Abstract
Purpose:
To evaluate respiratory muscle strength (PImax and PEmax) before and 24 and 48 h after conventional and single-port laparoscopic cholecystectomy.
Methods:
Forty women with symptomatic cholelithiasis (18 to 70 years) participated in the study. The patients were divided into two groups: 21 patients undergoing conventional laparoscopic cholecystectomy and 19 patients undergoing single-port laparoscopic cholecystectomy. Differences were considered to be significant when p<0.05.
Results:
The results showed a greater decline in PImax after 24 h in the group submitted to conventional laparoscopic cholecystectomy, with a significant difference between groups (p=0.0308).
Conclusion:
Recovery of the parameters studied was more satisfactory and respiratory muscle strength was less compromised in the group submitted to single-port laparoscopic cholecystectomy.
Key words:
Cholecystectomy, Laparoscopic; Cholelithiasis; Muscle Strength
Introduction
Cholelithiasis is defined as the presence of gallstones in the gallbladder lumen or extrahepatic biliary tree. The disease affects 10% to 20% of the adult population in developed countries and there is a female predilection, with a female-to-male ratio of approximately 2:111 Chen YY, Chen CC. Cholelithiasis. N Engl J Med. 2017 Jul 27;377(4):371. doi: 10.1056/NEJMicm1612408.
https://doi.org/10.1056/NEJMicm1612408...
. In the United States, it is estimated that 20 million people have gallstones, accounting for an annual cost of about US$ 6.2 billion22 Rangaswamy R, Singh CG, Singh HM, Punyabati P, Nyuwi KT. Impact of Biliary Calculi on the Liver. J Clin Diagn Res. 2017 Apr;11(4):PC04-PC07. doi: 10.7860/JCDR/2017/24680.9738.
https://doi.org/10.7860/JCDR/2017/24680....
.
Laparoscopic cholecystectomy has become the gold standard for the surgical treatment of benign biliary disease. The advantages of this procedure compared to laparotomy include smaller incisions, reduced surgical trauma, less postoperative pain, faster recovery times, shorter hospital stay, and faster return to normal activities and work, in addition to reducing the risk of surgical wound infections, incisional hernias and eviscerations33 Agabiti N, Stafoggia M, Davoli M, Fusco D, Barone AP, Perucci CA. Thirty-day complications after laparoscopic or open cholecystectomy: a population-based cohort study in Italy. BMJ Open. 2013 Feb 13;3(2). pii: e001943. doi: 10.1136/bmjopen-2012-001943.
https://doi.org/10.1136/bmjopen-2012-001...
.
After a few decades and several technical modifications, including a reduction in the number of ports, the use of smaller ports and natural orifice surgery, in 1997 Navarra et al.44 Navarra G, Pozza E, Occhionorelli S, Carcoforo P, Donini I. One-wound laparoscopic cholecys¬tectomy. Br J Surg. 1997 May;84(5):695. PMID: 9171771. described the use of a single incision for laparoscopic cholecystectomy as an alternative procedure to conventional four-port laparoscopic cholecystectomy. The patients benefit from the single-port approach since the procedure is faster and less traumatic, recovery is faster, and the risk of infection is lower55 Hajong R, Hajong D, Natung T, Anand M, Sharma G. A Comparative study of single incision versus conventional four ports laparoscopic cholecystectomy. J Clin Diagn Res. 2016 Oct;10(10):PC06-9. PMID: 27891389.,66 Shaikh AR, Ali SA, Munir A, Shaikh AA. Single incision laparoscopic cholecystectomy with conventional instruments and ports: initial experience at tertiary care public sector hospital. Pak J Med Sci. 2017 May-Jun;33(3):654-8. doi: 10.12669/pjms.333.12930.
https://doi.org/10.12669/pjms.333.12930...
.
Abdominal surgeries alter the respiratory muscle strength of the patient, reducing lung volumes and capacities and consequently compromising gas exchange. The causes of these changes are multiple and can be related to abdominal manipulation, the effects of general anesthesia, pain at the incision site, and time of stay in the bed. Altered inspiratory muscle strength leads to a reduction in inspired lung volume. This reduction, associated with the impairment of expiratory muscles, decreases expiratory flow and compromises the cough mechanism, favoring the retention of pulmonary secretions77 Katsura M1, Kuriyama A, Takeshima T, Fukuhara S, Furukawa TA. Preoperative inspiratory muscle training for postoperative pulmonary complications in adults undergoing cardiac and major abdominal surgery. Cochrane Database Syst Rev. 2015 Oct 5;(10):CD010356. doi: 10.1002/14651858.CD010356.pub2.
https://doi.org/10.1002/14651858.CD01035...
,88 Ávila AC, Fenili R. Incidence and risk factors for postoperative pulmonary complications in patients undergoing thoracic and abdominal surgeries. Rev Col Bras Cir. 2017 May-Jun;44(3):284-92. doi: 10.1590/0100-69912017003011.
https://doi.org/10.1590/0100-69912017003...
