Abstract
Purpose:
To performed a histomorphometric and radiological study to evaluate the effects of alendronate sodium administered locally in mandibular bone defects created in rabbits.
Methods:
Two circular defects 5 mm in diameter were created bilaterally in the mandibular corpus of 20 New Zealand rabbits (i.e., four defects per animal). Each defect received one of four treatments: no treatment (EC group), alendronate irrigation (AL group), autogenous bone grafting (AG group), or alendronate irrigation with autogenous bone grafting (AL+AG group). Histomorphometric and radiological assessments were conducted at 4 and 8 weeks after surgery.
Results:
Between-group comparisons of the new bone area, the value of the AL+AG group was significantly lower thanthe remaining three groups at 4 weeks postoperatively. In all groups, the new bone area was significantly larger at 8 weeks than at 4 weeks. The residual graft area at 4 and 8 weeks was significantly higher in the AL+AG group than in the AG group, although it was significantly smaller at 8 weeks than at 4 weeks in both these groups.
Conclusion:
The use of alendronate sodium in conjunction with autogenous bone grafting improves the osteoconductive properties of the graft, enhances graft retention in the defect, and improves ossification.
Key words:
Alendronate; Disphosphonates; Bone Transplantation; Rabbits
Introduction
In oral surgery, bone grafting is commonly performed to compensate for hard tissue loss, and approximately 2.2 million graft surgeries are annually performed worldwide11 Giannoudis P, Psarakis S, and Kontakis G. Can we accelerate fracture healing? A critical analysis of the literature. Injury. 2007;38(1):S81-9. doi: 10.1016/j.injury.2007.02.013.
https://doi.org/10.1016/j.injury.2007.02...
. Various types of grafts are used to repair defects that develop secondary to atrophy, injury, congenital malformations, or tumor surgeries22 Alfaro FH. Bone grafting in oral implantology: techniques and clinical applications. Spain: Quintessence Books; 2006.. While autogenous bone is the current gold standard for such grafts, this material, as all other graft materials used today, has various advantages and disadvantages33 Ashammakhi N, Ferretti P. Topics in tissue engineering. v.1. Finland: University of Oulu; 2003.. The operating surgeon should be aware of certain properties of the graft material in relation to bone metabolism and associated factors.
Bisphosphonates, which are frequently prescribed in the treatment of bone diseases, have been in use for approximately a century44 Marx RE, Cillo JE, Ulloa JJ. Oral bisphosphonate-induced osteonecrosis: risk factors, prediction of risk using serum CTX testing, prevention, and treatment. J Oral Maxillofac Surg. 2007;65(12):2397-410. doi: 10.1016/j.joms.2007.08.003.
https://doi.org/10.1016/j.joms.2007.08.0...
. Specifically, bisphosphonates were introduced for clinical use after their biological effects were reported in 1968, and they were first prescribed to prevent the formation of kidney stones55 Watts NB. Treatment of osteoporosis with bisphosphonates. Rheum Dis Clin North Am. 2001;27(1):197-214. doi: 10.1016/S0889-8529(05)70014-1.
https://doi.org/10.1016/S0889-8529(05)70...
. Subsequently, in 1971, bisphosphonates were used for the treatment of Paget’s disease44 Marx RE, Cillo JE, Ulloa JJ. Oral bisphosphonate-induced osteonecrosis: risk factors, prediction of risk using serum CTX testing, prevention, and treatment. J Oral Maxillofac Surg. 2007;65(12):2397-410. doi: 10.1016/j.joms.2007.08.003.
https://doi.org/10.1016/j.joms.2007.08.0...
.
Bisphosphonates affect bone metabolism by decreasing both the activity and number of osteoclasts, as well as indirectly inhibiting the activity of osteoclasts by altering the signals sent by osteoblasts to osteoclasts44 Marx RE, Cillo JE, Ulloa JJ. Oral bisphosphonate-induced osteonecrosis: risk factors, prediction of risk using serum CTX testing, prevention, and treatment. J Oral Maxillofac Surg. 2007;65(12):2397-410. doi: 10.1016/j.joms.2007.08.003.
https://doi.org/10.1016/j.joms.2007.08.0...
. In other words, bisphosphonates do not allow osteoblasts to release factors that attract osteoclasts to the resorption site, thereby prolonging their life. The main effect at the tissue level is a decrease in bone turnover secondary to the inhibition of bone resorption55 Watts NB. Treatment of osteoporosis with bisphosphonates. Rheum Dis Clin North Am. 2001;27(1):197-214. doi: 10.1016/S0889-8529(05)70014-1.
https://doi.org/10.1016/S0889-8529(05)70...
.
Alendronate sodium, which contains nitrogen, is a very potent drug from the bisphosphonate family. On systemic administration, alendronate inhibits bone resorption and increases osteoblast proliferation by affecting bone metabolism66 Fleisch H. Bisphosphonates in bone disease: from the laboratory to the patient. USA: Academic Press; 2000.. Alendronate-induced inhibition of bone resorption is dose-dependent and approximately 1000-fold stronger than that caused by etidronate, the first bisphosphonate-derived drug introduced into clinical use. Like other bisphosphonates, alendronate sodium is used for the treatment and prevention of Paget’s disease, fibrous dysplasia, and osteoporosis, which are characterized by increased bone turnover and resorption66 Fleisch H. Bisphosphonates in bone disease: from the laboratory to the patient. USA: Academic Press; 2000.. Recent studies investigating the local effects and systemic side effects of alendronate sodium have shown that locally administered alendronate solution is absorbed by the bone at a rate of 20%-30% at the site of administration, where it inhibits bone resorption77 Binderman I, Adut M, Yaffe A. Effectiveness of local delivery of alendronate in reducing alveolar bone loss following periodontal surgery in rats. J Periodontol. 2000;71(8):1236-40. doi: 10.1902/jop.2000.71.8.1236.
https://doi.org/10.1902/jop.2000.71.8.12...
.
In the present study, we used histomorphometric and radiological assessments to evaluate the effects of alendronate sodium administered locally at the site of mandibular bone defects created in rabbits. Our hypothesis was that the local administration of bisphosphonate derivatives in conjunction with autogenous bone grafting will decelerate the resorption of the bone graft and simultaneously accelerate ossification.
