Abstract
Purpose:
To investigate the cause of congenital anomalies resulted from gestational diabetes on fetal cardiac tissue in experimental animal study model.
Methods:
Totally 12 female Wistar albino rats were divided into two groups, each consisting of 6 rats. Streptozotocin (60 mg/kg) was administered intraperitoneally to the study group by dissolving in citrate solution. The rats with a blood glucose level of 200 mg/dL and above were considered to be diabetic rats. Total antioxidant status (TAS), total oxidative stress (TOS) and oxidative stress index (OSI) values were calculated in the cardiac tissues and maternal serum samples of the fetuses delivered by cesarean section after the mating process. The cardiac tissues were also subjected to histopathological examination.
Results:
TOS and OSI values in fetal cardiac tissues of the diabetic rats were found to be significantly higher than that of the control group (p=0.026 and p=0.005). Histopathological examination revealed that the mitotic index was lower and the cell organization was found to be damaged in the fetuses of the study group rats.
Conclusion:
Increased levels of free oxygen radicals considered to be due to hyperglycemia may cause congenital anomalies, especially during organogenesis period, by disrupting cell homeostasis and adversely affecting mitosis.
Key words:
Antioxidants; Oxidative Stress; Hyperglycemia; Diabetes, Gestational; Rats.
Introduction
Diabetes Mellitus (DM) is a chronic and multisystemic disease, characterized by glucose intolerance, and multiple mechanisms are responsible in its etiology. Another variant of this chronic disease, which can be explained by insufficient serum insulin level and/or the development of resistance to the insulin function in target tissues, is gestational diabetes (GDM) caused by altered hormone and metabolic status arising in pregnant women. Maternal hyperglycemia arising during pregnancy has been associated with fetal macrosomia, neonatal hypoglycemia, especially cardiac congenital anomalies and increased fetal mortality11 Yilmaz E, Celik O, Celik N, Celik E, Turkcuoglu I, Simsek Y, Minareci Y, Boz M, Aydin S. Maternal and fetal serum orexin-A levels in gestational diabetes mellitus. J Obstet Gynaecol Res. 2013;39:139-45. doi: 10.1111/j.1447-0756.2012.01955.x.
https://doi.org/10.1111/j.1447-0756.2012...
. At the same time, diabetic pregnant women also have increased the risk of hypertension, infection, cesarean section and operative delivery22 Rodrigues F, de Lucca L, Neme WS, Goncalves TL. Influence of gestational diabetes on the activity of d-aminolevulinate dehydratase and oxidative stress biomarkers. Redox Rep. 2017;17:1-5. doi: 10.1080/13510002.2017.1402981.
https://doi.org/10.1080/13510002.2017.14...
.
Although the etiology of GDM has not been fully understood, studies have shown that increased oxidative stress due to maternal hyperglycemia caused the development of fetal and maternal complications. Reactive oxygen radicals (ROR) result in the development of oxidative stress, and in cases where antioxidant mechanisms are insufficient, they lead to functional alteration or damage in structural patterns such as lipid, protein and DNA, at cellular level. It is known that this condition causes cellular damage by disrupting the sensitive balance in the cell cycle and leads to some congenital malformations characterized by GDM33 Baz B, Riveline JP, Gautier JF. Endocrinology of pregnancy. Gestational diabetes mellitus: definition, aetiological and clinical aspects. Eur J Endocrinol. 2016;174:43-51. doi: 10.1530/EJE-15-0378.
https://doi.org/10.1530/EJE-15-0378...
.
