Abstract
Purpose:
To investigate the potential protective effects of erdosteine against the harmful effects of ischemia-reperfusion injury on the liver in an experimental rat model.
Methods:
Forty rats were divided into 4 groups. In the sham group, only the hepatic pedicle was mobilized. No other manipulation or treatment was performed. In the other groups, ischemia was achieved by clamping the hepatic pedicle for 60 min. After that, 90 min reperfusion was provided. In the control group, no treatment was given. In the perioperative treatment group, 100 mg/kg erdosteine was administered 2 hours before ischemia induction. In the preoperative treatment group, 100 mg/kg/day erdosteine was administered daily for ten days before the operation. At the end of the procedures, blood and liver samples were obtained for biochemical and histopathological assessment.
Results:
Treatment with erdosteine ameliorated the histopathological abnormalities when compared with the control group. Furthermore, this treatment significantly decreased the serum liver function test values. It was also found that erdosteine ameliorated the oxidative stress parameters in both the perioperative and preoperative treatment groups.
Conclusion:
The current study is the first to have shown the favorable effects of erdosteine on the harmful effects of experimental hepatic ischemia-reperfusion injury.
Key words:
Ischemia; Reperfusion; Oxidative Stress; Pathology; Liver; Rats
Introduction
Ischemia-reperfusion injury (IRI) is an inevitable situation in a wide range of surgical settings and has an important impact on the clinical outcome. It occurs when the blood supply is interrupted for a prolonged period of time and is then restored. During the ischemic period, tissues are deprived of oxygen and nutrients, leading to cellular metabolism breakdown. After blood flow restoration (reperfusion), more severe damage is provoked in the ischemic tissue due to the production of reactive oxygen species (ROS), progressing to extended inflammation and necrotic and apoptotic events which finally result in organ injury11 Pantazi E, Bejaoui M, Folch-Puy E, Adam R, Roselló-Catafau J. Advances in treatment strategies for ischemia reperfusion injury. Expert Opin Pharmacother. 2016;17:169-79. doi: 10.1517/14656566.2016.1115015.
https://doi.org/10.1517/14656566.2016.11...
. Hepatic IRI occurs via a complex pathological network that features a combination of factors, including impairment of sinusoidal endothelial cells (SECs), activation of Kupffer cells (KCs), disturbance of microcirculation, oxidative stress, inflammation, activation of complementary factors, accumulation of leukocytes, apoptosis, and necrosis. From the point of clinical application, various experimental studies have focused on developing pharmacological strategies with the aim of disrupting the pathways of IRI. Unfortunately, promising agents and strategies against IRI have not yet become part of the clinical routine22 Yamanaka K, Houben P, Bruns H, Schultze D, Hatano E, Schemmer P. A systematic review of pharmacological treatment options used to reduce ischemia reperfusion injury in rat liver transplantation. PLoS One. 2015;10:e0122214. doi: 10.1371/journal.pone. 0122214.
https://doi.org/10.1371/journal.pone...
.
Erdosteine [N-(carboxymethylthioacetyl)-homocysteine thiolactone] was introduced as a mucolytic agent for chronic pulmonary diseases more than 10 years ago. Erdosteine contains two blocked sulphydryl groups, one of which, after hepatic metabolization and opening of the thiolactone ring, becomes available for pharmacological activity. The free sulphydryl group breaks the disulphide bridges of the high-molecular-weight mucus glycoproteins, resulting in reduced sputum physical properties. The favorable effects of erdosteine might, however, extend much further than the modulation of mucus viscosity and improved tracheobronchial clearance; the blocked sulphydryl group, after hepatic metabolization, also becomes available for free radical scavenging and antioxidant activity. This free radical scavenging activity could be widely exploited in COPD and other lung diseases where oxidative stress plays an important role in the pathogenesis of the disease. Recently, several spontaneous studies on erdosteine have appeared in the international literature. Experimental evidence in animals supports the protective effect of erdosteine in various types of tissue injuries mediated by products of oxidative stress33 Moretti M, Marchioni CF. An overview of erdosteine antioxidant activity in experimental research. Pharmacol Res. 2007;55:249-54. doi: 10.1016/j.phrs.2006. 12. 006.
https://doi.org/10.1016/j.phrs.2006...
. Erdosteine also has an anti-inflammatory effect which is mediated through the inhibition of LPS-induced NF-kB activation44 Park JS, Park MY, Cho YJ, Lee JH, Yoo CG, Lee CT, Lee SM. Anti-inflammatory Effect of erdosteine in lipopolysaccharide-stimulated RAW 264.7 cells. Inflammation. 2016;39:1573-81. doi: 10.1007/s10753-016-0393-4.
https://doi.org/10.1007/s10753-016-0393-...
.
The hepatoprotective effects of erdosteine against acetaminophen-induced liver damage, cyclosporine-A-induced hepatotoxicity, experimental biliary obstruction, cisplatin-induced hepatic oxidant injury, doxorubicin-induced hepatotoxicity, and vancomycin-induced oxidative stress in the rat liver have been shown in previous studies55 Saritas A, Kandis H, Baltaci D, Yildirim U, Kaya H, Karakus A, Colakoglu S, Memisogullari R, Kara IH. N-Acetyl cysteine and erdosteine treatment in acetaminophen-induced liver damage. Toxicol Ind Health. 2014;30:670-8. doi: 10. 1177/0748233712463780.
https://doi.org/10...
6 Erarslan E, Ekiz F, Uz B, Koca C, Turkcu UO, Bayrak R, Delibasi T. Effects of erdosteine on cyclosporine-A-induced hepatotoxicity in rats. Drug Chem Toxicol. 2011;34:32-7. doi: 10.3109/01480545.2010.483519.
https://doi.org/10.3109/01480545.2010.48...
7 Kuvandik G, Duru M, Nacar A, Yonden Z, Helvaci R, Koc A, Kozlu T, Kaya H, Sogüt S. Effects of erdosteine on acetaminophen-induced hepatotoxicity in rats. Toxicol Pathol. 2008;36:714-9. doi: 10.1177/0192623308320800.
https://doi.org/10.1177/0192623308320800...
8 Sener G, Sehirli AO, Toklu HZ, Yuksel M, Ercan F, Gedik N. Erdosteine treatment attenuates oxidative stress and fibrosis in experimental biliary obstruction. Pediatr Surg Int. 2007;23:233-41. doi: 10.1007/s00383-006-1872-8.
https://doi.org/10.1007/s00383-006-1872-...