Methods
This study was approved by the Local Ethics Committee of Animal Experiments of Hacettepe University (decision 2014/29-1 issued June 10, 2014).
Twenty adult New Zealand rabbits (10 male, 10 female) weighing an average of 3500 g each were used for the study. The experimental animals were housed in suitably sized cages maintained at a temperature of 22±2°C and a 12-h light/dark cycle. The animals were transferred to the laboratory environment at least a week before surgery in order to help them adapt to the new environment as well as to check their general health and ensure an optimal, infection-free environment. The animals were fed with standard laboratory food and water without any limitation. Each rabbit was housed in a separate cage to ensure comfortable access to water and food, an adequate range of motion, and a stress-free environment.
Alendronate sodium trihydrate (Cayman Chemical, Ann Arbor, MI, USA) was used for local administration to the defect. To promote guided bone regeneration, a collagen membrane (Collagene AT; Sistema AT, Padova, Italy) known to have delayed resorption was used.
Four defects were created in each animal. Specifically, two defects were created in the left mandibular corpus, and another two defects were created in the right mandibular corpus. Four treatment modalities were evaluated in this study, and each defect in the same animal underwent a different treatment. Accordingly, four groups of 20 defects each were designated as follows: defects in the EC group (empty control) were left empty; defects in the AG group received autogenous bone grafts only; defects in the AL group received local alendronate irrigation only; and defects in the AL+AG group received autogenous grafts with local alendronate irrigation. To prevent possible contamination during alendronate treatment, defects created on the right side of the mandible were included in the AG or EC groups, whereas those created on the left side were included in the AL+AG or AL groups. Moreover, to ensure standardization and prevent possible tissue intermingling during autogenous bone grafting, the grafts created in the posterior region were included in the AG or AL+AG groups, whereas those created in the anterior region were included in the EC or AL groups. Radiological and histomorphometric assessments were carried out at 4 and 8 weeks after surgery.
Preparation of the alendronate solution
In accordance with the manufacturer’s recommendations, alendronate sodium was stored in a dark environment at −20°C. On the day of surgery, an alendronate solution for local administration was prepared at a concentration of 1 mg/mL of alendronate sodium in saline. The prepared solution was stored under sterile conditions at 5°C for less than 24 h. When required, the solution was transferred to the working area in a sterile syringe and administered.
Surgical procedure
For anesthesia, ketamine hydrochloride 35 mg/kg (Alfamine; Alfasan Int.) and xylazine hydrochloride 2.5 mg/kg (Alfazyne; Alfasan Int.) were administered intramuscularly. The animals were shaved in the regions over the right and left mandibular corpora, and the surgical site was cleaned with povidone iodine (Batticon; Adeka Laboratories, Istanbul, Turkey). For hemostasis, 1 mL of local anesthetic solution (Ultracain D-S forte; Sanofi-Aventis, Istanbul, Turkey) was infiltrated in the relevant regions on the right and left sides. A 1.5-cm long, full-thickness incision including the periosteum was made over the right and left corpora using a No. 15 scalpel. Subsequently, the bone surface was exposed by dissection of several muscle attachments in the anterior part of the masseter muscle. A total of four bone defects, two on the right side and two on the left, were created using a trephine drill with an external diameter of 5 mm and an internal diameter of 4 mm, with sterile saline irrigation. The depth of each defect was 3 mm, and there was a minimum gap of 3 mm between defects (Figure 1A). The cortical bone removed from the right side was stored in a dappen dish and used as autologous graft material in the AG group (on the right side). Similarly, the cortical bone removed from the left side was stored in a dappen dish and used as autologous graft material in the AL+AG group (on the left side). The following protocol was applied:
Protocol for autogenous bone grafting of mandibular bone defects in a rabbit model. A. Two bone defects measuring 5 mm in diameter were created on each side of the mandible (four defects in total). B. The autogenous bone grafts were milled into small particles using a bone grinder. C. The particles obtained from one defect were used as autogenous graft material to fill the other defect on the same side, which was irrigated with saline or alendronate solution. D. The grafted area was covered with a collagen membrane, which was rehydrated with saline or alendronate solution.
-
EC group: The defect was irrigated with saline and covered with the collagen membrane, which was rehydrated with saline.
-
AG group: The disc-shaped area of cortical bone removed to create the other defect on the same side of the mandible was milled into small particles using a bone grinder (Figure 1B), and the particles were used to fill the defect, which was irrigated with saline (Figure 1C). Then, the grafted area was covered with the collagen membrane, which was rehydrated with saline (Figure 1D).
-
AL group: The defect was irrigated with the prepared alendronate solution and left uncovered for 5 min. Then, the defect was covered with the collagen membrane, which was rehydrated with the alendronate solution.
-
AL+AG group: The disc-shaped area of cortical bone removed to create the other defect on the same side of the mandible was milled into small particles using a bone grinder. The bone particles were dispersed in the prepared alendronate solution and left to sit for 5 min. The defect was irrigated with the alendronate solution mixed with bone particles and left uncovered for 5 min. Afterwards, the defect was covered with the collagen membrane, which was rehydrated with the same alendronate solution mixed with bone particles.
Finally, the skin and subcutaneous tissues were primarily sutured with resorbable, 16-mm, 3/8 circle, sharp 4-0 polyglactin sutures (coated Vicryl; Ethicon, Johnson & Johnson, Brussels, Belgium). The sutured areas were treated with a wound dressing spray (Opsite; Smith & Nephew, Mississauga, Canada) to prevent postoperative infection.
After surgery, each animal received a single daily intramuscular dose of meloxicam 1 mg/kg (Maxicam X4; Sanovel, Istanbul, Turkey) for analgesia and enrofloxacin 2.5 mg/kg (Baytril-K 5%; Bayer, Pittsburgh, PA, USA) for infection control during the first five postoperative days. During the experimental period, the animals were kept alive in the same cages as those used before surgery, under 12-h light/dark cycles. The average temperature and humidity of the environment were maintained at 22-24°C and 55%-70%, respectively. The wound areas were regularly checked, and adequate amounts of food and water were provided. One animal was excluded from the 8-week follow-up analysis because it died at 6 weeks after surgery.