.
We found no studies in the literature evaluating respiratory muscle strength (PImax and PEmax) before and after conventional and single-port laparoscopic cholecystectomy. Since single-port laparoscopic cholecystectomy is performed through a single point of entry, i.e., the surgical trauma is less, the objective of the present study was to evaluate whether impairment of respiratory muscle strength is lower in patients submitted to this procedure when compared to patients undergoing conventional four-port laparoscopic cholecystectomy.
Methods
The study was approved by the Research Ethics Committee of UFTM (Protocol nº 2503).
A prospective, cross-sectional study was conducted at the Department of Digestive Tract Surgery, University Hospital, Universidade Federal do Triângulo Mineiro. Forty women with symptomatic cholelithiasis, ranging in age from 18 to 70 years, participated in the study. The patients were divided into two groups: 21 patients were submitted to conventional laparoscopic cholecystectomy and 19 patients to single-port laparoscopic cholecystectomy.
The data of each patient (age and anthropometric variables) were recorded on an assessment form. Additionally, each patient provided written informed consent after receiving detailed information about the study.
Women with symptomatic cholelithiasis older than 18 years, who were able to understand the objective of the study and gave informed consent, were included in the study. The criteria for exclusion were pregnancy, BMI >35 kg/m2, neurological or cognitive deficit impairing participation in the respiratory muscle strength assessment, presence of respiratory infections, symptomatic allergic sinus disease and chronic obstructive pulmonary disease, a previous diagnosis of bronchial asthma, pleuropulmonary anomalies, chest wall deformities, a suspicion or confirmation of liver cirrhosis, coagulopathy (platelet count <50,000/µl), dual antiplatelet therapy (acetylsalicylic acid and clopidogrel), acute pancreatitis, and jaundice.
Surgical procedure
Anesthesia was standardized as follows: pre-anesthetic medication administered 3 h before surgery consisting of oral diazepam (10 mg), intravenous midazolam (5 mg) and volume expansion with 1,000 ml saline; induction of anesthesia: alfentanil (30 mg/kg), etomidate (0.3 mg/kg) and atracure (0.5 mg/kg); maintenance of anesthesia: continuous infusion of alfentanil (1 mg/kg/min) and isoflurane (0.5-1.5%); decurarization: intravenous atropine (1 mg) and prostigmin (2 mg).
Conventional laparoscopic cholecystectomy
The patient was placed in horizontal dorsal decubitus on the operating table and received general anesthesia. Perioperative monitoring consisted of cardioscopy, noninvasive blood pressure monitoring, pulse oximetry, and capnography.
Conventional laparoscopic cholecystectomy consisted of the insertion of 4 trocars, one 10-mm trocar in the supraumbilical region for placement of the optical system and one 5-mm trocar in the right flank for cranial traction of the gallbladder. The other two working trocars were placed in the same line, one 5-mm trocar in the right upper quadrant for removal of the gallbladder and the other 10-mm trocar in the epigastrium, on the left side of the round ligament, for dissection and hemostasis.
After the skin was cut in the supraumbilical region, the aponeurosis was dissected with Backhaus forceps and the abdominal wall was lifted. This maneuver of elevation of the abdominal wall facilitates Veress needle insertion and creation of a pneumoperitoneum. When the pressure reached 14 mmHg as indicated on the control panel of the insufflator, the trocar was carefully inserted, with the abdominal wall always firmly suspended. The other trocars were placed under optically guided direct vision, preferably of 30 degrees. The patient was then placed in the reverse Trendelenburg position and left lateral decubitus.
The operating field was exposed by pulling the gallbladder up and outward in the direction of the right hemidiaphragm. The hilum was exposed by lateral retraction, holding the infundibulum and retracting it to the right and downwards. The technique consisted of opening the peritoneum and dissection following the outer contour of the gallbladder from the infundibulum, the neck of the gallbladder and the cystic duct, avoiding the longitudinal axis of the hepatic hilum as well as part of Calot’s triangle, thus conferring surgical safety. The cystic duct was isolated, partially sectioned and always catheterized, if possible, for intraoperative cholangiography. After careful analysis of the cholangiogram, the cystic duct was ligated and completely sectioned. Next, the cystic artery was identified, isolated, and clipped with metal clips. The gallbladder was then removed from the liver bed.
Single-port laparoscopic cholecystectomy
The patient was placed in dorsal decubitus. After general anesthesia, discrete reverse Trendelenburg and left lateralization were performed, with the legs being placed in stirrups (French or European position). The surgeon was standing between the patient’s legs and its first assistant sitting on the left. The monitors were positioned at the level of the patient’s right shoulder.