9 Koc A, Duru M, Ciralik H, Akcan R, Sogut S. Protective agent, erdosteine, against cisplatin-induced hepatic oxidant injury in rats. Mol Cell Biochem. 2005;278:79-84. doi: 10.1007/s11010-005-6630-z.
https://doi.org/10.1007/s11010-005-6630-...
10 Yagmurca M, Bas O, Mollaoglu H, Sahin O, Nacar A, Karaman O, Songur A. Protective effects of erdosteine on doxorubicin-induced hepatotoxicity in rats. Arch Med Res. 2007;38:380-5. doi: 10.1016/j.arcmed.2007.01.007.
https://doi.org/10.1016/j.arcmed.2007.01...
-1111 Sahin M, Cam H, Olgar S, Tunc SE, Arslan C, Uz E, Yilmaz HR. Protective role of erdosteine on vancomycin-induced oxidative stress in rat liver. Mol Cell Biochem. 2006;291:155-60. doi: 10.1007/s11010-006-9209-4.
https://doi.org/10.1007/s11010-006-9209-...
.
Erdosteine has beneficial effects on ischemia-reperfusion injury in various organs including lung, brain, kidney, intestines, ovaries, and spinal cord1212 Kurtoglu T, Sacar M, Inan BK, Duver MH, Guler A, Ucak A, Us MH, Yilmaz AT. Erdosteine ameliorates lung injury induced by transient aortic occlusion in rats. Cardiovasc J Afr. 2007;18:367-70. PMID: 18092111.
13 Ozerol E, Bilgic S, Iraz M, Cigli A, Ilhan A, Akyol O. The protective effect of erdosteine on short-term global brain ischemia/reperfusion injury in rats. Prog Neuropsychopharmacol Biol Psychiatry. 2009 33:20-4. doi: 10.1016/j.pnpbp. 2008. 09.024.
https://doi.org/10.1016/j.pnpbp. 2008. 0...
14 Gurel A, Armutcu F, Cihan A, Numanoglu KV, Unalacak M. Erdosteine improves oxidative damage in a rat model of renal ischemia-reperfusion injury. Eur Surg Res. 2004;36:206-9. doi: 10.1159/000078854.
https://doi.org/10.1159/000078854...
15 Tunc T, Uysal B, Atabek C, Kesik V, Caliskan B, Oztas E, Ersoz N, Oter S, Guven A. Erdosteine and ebselen as useful agents in intestinal ischemia/reperfusion injury. J Surg Res. 2009;155: 210-6. doi: 10.1016/j.jss.2008.06.017.
https://doi.org/10.1016/j.jss.2008.06.01...
16 Ugurel V, Cicek AC, Cemek M, Demirtas S, Kocaman AT, Karaca T. Antioxidant and antiapoptotic effects of erdosteine in a rat model of ovarian ischemia-reperfusion injury. Iran J Basic Med Sci. 2017;20:53-8. doi: 10.22038/ijbms.2017.8093.
https://doi.org/10.22038/ijbms.2017.8093...
-1717 Ege E, Ilhan A, Gurel A, Akyol O, Ozen S. Erdosteine ameliorates neurological outcome and oxidative stress due to ischemia/reperfusion injury in rabbit spinal cord. Eur J Vasc Endovasc Surg. 2004;28:379-86. doi: 10.1016/j.ejvs.2004.06.004.
https://doi.org/10.1016/j.ejvs.2004.06.0...
. To the best of our knowledge, there is no evidences about the effect of erdosteine on hepatic ischemia-reperfusion in the current literature. The aim of the current experimental study was to investigate the potential effects of erdosteine on hepatic ischemia-reperfusion injury in a rat model.
Methods
This study was conducted in the Husnu Sakal Experimental Practice Center, Ankara, Turkey. The procedures in this experimental study were performed in accordance with the National Guidelines for the Use and Care of Laboratory Animals and the Animal Ethics Committee of Ankara Education and Research Hospital granted approval for the study (protocol number: 464).
The study was carried out on 40 Wistar-Albino female rats, each weighing 250±25 g. The rats were allowed to adapt to laboratory conditions for 1 week before experimental use. The animals were housed under standard laboratory conditions at a constant temperature (21°±2°C) with a 12-hour light-dark cycle. The animals were fed with standard laboratory chow and water ad libitum. Twelve hours before anesthesia, the animals were deprived of food, but continued to have free access to water until 2 hrs before anesthesia. All the animals were anesthetized with an intramuscular injection of 50 mg/kg ketamine hydrochloride (Ketalar®, Parke-Davis, Istanbul) and 5 mg/kg xylazine (Rompun®, Bayer, Istanbul).
The rats were randomly divided into 4 groups of 10: sham group, control group (ischemia-reperfusion) and treatment groups (preoperative group and perioperative group). In the sham group (Group 1), a midline incision was made, and the hepatic pedicle was mobilized. No other manipulation or treatment was performed in this group. In the other groups, ischemia was achieved by clamping the hepatic pedicle with a vascular clamp for 60 min. After the ischemic period, the clamp was opened and 90 min reperfusion of the liver was provided. In the control group (Group 2), no treatment was given. In the perioperative treatment group (Group 3), 100 mg/kg erdosteine was administered by orogastric tube to the rats 2 hours before ischemia induction. In the preoperative treatment group (Group 4), 100 mg/kg/day erdosteine (Erdostin®, Sandoz, Istanbul, Turkey) was administered by orogastric tube to the rats daily for ten days before the operation. At the end of the procedures, blood and liver samples were obtained for biochemical and histopathological assessment. All rats were euthanized after the procedure.
Serum alanine aminotransferase (ALT), aspartate aminotransferase (AST), gamma glutamyl transpeptidase (GGT) and alkaline phosphatase (ALP) measurements were taken with a clinical chemistry analyzer (Beckman Coulter 5800) using Beckman Coulter commercial kits.