Tissue processing
Half of the animals were sacrificed at the end of week 4 after surgery, whereas the remaining animals were sacrificed at the end of week 8 after surgery. Death was induced by administration of a lethal dose of intramuscular xylazine hydrochloride (30 mg/kg; Alfazyne; Alfasan Int.) and ketamine hydrochloride (70 mg/kg; Alfamine; Alfasan Int.). Each rabbit was numbered, and the mandible was removed via an incision made beneath. Following periosteal elevation, the regions of the defects were identified and separated from the rest of the mandible. The obtained pieces were reduced in size by cutting away excess bone but leaving sufficient amount of solid bone tissue around the defects themselves. The specimens collected from each animal were assigned to the appropriate groups and fixed in 10% buffered formaldehyde for 48 h.
Radiological analysis
The specimens were scanned using a micro-computed tomography (micro-CT) device (Skyscan 1174; Skyscan, Kontich, Belgium) with a pixel size of 40 μm. The X-ray tube voltage was set at 50 kV and the current was set at 800 μA. The exposure time was 2300 ms. X-ray projections were obtained at 0.70° intervals, with a scanning angular rotation of 180°. Crude data obtained at the screening stage were subsequently reconstructed using the NRecon software (NRecon version 1.6.9.4; Skyscan, Kontich, Belgium) provided by the manufacturer. The 8-bit gray images reconstructed using NRecon were transferred to the CTan software (version 1.13.5.1; Skycan). Basic values such as bone volume, bone surface area, soft tissue volume, soft tissue surface area, bone/soft tissue surface area ratio, and bone density were obtained for selected regions of interest.
Histomorphometric analysis
Histomorphometric analysis was performed by an examiner blinded to specimen treatment. Specimens were decalcified for 21 days in a 10% formic acid solution that was changed every 3 days. The tissues were washed with distilled water and dehydrated by passing through a series of alcohol solutions. Next, an elution process with a series of xylene solutions was performed. Subsequently, specimen blocks were prepared and embedded in paraffin. Then, 4-6-μm-thick transverse serial sections were obtained with 1/20 sampling, deparaffinized with xylene, incubated at 60°C overnight, dehydrated, and stained either with hematoxylin and eosin or with modified Movat’s pentachrome. All stained sections were covered with Canada balsam and mounted on glass slides for histological analysis. Histomorphometric analyses were performed using specialized image analysis software (Leica Qwin plus V3; Leica Microsystems, Wetzlar, Germany). Five different images were obtained from each specimen section under x100 magnification. In each image, the areas of new bone trabeculae, soft tissue, and graft filling the defect area were calculated in μm2. Only the mean values based on measurements in five images were retained.
Statistical analysis
The study data were analyzed using the software SPSS version 18.0 (SPSS Inc., Chicago, IL, USA). Descriptive statistics (means, medians, standard deviations), Mann-Whitney U tests (for comparisons of non-normally distributed, independent, continuous, two-group continuous variables), and Kruskal-Wallis tests (for comparisons of more than two groups) were used for analyses. The Wilcoxon test was performed to compare non-normally distributed, dependent, two-group continuous variables. The distribution of values for each variable was checked for normality using the Kolmogorow-Smirnov and Shapiro-Wilk tests. Type-1 error levels (alpha values) of <0.05 were considered to indicate statistical significance.
Results
Histological analysis of the specimens collected at 4 weeks postoperatively revealed new bone trabeculae and loose connective tissue with interconnected collagen fibrils. As the connective tissue approached the center of the defect, it began to show cellular appearance. New bone formation was essentially centripetal, i.e., it began from the periphery of the defect and increased toward the center, with activation of the injured periosteum. This centripetal tendency was more remarkable in specimens from the AL and AL+AG groups. Although new bone trabeculae were formed around the graft particles, the majority were localized at the periphery of the defect. Histological analysis of the specimens collected at 8 weeks postoperatively revealed a general histological appearance that was similar to that of observed at 4 weeks, although the number of bone trabeculae was greater at 8 weeks. Moreover, fewer graft particles remained between the trabeculae at 8 weeks than at 4 weeks. Although new bone formation was slightly more pronounced toward the center, the entire defect was never filled with new bone. In the AL+AG group specimens, although graft particles remained between the bone trabeculae at 8 weeks, they were fewer than noted at 4 weeks. However, at 8 weeks, there were more graft particles between the bone trabeculae in the AL+AG group specimens than in the AG group specimens (Figures 2 and 3).
Histological findings at 4 after surgery for mandibular bone defects in a rabbit model. A. At 4 weeks, specimens in the AL+AG group showed formation of anastomosing new bone trabeculae and evidence of graft material (hematoxylin and eosin [H&E], ×40). B. At 4 weeks, new bone trabeculae were observed towards the center of the defect at the continuation of the host bone trabeculae specimen in the AG group. The loose collagen ligament in the periphery of the defect was noted to be in a central collagenous structure (Movat’s modified pentachrome, ×40). C. In specimens from the AL group, the defect area is composed of new bone trabeculae and loose collagen connective tissue at 4 weeks (H&E, ×40). D. At 4 weeks, specimens in the EC group show that the defect area consists of anastomosing new bone trabeculae’s and loose collagen connective tissue (H&E, ×40). (EC group, no treatment; AL group, alendronate irrigation only; AG group, autogenous bone grafting only; AL+AG group, alendronate irrigation with autogenous bone grafting; n, new bone trabeculae; g, graft material; st, soft tissue; hb, host bone).
Histological findings at 8 weeks after surgery for mandibular bone defects in a rabbit model. A. At 8 weeks, specimens in the AL+AG group showed formation of anastomosing new bone trabeculae, graft particles, and cellular structured collagen connective tissue (hematoxylin and eosin [H&E], ×40). B. Specimens in the AG group at 8 weeks showed anastomosing new bone trabeculae (less than in the AL+AG group), graft particles, and cellular structured collagen connective tissue (H&E, ×40). C. At 8 weeks, AL group specimens show anastomosing new bone trabeculae’s that start from the periphery, and cellular structured connective tissue (Movat’s modified pentachrome, ×40). D. EC group specimens show the new bone trabeculae and collagen connective tissue, which was cellular structured in the center of the defect area and loose structured in the periphery at 8 weeks (H&E, ×40). (EC group, no treatment; AL group, alendronate irrigation only; AG group, autogenous bone grafting only; AL+AG group, alendronate irrigation with autogenous bone grafting; n, new bone trabeculae; g, graft material; st, soft tissue; hb, host bone).
