The temperature was maintained between 22 and 25°C and humidity was maintained at 50-70%. The tank was opened for 0.5 h/d to provide water and food and to change the padding. The female rats were interchanged with those of the control group every day to avoid oxygen toxicity. The control group was exposed to air (FiO2 of 21%), but the other methods and conditions were the same as those of the experimental group.
The pulmonary fibrosis scoring system was taken into concern to determine the chronic lung disease, which was implemented by optical microscope (100×) and the results ranged from 0 point to 8 points. Grading criteria: 0 were normal lung tissue; 1 were fibrosis of alveolar and terminal bronchiole, 2-3 was moderate fibrosis without any change of alveolar structure; 4-5 was moderate fibrosis with alveolar structure change, also with fiber bundle or fibrous nodule formation; 6-7 was severe fibrosis with severe alveolar structural changes; 8 was diffuse fibrosis,
Isolation, culture, and identification of LFs in vitro
We followed the method reported by Kelleher et al.88 Kelleher MD, Naureckas ET, Solway J, Hershenson MB. In vivo hyperoxic exposure increases cultured lung fibroblast proliferation and c-Ha-ras expression. Am J Respir Cell Mol Biol. 1995 Jan;12(1):19-26. doi: 10.1165/ajrcmb.12.1.7811467.
https://doi.org/10.1165/ajrcmb.12.1.7811...
but with the following modifications. Wistar rats22 Ambalavanan N, Mourani P. Pulmonary hypertension in bronchopulmonary dysplasia. Birth Defects Res A Clin Mol Teratol. 2014 Mar;100(3):240-6. doi: 10.1002/bdra.23241.
https://doi.org/10.1002/bdra.23241...
,33 Chelstowska S, Wajs J, Rzepecki P, Plusa T. The role of fibrocytes in pulmonary fibrosis. Pol Merkur Lekarski. 2014 Jan;36(211):39-41. PMID: 24645577. were selected at random on days 3, 7 and 14 after the experiment had commenced. pentobarbital sodium (3%) was injected intraperitoneally (1 mL/100 g body weight) to sacrifice the rats. The lungs were immediately removed under aseptic conditions, placed in pre-cooled sterile petri dishes containing D-Hank's solution, and washed repeatedly to remove residual blood. Non-lung stromal tissues were isolated simultaneously. The washed lung tissues were transferred to sterile beakers, each specimen was cut to a size of 1 mm33 Chelstowska S, Wajs J, Rzepecki P, Plusa T. The role of fibrocytes in pulmonary fibrosis. Pol Merkur Lekarski. 2014 Jan;36(211):39-41. PMID: 24645577., and was gently suspended in D-Hank's solution. The supernatant was aspirated until the tissues were naturally pelleted. This was repeated 2-3 times, before 0.25% trypsin was added. Tissues were digested for 30 min in a 37°C oscillating water bath and digestions were terminated by adding an equal volume of Dulbecco's modified Eagle medium (DMEM) containing 10% fetal calf serum. Digests were passed through a 200-mesh filter, transferred to a tube for centrifugation (4°C, 80 xg, 5 min), and the supernatant was discarded. The pellets were resuspended in DMEM containing 10% FBS, the cell concentrations were adjusted as 2×1055 Wang XQ, Mao LJ, Fang QH, Kobayashi T, Kim HJ, Sugiura H, Kawasaki S, Togo S, Kamio K, Liu X, Rennard SI. Sphingosylphosphorylcholine induces a-smooth muscle actin expression in human lung fibroblasts and fibroblasts mediated gel contraction via S1P2 receptor and Rho/Rho-Kinase pathway. Prostaglandins Other Lipid Mediat. 2014 Jan;108:23-30. doi: 10.1016/j.prostaglandins.2014.02.002.
https://doi.org/10.1016/j.prostaglandins...
cells/ml, and then cells were inoculated into 5 mL cell culture flasks. Flasks were placed in an incubator with 5% CO2 at 37°C for 12 h. Approximately 80% of the adherent cells changed into LFs. The culture medium was changed daily. The cell morphology and the basic growth conditions were observed using an inverted microscope (Olympus, Tokyo, Japan).