The evaluation of oxidative stress parameters was performed in the Biochemistry Department of Ankara Education and Research Hospital. Tissues were stored at -80°C until the assays. Tissue malondialdehyde (MDA), total sulphydryl groups (total-SH), tissue fluorescence oxidation products (TFOP) and total oxidative stress (TOS) levels were measured. MDA levels were calculated using the fluorometric method, as described by Wasowicz et al.1818 Wasowicz W, Nève J, Peretz A. Optimized steps in fluorometric determination of thiobarbituric acid-reactive substances in serum: importance of extraction pH and influence of sample preservation and storage. Clin Chem. 1993;39:2522-6. PMID: 8252725.. After the reaction of thiobarbituric acid (TBA) with MDA, the reaction product was extracted in butanol and was measured spectrofluorometrically at wavelengths of 525 nm for excitation and 547 nm for emission. A solution of 0-5 μmol/L 1,1’,3,3’ tetraethoxypropane was used as standard. For the measurement of tissue MDA levels, 50 μL of homogenate was added and introduced into 10 mL glass tubes containing 1 mL of distilled water. After the addition of 1 mL of the solution containing 29 mmol/L TBA in acetic acid and mixing, the samples were placed in a water bath and heated for 1 hr at 95°-100°C. The samples were then cooled, 25 μL of 5 mol/L hydrochloric acid (HCL) was added and the reaction mixture was extracted by agitation for 5 min with 3.5 mL n-butanol. After separation of the butanol phase by centrifugation at 1500x g for 10 min, the fluorescence of the butanol extract was measured with a fluorometer (HITACHI F-2500) at wave-lengths of 525 nm for excitation and 547 nm for emission. 0-5 μmol/L 1,1’,3,3’-tetraethoxypropane solutions were used as standard. MDA levels were presented as nmol/g wet tissue.
Total SH groups were measured spectrophotometrically using the Sedlak and Lindsay method1919 Sedlak J, Lindsay RH. Estimation of total, protein bound, and non-protein sulfhydryl groups in tissue with Ellman's reagent. Anal. Biochem. 1968;25:192-205. PMID: 4973948.. Aliquots of 250 μl of the supernatant fraction of the tissue homogenate were mixed in 5 mL test tubes with 750 μl of 0.2 M Tris buffer, pH 8.2, and 50 μl of 0.01 M 5,5′′-dithiobis (2 nitrobenzoic acid) (DTNB). With the addition of 3950 μl of absolute methanol, the mixture was brought to 5 mL. A reagent blank (without sample) and a sample blank (without DTNB) were prepared in a similar manner. The test tubes were stoppered with rubber caps, the color was allowed to develop for 15 min and the reaction mixtures were centrifuged at approximately 3,000g at room temperature for 15 min. The absorbance of supernatant fractions was read in a spectrophotometer at 412 nm1919 Sedlak J, Lindsay RH. Estimation of total, protein bound, and non-protein sulfhydryl groups in tissue with Ellman's reagent. Anal. Biochem. 1968;25:192-205. PMID: 4973948..
For TFOP measurements, tissue homogenates were extracted with ethanol-ether (3/1, v/v) and were measured with a spectrofluorometer at a wavelength of 360/430 (excitation/ emission wavelength)2020 Wu T, Willett WC, Rifai N, Rimm EB. Plasma fluorescent oxidation products as potential markers of oxidative stress for epidemiologic studies. Am J Epidemiol. 2007;166:552-60. PMID: 17615091..
Measurement of TOS was made using the calorimetric method based on the cumulative oxidation of the molecules in the sample from ferrous ion to ferric ion. The results were stated as μmol H2O2 Equivalent/ L2121 Verde V, Fogliano V, Ritieni A, Maiani G, Morisco F, Caporaso N. Use of N,N-dimethyl-p-phenylenediamine to evaluate the oxidative status of human plasma. Free Radic Res. 2002;36:869-73. PMID: 12420745..
The histopathological analyses were performed in the Pathology Department of Ankara Education and Research Hospital. For light microscopic analyses, the samples were obtained from the liver and fixed in 10% neutral buffered formalin solution for 2 days. The tissues were washed in running water and dehydrated with increasing concentrations of ethanol. After dehydration, the specimens were placed in xylene to obtain transparency and were then infiltrated with and embedded in paraffin. The embedded tissues were cut into 4-μm thick sections using a Leica RM 2125 RT microtome and stained with hematoxylin & eosin. Histopathological examinations were performed with a light microscope (Olympus BX51TF, Tokyo, Japan) by a pathologists blinded to the study design. Histopathological parameters including sinusoidal congestion, hepatocyte necrosis and liver cell vacuolization were evaluated semi-quantitatively according to the modified Suzuki scoring system from 0 to 42222 Suzuki S, Toledo-Peneyra LH, Rodriquez FJ, Cejalvo D. Neutrophil infiltration as an important factor in liver ischemia and reperfusion injury. Modulating effects of FK506 and cylosporine. Transplantation. 1993;55:1265-72. PMID: 7685932.. The scoring system of these parameters is given in Table 1.
Data analysis was performed using the Statistical Package for Social Sciences (SPSS) version 15.0 for Windows (SPSS Inc, Chicago, IL). All variables were normally distributed about the mean. Data were presented as mean±standard deviation (SD). Differences between the groups were evaluated using One-Way analysis of variance (ANOVA) or Kruskal-Wallis variance analysis, whichever was appropriate. When the p values from the variance analysis were statistically significant, the Tukey honestly significant difference (HSD) or Mann-Whitney U multiple comparison test was used to determine which group was different from the others. A value of p<0.05 was considered to be statistically significant.
Results
The mean levels of the liver function tests (AST, ALT, GGT and ALP) are summarized in Table 2. The differences between the sham and other groups were significant for all parameters (p<0.05). The highest liver function test values were found in the control group. The use of preoperative and perioperative erdosteine decreased these levels significantly when compared with the control group (p<0.05). There was no significant difference between the preoperative and postoperative erdosteine groups (p>0.05).
The mean levels of the oxidative stress parameters for the liver (MDA, total-SH, TFOP and TOS) are summarized in Table 3. For tissue MDA, total-SH, TFOP, and TOS levels, the differences between the sham and other groups were significant (p<0.05 for all parameters). The highest MDA, TFOP, and TOS values, and the lowest total-SH values were found in the control group. The use of erdosteine was observed to lower high values and increase low values of all the oxidative stress parameters. The differences between the control vs perioperative and preoperative erdosteine groups were statistically significant (p<0.05 for all parameters). Although the levels for MDA, TFOP, and TOS were lower, and total-SH levels were higher in the preoperative erdosteine group (Group 4) when compared with the perioperative erdosteine group (Group 3), there was no significant difference between these groups (p>0.05).
