Abstract
Purpose:
To investigate whether oxymatrine (OMT) prevents hepatic fibrosis in rats by regulating liver transforming growth factor β1 (TGF-β1) level.
Methods:
Hepatic fibrosis was induced in rats by thioacetamide (TAA). Blood was collected at the end of week 12 to determine the levels of alanine aminotransferase (ALT), aspartate aminotransferase (AST), and glutathione (GSH). Changes in liver tissue were observed after hematoxylin-eosin (HE) staining.
Results:
Fibrosis was confirmed by Masson’s collagen staining. Liver TGF-β1 level was determined by ELISA. OMT significantly reduced serum ALT and AST but increased GSH levels in rats with hepatic fibrosis. Moreover, it significantly improved liver histology in rats with TAA-induced hepatic fibrosis. It significantly decreased liver TGF-β1 level compared to that in the untreated group. It also significantly reduced collagen deposition in rats.
Conclusion:
Oxymatrine is effective in protecting rats from thioacetamide-induced hepatic fibrosis by regulating TGF-β1 expression.
Key words:
Thioacetamide; Fibrosis; Liver; Transforming Growth Factor beta 1; Rats.
Introduction
Liver fibrosis is characterized by the disruption of liver cell structure and function. It may occur due to a variety of pathogenic factors that may cause liver cell necrosis and inflammatory cell infiltration, resulting in diffuse liver disease due to the excessive deposition of collagen and an imbalance in extracellular matrix (ECM)11 Schwabe R, Bataller R. Liver fibrosis. Semin Liver Dis. 2015;35:95-6. doi: 10.1055/s-0035-1550053.
https://doi.org/10.1055/s-0035-1550053...
. The dysregulation of transforming growth factor β (TGF-β) signaling is implicated in liver fibrosis. It causes cancer in p53 knockout mice22 Morris SM, Baek JY, Koszarek A, Kanngurn S, Knoblaugh SE, Grady WM. Transforming growth factor-beta signaling promotes hepatocarcinogenesis induced by p53 loss. Hepatology. 2012;55:121-31. doi: 10.1002/hep.24653.
https://doi.org/10.1002/hep.24653...
. TGF-β turns stem cells cancerous by stimulating lysophosphatidylcholine, thereby causing liver cancer33 Wu K, Ding J, Chen C, Sun W, Ning BF, Wen W, Huang L, Han T, Yang W, Wang C, Li Z, Wu MC, Feng GS, Xie WF, Wang HY. Hepatic transforming growth factor beta gives rise to tumor-initiating cells and promotes liver cancer development. Hepatology. 2012;56:2255-67. doi: 10.1002/hep.26007.
https://doi.org/10.1002/hep.26007...
. Traditional Chinese medicines, such as prunella44 Hu YX, Yu CH, Wu F, Yu WY, Zhong YS, Ying HZ, Yu B. Antihepatofibrotic effects of aqueous extract of Prunella vulgaris on carbon tetrachloride-induced hepatic fibrosis in rats. Planta Med. 2016;82:97-105. doi: 10.1055/s-0035-1558112.
https://doi.org/10.1055/s-0035-1558112...
, skullcap55 Sun H, Che QM, Zhao X, Pu XP. Antifibrotic effects of chronic baicalein administration in a CCL4 liver fibrosis model in rats. Eur J Pharmacol. 2010;631:53-60. doi: 10.1016/j.ejphar.2010.01.002.
https://doi.org/10.1016/j.ejphar.2010.01...
, and resveratrol66 Ahmad A, Ahmad R. Resveratrol mitigate structural changes and hepatic stellate cell activation in N'-nitrosodimethylamine-induced liver fibrosis via restraining oxidative damage. Chem Biol Interact. 2014;221:1-12. doi: 10.1016/j.cbi.2014.07.007.
https://doi.org/10.1016/j.cbi.2014.07.00...