A transumbilical horizontal incision measuring about 1.5 cm was made for Veress needle insertion by puncture. When an umbilical hernia was present, the hernial ring was dissected for placement of the needle or trocar under direct vision. Next, the pneumoperitoneum was induced and maintained at 14 mmHg by insufflation of carbon dioxide (CO2). A 10-mm trocar was inserted for placement of the 30o optical system. A second, 5- or 10-mm, trocar was inserted under vision of the optical system above and to the right of the first trocar, accessing the fascia of the rectus abdominis muscles to position the hook or scissors, among other instruments used by the surgeon’s right hand. A third trocar (5 or 3 mm) was inserted to the left and at the same height as the second trocar for forceps placements.
When necessary, a 2-mm trocar was inserted in the right flank for placement of the grasping forceps in order to help with exposure of Calot’s triangle and dissection of the cystic duct and artery. If available, a 5-mm 30o optical system was used at the time of placement of the clips in the cystic duct and artery, permitting passage of the clipper through the 10-mm trocar previously inserted in the umbilical scar. If not available, a second 10-mm trocar was inserted at the position of the 5-mm trocar. After cholangiography, the cystic duct was clipped and cut, maintaining two clips to close its proximal stump and one clip at the distal stump (close to the gallbladder). The cystic artery was clamped in the same way. Next, antegrade dissection was performed and the gallbladder was released from the liver bed. The umbilical fascia was approximated, followed by intradermal suturing.
Evaluation of respiratory muscle strength
Respiratory muscle strength was evaluated based on the measurement of maximum respiratory pressures: maximum inspiratory pressure (PImax) and maximum expiratory pressure (PEmax). These parameters permit a simple, rapid and reproducible assessment of respiratory muscle strength. The measurements were performed according to Black and Hyatt99 Black LF, Hyatt RE. Maximal respiratory pressures: normal values and relationship to age and sex. Am Rev Respir Dis. 1969 May;99(5):696-702. doi: 10.1164/arrd.1969.99.5.696.
https://doi.org/10.1164/arrd.1969.99.5.6...
.
PImax and PEmax were measured using a manovacuometer (Comercial Médica; -300 to +300 cmH2O). For the measurement, the patient was sitting comfortably and used a nose clip and rigid plastic mouthpiece. The procedure was previously explained and shown to the patient; in addition, the importance of avoiding the escape of air between the mouth and mouthpiece during the maneuver was emphasized. For the measurement of PImax, the patient was asked to slowly and completely expire air until the residual volume in such a way that, with the orifice of the tube closed, a maximum inspiratory effort would occur. PEmax was obtained at the time when the patient inspired until total lung capacity and then performed a maximum expiratory effort against the orifice of the closed tube.
The measurements of PImax and PEmax were sustained for 2 seconds and performed in three intercalated steps, with a resting interval of 2 min. An interval of 5 min was allowed between the measurements of PImax and PEmax. Only the highest of the three values obtained for PImax and PEmax was considered. Verbal encouragement of the researcher permitted better execution of the technique.
For statistical analysis, the Student t-test was used for comparison between the two surgical techniques. Comparison of the same surgery with the pre-surgery condition and between 24 and 48 h post-surgery was done using ANOVA for repeated measures, followed by the Bonferroni post-test.
Results
Table 1 shows the mean ± standard deviation of the age and anthropometric variables of the groups undergoing conventional and single-port laparoscopic cholecystectomy. Height differed significantly between groups (p=0.022). However, this anthropometric variable is not related to the formation of gallstones.
When asked about the smoking habit in the initial protocol, three (14.3%) patients in the group undergoing conventional laparoscopic surgery and six (31.6%) in the group undergoing single-port surgery were smokers.
Results of PImax and PEmax - Respiratory muscle strength
All patients participating in the study underwent respiratory muscle strength assessment. Evaluation consisted of the measurement of maximum respiratory pressures (PImax and PEmax) before and 24 and 48 h after the surgical procedures.
Comparison of PImax in the group of patients submitted to conventional laparoscopic cholecystectomy before and 24 and 48 h after surgery showed mean values of 124.90 ± 6.59, 88.85 ± 5.73 and 103.66 ± 5.49 cmH2O, respectively (p<0.0001). In the group undergoing single-port cholecystectomy, mean PImax was 122.31 ± 6.51, 107.84 ± 6.25 and 118.42 ± 6.26 cmH2O before and 24 and 48 h after surgery, respectively (p<0.0001).
Preoperative mean PImax was 124.90 ± 6.59 cmH2O in the group undergoing conventional laparoscopic cholecystectomy versus 122.31 ± 6.51 cmH2O in the group undergoing single-port surgery. Comparison of postoperative mean PImax values obtained after 24 h showed a significant difference between the group undergoing conventional laparoscopic cholecystectomy and the group undergoing single-port surgery (88.85 ± 5.73 and 107.84 ± 6.25 cmH2O, respectively, p=0.0308). There was no significant difference in postoperative mean PImax values obtained after 48 h between the group undergoing conventional versus single-port laparoscopic cholecystectomy (103.66 ± 5.49 and 118.42 ± 6.26 cmH2O, p=0.0833) (Figure 1).