The mean histopathological scores of the groups are given in Table 4. Statistical analyses of the histopathological scores showed that the congestion and necrosis scores of the sham group were significantly lower than those of the other groups (p<0.05 for both scores, and each group). The congestion and necrosis scores in the perioperative and preoperative erdosteine-treated groups were determined to be significantly lower than the scores in the control group (p<0.05 for both parameters, and each group). There was no significant difference between the congestion scores of the perioperative and preoperative erdosteine groups (p>0.05), but the necrosis scores of the perioperative erdosteine group (Group 3) were significantly higher than those of the preoperative treated group (Group 4) (p<0.05). Vacuolization was detected in only 2 rats in the control group (mean vacuolization score of this group was 0.20 for this group), however, there was no significant difference between all of the groups, including the sham group (p>0.05).
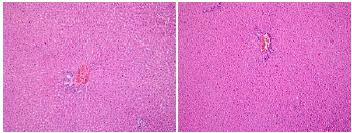
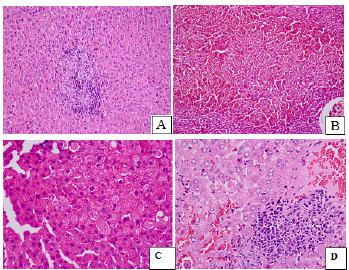
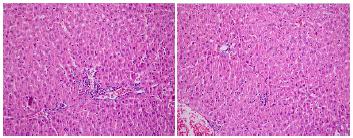
The histopathological findings are presented in Figures 1 to 3. Liver tissue sections from the sham group exhibited almost normal morphology. The structures of hepatocytes and portal areas were observed to be of normal appearance. The sinusoids were found to be of normal width and structure. No cell infiltration was observed (Figure 1-Sham group). In the control group, the integrity of hepatocyte cell cords was disrupted. Mononuclear inflammatory cells had infiltrated the parenchymal areas, sinuses were dilated, and there was diffuse congestion (Figure 2). Perioperative and preoperative treatment with erdosteine decreased the histopathological abnormalities when compared with the control group (Figure 3).
In Group 1 (Sham group) the structures of hepatocytes and portal areas were observed to be of normal appearance. Hepatocytes were observed in the form of cell cords located radially around the vena centralis. The sinusoids between them were observed at normal width and structure. Portal areas and lobules were regular with no inflammation or sinusoidal congestion (H&E, x100).
In the control group, the integrity of hepatocyte cell cords was disrupted. Mononuclear inflammatory cells (A, D), sinusoidal dilatation and diffuse congestion (B, D) and hepatocyte vacuolization (C) were observed to be more intense when compared with the erdosteine groups (H&E, x100, x200).
In erdosteine groups, rare mononuclear inflammatory cells and mild sinusoidal congestion were observed in the liver parenchyma (H&E, x200).
Discussion
Ischemia reperfusion injury (IRI) is referred to as the condition of hypoxia-mediated cellular damage to an organ resulting in paradoxical exacerbation following oxygen restoration. It involves a process that is dynamic in nature including two inter-related stages of inflammation-mediated reperfusion injury and local ischemic insult. The existence of this concept has been observed in different organ systems, such as the heart, kidney, central nervous system, liver, lung, skeletal muscle and intestine2323 Li J, Li RJ, Lv GY, Liu HQ. The mechanisms and strategies to protect from hepatic ischemia-reperfusion injury. Eur Rev Med Pharmacol Sci. 2015;19:2036-47. PMID: 26125267.. Liver ischemia and reperfusion is implicated in many clinical scenarios, including hemorrhagic shock and resuscitation, trauma, liver resections, and liver transplantation. After the introduction of vascular control techniques during hepatic surgery, liver ischemia and reperfusion has been recognized as one of the key elements that contributes to postoperative morbidity and mortality. Liver dysfunction and failure are serious postoperative complications which may ensue as a result of reperfusion injury2424 Nastos C, Kalimeris K, Papoutsidakis N, Tasoulis MK, Lykoudis PM, Theodoraki K, Nastou D, Smyrniotis V, Arkadopoulos N. Global consequences of liver ischemia/reperfusion injury. Oxid Med Cell Longev. 2014;2014:906965. doi: 10. 1155/ 2014/906965.
https://doi.org/10...
.
On restoring blood supply after ischemia, the liver is prone to further injury that aggravates the injury already caused by ischemia. This is called ischemia-reperfusion injury. Endothelial and Kupffer cell swelling, vasoconstriction, leukocyte infiltration, and platelet aggregation in sinusoids characterize IRI. The end-result is microcirculatory failure. The activation of Kupffer cells and neutrophils leads to the release of inflammatory cytokines and free radicals, which further aggravates the liver injury. Ischemia creates the basis for the subsequent production of toxic molecules after reperfusion, particularly reactive oxygen intermediates, the basis of the cascade of events that characterize IRI2525 Saidi RF, Kenari SK. Liver ischemia/reperfusion injury: an overview. J Invest Surg. 2014;27:366-79. doi: 10.3109/08941939.2014.932473.
https://doi.org/10.3109/08941939.2014.93...
. Hepatic IRI is not only a pathophysiological process involving the liver itself, but also a complex systemic process affecting multiple tissues and organs. Hepatic IRI can seriously impair liver function, even producing irreversible damage, which causes a cascade of multiple organ dysfunction2626 Guan LY, Fu PY, Li PD, Li ZN, Liu HY, Xin MG, Li W. Mechanisms of hepatic ischemia-reperfusion injury and protective effects of nitric oxide. World J Gastrointest Surg. 2014;6:122-8. doi: 10.4240/wjgs.v6.i7.122.
https://doi.org/10.4240/wjgs.v6.i7.122...
.
The overall mechanisms of IRI are highly complex and involve the interaction between different molecular pathways which could explain the difficulty of developing therapeutic strategies able to be translated to clinical investigations11 Pantazi E, Bejaoui M, Folch-Puy E, Adam R, Roselló-Catafau J. Advances in treatment strategies for ischemia reperfusion injury. Expert Opin Pharmacother. 2016;17:169-79. doi: 10.1517/14656566.2016.1115015.
https://doi.org/10.1517/14656566.2016.11...