, protect against hepatic fibrosis. Oxymatrine (OMT) is an alkaloid extracted from the roots of the Chinese herb Sophora subprostrata, and has anti-inflammatory, anti-bacterial, anti-viral, immunosuppressive, and anticancer potential. It effectively reduces alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels in chronic hepatitis, decreases apoptosis, reduces inflammatory cell infiltration, and liver cell necrosis, thereby preventing and treating hepatic fibrosis in rats77 Zeli G, Xiaoli L, Guoyu C. Clinical evaluation of hepatic fibrosis in chinese patients with nonalcoholic fatty liver disease with FibroScan. Chin Med. 2015;6:115-23.,88 Trebicka J, Anadol E, Elfimova N, Strack I, Roggendorf M, Viazov S, Wedemeyer I, Drebber U, Rockstroh J, Sauerbruch T, Dienes HP, Odenthal M. Hepatic and serum levels of miR-122 after chronic HCV-induced fibrosis. J Hepatol. 2013;58:234-9. doi: 10.1016/j.jhep.2012.10.015.
https://doi.org/10.1016/j.jhep.2012.10.0...
.
Therefore, the aim of the present study was to investigate whether OMT counters hepatic fibrosis by regulating TGF-β1 signaling, and to provide evidence for its clinical application.
Methods
This study was carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health. The animal use protocol has been reviewed and approved by the Institutional Animal Care and Use Committee (IACUC) of Ningxia Medical University.
Sixty Sprague-Dawley rats weighing 250±50g, were provided by the Experimental Animal Center of Ningxia Medical University [experimental animal production license number: SCXK (Ning) 2005-0001]. The rats were housed under standard conditions for one week with free access to water before the experiment.
Drugs and reagents
OMT (National Institutes for Food and Drug Control, Lot 201508) was diluted to the desired concentration with saline before use. Thioacetamide (TAA) was purchased from Aladdin Chemistry Co. Ltd (batch number: 43071). The serum levels of ALT and AST were determined spectrophotometrically using commercial kits (Huaxin Technology Co., Ltd., Panyu District, Guangzhou; lot numbers: 170814 and 170706) on an automatic biochemical analyzer (Japan SYSMEX, XT-2000i). ALT catalyzes the transamination of L-alanine and α-ketoglutarate to produce pyruvate and L-glutamate. When LDH is present in the reaction system, pyruvate and β-NADH react to form L-lactic acid and β-NAD, causing a decrease in the absorbance at 340 nm, which is positively proportional to ALT activity. AST catalyzes the transamination of α-glutaric acid and L-aspartic acid to produce L-glutamic acid and oxaloacetic acid. When MDH is present in the reaction system, oxaloacetic acid and β-NADH react to form L-malic acid and β-NAD. In the reaction, due to the formation of oxidized β-NAD from β-NADH, the absorbance at 340 nm decreases with AST activity. Trace enzyme-labeling method was used to determine glutathione (GSH) level using a commercial kit (Nanjing Jiancheng Bioengineering Institute, batch number: 20171026) and read on a microplate reader. GSH reacts with dithiobis-dinitrobenzoic acid to produce a yellow compound that can be detected at 405 nm. TGF-β1 kit was purchased from Shanghai Senxiong Biotech Co. Ltd.
Animal grouping and processing
Sixty healthy Sprague-Dawley rats were randomized into six groups of 10 each-normal, model (untreated), OMT treatment (high, medium, and low doses), and silymarin positive control groups. Except for the normal group, hepatic fibrosis was induced in all rats by intraperitoneal injection (200 mg/kg) of TAA three times a week for two weeks, twice a week for six weeks, and then once a week for four weeks. One week after the end of TAA administration, rats in the OMT treatment groups were orally administered OMT (10, 20, or 35 mg/kg), and those in the positive control group were administered silymarin (30 mg/kg) daily for 11 weeks. At the end of week 12, the rats were killed, and blood and liver tissue samples were collected; a part of the liver tissue was cryopreserved immediately, and another part of the same liver was fixed with formaldehyde (volume fraction, 0.10), and treated with hematoxylin-eosin (HE) and Masson’s collagen-specific staining.
Determination of serum markers
After standing for 1 h at room temperature, blood samples were centrifuged at 3,000 rpm for 15 min, and serum samples were separated. The levels of ALT, AST, and GSH were determined according to the manufacturers’ instructions.
Histopathological examination
Formaldehyde-fixed tissues were embedded in paraffin, and histological sections were prepared. They underwent HE and Masson’s collagen staining, and were observed for histopathological changes under a light microscope (magnification, 100×). Semi-quantitative analysis was conducted based on the results of Masson’s staining.
Determination of TGF-β1 level
TGF-β1 level in liver tissue homogenates was determined according to the kit manufacturer’s instructions.