In order to obtain an objective value in studies conducted on this topic, total oxidant status (TOS) is used to assess the elevated levels of free oxygen radicals caused by maternal hyperglucosemia due to uncontrolled GDM and likewise, total antioxidant status (TAS) value is used to assess the levels of substrates in enzymatic and non-enzymatic antioxidant defense mechanisms. The oxidative stress index (OSI, TOS/TAS), which is more specific than the TOS and TAS values and assesses the oxidative state of cells more sensitively, is another marker used for this purpose44 Usluogullari B, Usluogullari CA, Balkan F, Orkmez M. Role of serum levels of irisin and oxidative stress markers in pregnant women with and without gestational diabetes. Gynecol Endocrinol. 2017;33:405-7. doi: 10.1080/09513590.2017.1284789.
https://doi.org/10.1080/09513590.2017.12...
.
We aimed to investigate the effect of hyperglycemia on fetal cardiac tissue by assessing the TAS, TOS, OSI and cardiac cell microscopy in the cardiac tissue and maternal serum of fetuses of female Wistar albino rats that were made diabetic by the administration of streptozotocin (STZ).
Methods
After obtaining Ethics Committee approval from Inonu University, 12 weeks old 12 female Wistar albino rats, approximately 200-300 gr in weight, were used in this study which was conducted by researchers with the certificate for the use of laboratory animals. After all rats were evaluated to be healthy under veterinary control, they were placed in the laboratory environment and kept for approximately 14 days for adaptation period to the environment. The standard environment in plastic cages was designed in the form of 12 hours of daytime and 12 hours of night-time periods. The room temperature was 20°C and humidity level was kept in the range of 40 and 60%. Animals access to dry fodder was standard and feeding was provided in the form of ad libitum and water was provided with tap water until the end of the study. The study was conducted in accordance with the principles of the Declaration of Helsinki for animal experiments.
Preparation of experimental animals
STZ (60 mg/kg) was administered intraperitoneally by dissolving in citrate solution in order to produce diabetes in rats. Rats with a blood glucose level of 200 mg/dL and above were considered to be diabetic55 Thornberry NA, Rano TA, Peterson EP, Rasper DM, Timkey T, Garcia-Calvo M, Houtzager VM, Nordstrom PA, Roy S, Vaillancourt JP, Chapman KT, Nicholson DW. A combinatorial approach defines specificities of members of the caspase family and granzyme B. Functional relationships established for key mediators of apoptosis. J Biol Chem. 1997;272:17907-11. doi: 10.1074/jbc.272.29.17907.
https://doi.org/10.1074/jbc.272.29.17907...
. Our study was designed in the form of 2 groups and 12 rats, 6 in each group, were used and 2 rats in both groups were excluded from the study since pregnancy could not be obtained. The next morning after mating, vaginal smear samples were taken and the presence of sperm in light microscopy was determined to be the 0th day of pregnancy. The weight gain, blood glucose levels, fluid intake and consumption of maternal rats were monitored regularly in the diabetic group and the control group over the course of their pregnancies. Following the 20th day of pregnancy, abdomens were opened by cesarean incision and the fetuses were delivered in both groups.
The number of abortion, number of newborn fetuses, weight, crown rump lenght (CRL) placental weight and possible congenital anomalies were evaluated in the diabetic group and in the control group. In newborn rats, cardiac tissues were excised by making a thoracic incision and then cardiac tissue was weighed. The cellular and nuclear alterations, mitotic index were assesed immunhistochemically in the newborn cardiac tissue by using electron microscope. Also TAS, TOS and OSI levels were assayed biochemically. Oxidative stress caused by maternal diabetes has also been studied in maternal centrifuged serum.
Measurement of blood samples
During the study blood samples, after the delivery of newborns, maternal cardiac blood samples were taken. Blood glucose concentration was determined by enzymatic assay of glucose oxidase (Ames Glucometer-Miles Laboratories Inc., Elkhart, IN, USA)
Preparation of tissue supernatants and serum samples
0.9 ml of sodium phosphate buffer (pH:7 and 100 mM) was added on 0.1 gr of fetal cardiac tissue, and then it was subjected to 3-min homogenization at 14,000 rpm in ice. Thus, 10% homogenate was prepared. This homogenate was then centrifuged at 3500 rpm at +4ºC for 15 min and the supernatants were obtained. Blood samples were taken from the maternal rats between 8-10 o’clock in the morning and the serums were separated. These supernatant and serum samples were used for TAS and TOS measurements.