The bars represent the mean and lines indicate the standard error of the mean. * p<0.05 comparing the different types of surgery at the same time point (Student t-test); # p<0.05 comparing the same surgery with the pre-surgery condition (ANOVA for repeated measures followed by the Bonferroni post-test); Δ p<0.05 comparing the same surgery between 24 and 48h post-surgery (ANOVA for repeated measures followed by the Bonferroni post-test).
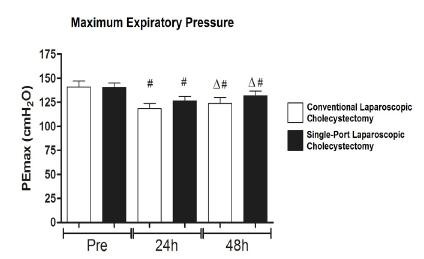
Comparison of mean PEmax in the group of patients submitted to conventional laparoscopic cholecystectomy before and 24 and 48 h after surgery showed values of 140.61 ± 6.44, 118.14 ± 5.75 and 123.90 ± 5.92 cmH2O, respectively (p<0.0001). In the group submitted to single-port surgery, mean PEmax was 140.26 ± 4.81, 126.42 ± 4.68 and 131.78 ± 4.77 cmH2O before and 24 and 48h after surgery, respectively (p<0.0001).
Preoperative mean PEmax was 140.61 ± 6.44 cmH2O in the group undergoing conventional laparoscopic cholecystectomy vs. 140.26 ± 4.81 cmH2O in the group undergoing single-port surgery. Comparison of postoperative PEmax obtained after 24h between the group undergoing conventional laparoscopic cholecystectomy and the group undergoing single-port laparoscopic cholecystectomy showed mean values of 118.14 ± 5.75 and 126.42 ± 4.68 cmH2O, respectively. Postoperative mean PEmax values obtained after 48 h were 123.90 ± 5.92 cmH2O in the group undergoing conventional laparoscopic cholecystectomy vs. 131.78 ± 4.77 cmH2O in the group undergoing single-port surgery (Figure 2).
The bars represent the mean and lines indicate the standard error of the mean. p<0.05 comparing the different types of surgery at the same time point (Student t-test); # p<0.05 comparing the same surgery with the pre-surgery condition (ANOVA for repeated measures followed by the Bonferroni post-test); Δ p<0.05 comparing the same surgery between 24 and 48h post-surgery (ANOVA for repeated measures followed by the Bonferroni post-test).
Discussion
It is now known that diaphragmatic paralysis is the main determinant of alterations in lung volumes and lung capacities. A reduction in lung volume and capacity is observed within the first 24 to 48 h after surgery, followed by a gradual increase in these values which usually return to normal by the second week. The ventilatory pattern returns to preoperative levels 7 to 10 days after surgery1010 Giovanetti EA, Boueri CA, Braga KF. Comparative study of pulmonary volume and oxigenation after use of Respiron e Voldyne in upper abdominal surgery. Reabilitar. 2004 Out 6(25):30-9. PMID: 413819..
The main physiopathological mechanism involved in the postoperative reduction of respiratory muscle strength is characterized by dysfunction of the diaphragm and intercostal muscles as a result of stimulation of reflexogenic areas manipulated at surgical sites close to the diaphragm1111 Berdah SV, Picaud R, Jammes Y. Short communication surface diaphragmatic electromyogram changes after laparotomy. Clin Physiol Funct Imaging. 2002 Mar;22(2):157-60. PMID: 12005159.. The stimulation of mesenteric nerves and afferent sympathetic fibers, as well as mechanisms of small bowel distension, inhibits efferent nerve impulses from the phrenic nerve. This reflex mechanism is the cause of diaphragmatic dysfunction and consequent postoperative restrictive ventilatory defects in cholecystectomies1212 Ravimohan SM, Kaman L, Jindal R, Singh R, Jindal SK. Postoperative pulmonary function in laparoscopic versus open cholecystectomy: prospective, comparative study. Indian J Gastroenterol. 2005 Jan-Feb;24(1):6-8. PMID: 15778517.,1313 Osman Y, Fusun A, Serpil A, Umit T, Ebru M, Bulent U, Mete D, Omer C. The comparison of pulmonary functions in open versus laparoscopic cholecystectomy. J Pak Med Assoc. 2009 Apr;59(4):201-4. PMID: 19402277..