Cell growth was monitored using microscopy. The culture medium was discarded when the cells were close to fusion (80%). D-Hank's solution was used to wash the cells and 1 mL 0.25% trypsin was added. Cells were exposed to trypsin for 2-5 min at room temperature until the cytoplasm retracted, cell bodies rounded, and cell gaps enlarged. The trypsin was then aspirated, and 10 mL of DMEM containing 10% fetal bovine serum was added to terminate the digestion. Cells were gently pipetted against the wall of the tube. After the cells were dissociated, the suspension was aliquoted into the flasks and the suspension was diluted 1:2 or 1:3 in culture medium. Flasks were placed into a CO2 incubator, and the medium was changed after 2 h to remove non-adherent cells. The third-generation cells at a purity of more than 95% were used in the experiment.
The second-generation LFs close to fusion were digested using trypsin, and the cell concentration was adjusted to 1×105 /mL. One milliliter of the cell suspension was added to six-well plates containing sterile coverslips. Plates were placed in 5% CO2 in a 37°C incubator, and the coverslips coated in cells were collected the following morning. Cells were washed in phosphate-buffered saline (PBS) three times and were fixed in 4% paraformaldehyde for 30 min. Vimentin expression was detected by immunohistochemistry, using the streptavidin-peroxidase method (Beijing Zhongshan Golden Bridge Biotechnology Co., Ltd.). After incubation for 10 minutes at room temperature with goat serum, the primary antibody of CAV-1 (1:160) was added with tissue for the whole night, then the secondary fluorescent antibody was added in darkroom, which was incubated for 90 minutes, before taken the picture, DAPI was added and incubated for 10 minutes at 37°C.
Caveolin-1 immunofluorescence staining
Cell-coated coverslips were washed in distilled water three times (5 min/wash). 5% normal goat serum was incubated with the coverslips at room temperature for 10 min. Vimentin was the antibody to test the caveolin-1 expression. Vimentin (1:100, Beijing Zhong Shan -Golden Bridge Biological Technology CO., LTD) and caveolin-1 antibodies (1:160, Santa Cruz Biotechnology, Santa Cruz, CA, USA) were applied and coverslips were placed at 4°C overnight. Coverslips were washed in PBS 3 times (5 min/wash). Fluorescent secondary antibodies (1:50) were added in a darkroom and were incubated with the coverslip for 90 min in a 37°C humidified chamber. Coverslips were washed in PBS 3 times (5 min/wash). Excess liquid was removed, and 50 μL 4′, 6-diamidino-2-phenylindole (DAPI) was added to each coverslip. Coverslips were incubated in DAPI for 10 min in a 37°C humidified chamber and were then washed in PBS 3 times (5 min/wash). Cells were examined and photographed using a fluorescence microscope (Olympus Corporation, Tokyo, Japan).
Western blot analysis of caveolin-1 levels in LFs
Proteins were extracted from the cells according to the protocol provided by the KGI whole protein extraction kit (Nanjing KGI Biotechnology Development Co., Ltd.). The BCA method was used to measure the protein concentration in the samples (Haibi Skylight Biotechnology Co., Ltd.). A 15% separating gel (20 mL) and 5% stacking gel (10 mL) were prepared, and 20 μL of protein sample was loaded into each well. Electrophoresis was performed (Bio-Rad-PAC300; Bio-Rad, Hercules, CA, USA) and proteins were then transferred to a nitrocellulose membrane. The membrane was washed once and then blocked using (Tris-buffered saline containing 0.1% Triton Tween 20) containing 5% nonfat milk for 1 h at room temperature. The membrane was washed in TBST 3 times (15 min/wash) and then incubated with caveolin-1 antibodies (1:200; Santa Cruz Biotechnology, Santa Cruz, CA, USA), at 4°C overnight.
Horseradish peroxidase-conjugated secondary antibodies were added (1:2000; Beijing Zhongshan Golden Bridge Biotechnology Co., Ltd.) for 2h at room temperature. Membranes were then washed 3 times in TBST (15 min/wash), and proteins were detected using enhanced chemiluminescence reagent.
Images were obtained using the MF-ChemiBIS 3.2 electrophoresis gel image analysis system (DNR Bio-Imaging Systems, Mahale HaHamisha, Israel). An imaging data analysis system (Universal Imaging Proportion, Molecular Devices, Sunnyvale, CA, USA) was used to record the gray value of each protein band and quantitative analysis was then performed. Protein content = gray value of sample protein/gray value of β-actin in the same sample.