. Many factors, including anaerobic metabolism, mitochondrial damage, oxidative stress, intracellular Ca2+ overload, cytokines and chemokines produced by KCs and neutrophils, and NO, are all involved in the regulation of liver IRI processes. The most important pathways of liver IRI are initiated by oxidative stress, anaerobic metabolism and acidosis, resulting in further cellular damage through the induction of apoptosis, immune responses, and cytokine regulation2626 Guan LY, Fu PY, Li PD, Li ZN, Liu HY, Xin MG, Li W. Mechanisms of hepatic ischemia-reperfusion injury and protective effects of nitric oxide. World J Gastrointest Surg. 2014;6:122-8. doi: 10.4240/wjgs.v6.i7.122.
https://doi.org/10.4240/wjgs.v6.i7.122...
.
The early phase of reperfusion occurs from the first minutes after ischemia and up to 6 hours later. Immediately after reperfusion, cellular swelling takes place due to disturbance of the Na/K/ATPase function. In addition, reactive oxidant species (ROS) can be found inducing oxidative stress to the liver as well as to distant organs. ROS activate Kuppfer cells, promoting even further ROS as well as cytokine production2424 Nastos C, Kalimeris K, Papoutsidakis N, Tasoulis MK, Lykoudis PM, Theodoraki K, Nastou D, Smyrniotis V, Arkadopoulos N. Global consequences of liver ischemia/reperfusion injury. Oxid Med Cell Longev. 2014;2014:906965. doi: 10. 1155/ 2014/906965.
https://doi.org/10...
. Reactive oxygen species are largely generated from mitochondrial energy metabolism via oxidative phosphorylation in the respiratory chain. Due to the existence of antioxidant systems, the redox balance is well maintained. However, ischemia/reperfusion increases the production of ROS from various sources and results in the disturbance of this delicate balance. The increase in ROS consumes endogenous antioxidants and induces the expression of antioxidant enzymes in order to maintain the redox balance. When the injury is pronounced or persistent, compensatory responses become inadequate to correct the imbalanced redox state, giving rise to oxidant stress and leading to inflammatory responses and hepatic damage. Formation of ROS and oxidant stress are the disease mechanisms most commonly invoked in hepatic IRI. The injury process is more extensive during the reperfusion period than during ischemia. Growing evidence supports the notion that oxidant stress is the major initiator in eliciting signaling pathways that lead to the onset of necrosis/apoptosis during the hepatic I/R procedure, especially in the early stage of the process. The destructive effects of I/R result from the acute generation of ROS subsequent to reoxygenation. These ROS inflict direct tissue damage and initiate a cascade of deleterious cellular responses leading to inflammation, cell death, and organ failure. The initial phase of hepatic IRI (< 2 h after reperfusion) is characterized by oxidative stress. At this stage, the excessive ROS cause tissue damage and cell death by binding and altering cellular macromolecules (including DNA, proteins, and lipids), thus affecting their function. The key event in the initial phase of reperfusion injury is activation of the macrophages that are the primary source of extracellular ROS, leading to endothelial injury and further release of proinflammatory cytokines2727 Elias-Miró M, Jiménez-Castro MB, Rodés J, Peralta C. Current knowledge on oxidative stress in hepatic ischemia/reperfusion. Free Radic Res. 2013;47:555-68. doi: 10.3109/ 10715762.2013.811721.
https://doi.org/10.3109/...
.
Although various experimental studies have focused on developing pharmacological strategies with the aim of disrupting the pathways of IRI, none of the pharmacological agents have become part of the clinical routine22 Yamanaka K, Houben P, Bruns H, Schultze D, Hatano E, Schemmer P. A systematic review of pharmacological treatment options used to reduce ischemia reperfusion injury in rat liver transplantation. PLoS One. 2015;10:e0122214. doi: 10.1371/journal.pone. 0122214.
https://doi.org/10.1371/journal.pone...
.
Erdosteine [N-(carboxymethylthioacetyl)-homocysteine thiolactone] was developed as a mucolytic agent for chronic pulmonary diseases. Erdosteine molecule is characterized by the presence of two sulphur atoms. The molecule is stable only in the dry state or in the acidic media of the stomach. When passing to a more alkaline environment, the thiolactone ring slowly opens, achieving, in the blood stream, the complete transformation to metabolite I, N-thiodiglycolylhomocysteine, which is the active metabolite of erdosteine by its free thiol group. The free sulphydryl group in metabolite I results in a multifactorial mechanism of action including mucolytic, antibacterial, antioxidant and anti-inflammatory activities. The free radical scavenging activity could be widely exploited in COPD and other lung diseases where oxidative stress plays an important role in the pathogenesis of the disease. Experimental evidence in animals supports the protective effect of erdosteine in various types of tissue injuries mediated by products of oxidative stress. It prevents the accumulation of free oxygen radicals when their production is accelerated and increases antioxidant cellular protective mechanisms. The final result is a protective effect on tissues that reduces lipid peroxidation, neutrophil infiltration and cell apoptosis mediated by noxious agents33 Moretti M, Marchioni CF. An overview of erdosteine antioxidant activity in experimental research. Pharmacol Res. 2007;55:249-54. doi: 10.1016/j.phrs.2006. 12. 006.
https://doi.org/10.1016/j.phrs.2006...
,2828 Moretti M. Erdosteine: its relevance in COPD treatment. Expert Opin Drug Metab Toxicol. 2009;5:333-43. doi: 10.1517/17425250902814790.
https://doi.org/10.1517/1742525090281479...
. The hepatoprotective effect of erdosteine has also been presented in many studies55 Saritas A, Kandis H, Baltaci D, Yildirim U, Kaya H, Karakus A, Colakoglu S, Memisogullari R, Kara IH. N-Acetyl cysteine and erdosteine treatment in acetaminophen-induced liver damage. Toxicol Ind Health. 2014;30:670-8. doi: 10. 1177/0748233712463780.
https://doi.org/10...
6 Erarslan E, Ekiz F, Uz B, Koca C, Turkcu UO, Bayrak R, Delibasi T. Effects of erdosteine on cyclosporine-A-induced hepatotoxicity in rats. Drug Chem Toxicol. 2011;34:32-7. doi: 10.3109/01480545.2010.483519.
https://doi.org/10.3109/01480545.2010.48...