Measurement of TAS
Plasma and tissue TAS levels were determined using a commercially available kit developed by Erel66 Simsek Y, Gul M, Yilmaz E, Ozerol IH, Ozerol E, Parlakpinar H. Atorvastatin exerts anti-nociceptive activity and decreases serum levels of high-sensitivity C-reactive protein and tumor necrosis factor-a in a rat endometriosis model. Arch Gynecol Obstet. 2014;290:999-1006. doi: 10.1007/s00404-014-3295-4.
https://doi.org/10.1007/s00404-014-3295-...
, (REL assay diagnostics, Mega Tip, Gaziantep, Turkey). In this method, hydroxyl radical, which is the most potent radical, is produced via Fenton reaction. In the classical Fenton reaction, the hydroxyl radical is produced by mixing of ferrous ion solution and hydrogen peroxide solution. In the most recently developed assay by Erel, the same reaction is used. In the assay, ferrous ion solution, which is present in the reagent 1, is mixed with hydrogen peroxide, which is present in the reagent 2. The sequentially produced radicals such as brown-colored dianisidinyl radical cation, produced by the hydroxyl radical, are also potent radicals. In this assay, antioxidative effect of the sample against the potent-free radical reactions, which is initiated by the produced hydroxyl radical, is measured77 Karsen H, Binici I, Sunnetcioglu M, Baran AI, Ceylan MR, Selek S, Celik H. Association of paraoxonase activity and atherosclerosis in patients with chronic hepatitis B. Afr Health Sci. 2012;12:114-8. doi: 10.4314/ahs.v12i2.6.
https://doi.org/10.4314/ahs.v12i2.6...
. The assay has got excellent precision values which are lower than 3%. The results are expressed as millimoles of Trolox (Sigma-Aldrich Chemical Co., Deutschland, Germany) equivalent per liter.
Measurement of TOS
Plasma and tissue TOS levels were determined using a commercially available kit, developed by Erel66 Simsek Y, Gul M, Yilmaz E, Ozerol IH, Ozerol E, Parlakpinar H. Atorvastatin exerts anti-nociceptive activity and decreases serum levels of high-sensitivity C-reactive protein and tumor necrosis factor-a in a rat endometriosis model. Arch Gynecol Obstet. 2014;290:999-1006. doi: 10.1007/s00404-014-3295-4.
https://doi.org/10.1007/s00404-014-3295-...
(REL assay diagnostics, Mega Tip, Gaziantep, Turkey). In this method, oxidants present in the sample oxidize the ferrous ion-odianisidine complex to ferric ion. The oxidation reaction is enhanced by glycerol molecules, which are abundantly present in the reaction medium. The ferric ion makes a colored complex with xylenol orange in an acidic medium. The color intensity, which can be measured spectrophotometrically, is related to the total amount of oxidant molecules present in the sample77 Karsen H, Binici I, Sunnetcioglu M, Baran AI, Ceylan MR, Selek S, Celik H. Association of paraoxonase activity and atherosclerosis in patients with chronic hepatitis B. Afr Health Sci. 2012;12:114-8. doi: 10.4314/ahs.v12i2.6.
https://doi.org/10.4314/ahs.v12i2.6...
. The assay is calibrated with hydrogen peroxide, and the results are expressed in micromolar hydrogen peroxide (H2O2) equivalent per liter (μmol H2O2 equiv/L) and and other derivatives of peroxides, produced physiologically in organisms and occurring in higher concentrations in the tissues under some pathologic conditions, and diffuse into plasma. The level of total peroxide was measured and expressed as TOS in this study.