Miranda et al.1414 Miranda AS, Novaes RD, Ferreira AE, Neves MP, Corrêa CL, Mendonça VA. Assessment of respiratory muscle strength, peak expiratory flow and pain after open cholecystectomy. Acta Gastroenterol Latinoam. 2009 Mar;39(1):38-46. PMID: 19408738. investigated the behavior and correlation between pain and respiratory muscle strength before and after open cholecystectomy. The mean percent difference observed at each time point analyzed showed that the most marked reduction in PImax and PEmax occurred on the first day after open cholecystectomy1414 Miranda AS, Novaes RD, Ferreira AE, Neves MP, Corrêa CL, Mendonça VA. Assessment of respiratory muscle strength, peak expiratory flow and pain after open cholecystectomy. Acta Gastroenterol Latinoam. 2009 Mar;39(1):38-46. PMID: 19408738.. The authors concluded that impairment of respiratory muscle strength occurs mainly on the first postoperative day after open cholecystectomy, with improvement of this parameter as early as on the second postoperative day1515 Ramos GC, Pereira E, Neto SG, De Oliveira EC. Pulmonary performance test after conventional and laparoscopic cholecystectomy. Rev Col Bras Cir. 2007;34(5):326-30. doi: 10.1590/S0100-69912007000500009.
https://doi.org/10.1590/S0100-6991200700...
,1616 Keus F, Ahmed Ali U, Noordergraaf GJ, Roukema JA, Gooszen HG, van Laarhoven CJ. Laparoscopic vs small incision cholecystectomy: implications for pulmonary function and pain. A randomized clinical trial. Acta Anaesthesiol Scand. 2008 Mar;52(3):363-73. doi: 10.1111/j.1399-6576.2007.01488.x.
https://doi.org/10.1111/j.1399-6576.2007...
.
Rovina et al.1717 Rovina N, Bouros D, Tzanakis N, Velegrakis M, Kandilakis S, Vlasserou F, Siafakas NM. Effects of laparoscopy cholecystectomy on global respiratory muscle strength. Am J Respir Crit Care Med. 1996 Jan;153(1):458-61. doi: 10.1164/ajrccm.153.1.8542159.
https://doi.org/10.1164/ajrccm.153.1.854...
evaluated PImax and PEmax after conventional laparoscopic and open cholecystectomy. The results showed a significant reduction in PaO2 (p<0.007) after 24 h in the group submitted to open surgery1818 Saia M, Mantoan D, Buja A, Bertoncello C, Baldovin T, Callegaro G, Baldo V. Time trend and variability of open versus laparoscopic cholecystectomy in patients with symptomatic gallstone disease. Surg Endosc. 2013 Sep;27(9):3254-61. doi: 10.1007/s00464-013-2902-y.
https://doi.org/10.1007/s00464-013-2902-...
,1919 Agabiti N, Stafoggia M, Davoli M, Fusco D, Barone AP, Perucci CA. Thirty-day complications after laparoscopic or open cholecystectomy: a population-based cohort study in Italy. BMJ Open. 2013 Feb 13;3(2). pii: e001943. doi: 10.1136/bmjopen-2012-001943.
https://doi.org/10.1136/bmjopen-2012-001...
. PImax was significantly reduced 24 and 48 h after surgery (p<0.01 and p<0.005, respectively) in both groups, but this reduction was significantly lower in the group submitted to laparoscopic cholecystectomy. A significant reduction in PEmax was observed in the group undergoing open cholecystectomy 48 h after the surgical procedure (p<0.0001)2020 Siafakas NM, Mitrouska I, Argiana E, Bouros D. Effects of surgery on the function of the respiratory muscles. Monaldi Arch Chest Dis. 1999 Dec;54(6):526-31. PMID: 10695325..
Diaphragmatic dysfunction is responsible for many pulmonary complications, including atelectasis and pneumonia. According to the authors, diaphragmatic dysfunction is due to several factors, such as the site of surgical incision by affecting muscle integrity and the duration of the surgical procedure during which the patient remains in a supine position. Furthermore, anesthesia can cause neuromuscular blockade and a reduction in respiratory muscle contraction. Ford et al.2121 Ford GT, Whitelaw WA, Rosenal TW, Cruse PJ, Guenter CA. Diaphragm function after upper abdominal surgery in humans. Am Rev Respir Dis. 1983 Apr;127(4):431-6. doi: 10.1164/arrd.1983.127.4.431.
https://doi.org/10.1164/arrd.1983.127.4....
suggested diaphragmatic dysfunction to be due to direct secondary injury to the abdominal muscles, causing mechanical failure and pain at the incision site which contribute to respiratory alterations. Ayoub et al.2222 Ayoub J, Cohendy R, Prioux J, Ahmaidi S, Bourgeois JM, Dauzat M, Ramonatxo M, Préfaut C. Diaphragm movement before and after cholecystectomy: a sonographic study. Anesth Analg. 2001 Mar;92(3):755-61. PMID: 11226114. also reported a decrease in diaphragmatic excursion during the course of upper abdominal surgeries due to surgical incision, reducing the inspiratory amplitude, especially in conventional cholecystectomy.