Statistical analysis
SPSS v11.5 software was used for statistical analysis. Normally distributed values are presented as the mean±standard deviation (SD); differences were compared with one-way ANOVA and the Student’s t test. Ranked values were compared by the nonparametric rank sum test. P<0.05 was considered as statistically significant.
Results
Effects of OMT on serum ALT and AST levels in rats with TAA-induced hepatic fibrosis
The results are shown in Table 1. Serum ALT and AST levels increased significantly in the model group compared to those in the normal group (P<0.01), and significantly decreased in both the OMT treatment and positive control groups compared to those in the model group (P<0.05).
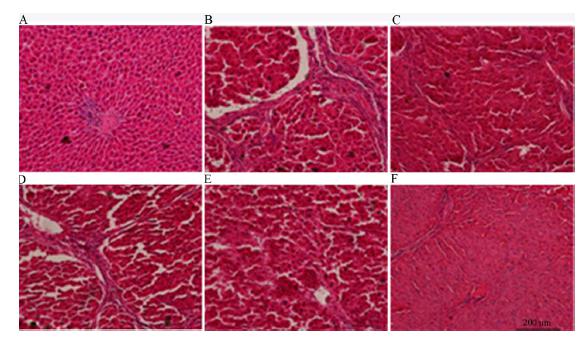
Effects of OMT on liver histopathology in rats with TAA-induced hepatic fibrosis
HE staining
Histopathological observations after HE staining are shown in Figure 1. Images of the normal group revealed structurally intact hepatic lobules, no expansion of central venous and venous sinusoids, the regular distribution of liver cells, and no degeneration or fibrosis. Fibrous deposition spreading outside the central lobule and the formation of pseudolobules were observed in the model group. OMT and silymarin treatment reduced fibrous deposition, the thickness of the fibrous septa, inflammatory cell infiltration, and the number of fat vacuoles.
Masson’s staining
Histopathological observations after Masson’s collagen staining are shown in Figure 2 and Table 2. Images of the normal group reveal structurally intact hepatic lobules, the regular distribution of liver cells, and only a small part of the vascular wall stained blue. Model group images indicated a high level of fibrous deposition, which was stained blue, thick fibrous septa, significant pseudolobule formation, and numerous fat vacuoles. OMT and silymarin treatment reduced fibrous deposition, the thickness of the fibrous septa, and inflammatory cell infiltration, and the number of fat vacuoles.
Hepatic fibrosis grading standard
The results are shown in Table 2. The severity of hepatic fibrosis was graded; 0: no fibrosis; 1: mild fibrosis, with fibrous deposition only in the central lobule; 2: moderate fibrosis, with fibrous deposition extended outside the central lobule but not to the lobular margin; 3: severe fibrosis, with fibrous deposition extended to the lobular margin; 4: early fibrosis.
Effects of OMT on TGF-β1 level in liver tissue homogenates in rats with TAA-induced hepatic fibrosis
Almost no expression of TGF-β1 was observed in normal liver tissue. The expression of TGF-β1 was significantly higher in liver tissues of rats with TAA-induced hepatic fibrosis than in liver tissues of normal rats (P<0.01), and it significantly decreased after OMT treatment (P<0.01, Table 3).
Effects of OMT on GSH levels in liver tissue homogenates in rats with TAA-induced hepatic fibrosis
Compared with the normal group, GSH levels were significantly decreased in liver tissues of rats with TAA-induced hepatic fibrosis (P<0.05), and were significantly increased after OMT treatment (P<0.05, Table 4).
Discussion
Liver fibrosis is a common pathological basis for all chronic liver diseases culminating into cirrhosis99 Wallace K, Burt AD, Wright MC. Liver fibrosis. Biochem J. 2008;411:1-18. doi: 10.1042/BJ20071570.
https://doi.org/10.1042/BJ20071570...
,1010 Rockey DC. Antifibrotic therapy in chronic liver disease. Clin Gastroenterol Hepatol. 2005;3:95-107. doi: 10.1016/S1542-3565(04)00445-8.
https://doi.org/10.1016/S1542-3565(04)00...