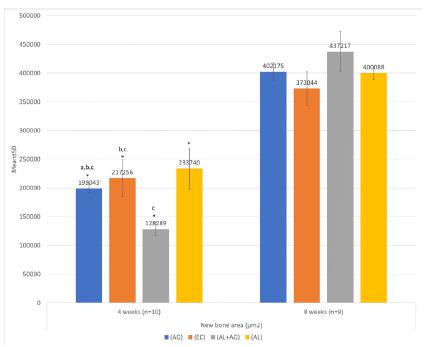
The new bone area at 4 weeks differed significantly among the four groups (p < 0.001). Specifically, there was a significant difference between the AL+AG group and the remaining three groups (p < 0.05). However, there was no significant difference among the four groups at 8 weeks (p < 0.126). In addition, for all groups, the new bone area was significantly greater at 8 weeks than at 4 weeks (p < 0.05; Figure 4).
New bone area measured by histomorphometry at 4 and 8 weeks after surgery for mandibular bone defects in a rabbit model. (EC group, no treatment; AL group, alendronate irrigation only; AG group, autogenous bone grafting only; AL+AG group, alendronate irrigation with autogenous bone grafting; *significantly different from values obtained at 8 weeks after surgery; a, significantly different from values obtained in the EC group; b, significantly different from values obtained in the AL+AG group; c, significantly different from values obtained in the AL group).
The residual graft area was significantly different between the AL+AG and AG groups at both 4 and 8 weeks (p < 0.05). The residual graft area at 4 weeks was statistically higher in the AG group, whereas it was found to be statistically higher in the AL + AG group at the 8 weeks. However, in both groups, the residual graft area at 8 weeks was significantly lower than that at 4 weeks (p < 0.05; Figure 5).
Residual graft area measured by histomorphometry at 4 and 8 weeks after surgery for mandibular bone defects in a rabbit model. (EC group, no treatment; AL group, alendronate irrigation only; AG group, autogenous bone grafting only; AL+AG group, alendronate irrigation with autogenous bone grafting; *significantly different from values obtained at 8 weeks after surgery; b, significantly different from values obtained in the AL+AG group).
The total radiographic percentage of bone in the EC, AL+AG, and AL groups was significantly higher at 8 weeks than at 4 weeks (p = 0.008), whereas no significant increase was noted in the AG group (p = 0.260; Figures 6 and 7).
Comparison of values for the percentage of total bone derived from radiographs obtained at 4 and 8 weeks after surgery. (EC group, no treatment; AL group, alendronate irrigation only; AG group, autogenous bone grafting only; AL+AG group, alendronate irrigation with autogenous bone grafting; *significantly different from values obtained at 8 weeks after surgery).
The bone density was not significantly different among the groups (p > 0.05). However, the bone density in the EC group was significantly lower at 8 weeks than at 4 weeks (p = 0.008), whereas no significant difference between the 4- and 8-week values of bone density was noted for the other three groups (p > 0.05; Figure 8).
Bone density values on radiographs obtained at 4 and 8 weeks after surgery for mandibular bone defects in a rabbit model. (EC group, no treatment; AL group, alendronate irrigation only; AG group, autogenous bone grafting only; AL+AG group, alendronate irrigation with autogenous bone grafting; *significantly different from values obtained at 8 weeks after surgery).
Discussion
The present study began with the hypothesis that the local administration of bisphosphonate derivatives in conjunction with autogenous bone grafting would decelerate the resorption of the bone graft and simultaneously accelerate ossification. Our results confirmed this hypothesis.
Previous studies in rabbit models focused on defects created in the mandible, calvaria, femur, tibia, fibula, and radius. In the present study, which also employed a rabbit model, we focused on defects in the mandible because this bone exhibits embryologic development characteristics similar to those of maxillofacial bones in humans.
Examination of the callus of injured mandibles typically reveals cells originating only from the neural crest, whereas cells of mesodermal origin are normally encountered in the callus of injured tibia bones; furthermore, when progenitor cells with mesodermal origin are placed in the injured area of the mandible, they do not transform into osteoblasts88 Leucht P, Kim JB, Amasha R, James AW, Girod S, Helms JA. Embryonic origin and Hox status determine progenitor cell fate during adult bone regeneration. Development. 2008;135(17):2845-54. doi: 10.1242/dev.023788.
https://doi.org/10.1242/dev.023788...
. On the basis of these previous findings, we chose to use the bone obtained from the creation of defects as the autogenous bone graft material for filling the other defect on the same side of the rabbit mandibles, considering that the embryonic roots would be the same. In this manner, the need for an additional surgical site (for graft harvesting) was also eliminated.
In a study by Aloise et al.99 Aloise AC, Pelegrine AA, Zimmermann A, Oliveira RME, Ferreiraet LM. Repair of critical-size bone defects using bone marrow stem cells or autogenous bone with or without collagen membrane: a histomorphometric study in rabbit calvaria. Int J Oral Maxillofac Implants. 2015;30(1):208-15. doi: 10.11607/jomi.4010.
https://doi.org/10.11607/jomi.4010...
which was conducted on the rabbit calvarium, defects on one side were randomly covered with a membrane after grafting, while those on the other side were left uncovered. Histological analysis at the end of 8 weeks after surgery indicated better ossification when the membrane was used99 Aloise AC, Pelegrine AA, Zimmermann A, Oliveira RME, Ferreiraet LM. Repair of critical-size bone defects using bone marrow stem cells or autogenous bone with or without collagen membrane: a histomorphometric study in rabbit calvaria. Int J Oral Maxillofac Implants. 2015;30(1):208-15. doi: 10.11607/jomi.4010.
https://doi.org/10.11607/jomi.4010...
. Of the several barrier materials used for guided bone regeneration, the most common are polytetrafluoroethylene), expanded polytetrafluoroethylene, collagen, freeze-dried dura mater, polyglactin 910, polylactic acid, polyglycolic acid, polyorthoester, polyurethane, polyhydroxybutyrate, calcium sulfate, microtitanium mesh, and titanium foils1010 Hammerle CH, Jung RE. Bone augmentation by means of barrier membranes. Periodontol 2000. 2003;33:36-53. PMID: 12950840..