Flow cytometric analysis of cell cycle
Cells belonging to each group were collected in the logarithmic growth phase and digested in 0.25% trypsin (Santa Cruz). Prepared cell suspensions were adjusted to 1×1066 Cai L, Yi F, Dai Z, Huang X, Zhao YD, Mirza MK, Xu J, Vogel SM, Zhao YY. Loss of caveolin-1 and adiponectin induces severe inflammatory lung injury following LPS challenge through excessive oxidative/nitrative stress. Am J Physiol Lung Cell Mol Physiol. 2014 Mar 15;306(6):L566-73. doi: 10.1152/ajplung.00182.2013.
https://doi.org/10.1152/ajplung.00182.20...
cells/mL and then centrifuged for 5 min at 1000 rpm at 4°C. Cells were washed twice in PBS and fixed in 5 mL of 70% ethanol overnight at 4°C. Subsequently, the cells were centrifuged the following day and resuspended in PBS. RNase (Sigma, St. Louis, MO, USA) was added to a final concentration of 50 μg/mL, and cells were placed in a 37°C water bath to allow the digest to proceed for 30 min. Propidium iodide (Sigma) was added to the cells at a final concentration of 100 μg/mL. Cells were stained in an ice bath in the dark for 30 min. Cells were then analyzed using a flow cytometer (BD, Franklin Lakes, USA) to detect changes in the cell cycle (BD CellQuest software).
Statistical analysis
SPSS13.0 software was used to perform statistical analyses. Data are presented as the mean ± standard deviation. The two samples were compared using t-tests. Single factor analysis of variance was used to compare samples. P<0.05 was considered statistically significant.
Results
Evaluation of the chronic lung disease in rats model
According to the score system, the results of the control group and hyperoxic group showed that on day 14, there was a significant difference between 2 groups, and the score was higher in hyperoxic group (Table 1).
Morphology and identification of primary cultured rat LFs
Dynamic LFs were shuttle- or star-shaped, but were not stained by trypan blue. The LFs possessed long processes and oval nuclei and showed radial or fence-like arrangement in the center of the culture. No significant changes in cell morphology were observed in the control and hyperoxic groups. Immunohistochemical analysis showed that vimentin was present in cells with brown cytoplasm and silk nets. The purity of cells the third-passage cells were greater than 95%. Up to 95% of cells were stained using immunofluorescence (Figure 1).
Localization of caveolin-1 in hyperoxic LFs
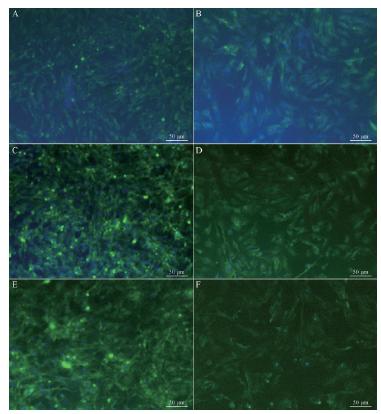
Caveolin-1 was expressed in both the nuclei and cytoplasm of LFs. On day 3, the hyperoxic group had no significant difference in caveolin-1 level compared to the corresponding control group. Caveolin-1 expression in hyperoxic cells on days 7 and 14, however, was significantly lower than in that the control group (P < 0.05). Caveolin-1 expression did not significantly differ in the control group on different days. No significant difference was observed in the hyperoxic group on days 3 and 7; however, caveolin-1 expression in hyperoxic cells was significantly lower on day 14 than on days 3 and 7 (Figure 2).
Caveolin-1 by immunofluorescent staining (A: control 3 d; B: experiment 3 d; C: control 7 d; D: experiment 7 d; E: control 14 d; F: experiment 14 d, x200).
Western blot
The hyperoxic group on day 3 showed no significant difference in caveolin-1 level from that of the corresponding control group. However, caveolin-1 expression in the hyperoxic group on days 14 was significantly lower than that of the control group (P<0.05). Caveolin-1 expression was not significantly different in the control group on different days. No significant difference in caveolin-1 level was observed in the hyperoxic group on days 3 and 7, but caveolin-1 expression was significantly lower in the hyperoxic group on day 14 than on days 3 and 7 (Figure 3, Table 2).