. TAA-induced rat hepatic fibrosis model is a classic model for studying liver damage and liver fibrosis. TAA causes lipid destruction and protein denaturation on the cell membrane of rat liver cells, leading to intracellular enzyme leakage and increased serum ALT and AST levels1111 Niu L, Cui X, Qi Y, Xie D, Wu Q, Chen X, Ge J, Liu Z. Involvement of TGF-ß1/Smad3 signaling in carbon tetrachloride-induced acute liver injury in mice. PLoS One. 2016;11:1-18. doi: 10.1371/journal.pone.0156090.
https://doi.org/10.1371/journal.pone.015...
. ALT and AST levels correlated directly with the severity of hepatic fibrosis and cirrhosis. A combination of ferulic acid and OMT significantly reduces serum ALT and AST levels in alcohol-induced liver injury in rats1212 Pei X, Wang W, Miao N, Xu M, Zhang C, Sun M, Xu M, Liu Z. The protective effects of the combination of sodium ferulate and oxymatrine on ethanol-induced liver damage in mice. Environ Toxicol Pharmacol. 2014;37:423-30. doi: 10.1016/j.etap.2013.12.005.
https://doi.org/10.1016/j.etap.2013.12.0...
. Indoline derivatives significantly reduce serum ALT and AST levels in an acute liver injury mouse model1313 Finkin-Groner E, Finkin S, Zeeli S, Weinstock M. Indoline derivatives mitigate liver damage in a mouse model of acute liver injury. Pharmacol Rep. 2017;69:894-902. doi: 10.1016/j.pharep.2017.03.025.
https://doi.org/10.1016/j.pharep.2017.03...
. Acute liver injury was identified by significantly elevated serum ALT and AST levels in the present study. Compared with the model group, each dose of OMT significantly decreased these levels, indicating that OMT at all doses reduces liver injury, improves liver function, and delays the onset of liver fibrosis.
As a member of the antioxidant system and a free radical scavenger, GSH attenuates the toxic effects of various peroxides; however, it may be overwhelmed by excessive oxidative stress1414 Batool R, Khan MR, Majid M. Euphorbia dracunculoides L. abrogates carbon tetrachloride induced liver and DNA damage in rats. BMC Complement Altern Med. 2017;17:223. doi: 10.1186/s12906-017-1744-x.
https://doi.org/10.1186/s12906-017-1744-...
. Therefore, decreased GSH level is a complementary indicator of the extent of liver injury. Serum ALT and AST levels increase significantly in CCl4-induced liver injury in rats, whereas GSH levels significantly decrease1515 Younis T, Khan MR, Sajid M. Protective effects of Fraxinus xanthoxyloides (wall.) leaves against CCl4 induced hepatic toxicity in rat. BMC Complement Altern Med. 2016;16:407. doi: 10.1186/s12906-016-1398-0.
https://doi.org/10.1186/s12906-016-1398-...
. Galloyl anhydride from Hypericum perforatum significantly increases serum GSH levels to prevent oxidative stress-induced rat liver injury1616 Wang P, Gao YM, Sun X, Guo N, Li J, Wang W, Yao LP, Fu YJ. Hepatoprotective effect of 2'- O -galloylhyperin against oxidative stress-induced liver damage through induction of Nrf2/ARE-mediated antioxidant pathway. Food Chem Toxicol. 2017;102:129-42. doi: 10.1016/j.fct.2017.02.016.
https://doi.org/10.1016/j.fct.2017.02.01...
. In the present study, OMT restored the reduced serum GSH levels in a dose-dependent manner. TAA-induced liver fibrosis in rats causes hepatocytes to rapidly utilize GSH to counter the initial surge in toxic free radicals produced1414 Batool R, Khan MR, Majid M. Euphorbia dracunculoides L. abrogates carbon tetrachloride induced liver and DNA damage in rats. BMC Complement Altern Med. 2017;17:223. doi: 10.1186/s12906-017-1744-x.
https://doi.org/10.1186/s12906-017-1744-...
, resulting in significantly reduced serum GSH levels in the model group. OMT remarkably increased rat serum GSH level in a dose-dependent manner, thereby protecting against liver injury and delaying the onset of liver fibrosis. Toxic free radicals were reduced, and a reduced amount of GSH was consumed by the hepatocytes, thereby increasing GSH level.
TGF-β1 is a strong pro-collagenogenic factor and contributes to the development of hepatic fibrosis1717 Yao X, Xu K. Compiled: the basic and clinical of liver fibrosis. Shanghai: Shanghai Science and Technology Education Press; 2003.,1818 Derynck R, Zhang YE. Smad-dependent and Smad-independent pathways in TGF-b family signaling. Nature. 2003;425:577-84. doi: 10.1038/nature02006.
https://doi.org/10.1038/nature02006...