Measurement of OSI
The ratio of TOS to TAS was accepted as the OSI. For calculation, the resulting unit of TAS was converted to mol/L, and the OSI value was calculated according to the following formula; OSI (arbitrary unit) = TOS (mmol H2O2 equiv/L)/TAC (mmol Trolox equiv/L)66 Simsek Y, Gul M, Yilmaz E, Ozerol IH, Ozerol E, Parlakpinar H. Atorvastatin exerts anti-nociceptive activity and decreases serum levels of high-sensitivity C-reactive protein and tumor necrosis factor-a in a rat endometriosis model. Arch Gynecol Obstet. 2014;290:999-1006. doi: 10.1007/s00404-014-3295-4.
https://doi.org/10.1007/s00404-014-3295-...
.
Histologic examination
All the specimens were also sent for histologic investigation in order to confirm the tissue diagnosis. For light microscopic evaluation, neonatal heart and brain samples were mixed in phosphate buffered 10% formalin and prepared for routine paraffin embedding. Sections of tissues were cut at 5 µm, mounted on slides, stained with hematoxylin and eosin (H&E) and examined by a Leica DFC280 light microscope and Leica Q Win Image Analyses System (Leica Microsystems Imaging Solutions, Cambridge, UK). Tissue sections were then examined microscopically by a pathologist blind to clinical data and experimental procedure. The major histopathological findings of cellular damage, vacuolation, necrosis, vascularity and leucocyte infiltration were evaluated. The histological score of the organ was calculated as the sum of the scores (0-3) given for each criterion, using the semiquantitative scale. Histological scorings were made at magnification of 40£ from 20 random fields per section from each specimen.
Statistical analysis
Statistical analysis of the data was made using the SPSS version 15.0 software for Windows. The data was checked by the Kolmogorov-Smirnov test for normal distribution. The independent samples t-test (morphometric findings, blood TOS level, tissue OSI level) was used for the comparison of normally distributed parameters, the Mann-Whitney U test and Fisher’s exact test were used for the comparison of non-normally distributed and categorical parameters (blood TAS, tissue TAS, tissue TOS, blood OSI and mitotic index) without normal distribution. A value of P<0.05 was accepted to be the limit of statistical significance.
Results
A total of 12 Wistar rats were divided into two groups, each consisting of 6 rats, and 6 rats that were made diabetic by the administration of STZ formed the study group. The mean blood glucose level of rats was 275 (68-363) mg/dL in the study group and was 89 (66-105) mg/dL in the control group . When the neonatal results were analyzed, the macrosomic effect of diabetes was clearly revealed in the study group (Table 1).
Comparison of fetal morphometric results of diabetic and control groups. CRL: Crown-rumb length.
Cardiac blood samples of the rats delivered by cesarean section were analyzed for the assessment of TAS and TOS values. The mean TOS value was found to be 1.4±2.77 in the study groupand 1.2±0.15 μmol eq./L in the control group. When the maternal serum TAS values were analyzed, the TAS value was assessed to be 0.86±0.87 mmol Trolox equivalent/L in diabetic rats and 0.58±2.53 mmol Trolox equivalent/L in the control group. The results of serum OSI were found to be 0.7 in the study group and 0.3 in the control group. When the results of maternal serum oxidant status were analyzed, it was found that the TOS and OSI values were higher in diabetic rats than in the control group but were not statistically significant.
Tissue TAS, TOS and OSI levels were assessed by measuring the cardiac tissues of fetuses delivered in the study and control groups at 530 nm with spectrophotometric method. The cardiac tissue TOS and OSI values were found to be statistically significantly higher in fetuses delivered from diabetic rats compared to the control group (Table 2).