Cell cycle changes of LF
Flow cytometry (FCM) analysis showed that the percentage of LFs in the G0/G1 phase in the hyperoxic cells on day 7 was lower compared to the control group on day 7, while the proportion of the S phase cells increased (P<0.05). These changes were more significant (P<0.01) in the hypoxic group on day 14 (Figure 4, Table 3).
S-phase fraction (A: control 3 d, B: control 7 d, C: control 14 d, D: experiment 3d; E: experiment 7 d, F: experiment 14 d) (
 vs the control, P<0.05).
vs the control, P<0.05).
Discussion
CLD, caused by prolonged inhalation of high concentrations of oxygen, is a primary disease threatening the health and life of preterm children99 Lodha A, Sauvé R, Bhandari V, Tang S, Christianson H, Bhandari A, Amin H, Singhal N. Need for supplemental oxygen at discharge in infants with bronchopulmonary dysplasia is not associated with worse neurodevelopmental outcomes at 3 years corrected age. PLoS One. 2014 Mar 19;9(3):e90843. doi: 10.1371/journal.pone.0090843.
https://doi.org/10.1371/journal.pone.009...
,1010 Murthy K, Savani RC, Lagatta JM, Zaniletti I, Wadhawan R, Truog W, Grover TR, Zhang H, Asselin JM, Durand DJ, Short BL, Pallotto EK, Padula MA, Dykes FD, Reber KM, Evans JR. Predicting death or tracheostomy placement in infants with severe bronchopulmonary dysplasia. J Perinatol. 2014 Jul;34(7):543-8. doi: 10.1038/jp.2014.35.
https://doi.org/10.1038/jp.2014.35...
. Although the exact pathogenesis of CLD is not well understood, the outcome of this pathological process is clear-interstitial pulmonary fibrosis1111 Jain D, Bancalari E. Bronchopulmonary dysplasia: Clinical perspective. Birth Defects Res A Clin Mol Teratol. 2014 Mar;100(3):134-44. doi: 10.1002/bdra.23229.
https://doi.org/10.1002/bdra.23229...
. Prevention and treatment of interstitial pulmonary fibrosis is a significant challenge in the treatment of CLD. LFs, the effector cells of pulmonary fibrosis, are capable of synthesizing extracellular matrix and cytokines1212 Loomis-King H, Flaherty KR, Moore BB. Pathogenesis, current treatments and future directions for idiopathic pulmonary fibrosis. Curr Opin Pharmacol. 2013 Jun;13(3):377-85. doi: 10.1016/j.coph.2013.03.015.
https://doi.org/10.1016/j.coph.2013.03.0...
and initiating the inflammatory response; these cells have a pro-fibrotic effect1313 Habiel DM, Hogaboam C. Heterogeneity in fibroblast proliferation and survival in idiopathic pulmonary fibrosis. Front Pharmacol. 2014 Jan 23;5:2. doi: 10.3389/fphar.2014.00002.
https://doi.org/10.3389/fphar.2014.00002...
. Pulmonary fibrosis is associated with excessively proliferating LFs that deposit in the interstitial lung or replace the normal lung epithelium1414 Zhang L, Li Y, Liang C, Yang W. CCN5 overexpression inhibits profibrotic phenotypes via the PI3K/Akt signaling pathway in lung fibroblasts isolated from patients with idiopathic pulmonary fibrosis and in an in vivo model of lung fibrosis. Int J Mol Med. 2014 Feb;33(2):478-86. doi: 10.3892/ijmm.2013.1565.
https://doi.org/10.3892/ijmm.2013.1565...
. LF hyper proliferation is a characteristic of CLD pulmonary fibrosis1515 Vuga LJ, Milosevic J, Pandit K, Ben-Yehudah A, Chu Y, Richards T, Sciurba J, Myerburg M, Zhang Y, Parwani AV, Gibson KF, Kaminski N. Cartilage oligomeric matrix protein in idiopathic pulmonary fibrosis. PLoS One. 2013 Dec 20;8(12):e83120. doi: 10.1371/journal.pone.0083120.
https://doi.org/10.1371/journal.pone.008...
.