Gastaldi et al.2323 Gastaldi AC, Magalhães CMB, Baraúna MA, Silva EMC, Souza HCD. Benefits of postoperative respiratory kinesiotherapy following laparoscopic cholecystectomy. Rev Bras Fisioter. 2008;12(2):100-6. doi: 10.1590/S1413-35552008000200005.
https://doi.org/10.1590/S1413-3555200800...
observed a reduction in respiratory muscle strength on the first postoperative day after laparoscopic cholecystectomy similar to the findings of other studies. However, PImax (p<0.05) and PEmax (p<0.05) showed an early return to normal levels by the third or fourth postoperative day. Similarly, other studies reported a reduction in respiratory muscle function between the first and third postoperative day. This reduction persisted for one week before returning to normal, suggesting the occurrence of diaphragmatic dysfunction2424 Simonneau G, Vivien A, Sartene R, Kunstlinger F, Samii K, Noviant Y, Duroux P. Diaphragm dysfunction induced by upper abdominal surgery. Role of postoperative pain. Am Rev Respir Dis. 1983 Nov;128(5):899-903. doi: 10.1164/arrd.1983.128.5.899.
https://doi.org/10.1164/arrd.1983.128.5....
,2525 Ford GT, Rosenal TW, Clergue F, Whitelaw WA. Respiratory physiology in upper abdominal surgery. Clin Chest Med. 1993 Jun;14(2):237-52. PMID: 8519170..
Postoperative respiratory complications prolong the length of hospital stay of patients, with a consequent increase in hospital costs and significantly contributing to patient mortality2626 Vijayaraghavan N, Sistla SC, Kundra P, Ananthanarayan PH, Karthikeyan VS, Ali SM, Sasi SP, Vikram K. Comparison of standard-pressure and low-pressure pneumoperitoneum in laparoscopic cholecystectomy: a double blinded randomized controlled study. Surg Laparosc Endosc Percutan Tech. 2014 Apr;24(2):127-33. doi: 10.1097/SLE.0b013e3182937980.
https://doi.org/10.1097/SLE.0b013e318293...
.
Erice et al.2727 Erice F, Fox GS, Salib YM, Romano E, Meakins JL, Magder SA. Diaphragmatic function before and after laparoscopic cholecystectomy. Anesthesiology. 1993 Nov;79(5):966-75. PMID: 8239015., comparing laparoscopic and conventional cholecystectomy, showed that surgical trauma to the abdominal wall can increase the incidence of ventilatory defects by reducing diaphragmatic excursion. Diaphragmatic dysfunction, in turn, decreases ventilation of the lower lobe of the lung on the affected side and causes a reduction in tidal volume, vital capacity and PImax. These alterations can persist for 48 h after surgery.
Agrelli et al.2828 Agrelli TF, de Carvalho Ramos M, Guglielminetti R, Silva AA, Crema E. Preoperative ambulatory inspiratory muscle training in patients undergoing esophagectomy. A pilot study. Int Surg. 2012 Jul-Sep;97(3):198-202. doi: 10.9738/CC136.1.
https://doi.org/10.9738/CC136.1...
observed a significant increase in PImax was observed after inspiratory muscle training when compared with baseline values (from -55.059 ± 18.359 to -76.286 ± 16.786). Preoperative ambulatory inspiratory muscle training was effective in increasing respiratory muscle strength in patients undergoing esophagectomy and contributed to the prevention of postoperative complications.
The present results showed a significant reduction in PImax 24 h after the surgical procedures, with the observation of an important increase in these values after 48 h. The values were more satisfactory after 24 h for the group undergoing single-port laparoscopic cholecystectomy compared to the group submitted to conventional laparoscopic cholecystectomy.
With respect to PEmax, in the present study more satisfactory values were obtained 24 and 48 h after surgery for the group undergoing single-port laparoscopic cholecystectomy. No significant difference in PEmax values was observed between groups at the time points analyzed (pre, 24 and 48 h after surgery).
Smoking is also an important factor that contributes to the deterioration of lung function, since PImax and PEmax not only depend on the strength of respiratory muscles, but also on the lung volumes in which the measurements are performed and the corresponding elastic retraction pressure of the respiratory system2929 Cosa D, Gonçalves HA, Lima LP, Ike D, Cancelliero KM, Montebelo MI. New reference values for maximal respiratory pressures in the Brazilian population: corrections. J Bras Pneumol. 2010 Sep-Oct;36(5):667. PMID: 21085835.. In the present study, the number of smokers was larger in the group submitted to single-port laparoscopic cholecystectomy; however, the results of this group were better, suggesting that a single incision can preserve the integrity of the respiratory muscles and prevent pulmonary complications.
Conclusions
The present results showed a significant difference in PImax values obtained 24h after surgery between the surgical techniques. More satisfactory results and less compromised respiratory muscle strength were observed in the group submitted to single-port laparoscopic cholecystectomy.
References
-
1Chen YY, Chen CC. Cholelithiasis. N Engl J Med. 2017 Jul 27;377(4):371. doi: 10.1056/NEJMicm1612408.