In the present study, we used a collagen membrane as a carrier for the alendronate solution. In the field of ophthalmology, collagen is used in membrane form as a carrier for drugs, in sponge form for managing wounds and burns, in gel form combined with liposomes for continuous drug delivery, in nanoparticle form for gene transfer, and in simple matrix form for cell culture systems1111 Lee CH, Singla A, Lee Y. Biomedical applications of collagen. Int J Pharm. 2001;221(1-2):1-22. PMID: 11397563.. Friess investigated controlled drug release systems including collagen and reported ophthalmological barriers and sponges as the most effective for delivering antibiotics. The success of collagen in the local delivery of drugs has been attributed to its high biocompatibility, low antigenicity, ability to improve cellular penetration, processability in liquids, and ability to dissociate into physiologically tolerated components1212 Friess W. Collagen--biomaterial for drug delivery. Eur J Pharm Biopharm. 1998;45(2):113-36. PMID: 9704909.. The effects of collagen on osteoblasts was evaluated by Takayama and Mizumachi for a collagen membrane loaded on lactoferrin, which is an osteogenic growth factor; the authors concluded that such lactoferrin-loaded collagen membranes accelerated osteogenic differentiation in MG63 cells by increasing osteocalcin production and alkaline phosphatase activity1313 Takayama Y, Mizumachi K. Effect of lactoferrin-embedded collagen membrane on osteogenic differentiation of human osteoblast-like cells. J Biosci Bioeng. 2009;107(2):191-5. doi: 10.1016/j.jbiosc.2008.09.018.
https://doi.org/10.1016/j.jbiosc.2008.09...
. In the present study, we used collagen membranes because of their high biocompatibility and the ability to increase the local concentration of alendronate in the defect.
With the aim of accelerating the ossification of bone grafts used in oral surgery, studies have investigated the bone tissue interactions of several chemical and biological agents ranging from statins to stem cells and from bone morphogenetic proteins to bisphosphonates, administered locally or systemically. Oral bisphosphonates are used at low doses for the treatment of osteoporosis, whereas parenteral bisphosphonates are used at high doses for the treatment of multiple myeloma and prevention of bone metastasis in various types of cancer44 Marx RE, Cillo JE, Ulloa JJ. Oral bisphosphonate-induced osteonecrosis: risk factors, prediction of risk using serum CTX testing, prevention, and treatment. J Oral Maxillofac Surg. 2007;65(12):2397-410. doi: 10.1016/j.joms.2007.08.003.
https://doi.org/10.1016/j.joms.2007.08.0...
,1414 Ruggiero SL, Mehrotra B, Rosenberg TJ, Engroff SL. Osteonecrosis of the jaws associated with the use of bisphosphonates: a review of 63 cases. J Oral Maxillofac Surg. 2004;62(5):527-34. PMID: 15122554.. Although positive results with regard to the treatment of cancer have been observed, the long-term use of bisphosphonates is considered to have negative effects on bone turnover because osteoclastic activity is substantially suppressed, leading to decreased osteoblastic activity1515 Zahrowski JJ. Bisphosphonate treatment: an orthodontic concern calling for a proactive approach. Am J Orthod Dentofacial Orthop. 2007;131(3):311-20. doi: 0.1016/j.ajodo.2006.09.035.. Alendronate has high potency in the physiological context but relatively low potency compared to other bisphosphonates, which is why alendronate has become a popular bisphosphonate derivative in recent years. Therefore, we used alendronate in the present study.
Recently, numerous studies have focused on the local administration of bisphosphonates to eliminate the complications associated with systemic administration of compounds with such high potency. These studies have shown that concentrations that affect the relevant area after systemic administration can be achieved with local administration1616 Srisubut S, Teerakapong A, Vattraphodes T, Taweechaisupaponget S. Effect of local delivery of alendronate on bone formation in bioactive glass grafting in rats. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2007;104(4):e11-6. doi: 0.1016/j.tripleo.2007.04.022.,1717 Agholme F, Aspenberg P. Experimental results of combining bisphosphonates with allograft in a rat model. J Bone Joint Surg Br. 2009;91(5):670-5. doi: 10.1302/0301-620X.91B5.21867.
https://doi.org/10.1302/0301-620X.91B5.2...
. Moreover, Küçük et al.1818 Küçük D, Ay S, Kara MI, Avunduk MC, Gümus C. Comparison of local and systemic alendronate on distraction osteogenesis. Int J Oral Maxillofac Surg. 2011;40(12):1395-400. doi: 10.1016/j.ijom.2011.08.004.
https://doi.org/10.1016/j.ijom.2011.08.0...
and Toker et al.1919 Toker H, Ozdemir H, Ozer H, Eren K. A comparative evaluation of the systemic and local alendronate treatment in synthetic bone graft: a histologic and histomorphometric study in a rat calvarial defect model. Oral Surg Oral Med Oral Pathol Oral Radiol. 2012;114(5 Suppl):S146-52. doi: 10.1016/j.oooo.2011.09.027.
https://doi.org/10.1016/j.oooo.2011.09.0...
found no significant difference between systemic and local administration of alendronate sodium in the rabbit model of distraction and rat model of calvarium defect, respectively. Because bisphosphonates inhibit osteoclastic activity, their local use is preferable in cases where bone healing is important. The fact that systemic side effects are absent or minimal with local administration of bisphosphonates has increased the popularity of this approach. With this aim in mind, the present study used locally administered alendronate, which was considered to have a positive effect on local bone healing while minimizing the risk for systemic side effects. To further promote healing, we also used collagen membranes, with or without autogenous bone particles.
In their study on tibial implants, Jakobsen et al.2020 Jakobsen T, Baas J, Kold S, Bechtold JE, Elmengaard B, Søballe K. Local bisphosphonate treatment increases fixation of hydroxyapatite-coated implants inserted with bone compaction. J Orthop Res. 2009;27(2):189-94. doi: 10.1002/jor.20745.
https://doi.org/10.1002/jor.20745...