7 Kuvandik G, Duru M, Nacar A, Yonden Z, Helvaci R, Koc A, Kozlu T, Kaya H, Sogüt S. Effects of erdosteine on acetaminophen-induced hepatotoxicity in rats. Toxicol Pathol. 2008;36:714-9. doi: 10.1177/0192623308320800.
https://doi.org/10.1177/0192623308320800...
8 Sener G, Sehirli AO, Toklu HZ, Yuksel M, Ercan F, Gedik N. Erdosteine treatment attenuates oxidative stress and fibrosis in experimental biliary obstruction. Pediatr Surg Int. 2007;23:233-41. doi: 10.1007/s00383-006-1872-8.
https://doi.org/10.1007/s00383-006-1872-...
9 Koc A, Duru M, Ciralik H, Akcan R, Sogut S. Protective agent, erdosteine, against cisplatin-induced hepatic oxidant injury in rats. Mol Cell Biochem. 2005;278:79-84. doi: 10.1007/s11010-005-6630-z.
https://doi.org/10.1007/s11010-005-6630-...
10 Yagmurca M, Bas O, Mollaoglu H, Sahin O, Nacar A, Karaman O, Songur A. Protective effects of erdosteine on doxorubicin-induced hepatotoxicity in rats. Arch Med Res. 2007;38:380-5. doi: 10.1016/j.arcmed.2007.01.007.
https://doi.org/10.1016/j.arcmed.2007.01...
-1111 Sahin M, Cam H, Olgar S, Tunc SE, Arslan C, Uz E, Yilmaz HR. Protective role of erdosteine on vancomycin-induced oxidative stress in rat liver. Mol Cell Biochem. 2006;291:155-60. doi: 10.1007/s11010-006-9209-4.
https://doi.org/10.1007/s11010-006-9209-...
.
Saritas et al.55 Saritas A, Kandis H, Baltaci D, Yildirim U, Kaya H, Karakus A, Colakoglu S, Memisogullari R, Kara IH. N-Acetyl cysteine and erdosteine treatment in acetaminophen-induced liver damage. Toxicol Ind Health. 2014;30:670-8. doi: 10. 1177/0748233712463780.
https://doi.org/10...
investigated the efficacy of erdosteine usage in acetaminophen-induced liver damage and compared it with N-acetyl cysteine (NAC) in the treatment and prevention of liver toxicity due to acetaminophen overdose. Early treatment with a single dose of erdosteine was reported to be beneficial rather than NAC administration in the prevention of liver damage induced by acetaminophen. Kuvandik et al.77 Kuvandik G, Duru M, Nacar A, Yonden Z, Helvaci R, Koc A, Kozlu T, Kaya H, Sogüt S. Effects of erdosteine on acetaminophen-induced hepatotoxicity in rats. Toxicol Pathol. 2008;36:714-9. doi: 10.1177/0192623308320800.
https://doi.org/10.1177/0192623308320800...
also showed the hepatoprotective effect of erdosteine against acetaminophen-induced hepatotoxicity, and concluded that this effect could be attributed to the antioxidant and radical scavenging effects of erdosteine.
Erarslan et al.66 Erarslan E, Ekiz F, Uz B, Koca C, Turkcu UO, Bayrak R, Delibasi T. Effects of erdosteine on cyclosporine-A-induced hepatotoxicity in rats. Drug Chem Toxicol. 2011;34:32-7. doi: 10.3109/01480545.2010.483519.
https://doi.org/10.3109/01480545.2010.48...
determined the protective effects of erdosteine on cyclosporine A-induced liver injury. Erdosteine was found to significantly improve the functional and histological parameters and attenuate the oxidative stress induced by cyclosporine A. There are many studies in the literature in which the hepatoprotective effect of erdosteine has been demonstrated against hepatotoxic drugs including cisplatin, doxorubicin, and vancomycin99 Koc A, Duru M, Ciralik H, Akcan R, Sogut S. Protective agent, erdosteine, against cisplatin-induced hepatic oxidant injury in rats. Mol Cell Biochem. 2005;278:79-84. doi: 10.1007/s11010-005-6630-z.
https://doi.org/10.1007/s11010-005-6630-...
10 Yagmurca M, Bas O, Mollaoglu H, Sahin O, Nacar A, Karaman O, Songur A. Protective effects of erdosteine on doxorubicin-induced hepatotoxicity in rats. Arch Med Res. 2007;38:380-5. doi: 10.1016/j.arcmed.2007.01.007.
https://doi.org/10.1016/j.arcmed.2007.01...
-1111 Sahin M, Cam H, Olgar S, Tunc SE, Arslan C, Uz E, Yilmaz HR. Protective role of erdosteine on vancomycin-induced oxidative stress in rat liver. Mol Cell Biochem. 2006;291:155-60. doi: 10.1007/s11010-006-9209-4.
https://doi.org/10.1007/s11010-006-9209-...
.
Moreover, erdosteine has been shown to be effective for protecting the liver fibrosis and oxidative injury due to biliary obstruction. Sener et al.88 Sener G, Sehirli AO, Toklu HZ, Yuksel M, Ercan F, Gedik N. Erdosteine treatment attenuates oxidative stress and fibrosis in experimental biliary obstruction. Pediatr Surg Int. 2007;23:233-41. doi: 10.1007/s00383-006-1872-8.
https://doi.org/10.1007/s00383-006-1872-...
reported that serum AST, ALT, LDH, and plasma cytokines were elevated in the bile duct ligated (BDL) group and were significantly decreased by erdosteine treatment. It was also shown that erdosteine administration alleviated the BDL-induced oxidative injury of the liver and improved the hepatic functions, and it was suggested that these effects were associated with the antioxidant and antifibrotic properties of erdosteine.
Erdosteine also has preventive effects against ischemia-reperfusion injury. These effects have been shown in various studies for different organs such as the lungs, brain, kidney, ovaries, spinal cord, and intestines. The authors of these studies concluded that the ischemia-reperfusion protective effect of erdosteine was mainly due to the antioxidant activity of this drug1212 Kurtoglu T, Sacar M, Inan BK, Duver MH, Guler A, Ucak A, Us MH, Yilmaz AT. Erdosteine ameliorates lung injury induced by transient aortic occlusion in rats. Cardiovasc J Afr. 2007;18:367-70. PMID: 18092111.