» https://doi.org/10.1056/NEJMicm1612408 -
2Rangaswamy R, Singh CG, Singh HM, Punyabati P, Nyuwi KT. Impact of Biliary Calculi on the Liver. J Clin Diagn Res. 2017 Apr;11(4):PC04-PC07. doi: 10.7860/JCDR/2017/24680.9738.
» https://doi.org/10.7860/JCDR/2017/24680.9738 -
3Agabiti N, Stafoggia M, Davoli M, Fusco D, Barone AP, Perucci CA. Thirty-day complications after laparoscopic or open cholecystectomy: a population-based cohort study in Italy. BMJ Open. 2013 Feb 13;3(2). pii: e001943. doi: 10.1136/bmjopen-2012-001943.
» https://doi.org/10.1136/bmjopen-2012-001943 -
4Navarra G, Pozza E, Occhionorelli S, Carcoforo P, Donini I. One-wound laparoscopic cholecys¬tectomy. Br J Surg. 1997 May;84(5):695. PMID: 9171771.
-
5Hajong R, Hajong D, Natung T, Anand M, Sharma G. A Comparative study of single incision versus conventional four ports laparoscopic cholecystectomy. J Clin Diagn Res. 2016 Oct;10(10):PC06-9. PMID: 27891389.
-
6Shaikh AR, Ali SA, Munir A, Shaikh AA. Single incision laparoscopic cholecystectomy with conventional instruments and ports: initial experience at tertiary care public sector hospital. Pak J Med Sci. 2017 May-Jun;33(3):654-8. doi: 10.12669/pjms.333.12930.
» https://doi.org/10.12669/pjms.333.12930 -
7Katsura M1, Kuriyama A, Takeshima T, Fukuhara S, Furukawa TA. Preoperative inspiratory muscle training for postoperative pulmonary complications in adults undergoing cardiac and major abdominal surgery. Cochrane Database Syst Rev. 2015 Oct 5;(10):CD010356. doi: 10.1002/14651858.CD010356.pub2.
» https://doi.org/10.1002/14651858.CD010356.pub2 -
8Ávila AC, Fenili R. Incidence and risk factors for postoperative pulmonary complications in patients undergoing thoracic and abdominal surgeries. Rev Col Bras Cir. 2017 May-Jun;44(3):284-92. doi: 10.1590/0100-69912017003011.
» https://doi.org/10.1590/0100-69912017003011 -
9Black LF, Hyatt RE. Maximal respiratory pressures: normal values and relationship to age and sex. Am Rev Respir Dis. 1969 May;99(5):696-702. doi: 10.1164/arrd.1969.99.5.696.
» https://doi.org/10.1164/arrd.1969.99.5.696 -
10Giovanetti EA, Boueri CA, Braga KF. Comparative study of pulmonary volume and oxigenation after use of Respiron e Voldyne in upper abdominal surgery. Reabilitar. 2004 Out 6(25):30-9. PMID: 413819.
-
11Berdah SV, Picaud R, Jammes Y. Short communication surface diaphragmatic electromyogram changes after laparotomy. Clin Physiol Funct Imaging. 2002 Mar;22(2):157-60. PMID: 12005159.
-
12Ravimohan SM, Kaman L, Jindal R, Singh R, Jindal SK. Postoperative pulmonary function in laparoscopic versus open cholecystectomy: prospective, comparative study. Indian J Gastroenterol. 2005 Jan-Feb;24(1):6-8. PMID: 15778517.
-
13Osman Y, Fusun A, Serpil A, Umit T, Ebru M, Bulent U, Mete D, Omer C. The comparison of pulmonary functions in open versus laparoscopic cholecystectomy. J Pak Med Assoc. 2009 Apr;59(4):201-4. PMID: 19402277.
-
14Miranda AS, Novaes RD, Ferreira AE, Neves MP, Corrêa CL, Mendonça VA. Assessment of respiratory muscle strength, peak expiratory flow and pain after open cholecystectomy. Acta Gastroenterol Latinoam. 2009 Mar;39(1):38-46. PMID: 19408738.
-
15Ramos GC, Pereira E, Neto SG, De Oliveira EC. Pulmonary performance test after conventional and laparoscopic cholecystectomy. Rev Col Bras Cir. 2007;34(5):326-30. doi: 10.1590/S0100-69912007000500009.
» https://doi.org/10.1590/S0100-69912007000500009 -
16Keus F, Ahmed Ali U, Noordergraaf GJ, Roukema JA, Gooszen HG, van Laarhoven CJ. Laparoscopic vs small incision cholecystectomy: implications for pulmonary function and pain. A randomized clinical trial. Acta Anaesthesiol Scand. 2008 Mar;52(3):363-73. doi: 10.1111/j.1399-6576.2007.01488.x.