. Moreover, the expression of TGF-β1 in liver tissues correlates with the pathological degree of liver fibrosis1919 Yoshida K, Murata M, Yamaguchi T, Matsuzaki K. TGF-ß/Smad signaling during hepatic fibro-carcinogenesis (Review). Int J Oncol. 2014;45:1363-71. doi: 10.3892/ijo.2014.2552.
https://doi.org/10.3892/ijo.2014.2552...
. Liver TGF-β1 level increases significantly due to hepatic fibrosis caused by Echinococcus granulosus infection2020 Liu Y, Abudounnasier G, Zhang T, Liu X, Wang Q, Yan Y, Ding J, Wen H, Yimiti D, Ma X. Increased expression of TGF-ß1 in correlation with liver fibrosis during Echinococcus granulosus infection in mice. Korean J Parasitol. 2016;54:519-25. doi: 10.3347/kjp.2016.54.4.519.
https://doi.org/10.3347/kjp.2016.54.4.51...
. It also significantly increases in CCl4-induced rat liver fibrosis; however, OMT restores TGF-β1 level after 4-8 weeks, thereby reducing the pathological degree of liver fibrosis2121 Yu JL, Li JH, Chengz RG, Ma YM, Wang XJ, Liu JC. Effect of matrine on transforming growth factor ß1 and hepatocyte growth factor in rat liver fibrosis model. Asian Pac J Trop Med. 2014;7:390-3. doi: 10.1016/S1995-7645(14)60062-6.
https://doi.org/10.1016/S1995-7645(14)60...
. However, OMT is also reported to be toxic and aggravates liver damage, despite its anti-liver fibrosis effect2222 Lu H, Zhang L, Gu LL, Hou BY, Du GH. oxymatrine induces liver injury through JNK Signalling pathway mediated by TNF-a in vivo. Basic Clin Pharmacol Toxicol. 2016;119:405-11. doi: 10.1111/bcpt.12608.
https://doi.org/10.1111/bcpt.12608...
. It is well known that the drug is a double-edged sword that it has some toxic effects though it has therapeutic effects. Accordingly, TGF-β1 levels in rats treated with 90 and 45 mg/kg of OMT were higher than those in rats treated with 22.5 mg/kg OMT. Thus, we reduced the dose of OMT in the present study. In this experiment, TGF-β1 was not expressed in the liver tissues of normal rats, whereas its expression was significantly increased in the liver tissues of rats in the model group (P<0.01). OMT significantly downregulated the expression of TGF-β1 in rat liver tissues (P<0.01). The regulatory effect of TGF-β1 on hepatic fibrosis is mediated by intracellular signal transduction pathways, and the Smad family is the only TGF-β1 substrate family identified1717 Yao X, Xu K. Compiled: the basic and clinical of liver fibrosis. Shanghai: Shanghai Science and Technology Education Press; 2003.. After receptor binding, TGF-β1 activates target cells through the Smad signaling pathway to exert biological effects. The TGF-β1/Smad signaling pathway induces hepatic stellate cell activation and ECM production, and contributes to the development of hepatic fibrosis1919 Yoshida K, Murata M, Yamaguchi T, Matsuzaki K. TGF-ß/Smad signaling during hepatic fibro-carcinogenesis (Review). Int J Oncol. 2014;45:1363-71. doi: 10.3892/ijo.2014.2552.
https://doi.org/10.3892/ijo.2014.2552...
20 Liu Y, Abudounnasier G, Zhang T, Liu X, Wang Q, Yan Y, Ding J, Wen H, Yimiti D, Ma X. Increased expression of TGF-ß1 in correlation with liver fibrosis during Echinococcus granulosus infection in mice. Korean J Parasitol. 2016;54:519-25. doi: 10.3347/kjp.2016.54.4.519.
https://doi.org/10.3347/kjp.2016.54.4.51...
21 Yu JL, Li JH, Chengz RG, Ma YM, Wang XJ, Liu JC. Effect of matrine on transforming growth factor ß1 and hepatocyte growth factor in rat liver fibrosis model. Asian Pac J Trop Med. 2014;7:390-3. doi: 10.1016/S1995-7645(14)60062-6.
https://doi.org/10.1016/S1995-7645(14)60...