,2121 Jakobsen T, Kold S, Bechtold J, Søballe K. Local alendronate increases fixation of implants inserted with bone compaction: 12-week canine study. J Orthop Res. 2007;25(4):432-41. doi: 10.1002/jor.20276.
https://doi.org/10.1002/jor.20276...
used alendronate solution with local administration, and determined the optimal concentration to be 2 mg/mL. For the repair of skull defects in rats, Toker et al.1919 Toker H, Ozdemir H, Ozer H, Eren K. A comparative evaluation of the systemic and local alendronate treatment in synthetic bone graft: a histologic and histomorphometric study in a rat calvarial defect model. Oral Surg Oral Med Oral Pathol Oral Radiol. 2012;114(5 Suppl):S146-52. doi: 10.1016/j.oooo.2011.09.027.
https://doi.org/10.1016/j.oooo.2011.09.0...
used alendronate solution at the concentration of 1 mg/mL. Mathijssen et al.2222 Mathijssen NM, Hannink G, Pilot P, Schreurs BW, Bloem RM, Buma P. Impregnation of bone chips with alendronate and cefazolin, combined with demineralized bone matrix: a bone chamber study in goats. BMC Musculoskelet Disord. 2012;13:44. doi: 10.1186/1471-2474-13-44.
https://doi.org/10.1186/1471-2474-13-44...
investigated the effect of the concentration of locally administered alendronate solution in their study on goats, and determined the optimal concentration as 1 mg/mL. The achievement of lower success rates with higher concentrations was attributed to the toxicity of bisphosphonates at high doses and to exaggerated bisphosphonate-induced osteoconduction, which failed to allow sufficient space for the formation of new bone tissue2222 Mathijssen NM, Hannink G, Pilot P, Schreurs BW, Bloem RM, Buma P. Impregnation of bone chips with alendronate and cefazolin, combined with demineralized bone matrix: a bone chamber study in goats. BMC Musculoskelet Disord. 2012;13:44. doi: 10.1186/1471-2474-13-44.
https://doi.org/10.1186/1471-2474-13-44...
. Therefore, in the present study, we prepared the alendronate solution by adding 1 mg alendronate sodium per 1 mL of saline.
Tanaka et al.2323 Tanaka T, Saito M, Chazono M, Kumagae Y, Kikuchi T, Kitasato S, Marumo K. Effects of alendronate on bone formation and osteoclastic resorption after implantation of beta-tricalcium phosphate. J Biomed Mater Res A. 2010;93(2):469-74. doi: 10.1002/jbm.a.32560.
https://doi.org/10.1002/jbm.a.32560...
combined alendronate with β-tricalcium phosphate to promote the antiresorptive effects of alendronate, while Zhao et al.2424 Zhao J, Tang H, Wang J, Li G. Local treatment of osteoporosis with alendronate-loaded calcium phosphate cement. Chin Med J (Engl). 2014;127(22):3906-14. PMID: 25421189. combined alendronate with calcium phosphate cement for local application. Hur et al.2525 Hur W, Park M, Lee JY, Kim MH, Lee SH, Park CG, Kim SN, Min HS, Min HJ, Chai JH, Lee SJ, Kim S, Choi TH, Choy YB. Bioabsorbable bone plates enabled with local, sustained delivery of alendronate for bone regeneration. J Control Release. 2016;222:97-106. PMID: 26682503. found that alendronate sodium loaded on resorbable bone plaques provided enhanced bone regeneration. In the in vitro study by Boanini et al.2626 Boanini E, Torricelli P, Gazzano M, Fini M, Bigi A. The effect of alendronate doped calcium phosphates on bone cells activity. Bone. 2012;51(5):944-52. doi: 10.1016/j.bone.2012.07.020.
https://doi.org/10.1016/j.bone.2012.07.0...
, the activity of bone cells was found to be elevated in the presence of alendronate loaded on octacalcium phosphate. In the study by Bobyn et al.2727 Bobyn JD, Thompson R, Lim L, Pura JA, Bobyn K, Tanzer M. Local alendronic acid elution increases net periimplant bone formation: a micro-CT analysis. Clin Orthop Relat Res. 2014;472(2):687-94. doi: 10.1007/s11999-013-3120-6.
https://doi.org/10.1007/s11999-013-3120-...
, locally administered alendronate sodium with porous titanium intramedullary implants placed in the femur bone of dogs was found to induce a significant increase in peri-implant bone mass, as detected radiologically.
Although alendronate is often combined with many different graft materials, its administration with autogenous grafts, which are considered the gold standard for bone defect repair, is more valuable because it slows graft resorption and prolongs the osteoconductive effects. While administration of alendronate with synthetic grafts is useful because it increases osteoblastic activity secondary to osteoclastic inhibition, its administration with autogenous grafts is necessary to slow down the resorption of such grafts. Johansson et al.2828 Johansson B, Grepe A, Wannfors K, Hirsch JM. A clinical study of changes in the volume of bone grafts in the atrophic maxilla. Dentomaxillofac Radiol. 2001;30(3):157-61. doi: 10.1038/sj/dmfr/4600601.
https://doi.org/10.1038/sj/dmfr/4600601...
found that patients who underwent maxillary sinus augmentation with autogenous grafts had 49.5% graft resorption at 6 months after surgery. In the present study, alendronate sodium was combined with autogenous bone grafting in an effort to improve graft retention in the defect (i.e., ensure that the autogenous bone would remain in the defect for a longer period without resorption). Histomorphometric analysis at 8 weeks after surgery showed that the percentage of newly formed bone and residual graft area were 61.43% and 14.21%, respectively, in defects that received only autogenous bone grafts (AG group), compared to 71.3% and 19.43%, respectively, in defects that received both alendronate irrigation and autogenous bone grafting (AL+AG group). Interestingly, at 4 weeks after surgery, the histomormphometric outcomes (including residual graft area, new bone area, and percentage of new bone) and radiologic outcomes (including the percentage of total bone and bone volume) were less favorable in the AL+AG group than in the AG group. The discrepancy between the findings at 4 and 8 weeks can be explained by considering the pH of alendronate solution (4.0-5.0), which takes time to be buffered by body fluids. This delay can be considered a disadvantage for living cells in autogenous grafts. However, at 8 weeks, this disadvantage had disappeared, and the AL+AG group was found to be superior to the AG group regarding all outcomes analyzed. The positive effect of alendronate can also be distinguished when examining the outcomes of groups without autogenous grafts. Specifically, the AL group (alendronate irrigation only) was found to be superior to the EC group (no treatment) in terms of both histomorphometric and radiological outcomes, at both 4 weeks and at 8 weeks after surgery. Also in the EC and AL group, new bone area and percentage of new bone values was higher than the AG and AL+AG groups, which used autogenous graft at 4 weeks specimens. It may make sense to consider the reason for this as the volume of the empty defect area. Because in the grafted groups, the defect area was filled with grafts at first, while in non-grafted defects, there was only a sufficient amount of space available for the new bone formation to be observed. At the same time, this data is a precious data about the fact that the remaining vacancies,do not exceed the critical-size, will become self-bony.