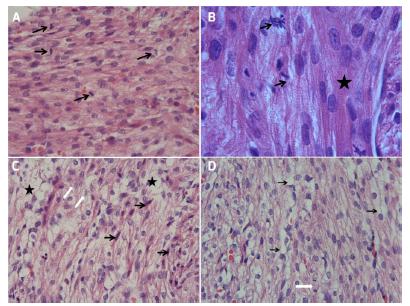
The tissues taken from the fetal cardiac tissues were also histopathologically evaluated. Centrally-localized euchromatic nuclei and eosinophilic stained granular-filamentous cytoplasmic content were noted in cardiomyocytes oriented in different directions within the myocardial layer. Longitudinal sections showed typical transverse bandings and myofibril bundles in the cytoplasm of the cardiomyocytes. Cardiac tissue examination of fetuses delivered from the diabetic rats revealed that the nuclei of cardiomyocytes had a large, euchromatic structure and chromatolysis was present in some of the nuclei. In addition, cells with heterochromatic-pyknotic nucleus were seen in some areas. The cytoplasm of the cardiomyocytes generally contained edematous large vacuoles, cytoplasmic staining intensity was reduced and their borders were irregular the cytoplasm. Hydropic degeneration was detected in the cardiomyocytes in some areas within the myocardium. Typical regular myofibril organization was not observed in the cytoplasm of the cardiomyocytes. When the mitotic indices of both groups were calculated, the mitotic index of the control group (1.22±0.28) was found to be significantly higher than that of the study group (0.47±0.42) (p=0.001).
The sections were found to have partial mitotic figures at reduced rate (Figure 1).
Examination of the cardiac tissues of fetuses of Wistar rats included in the control and study groups with Hematoxylin-Eosin stain. A. Control group light microscopy (H&E x40), black arrows demonstrate the mitotic figures. B. Group light microscopy (H&E x100), black arrows demonstrate the mitotic figures, and the star-marked area demonstrates the transverse banding in cardiomyocytes. C. Gestational diabetes group, light microscopy (H&E x40), black arrows demonstrate the pyknotic nucleus which is also the indicator of apoptosis, white arrows demonstrate the chromatolysis in nucleus, star symbol demonstrates the cytoplasmic hydropic alteration. D. Diabetes group, light microscopy (H&E x40), black arrows demonstrate the cytoplasmic vacuolation, white arrows demonstrate the advanced stage mitotic figure.
Discussion
The mitosis and apoptosis microbalance, which forms the basis of the cell cycle, can be obtained by neutralizing the formed free oxygen radicals with enzymatic and/or nonenzymatic antioxidant activities. The impairment of this cell balance, which is essential for a healthy cell cycle, in favor of free oxygen radicals is associated with many pathological conditions, especially with the formation of tumorogenesis, leading to lipid peroxidation and DNA damage88 Levine AS, Sun L, Tan R, Gao Y, Yang L, Chen H, Teng Y, Lan L. The oxidative DNA damage response: A review of research undertaken with Tsinghua and Xiangya students at the University of Pittsburgh. Sci China Life Sci. 2017;60:1077-80. doi: 10.1007/s11427-017-9184-6.
https://doi.org/10.1007/s11427-017-9184-...
. In studies conducted, the oxidative damage has been shown to have caused some congenital malformations, disrupting the structure of cell lipids, proteins, nucleic acids and hydrocarbons, especially affecting the development of tissue negatively during embryogenesis99 Gęgotek A, Nikliński J, Žarković N, Žarković K, Waeg G, Łuczaj W, Charkiewicz R, Skrzydlewska E. Lipid mediators involved in the oxidative stress and antioxidant defence of human lung cancer cells. Redox Biol. 2016;9:210-19. doi: 10.1016/j.redox.2016.08.010.
https://doi.org/10.1016/j.redox.2016.08....
. A study conducted by Pietryga et al.1010 Pietryga M, Dydowicz P, Tobola K, Napierala M, Miechowicz I, Gasiorowska A, Brazert M, Florek E. Selected oxidative stress biomarkers in antenatal diagnosis as 11-14 gestational weeks. Free Radic Biol Med. 2017;108:517-23. doi: 10.1016/j.freeradbiomed.2017.04.020.
https://doi.org/10.1016/j.freeradbiomed....
found that free oxygen radicals were significantly higher in the serum of women, who were found to have chromosomal anomaly and congenital fetal malformation, than that of the control group.