22 Lu H, Zhang L, Gu LL, Hou BY, Du GH. oxymatrine induces liver injury through JNK Signalling pathway mediated by TNF-a in vivo. Basic Clin Pharmacol Toxicol. 2016;119:405-11. doi: 10.1111/bcpt.12608.
https://doi.org/10.1111/bcpt.12608...
23 Tahashi Y, Matsuzaki K, Date M, Yoshida K, Furukawa F, Sugano Y, Matsushita M, Himeno Y, Inagaki Y, Inoue K. Differential regulation of TGF-beta signal in hepatic stellate cells between acute and chronic rat liver injury. Hepatology. 2002;35:49-61. doi: 10.1053/jhep.2002.30083.
https://doi.org/10.1053/jhep.2002.30083...
24 Cheng D, Li J, Zhen W, Chen Z, Wang X. Effect of exogenous transforming growth factor ß1 on activation of primary rat hepatic stellate cells. World Chinese Digest. 2007;15:211-5.
25 Wu X, Zeng W, Jang M, Qin J, Xu H. The status and significance of smads gene expression in hepatic fibrosis rats. World Chinese Digest. 2008;16:1037-41.-2626 Liu H, Wei W. TGF-ß signal pathway and anti TGFß strategies fortreatment of liver fibrosis. Chin Pharmacol Bull. 2007;23:561-5..
Histopathological examination is the gold standard for confirming a pathological condition; therefore, we performed HE and Masson’s collagen staining on liver tissues of each group. Results of histopathological examination were in accordance with quantitative results of previous experiments. TAA significantly increases TGF-β1 expression in the rat liver, which leads to the activation of myofibroblasts by stellate cells and significant hepatocyte death, causing hepatic fibrosis1414 Batool R, Khan MR, Majid M. Euphorbia dracunculoides L. abrogates carbon tetrachloride induced liver and DNA damage in rats. BMC Complement Altern Med. 2017;17:223. doi: 10.1186/s12906-017-1744-x.
https://doi.org/10.1186/s12906-017-1744-...
. HE staining confirmed fibrous deposition, which spread to the center of the leaflets and pseudolobule formation in rat liver tissues in the model group. Masson’s collagen staining showed extensive fibrous deposition (stained blue) in liver tissues of rats in the model group, and the fibrous septa were thick. Pseudolobules formed exhibited numerous fat vacuoles, which might have increased serum ALT and AST levels and decreased GSH level. OMT treatment significantly decreased serum TGF-β1 levels, fibrous deposition in rat liver tissues, the thickness of the fibrous septa, inflammatory cell infiltration, and the formation of fat vacuoles. Masson’s collagen staining also confirmed that inflammatory cell infiltration and the number of fat vacuoles were significantly reduced, fibrous deposition was alleviated, and the fibrous septa were narrowed by OMT compared with those in the model group. Decreased TGF-β1 level and TGF-β1/Smad signaling inhibit the activation of hepatic stellate cells, thereby reducing hepatic fibrosis2727 Chen Q, Zhang H, Cao Y, Li Y, Sun S, Zhang J, Zhang G. Schisandrin B attenuates CCl4-induced liver fibrosis in rats by regulation of Nrf2-ARE and TGF-ß/Smad signaling pathways. Drug Des Devel Ther. 2017;11:2179-91. doi: 10.2147/DDDT.S137507.
https://doi.org/10.2147/DDDT.S137507...
. Accordingly, histopathological examination visually verified that OMT can effectively reduce liver damage.
Conclusions
Oxymatrine significantly reduces TGF-β1 expression in rat liver tissues and prevents hepatic fibrosis. Its effects on TGF-β1 substrates, such as the Smad family, and other intracellular signaling pathways involved in hepatic fibrosis remain to be further studied.
References
-
1Schwabe R, Bataller R. Liver fibrosis. Semin Liver Dis. 2015;35:95-6. doi: 10.1055/s-0035-1550053.
» https://doi.org/10.1055/s-0035-1550053 -
2Morris SM, Baek JY, Koszarek A, Kanngurn S, Knoblaugh SE, Grady WM. Transforming growth factor-beta signaling promotes hepatocarcinogenesis induced by p53 loss. Hepatology. 2012;55:121-31. doi: 10.1002/hep.24653.