In this study, we did not use critical-size defects because we focused on evaluating the effect of alendronate on the rate and nature of early healing, whereas critical defects would require longer-term observation. At the same time, two defects in each animal were not treated with autogenous graft, so that the direct effect of alendronate sodium on the tissues could be examined. In particular, we wished to determine whether the absence of the graft would affect the dissolution of alendronate sodium in the tissue fluids and subsequent effect on osteoclastic activity. Our findings demonstrated the efficacy of locally administered alendronate, alone and in conjunction with autogenous bone grafts. At 4 weeks after surgery, the mean new bone formation rate was 26.25% in defects that received alendronate irrigation only (AL group), and 27.73% in defects that received alendronate irrigation with autogenous bone grafts (AL+AG group). These values were 44.17% and 48.03%, respectively, at 8 weeks after surgery. Our results demonstrate that, rather than alendronate administration alone, the combination with autogenous bone grafting will provide maximum benefits in terms of osteoinduction, osteoconduction, and osteogenesis.
The most reliable methods for the measurement of newly formed bone and soft tissue are two-dimensional histomorphometry and three-dimensional micro-CT. However, compared to micro-CT analysis, histomorphometric analysis requires larger cross-sections. Therefore, studies conducted using micro-CT analysis have been reported to give more accurate and detailed results2929 Deluiz D, Oliveira LS, Pires FR, Tinocoet EMB. Time-dependent changes in fresh-frozen bone block grafts: tomographic, histologic, and histomorphometric findings. Clin Implant Dent Relat Res. 2015;17(2):296-306. doi: 10.1111/cid.12108.
https://doi.org/10.1111/cid.12108...
. In the present study, the findings of micro-CT correlated with those of histomorphometry.
Because no study has evaluated the outcome of local administration of alendronate sodium in the human bone, the effects of alendronate on bone healing in humans remain unclear. However, the present study and similar studies are important because the outcomes may be translated in terms of applicability to humans. In the present study, a solution of alendronate sodium and sterile saline was used. To confirm the effectiveness and safety of alendronate sodium, as well as to determine the optimal conditions regarding its storage and use, further study should focus on investigating the safety and effectiveness of alendronate in different solutions, in mixtures with buffering solutions that attenuate the harmful effect on living tissues, and in different delivery forms (solid, liquid, gel, etc.).
Conclusions
The local administration of alendronate sodium in conjunction with autogenous bone grafts improves the osteoconductive properties of the graft, allows the graft to remain in the defect for a longer period (i.e., improves graft retention), and promotes better ossification in the bone defect, ultimately accelerating the formation and enhancing the quality of new bone. However, further studies on the local use of alendronate with synthetic grafts and xenografts are required, as well as studies determining the optimal concentration of the solution for local use.
References
-
1Giannoudis P, Psarakis S, and Kontakis G. Can we accelerate fracture healing? A critical analysis of the literature. Injury. 2007;38(1):S81-9. doi: 10.1016/j.injury.2007.02.013.
» https://doi.org/10.1016/j.injury.2007.02.013 -
2Alfaro FH. Bone grafting in oral implantology: techniques and clinical applications. Spain: Quintessence Books; 2006.
-
3Ashammakhi N, Ferretti P. Topics in tissue engineering. v.1. Finland: University of Oulu; 2003.
-
4Marx RE, Cillo JE, Ulloa JJ. Oral bisphosphonate-induced osteonecrosis: risk factors, prediction of risk using serum CTX testing, prevention, and treatment. J Oral Maxillofac Surg. 2007;65(12):2397-410. doi: 10.1016/j.joms.2007.08.003.
» https://doi.org/10.1016/j.joms.2007.08.003 -
5Watts NB. Treatment of osteoporosis with bisphosphonates. Rheum Dis Clin North Am. 2001;27(1):197-214. doi: 10.1016/S0889-8529(05)70014-1.
» https://doi.org/10.1016/S0889-8529(05)70014-1 -
6Fleisch H. Bisphosphonates in bone disease: from the laboratory to the patient. USA: Academic Press; 2000.
-
7Binderman I, Adut M, Yaffe A. Effectiveness of local delivery of alendronate in reducing alveolar bone loss following periodontal surgery in rats. J Periodontol. 2000;71(8):1236-40. doi: 10.1902/jop.2000.71.8.1236.
» https://doi.org/10.1902/jop.2000.71.8.1236 -
8Leucht P, Kim JB, Amasha R, James AW, Girod S, Helms JA. Embryonic origin and Hox status determine progenitor cell fate during adult bone regeneration. Development. 2008;135(17):2845-54. doi: 10.1242/dev.023788.
» https://doi.org/10.1242/dev.023788 -
9Aloise AC, Pelegrine AA, Zimmermann A, Oliveira RME, Ferreiraet LM. Repair of critical-size bone defects using bone marrow stem cells or autogenous bone with or without collagen membrane: a histomorphometric study in rabbit calvaria. Int J Oral Maxillofac Implants. 2015;30(1):208-15. doi: 10.11607/jomi.4010.
» https://doi.org/10.11607/jomi.4010 -
10Hammerle CH, Jung RE. Bone augmentation by means of barrier membranes. Periodontol 2000. 2003;33:36-53. PMID: 12950840.
-
11Lee CH, Singla A, Lee Y. Biomedical applications of collagen. Int J Pharm. 2001;221(1-2):1-22. PMID: 11397563.
-
12Friess W. Collagen--biomaterial for drug delivery. Eur J Pharm Biopharm. 1998;45(2):113-36. PMID: 9704909.
-
13Takayama Y, Mizumachi K. Effect of lactoferrin-embedded collagen membrane on osteogenic differentiation of human osteoblast-like cells. J Biosci Bioeng. 2009;107(2):191-5. doi: 10.1016/j.jbiosc.2008.09.018.