Directly culturing LFs in vitro in the presence of high concentrations of oxygen inhibits cell proliferation; however, the opposite effects are observed in the LFs derived from the tissues of humans or animals that have inhaled high concentrations of oxygen. Therefore, primary cell culture of LFs derived from CLD neonatal rats is the only feasible method to study the cellular mechanism by which LFs undergo excessive proliferation. The biological characteristics of the primary cell cultures obtained from the donors did not change significantly, maintaining diploid genetic characteristics. The cells closely reflected in vivo growth characteristics, which is extremely important for the study of cell growth, differentiation, metabolism, and mechanisms of physiological and pathological changes. It is generally held that extracted primary LFs that show no significant differences in biological traits over less than four generations can be used for experiments. Therefore, in this study, we examined the biological behavior of the third-generation LF cells by establishing a reliable primary cell culture method for LFs derived from CLD newborn rats to create a platform to study the mechanism of CLD lung fibrosis in vitro.
Tissue injury and fibrosis replacement after injury are pathophysiological processes involving tissue repair1616 Woodcock HV, Molyneaux PL, Maher TM. Reducing lung function decline in patients with idiopathic pulmonary fibrosis: potential of nintedanib. Drug Des Devel Ther. 2013 Jun 19;7:503-10. doi: 10.2147/DDDT.S38833.
https://doi.org/10.2147/DDDT.S38833...
. How LF proliferation is temporally and appropriately regulated such that it does not affect tissue repair but initiates fibrosis is not clear. Exploring the mechanism of cell cycle regulation in proliferating LFs could possibly address this question. A growing body of research on the cell cycle suggests that cell proliferation is closely linked to the cell cycle. The cell cycle is a fundamental cellular process. A complete cell cycle includes G1, S, G2, and M phases. The proportion of cells in G1 phase, the early DNA synthesis stage, is lower in highly proliferative cells. The proportion of cells in the DNA synthesis stage, S phase, is higher in highly proliferative cells. G2 and M phases are the late DNA synthesis and mitosis stages, respectively. Two important cell cycle checkpoints, G1/S and G2/M, regulate two key processes in the cell cycle, DNA replication and mitosis, respectively1717 Yunlan L, Juan Z, Qingshan L. Antitumor activity of di-n-butyl-(2, 6-difluorobenzohydroxamato)tin(iv) against human gastric carcinoma SGC-7901 cells via G2/M cell cycle arrest and cell apoptosis. PLoS One. 2014 Mar 18;9(3):e90793. doi: 10.1371/journal.pone.0090793.
https://doi.org/10.1371/journal.pone.009...
. The G1/S checkpoint is more important because it is the only point in the cycle that can be influenced by extrinsic proliferative and inhibitory information. Once cells undergo G1/S and transition into S phase, DNA replication is successfully completed and cells easily undergo the other checkpoints and complete mitosis. DNA was marked by nucleic acid staining and analyzed using flow cytometry to calculate the distribution of cells in G0/G1, S, and G2/M phases. The proportion of highly proliferative cells in S phase was elevated. In this study, the LF cells derived from newborn rats that had inhaled 90% oxygen for 3, 7, and 14 d were cultured to evaluate their proliferative state. FCM analysis showed that the proportion of LF cells in G1 phase of the cell cycle decreased with the increased exposure time to high oxygen, while the proportion of cells in the S phase increased. No significant changes were observed in the proportion of cells in the G2/M phase. These findings indicated that high oxygen weakened the G1/S checkpoint, allowing more cells to transition from G1 to S phase and ultimately promoting fibroblast proliferation.
Caveolin-1 is highly expressed in type I alveolar epithelial cells, endothelial cells, and fibroblasts1818 Vandermeulen E, Ruttens D, Verleden SE, Vos R, Van Raemdonck DE, Kastelijn EA, Wauters E, Lambrechts D, Nawrot TS, Cox B, Verleden GM, Vanaudenaerde BM. Genetic variation in caveolin-1 affects survival after lung transplantation. Transplantation. 2014 Aug 15;98(3):354-9. doi: 10.1097/TP.0000000000000058.
https://doi.org/10.1097/TP.0000000000000...