» https://doi.org/10.1002/hep.24653 -
3Wu K, Ding J, Chen C, Sun W, Ning BF, Wen W, Huang L, Han T, Yang W, Wang C, Li Z, Wu MC, Feng GS, Xie WF, Wang HY. Hepatic transforming growth factor beta gives rise to tumor-initiating cells and promotes liver cancer development. Hepatology. 2012;56:2255-67. doi: 10.1002/hep.26007.
» https://doi.org/10.1002/hep.26007 -
4Hu YX, Yu CH, Wu F, Yu WY, Zhong YS, Ying HZ, Yu B. Antihepatofibrotic effects of aqueous extract of Prunella vulgaris on carbon tetrachloride-induced hepatic fibrosis in rats. Planta Med. 2016;82:97-105. doi: 10.1055/s-0035-1558112.
» https://doi.org/10.1055/s-0035-1558112 -
5Sun H, Che QM, Zhao X, Pu XP. Antifibrotic effects of chronic baicalein administration in a CCL4 liver fibrosis model in rats. Eur J Pharmacol. 2010;631:53-60. doi: 10.1016/j.ejphar.2010.01.002.
» https://doi.org/10.1016/j.ejphar.2010.01.002 -
6Ahmad A, Ahmad R. Resveratrol mitigate structural changes and hepatic stellate cell activation in N'-nitrosodimethylamine-induced liver fibrosis via restraining oxidative damage. Chem Biol Interact. 2014;221:1-12. doi: 10.1016/j.cbi.2014.07.007.
» https://doi.org/10.1016/j.cbi.2014.07.007 -
7Zeli G, Xiaoli L, Guoyu C. Clinical evaluation of hepatic fibrosis in chinese patients with nonalcoholic fatty liver disease with FibroScan. Chin Med. 2015;6:115-23.
-
8Trebicka J, Anadol E, Elfimova N, Strack I, Roggendorf M, Viazov S, Wedemeyer I, Drebber U, Rockstroh J, Sauerbruch T, Dienes HP, Odenthal M. Hepatic and serum levels of miR-122 after chronic HCV-induced fibrosis. J Hepatol. 2013;58:234-9. doi: 10.1016/j.jhep.2012.10.015.
» https://doi.org/10.1016/j.jhep.2012.10.015 -
9Wallace K, Burt AD, Wright MC. Liver fibrosis. Biochem J. 2008;411:1-18. doi: 10.1042/BJ20071570.
» https://doi.org/10.1042/BJ20071570 -
10Rockey DC. Antifibrotic therapy in chronic liver disease. Clin Gastroenterol Hepatol. 2005;3:95-107. doi: 10.1016/S1542-3565(04)00445-8.
» https://doi.org/10.1016/S1542-3565(04)00445-8 -
11Niu L, Cui X, Qi Y, Xie D, Wu Q, Chen X, Ge J, Liu Z. Involvement of TGF-ß1/Smad3 signaling in carbon tetrachloride-induced acute liver injury in mice. PLoS One. 2016;11:1-18. doi: 10.1371/journal.pone.0156090.
» https://doi.org/10.1371/journal.pone.0156090 -
12Pei X, Wang W, Miao N, Xu M, Zhang C, Sun M, Xu M, Liu Z. The protective effects of the combination of sodium ferulate and oxymatrine on ethanol-induced liver damage in mice. Environ Toxicol Pharmacol. 2014;37:423-30. doi: 10.1016/j.etap.2013.12.005.
» https://doi.org/10.1016/j.etap.2013.12.005 -
13Finkin-Groner E, Finkin S, Zeeli S, Weinstock M. Indoline derivatives mitigate liver damage in a mouse model of acute liver injury. Pharmacol Rep. 2017;69:894-902. doi: 10.1016/j.pharep.2017.03.025.
» https://doi.org/10.1016/j.pharep.2017.03.025 -
14Batool R, Khan MR, Majid M. Euphorbia dracunculoides L. abrogates carbon tetrachloride induced liver and DNA damage in rats. BMC Complement Altern Med. 2017;17:223. doi: 10.1186/s12906-017-1744-x.
» https://doi.org/10.1186/s12906-017-1744-x -
15Younis T, Khan MR, Sajid M. Protective effects of Fraxinus xanthoxyloides (wall.) leaves against CCl4 induced hepatic toxicity in rat. BMC Complement Altern Med. 2016;16:407. doi: 10.1186/s12906-016-1398-0.