13 Ozerol E, Bilgic S, Iraz M, Cigli A, Ilhan A, Akyol O. The protective effect of erdosteine on short-term global brain ischemia/reperfusion injury in rats. Prog Neuropsychopharmacol Biol Psychiatry. 2009 33:20-4. doi: 10.1016/j.pnpbp. 2008. 09.024.
https://doi.org/10.1016/j.pnpbp. 2008. 0...
14 Gurel A, Armutcu F, Cihan A, Numanoglu KV, Unalacak M. Erdosteine improves oxidative damage in a rat model of renal ischemia-reperfusion injury. Eur Surg Res. 2004;36:206-9. doi: 10.1159/000078854.
https://doi.org/10.1159/000078854...
15 Tunc T, Uysal B, Atabek C, Kesik V, Caliskan B, Oztas E, Ersoz N, Oter S, Guven A. Erdosteine and ebselen as useful agents in intestinal ischemia/reperfusion injury. J Surg Res. 2009;155: 210-6. doi: 10.1016/j.jss.2008.06.017.
https://doi.org/10.1016/j.jss.2008.06.01...
16 Ugurel V, Cicek AC, Cemek M, Demirtas S, Kocaman AT, Karaca T. Antioxidant and antiapoptotic effects of erdosteine in a rat model of ovarian ischemia-reperfusion injury. Iran J Basic Med Sci. 2017;20:53-8. doi: 10.22038/ijbms.2017.8093.
https://doi.org/10.22038/ijbms.2017.8093...
-1717 Ege E, Ilhan A, Gurel A, Akyol O, Ozen S. Erdosteine ameliorates neurological outcome and oxidative stress due to ischemia/reperfusion injury in rabbit spinal cord. Eur J Vasc Endovasc Surg. 2004;28:379-86. doi: 10.1016/j.ejvs.2004.06.004.
https://doi.org/10.1016/j.ejvs.2004.06.0...
.
In the light of these studies which have confirmed that erdosteine has evident hepatoprotective effects and positive effects on the harmful effects of ischemia-reperfusion, in the current study the effect of erdosteine was evaluated in a rat model of hepatic ischemia-reperfusion. The results of the present study showed that erdosteine ameliorated the harmful effects of ischemia-reperfusion injury on the liver. Perioperative and preoperative treatment with erdosteine reduced congestion and necrosis to a statistically significant degree when compared with the control group. Inflammatory cell infiltration was also seen to be less in the treatment groups. Furthermore, this treatment significantly decreased the serum liver function test values. For an explanation of the possible mechanism of this positive effect, the oxidative stress parameters were evaluated and it was found that erdosteine ameliorated the oxidative stress parameters in both the perioperative and preoperative treatment groups.
Conclusions
The current study is the first to have investigated the effect of erdosteine on the harmful effects of experimental hepatic ischemia-reperfusion injury. The perioperative and postoperative addition of erdosteine had positive effects on this serious injury when compared with the control group. Although the preoperative treatment group had better results than the perioperative group, the difference was not statistically significant. According to these results, it was concluded that after prospective randomized clinical trials, erdosteine could be used for protection against the destructive effects of hepatic ischemia-reperfusion injury in both emergent and elective hepatic surgical operations.
References
-
1Pantazi E, Bejaoui M, Folch-Puy E, Adam R, Roselló-Catafau J. Advances in treatment strategies for ischemia reperfusion injury. Expert Opin Pharmacother. 2016;17:169-79. doi: 10.1517/14656566.2016.1115015.
» https://doi.org/10.1517/14656566.2016.1115015 -
2Yamanaka K, Houben P, Bruns H, Schultze D, Hatano E, Schemmer P. A systematic review of pharmacological treatment options used to reduce ischemia reperfusion injury in rat liver transplantation. PLoS One. 2015;10:e0122214. doi: 10.1371/journal.pone. 0122214.
» https://doi.org/10.1371/journal.pone -
3Moretti M, Marchioni CF. An overview of erdosteine antioxidant activity in experimental research. Pharmacol Res. 2007;55:249-54. doi: 10.1016/j.phrs.2006. 12. 006.
» https://doi.org/10.1016/j.phrs.2006 -
4Park JS, Park MY, Cho YJ, Lee JH, Yoo CG, Lee CT, Lee SM. Anti-inflammatory Effect of erdosteine in lipopolysaccharide-stimulated RAW 264.7 cells. Inflammation. 2016;39:1573-81. doi: 10.1007/s10753-016-0393-4.
» https://doi.org/10.1007/s10753-016-0393-4 -
5Saritas A, Kandis H, Baltaci D, Yildirim U, Kaya H, Karakus A, Colakoglu S, Memisogullari R, Kara IH. N-Acetyl cysteine and erdosteine treatment in acetaminophen-induced liver damage. Toxicol Ind Health. 2014;30:670-8. doi: 10. 1177/0748233712463780.
» https://doi.org/10 -
6Erarslan E, Ekiz F, Uz B, Koca C, Turkcu UO, Bayrak R, Delibasi T. Effects of erdosteine on cyclosporine-A-induced hepatotoxicity in rats. Drug Chem Toxicol. 2011;34:32-7. doi: 10.3109/01480545.2010.483519.
» https://doi.org/10.3109/01480545.2010.483519 -
7Kuvandik G, Duru M, Nacar A, Yonden Z, Helvaci R, Koc A, Kozlu T, Kaya H, Sogüt S. Effects of erdosteine on acetaminophen-induced hepatotoxicity in rats. Toxicol Pathol. 2008;36:714-9. doi: 10.1177/0192623308320800.
» https://doi.org/10.1177/0192623308320800 -
8Sener G, Sehirli AO, Toklu HZ, Yuksel M, Ercan F, Gedik N. Erdosteine treatment attenuates oxidative stress and fibrosis in experimental biliary obstruction. Pediatr Surg Int. 2007;23:233-41. doi: 10.1007/s00383-006-1872-8.
» https://doi.org/10.1007/s00383-006-1872-8 -
9Koc A, Duru M, Ciralik H, Akcan R, Sogut S. Protective agent, erdosteine, against cisplatin-induced hepatic oxidant injury in rats. Mol Cell Biochem. 2005;278:79-84. doi: 10.1007/s11010-005-6630-z.