. Studies have shown that caveolin-1 is associated with cell proliferation. Under normal physiological conditions, lung fibroblast growth is tightly regulated and lung collagen synthesis and degradation are in equilibrium. Lung fibroblasts play an important role in chronic pulmonary fibrosis formation caused by various factors. Zundel et al.1919 Zundel W, Swiersz LM, Giaccia A. Caveolin 1-mediated regulation of receptor tyrosine kinase-associated phosphatidylinositol 3-kinase activity by ceramide. Mol Cell Biol. 2000 Mar;20(5):1507-14. PMID: 10669728. observed that a decrease in caveolin-1 expression may significantly downregulate the ceramide-induced PI3K signaling pathway, allowing Rat1 fibroblasts to resist ceramide-induced apoptosis. However, increased caveolin-1 expression may alter PI3K activity, increasing the sensitivity of Rat1 fibroblasts to ceramide-induced apoptosis. Using antisense technology to decrease caveolin-1 expression in NIH/3T3, Liu et al.2020 Liu J, Lee P, Galbiati F, Kitsis RN, Lisanti MP. Caveolin-1 expression sensitizes fibroblastic and epithelial cells to apoptotic stimulation. Am J Physiol Cell Physiol. 2001 Apr;280(4):C823-35. PMID: 11245599. found that cells with reduced caveolin-1 expression resist staurosporine-induced cell apoptosis, while increased expression restores NIH/3T3 cell sensitivity to apoptotic stimuli. After administering the PI3K/PKB pathway inhibitor LY2294002, NIH/3T3 cells have enhanced sensitivity to apoptotic stimuli. Wang et al2121 Wang XM, Zhang Y, Kim HP, Zhou Z, Feghali-Bostwick CA, Liu F, Ifedigbo E, Xu X, Oury TD, Kaminski N, Choi AM. Caveolin-1: a critical regulator of lung fibrosis in idiopathic pulmonary fibrosis. J Exp Med. 2006 Dec 25;203(13):2895-906. doi: 10.1084/jem.20061536.
https://doi.org/10.1084/jem.20061536...
reported that caveolin-1 expression is reduced in the lung tissues of patients with idiopathic pulmonary fibrosis. Stimulating primary cultured lung tissue fibroblasts and MRC25 with TGF-β decreases the mRNA and protein expression of caveolin-1 in a time- and dose-dependent manner. Bleomycin or TGF-β1 administered after caveolin-1 siRNA leads to increased α-SMA expression, stimulating lung fibroblasts to transform into myofibroblasts. Conversely, an increase in caveolin-1 expression inhibits cell transformation2222 Rathor N, Zhuang R, Wang JY, Donahue JM, Turner DJ, Rao JN. Src-mediated caveolin-1 phosphorylation regulates intestinal epithelial restitution by altering Ca2+ influx after wounding. Am J Physiol Gastrointest Liver Physiol. 2014 Apr 15;306(8):G650-8. doi: 10.1152/ajpgi.00003.2014.
https://doi.org/10.1152/ajpgi.00003.2014...
. In this study, LF cells from newborn rats that inhaled 90% oxygen for 3, 7, and 14 d were cultured. The role of caveolin-1 expression level in lungs fibroblast proliferation and the occurrence and development of pulmonary fibrosis through dynamic studying the caveolin-1 expression level of LF at different stages of development in hyperoxia-induced CLD rats. With longer exposure to high oxygen, caveolin-1 expression was significantly lower in the hyperoxic group than in the control group on days 7 and 14. These data were consistent with the above report. LF cell proliferation showed the opposite trend. We observed that caveolin-1 downregulation was correlated to LF cell proliferation, suggesting caveolin-1 is involved in the incidence and development of CLD rat lung fibrosis. However, the mechanism underlying this effect is not clear and warrants further study.
Conclusions
After exposure to hyperoxic conditions for 7 d, LFs showed a marked increase in proliferation, while the expression of caveolin-1 began to decrease. The level of caveolin-1 expression was inversely related to LF proliferation.
References
-
1Jensen EA, Schmidt B. Epidemiology of bronchopulmonary dysplasia. Birth Defects Res A Clin Mol Teratol. 2014 Mar;100(3):145-57. doi: 10.1002/bdra.23235.