» https://doi.org/10.1186/s12906-016-1398-0 -
16Wang P, Gao YM, Sun X, Guo N, Li J, Wang W, Yao LP, Fu YJ. Hepatoprotective effect of 2'- O -galloylhyperin against oxidative stress-induced liver damage through induction of Nrf2/ARE-mediated antioxidant pathway. Food Chem Toxicol. 2017;102:129-42. doi: 10.1016/j.fct.2017.02.016.
» https://doi.org/10.1016/j.fct.2017.02.016 -
17Yao X, Xu K. Compiled: the basic and clinical of liver fibrosis. Shanghai: Shanghai Science and Technology Education Press; 2003.
-
18Derynck R, Zhang YE. Smad-dependent and Smad-independent pathways in TGF-b family signaling. Nature. 2003;425:577-84. doi: 10.1038/nature02006.
» https://doi.org/10.1038/nature02006 -
19Yoshida K, Murata M, Yamaguchi T, Matsuzaki K. TGF-ß/Smad signaling during hepatic fibro-carcinogenesis (Review). Int J Oncol. 2014;45:1363-71. doi: 10.3892/ijo.2014.2552.
» https://doi.org/10.3892/ijo.2014.2552 -
20Liu Y, Abudounnasier G, Zhang T, Liu X, Wang Q, Yan Y, Ding J, Wen H, Yimiti D, Ma X. Increased expression of TGF-ß1 in correlation with liver fibrosis during Echinococcus granulosus infection in mice. Korean J Parasitol. 2016;54:519-25. doi: 10.3347/kjp.2016.54.4.519.
» https://doi.org/10.3347/kjp.2016.54.4.519 -
21Yu JL, Li JH, Chengz RG, Ma YM, Wang XJ, Liu JC. Effect of matrine on transforming growth factor ß1 and hepatocyte growth factor in rat liver fibrosis model. Asian Pac J Trop Med. 2014;7:390-3. doi: 10.1016/S1995-7645(14)60062-6.
» https://doi.org/10.1016/S1995-7645(14)60062-6 -
22Lu H, Zhang L, Gu LL, Hou BY, Du GH. oxymatrine induces liver injury through JNK Signalling pathway mediated by TNF-a in vivo. Basic Clin Pharmacol Toxicol. 2016;119:405-11. doi: 10.1111/bcpt.12608.
» https://doi.org/10.1111/bcpt.12608 -
23Tahashi Y, Matsuzaki K, Date M, Yoshida K, Furukawa F, Sugano Y, Matsushita M, Himeno Y, Inagaki Y, Inoue K. Differential regulation of TGF-beta signal in hepatic stellate cells between acute and chronic rat liver injury. Hepatology. 2002;35:49-61. doi: 10.1053/jhep.2002.30083.
» https://doi.org/10.1053/jhep.2002.30083 -
24Cheng D, Li J, Zhen W, Chen Z, Wang X. Effect of exogenous transforming growth factor ß1 on activation of primary rat hepatic stellate cells. World Chinese Digest. 2007;15:211-5.
-
25Wu X, Zeng W, Jang M, Qin J, Xu H. The status and significance of smads gene expression in hepatic fibrosis rats. World Chinese Digest. 2008;16:1037-41.
-
26Liu H, Wei W. TGF-ß signal pathway and anti TGFß strategies fortreatment of liver fibrosis. Chin Pharmacol Bull. 2007;23:561-5.
-
27Chen Q, Zhang H, Cao Y, Li Y, Sun S, Zhang J, Zhang G. Schisandrin B attenuates CCl4-induced liver fibrosis in rats by regulation of Nrf2-ARE and TGF-ß/Smad signaling pathways. Drug Des Devel Ther. 2017;11:2179-91. doi: 10.2147/DDDT.S137507.
» https://doi.org/10.2147/DDDT.S137507
-
Financial sources:
Ningxia Natural Science Foundation (Grant No.: NZ16057) and School Level Project of Ningxia Medical University (Grant No.: XY2017182)
-
1
Research performed at Laboratory Animal Center of Ningxia Medical University, Yinchuan, China.
Publication Dates
-
Publication in this collection
Mar 2018
History
-
Received
22 Nov 2017 -
Reviewed
23 Jan 2018 -
Accepted
20 Feb 2018