» https://doi.org/10.1016/j.jbiosc.2008.09.018 -
14Ruggiero SL, Mehrotra B, Rosenberg TJ, Engroff SL. Osteonecrosis of the jaws associated with the use of bisphosphonates: a review of 63 cases. J Oral Maxillofac Surg. 2004;62(5):527-34. PMID: 15122554.
-
15Zahrowski JJ. Bisphosphonate treatment: an orthodontic concern calling for a proactive approach. Am J Orthod Dentofacial Orthop. 2007;131(3):311-20. doi: 0.1016/j.ajodo.2006.09.035.
-
16Srisubut S, Teerakapong A, Vattraphodes T, Taweechaisupaponget S. Effect of local delivery of alendronate on bone formation in bioactive glass grafting in rats. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2007;104(4):e11-6. doi: 0.1016/j.tripleo.2007.04.022.
-
17Agholme F, Aspenberg P. Experimental results of combining bisphosphonates with allograft in a rat model. J Bone Joint Surg Br. 2009;91(5):670-5. doi: 10.1302/0301-620X.91B5.21867.
» https://doi.org/10.1302/0301-620X.91B5.21867 -
18Küçük D, Ay S, Kara MI, Avunduk MC, Gümus C. Comparison of local and systemic alendronate on distraction osteogenesis. Int J Oral Maxillofac Surg. 2011;40(12):1395-400. doi: 10.1016/j.ijom.2011.08.004.
» https://doi.org/10.1016/j.ijom.2011.08.004 -
19Toker H, Ozdemir H, Ozer H, Eren K. A comparative evaluation of the systemic and local alendronate treatment in synthetic bone graft: a histologic and histomorphometric study in a rat calvarial defect model. Oral Surg Oral Med Oral Pathol Oral Radiol. 2012;114(5 Suppl):S146-52. doi: 10.1016/j.oooo.2011.09.027.
» https://doi.org/10.1016/j.oooo.2011.09.027 -
20Jakobsen T, Baas J, Kold S, Bechtold JE, Elmengaard B, Søballe K. Local bisphosphonate treatment increases fixation of hydroxyapatite-coated implants inserted with bone compaction. J Orthop Res. 2009;27(2):189-94. doi: 10.1002/jor.20745.
» https://doi.org/10.1002/jor.20745 -
21Jakobsen T, Kold S, Bechtold J, Søballe K. Local alendronate increases fixation of implants inserted with bone compaction: 12-week canine study. J Orthop Res. 2007;25(4):432-41. doi: 10.1002/jor.20276.
» https://doi.org/10.1002/jor.20276 -
22Mathijssen NM, Hannink G, Pilot P, Schreurs BW, Bloem RM, Buma P. Impregnation of bone chips with alendronate and cefazolin, combined with demineralized bone matrix: a bone chamber study in goats. BMC Musculoskelet Disord. 2012;13:44. doi: 10.1186/1471-2474-13-44.
» https://doi.org/10.1186/1471-2474-13-44 -
23Tanaka T, Saito M, Chazono M, Kumagae Y, Kikuchi T, Kitasato S, Marumo K. Effects of alendronate on bone formation and osteoclastic resorption after implantation of beta-tricalcium phosphate. J Biomed Mater Res A. 2010;93(2):469-74. doi: 10.1002/jbm.a.32560.
» https://doi.org/10.1002/jbm.a.32560 -
24Zhao J, Tang H, Wang J, Li G. Local treatment of osteoporosis with alendronate-loaded calcium phosphate cement. Chin Med J (Engl). 2014;127(22):3906-14. PMID: 25421189.
-
25Hur W, Park M, Lee JY, Kim MH, Lee SH, Park CG, Kim SN, Min HS, Min HJ, Chai JH, Lee SJ, Kim S, Choi TH, Choy YB. Bioabsorbable bone plates enabled with local, sustained delivery of alendronate for bone regeneration. J Control Release. 2016;222:97-106. PMID: 26682503.
-
26Boanini E, Torricelli P, Gazzano M, Fini M, Bigi A. The effect of alendronate doped calcium phosphates on bone cells activity. Bone. 2012;51(5):944-52. doi: 10.1016/j.bone.2012.07.020.
» https://doi.org/10.1016/j.bone.2012.07.020 -
27Bobyn JD, Thompson R, Lim L, Pura JA, Bobyn K, Tanzer M. Local alendronic acid elution increases net periimplant bone formation: a micro-CT analysis. Clin Orthop Relat Res. 2014;472(2):687-94. doi: 10.1007/s11999-013-3120-6.
» https://doi.org/10.1007/s11999-013-3120-6 -
28Johansson B, Grepe A, Wannfors K, Hirsch JM. A clinical study of changes in the volume of bone grafts in the atrophic maxilla. Dentomaxillofac Radiol. 2001;30(3):157-61. doi: 10.1038/sj/dmfr/4600601.
» https://doi.org/10.1038/sj/dmfr/4600601 -
29Deluiz D, Oliveira LS, Pires FR, Tinocoet EMB. Time-dependent changes in fresh-frozen bone block grafts: tomographic, histologic, and histomorphometric findings. Clin Implant Dent Relat Res. 2015;17(2):296-306. doi: 10.1111/cid.12108.
» https://doi.org/10.1111/cid.12108
-
Financial source:
none
-
1
Research performed at Hacettepe University, Faculty of Medicine, Experimental Animal Research Laboratory; Gazi University, Faculty of Dentistry, Department of Oral Pathology, and Hacettepe University, Faculty of Medicine, Department of Anatomy, Turkey.
-
Erratum
Manuscript: Effects of local alendronate administration on bone defect healing. Histomorphometric and radiological evaluation in a rabbit model1Publication: Acta Cir. Bras. vol.32 no.9 São Paulo Sept. 2017http://dx.doi.org/10.1590/s0102-865020170090000010Where was it read:Taha Özer IAlper Akta IIEmre Bari IIIHakan Hamdi Çelik IVAlper Vatansever VRead:Taha Özer IAlper Aktas IIEmre Baris IIIHakan Hamdi Çelik IVAlper Vatansever V
Publication Dates
-
Publication in this collection
Sept 2017
History
-
Received
12 May 2017 -
Reviewed
14 July 2017 -
Accepted
18 Aug 2017