» https://doi.org/10.1002/bdra.23235 -
2Ambalavanan N, Mourani P. Pulmonary hypertension in bronchopulmonary dysplasia. Birth Defects Res A Clin Mol Teratol. 2014 Mar;100(3):240-6. doi: 10.1002/bdra.23241.
» https://doi.org/10.1002/bdra.23241 -
3Chelstowska S, Wajs J, Rzepecki P, Plusa T. The role of fibrocytes in pulmonary fibrosis. Pol Merkur Lekarski. 2014 Jan;36(211):39-41. PMID: 24645577.
-
4Ansarie M, Naseem A, Kasmani A, Ahmed R, Azeemuddin M. Profile of interstitial lung diseases in pakistan, karachi pulmonology clinics registry data: jan 2012 - aug 2013. Chest. 2014;145(3 Suppl):241A.
-
5Wang XQ, Mao LJ, Fang QH, Kobayashi T, Kim HJ, Sugiura H, Kawasaki S, Togo S, Kamio K, Liu X, Rennard SI. Sphingosylphosphorylcholine induces a-smooth muscle actin expression in human lung fibroblasts and fibroblasts mediated gel contraction via S1P2 receptor and Rho/Rho-Kinase pathway. Prostaglandins Other Lipid Mediat. 2014 Jan;108:23-30. doi: 10.1016/j.prostaglandins.2014.02.002.
» https://doi.org/10.1016/j.prostaglandins.2014.02.002 -
6Cai L, Yi F, Dai Z, Huang X, Zhao YD, Mirza MK, Xu J, Vogel SM, Zhao YY. Loss of caveolin-1 and adiponectin induces severe inflammatory lung injury following LPS challenge through excessive oxidative/nitrative stress. Am J Physiol Lung Cell Mol Physiol. 2014 Mar 15;306(6):L566-73. doi: 10.1152/ajplung.00182.2013.
» https://doi.org/10.1152/ajplung.00182.2013 -
7Reese C, Perry B, Heywood J, Bonner M, Visconti RP, Lee R, Hatfield CM, Silver RM, Hoffman S, Tourkina E. Caveolin-1 deficiency predisposes African-Americans to SSc-ILD. Arthritis Rheumatol. 2014 Jul;66(7):1909-19. doi: 10.1002/art.38572.
» https://doi.org/10.1002/art.38572 -
8Kelleher MD, Naureckas ET, Solway J, Hershenson MB. In vivo hyperoxic exposure increases cultured lung fibroblast proliferation and c-Ha-ras expression. Am J Respir Cell Mol Biol. 1995 Jan;12(1):19-26. doi: 10.1165/ajrcmb.12.1.7811467.
» https://doi.org/10.1165/ajrcmb.12.1.7811467 -
9Lodha A, Sauvé R, Bhandari V, Tang S, Christianson H, Bhandari A, Amin H, Singhal N. Need for supplemental oxygen at discharge in infants with bronchopulmonary dysplasia is not associated with worse neurodevelopmental outcomes at 3 years corrected age. PLoS One. 2014 Mar 19;9(3):e90843. doi: 10.1371/journal.pone.0090843.
» https://doi.org/10.1371/journal.pone.0090843 -
10Murthy K, Savani RC, Lagatta JM, Zaniletti I, Wadhawan R, Truog W, Grover TR, Zhang H, Asselin JM, Durand DJ, Short BL, Pallotto EK, Padula MA, Dykes FD, Reber KM, Evans JR. Predicting death or tracheostomy placement in infants with severe bronchopulmonary dysplasia. J Perinatol. 2014 Jul;34(7):543-8. doi: 10.1038/jp.2014.35.
» https://doi.org/10.1038/jp.2014.35 -
11Jain D, Bancalari E. Bronchopulmonary dysplasia: Clinical perspective. Birth Defects Res A Clin Mol Teratol. 2014 Mar;100(3):134-44. doi: 10.1002/bdra.23229.
» https://doi.org/10.1002/bdra.23229 -
12Loomis-King H, Flaherty KR, Moore BB. Pathogenesis, current treatments and future directions for idiopathic pulmonary fibrosis. Curr Opin Pharmacol. 2013 Jun;13(3):377-85. doi: 10.1016/j.coph.2013.03.015.