» https://doi.org/10.1111/j.1399-6576.2007.01488.x -
17Rovina N, Bouros D, Tzanakis N, Velegrakis M, Kandilakis S, Vlasserou F, Siafakas NM. Effects of laparoscopy cholecystectomy on global respiratory muscle strength. Am J Respir Crit Care Med. 1996 Jan;153(1):458-61. doi: 10.1164/ajrccm.153.1.8542159.
» https://doi.org/10.1164/ajrccm.153.1.8542159 -
18Saia M, Mantoan D, Buja A, Bertoncello C, Baldovin T, Callegaro G, Baldo V. Time trend and variability of open versus laparoscopic cholecystectomy in patients with symptomatic gallstone disease. Surg Endosc. 2013 Sep;27(9):3254-61. doi: 10.1007/s00464-013-2902-y.
» https://doi.org/10.1007/s00464-013-2902-y -
19Agabiti N, Stafoggia M, Davoli M, Fusco D, Barone AP, Perucci CA. Thirty-day complications after laparoscopic or open cholecystectomy: a population-based cohort study in Italy. BMJ Open. 2013 Feb 13;3(2). pii: e001943. doi: 10.1136/bmjopen-2012-001943.
» https://doi.org/10.1136/bmjopen-2012-001943 -
20Siafakas NM, Mitrouska I, Argiana E, Bouros D. Effects of surgery on the function of the respiratory muscles. Monaldi Arch Chest Dis. 1999 Dec;54(6):526-31. PMID: 10695325.
-
21Ford GT, Whitelaw WA, Rosenal TW, Cruse PJ, Guenter CA. Diaphragm function after upper abdominal surgery in humans. Am Rev Respir Dis. 1983 Apr;127(4):431-6. doi: 10.1164/arrd.1983.127.4.431.
» https://doi.org/10.1164/arrd.1983.127.4.431 -
22Ayoub J, Cohendy R, Prioux J, Ahmaidi S, Bourgeois JM, Dauzat M, Ramonatxo M, Préfaut C. Diaphragm movement before and after cholecystectomy: a sonographic study. Anesth Analg. 2001 Mar;92(3):755-61. PMID: 11226114.
-
23Gastaldi AC, Magalhães CMB, Baraúna MA, Silva EMC, Souza HCD. Benefits of postoperative respiratory kinesiotherapy following laparoscopic cholecystectomy. Rev Bras Fisioter. 2008;12(2):100-6. doi: 10.1590/S1413-35552008000200005.
» https://doi.org/10.1590/S1413-35552008000200005 -
24Simonneau G, Vivien A, Sartene R, Kunstlinger F, Samii K, Noviant Y, Duroux P. Diaphragm dysfunction induced by upper abdominal surgery. Role of postoperative pain. Am Rev Respir Dis. 1983 Nov;128(5):899-903. doi: 10.1164/arrd.1983.128.5.899.
» https://doi.org/10.1164/arrd.1983.128.5.899 -
25Ford GT, Rosenal TW, Clergue F, Whitelaw WA. Respiratory physiology in upper abdominal surgery. Clin Chest Med. 1993 Jun;14(2):237-52. PMID: 8519170.
-
26Vijayaraghavan N, Sistla SC, Kundra P, Ananthanarayan PH, Karthikeyan VS, Ali SM, Sasi SP, Vikram K. Comparison of standard-pressure and low-pressure pneumoperitoneum in laparoscopic cholecystectomy: a double blinded randomized controlled study. Surg Laparosc Endosc Percutan Tech. 2014 Apr;24(2):127-33. doi: 10.1097/SLE.0b013e3182937980.
» https://doi.org/10.1097/SLE.0b013e3182937980 -
27Erice F, Fox GS, Salib YM, Romano E, Meakins JL, Magder SA. Diaphragmatic function before and after laparoscopic cholecystectomy. Anesthesiology. 1993 Nov;79(5):966-75. PMID: 8239015.
-
28Agrelli TF, de Carvalho Ramos M, Guglielminetti R, Silva AA, Crema E. Preoperative ambulatory inspiratory muscle training in patients undergoing esophagectomy. A pilot study. Int Surg. 2012 Jul-Sep;97(3):198-202. doi: 10.9738/CC136.1.
» https://doi.org/10.9738/CC136.1 -
29Cosa D, Gonçalves HA, Lima LP, Ike D, Cancelliero KM, Montebelo MI. New reference values for maximal respiratory pressures in the Brazilian population: corrections. J Bras Pneumol. 2010 Sep-Oct;36(5):667. PMID: 21085835.
-
Financial sources:
FAPEMIG, CNPq, FUNEPU, and UFTM
-
1
Research performed at Department of Surgery, University Hospital, Universidade Federal do Triângulo Mineiro (UFTM), Uberaba-MG, Brazil.
Publication Dates
-
Publication in this collection
Oct 2017
History
-
Received
16 June 2017 -
Reviewed
14 Aug 2017 -
Accepted
18 Sept 2017