» https://doi.org/10.1007/s11010-005-6630-z -
10Yagmurca M, Bas O, Mollaoglu H, Sahin O, Nacar A, Karaman O, Songur A. Protective effects of erdosteine on doxorubicin-induced hepatotoxicity in rats. Arch Med Res. 2007;38:380-5. doi: 10.1016/j.arcmed.2007.01.007.
» https://doi.org/10.1016/j.arcmed.2007.01.007 -
11Sahin M, Cam H, Olgar S, Tunc SE, Arslan C, Uz E, Yilmaz HR. Protective role of erdosteine on vancomycin-induced oxidative stress in rat liver. Mol Cell Biochem. 2006;291:155-60. doi: 10.1007/s11010-006-9209-4.
» https://doi.org/10.1007/s11010-006-9209-4 -
12Kurtoglu T, Sacar M, Inan BK, Duver MH, Guler A, Ucak A, Us MH, Yilmaz AT. Erdosteine ameliorates lung injury induced by transient aortic occlusion in rats. Cardiovasc J Afr. 2007;18:367-70. PMID: 18092111.
-
13Ozerol E, Bilgic S, Iraz M, Cigli A, Ilhan A, Akyol O. The protective effect of erdosteine on short-term global brain ischemia/reperfusion injury in rats. Prog Neuropsychopharmacol Biol Psychiatry. 2009 33:20-4. doi: 10.1016/j.pnpbp. 2008. 09.024.
» https://doi.org/10.1016/j.pnpbp. 2008. 09.024 -
14Gurel A, Armutcu F, Cihan A, Numanoglu KV, Unalacak M. Erdosteine improves oxidative damage in a rat model of renal ischemia-reperfusion injury. Eur Surg Res. 2004;36:206-9. doi: 10.1159/000078854.
» https://doi.org/10.1159/000078854 -
15Tunc T, Uysal B, Atabek C, Kesik V, Caliskan B, Oztas E, Ersoz N, Oter S, Guven A. Erdosteine and ebselen as useful agents in intestinal ischemia/reperfusion injury. J Surg Res. 2009;155: 210-6. doi: 10.1016/j.jss.2008.06.017.
» https://doi.org/10.1016/j.jss.2008.06.017 -
16Ugurel V, Cicek AC, Cemek M, Demirtas S, Kocaman AT, Karaca T. Antioxidant and antiapoptotic effects of erdosteine in a rat model of ovarian ischemia-reperfusion injury. Iran J Basic Med Sci. 2017;20:53-8. doi: 10.22038/ijbms.2017.8093.
» https://doi.org/10.22038/ijbms.2017.8093 -
17Ege E, Ilhan A, Gurel A, Akyol O, Ozen S. Erdosteine ameliorates neurological outcome and oxidative stress due to ischemia/reperfusion injury in rabbit spinal cord. Eur J Vasc Endovasc Surg. 2004;28:379-86. doi: 10.1016/j.ejvs.2004.06.004.
» https://doi.org/10.1016/j.ejvs.2004.06.004 -
18Wasowicz W, Nève J, Peretz A. Optimized steps in fluorometric determination of thiobarbituric acid-reactive substances in serum: importance of extraction pH and influence of sample preservation and storage. Clin Chem. 1993;39:2522-6. PMID: 8252725.
-
19Sedlak J, Lindsay RH. Estimation of total, protein bound, and non-protein sulfhydryl groups in tissue with Ellman's reagent. Anal. Biochem. 1968;25:192-205. PMID: 4973948.
-
20Wu T, Willett WC, Rifai N, Rimm EB. Plasma fluorescent oxidation products as potential markers of oxidative stress for epidemiologic studies. Am J Epidemiol. 2007;166:552-60. PMID: 17615091.
-
21Verde V, Fogliano V, Ritieni A, Maiani G, Morisco F, Caporaso N. Use of N,N-dimethyl-p-phenylenediamine to evaluate the oxidative status of human plasma. Free Radic Res. 2002;36:869-73. PMID: 12420745.
-
22Suzuki S, Toledo-Peneyra LH, Rodriquez FJ, Cejalvo D. Neutrophil infiltration as an important factor in liver ischemia and reperfusion injury. Modulating effects of FK506 and cylosporine. Transplantation. 1993;55:1265-72. PMID: 7685932.
-
23Li J, Li RJ, Lv GY, Liu HQ. The mechanisms and strategies to protect from hepatic ischemia-reperfusion injury. Eur Rev Med Pharmacol Sci. 2015;19:2036-47. PMID: 26125267.
-
24Nastos C, Kalimeris K, Papoutsidakis N, Tasoulis MK, Lykoudis PM, Theodoraki K, Nastou D, Smyrniotis V, Arkadopoulos N. Global consequences of liver ischemia/reperfusion injury. Oxid Med Cell Longev. 2014;2014:906965. doi: 10. 1155/ 2014/906965.
» https://doi.org/10 -
25Saidi RF, Kenari SK. Liver ischemia/reperfusion injury: an overview. J Invest Surg. 2014;27:366-79. doi: 10.3109/08941939.2014.932473.
» https://doi.org/10.3109/08941939.2014.932473 -
26Guan LY, Fu PY, Li PD, Li ZN, Liu HY, Xin MG, Li W. Mechanisms of hepatic ischemia-reperfusion injury and protective effects of nitric oxide. World J Gastrointest Surg. 2014;6:122-8. doi: 10.4240/wjgs.v6.i7.122.
» https://doi.org/10.4240/wjgs.v6.i7.122 -
27Elias-Miró M, Jiménez-Castro MB, Rodés J, Peralta C. Current knowledge on oxidative stress in hepatic ischemia/reperfusion. Free Radic Res. 2013;47:555-68. doi: 10.3109/ 10715762.2013.811721.
» https://doi.org/10.3109/ -
28Moretti M. Erdosteine: its relevance in COPD treatment. Expert Opin Drug Metab Toxicol. 2009;5:333-43. doi: 10.1517/17425250902814790.
» https://doi.org/10.1517/17425250902814790
-
Financial source:
none
-
1
Research performed at Ankara Education and Research Hospital, Department of General Surgery, Ankara, Turkey.
Publication Dates
-
Publication in this collection
Oct 2017
History
-
Received
12 June 2017 -
Reviewed
14 Aug 2017 -
Accepted
18 Sept 2017