» https://doi.org/10.1016/j.coph.2013.03.015 -
13Habiel DM, Hogaboam C. Heterogeneity in fibroblast proliferation and survival in idiopathic pulmonary fibrosis. Front Pharmacol. 2014 Jan 23;5:2. doi: 10.3389/fphar.2014.00002.
» https://doi.org/10.3389/fphar.2014.00002 -
14Zhang L, Li Y, Liang C, Yang W. CCN5 overexpression inhibits profibrotic phenotypes via the PI3K/Akt signaling pathway in lung fibroblasts isolated from patients with idiopathic pulmonary fibrosis and in an in vivo model of lung fibrosis. Int J Mol Med. 2014 Feb;33(2):478-86. doi: 10.3892/ijmm.2013.1565.
» https://doi.org/10.3892/ijmm.2013.1565 -
15Vuga LJ, Milosevic J, Pandit K, Ben-Yehudah A, Chu Y, Richards T, Sciurba J, Myerburg M, Zhang Y, Parwani AV, Gibson KF, Kaminski N. Cartilage oligomeric matrix protein in idiopathic pulmonary fibrosis. PLoS One. 2013 Dec 20;8(12):e83120. doi: 10.1371/journal.pone.0083120.
» https://doi.org/10.1371/journal.pone.0083120 -
16Woodcock HV, Molyneaux PL, Maher TM. Reducing lung function decline in patients with idiopathic pulmonary fibrosis: potential of nintedanib. Drug Des Devel Ther. 2013 Jun 19;7:503-10. doi: 10.2147/DDDT.S38833.
» https://doi.org/10.2147/DDDT.S38833 -
17Yunlan L, Juan Z, Qingshan L. Antitumor activity of di-n-butyl-(2, 6-difluorobenzohydroxamato)tin(iv) against human gastric carcinoma SGC-7901 cells via G2/M cell cycle arrest and cell apoptosis. PLoS One. 2014 Mar 18;9(3):e90793. doi: 10.1371/journal.pone.0090793.
» https://doi.org/10.1371/journal.pone.0090793 -
18Vandermeulen E, Ruttens D, Verleden SE, Vos R, Van Raemdonck DE, Kastelijn EA, Wauters E, Lambrechts D, Nawrot TS, Cox B, Verleden GM, Vanaudenaerde BM. Genetic variation in caveolin-1 affects survival after lung transplantation. Transplantation. 2014 Aug 15;98(3):354-9. doi: 10.1097/TP.0000000000000058.
» https://doi.org/10.1097/TP.0000000000000058 -
19Zundel W, Swiersz LM, Giaccia A. Caveolin 1-mediated regulation of receptor tyrosine kinase-associated phosphatidylinositol 3-kinase activity by ceramide. Mol Cell Biol. 2000 Mar;20(5):1507-14. PMID: 10669728.
-
20Liu J, Lee P, Galbiati F, Kitsis RN, Lisanti MP. Caveolin-1 expression sensitizes fibroblastic and epithelial cells to apoptotic stimulation. Am J Physiol Cell Physiol. 2001 Apr;280(4):C823-35. PMID: 11245599.
-
21Wang XM, Zhang Y, Kim HP, Zhou Z, Feghali-Bostwick CA, Liu F, Ifedigbo E, Xu X, Oury TD, Kaminski N, Choi AM. Caveolin-1: a critical regulator of lung fibrosis in idiopathic pulmonary fibrosis. J Exp Med. 2006 Dec 25;203(13):2895-906. doi: 10.1084/jem.20061536.
» https://doi.org/10.1084/jem.20061536 -
22Rathor N, Zhuang R, Wang JY, Donahue JM, Turner DJ, Rao JN. Src-mediated caveolin-1 phosphorylation regulates intestinal epithelial restitution by altering Ca2+ influx after wounding. Am J Physiol Gastrointest Liver Physiol. 2014 Apr 15;306(8):G650-8. doi: 10.1152/ajpgi.00003.2014.
» https://doi.org/10.1152/ajpgi.00003.2014
-
Financial source:
none -
1
Research performed at Department of Pediatrics, Shengjing Hospital, China Medical University, Shenyang, China.
Publication Dates
-
Publication in this collection
May 2017
History
-
Received
15 Jan 2017 -
Reviewed
18 Mar 2017 -
Accepted
19 Apr 2017