GDM is a chronic disorder, diagnosed during the second trimester of pregnancy and characterized by maternal hyperglycemia, leading to various congenital anomalies, especially cardiac malformations in fetus. It is believed that free oxygen radicals play a vital role in this regard, although it has still not been clearly understood how they cause congenital malformations. The levels of antioxidant enzymes and TAS value have been shown to be significantly lower in the serum of patients diagnosed with GDM than that of the control group1111 Karacay O, Sepici-Dincel A, Karcaaltincaba D, Sahin D, Yalvac S, Akyol M, Kandemir O, Altan N. A quantitative evaluation of total antioxidant status and oxidative stress markers in preeclampsia and gestational diabetic patients in 24-36 weeks of gestation. Diabetes Res Clin Pract. 2010;89:231-8. doi: 10.1016/j.diabres.2010.04.015.
https://doi.org/10.1016/j.diabres.2010.0...
.
In the literature, the fetal effects of hyperglycemia have been tried to be explained in animal models that were made diabetic by injecting STZ which causes direct damage in pancreatic β-cells. In a study conducted on Wistar rats that were experimentally made diabetic, the levels of antioxidant enzyme were found to be lower in fetuses found to have congenital anomaly than in the control group1212 Damasceno DC, Kiss AC, Sinzato YK, de Campos KE, Rudge MV, Calderon IM, Volpato GT. Maternal-fetal outcome, lipid profile and oxidative stress of diabetic rats neonatally exposed to streptozotocin. Exp Clin Endocrinol Diabetes. 2011;119:408-13. doi: 10.1055/s-0030-1269886.
https://doi.org/10.1055/s-0030-1269886...
. Similar results were also obtained in the study of Zhao et al.1313 Zhao J, Hakvoort TB, Willemsen AM, Jongejan A, Sokolovic M, Bradley EJ, de Boer VC, Baas F, van Kampen AH, Lamers WH. Effect of hyperglycemia on gene expression during early organogenesis in mice. PLoS One. 2016;11(7):e0158035. doi: 10.1371/journal.pone.0158035.
https://doi.org/10.1371/journal.pone.015...
, free oxygen radicals were found to be higher in the anomalous fetuses of diabetic rats in proportion to the level of hyperglycemia. An animal model study investigated the results of N-acetylcysteine administration, an antioxidant derivative, in diabetic rats showed that serum superoxide levels were significantly reduced in diabetic animals received antioxidants, and that fetal cardiac cell proliferation was improved, and that myocardial tissue thickness increased significantly1414 Moazzen H, Lu X, Ma NL, Velenosi TJ, Urquhart BL, Wisse LJ, Gittenberger-de Groot AC, Feng Q. N-Acetylcysteine prevents congenital heart defects induced by pregestational diabetes. Cardiovasc Diabetol. 2014;13:1-13. doi: 10.1186/1475-2840-13-46.
https://doi.org/10.1186/1475-2840-13-46...
.
Free oxygen radicals that form in fetuses of diabetic rats and are found at higher rates in other malformed organs, especially in cardiac organs, can be directly associated with these congenital malformations. Regular mitosis function, which is responsible for the development of healthy organs, especially during organogenesis period, does not function properly due to the direct effect of free radicals. These radicals directly affect the DNA structure of cell and cause mitotic arrest, which ultimately leads to cell vacuolation, chromatolysis and pyknotic nucleus development, negatively affecting the organogenesis1515 Wang GF, Dong Q, Bai Y, Yuan J, Xu Q, Cao C, Liu X. Oxidative stress induces mitotic arrest by inhibiting Aurora A-involved mitotic spindle formation. Free Radic Biol Med. 2017;103:177-87. doi: 10.1016/j.freeradbiomed.2016.12.031.
https://doi.org/10.1016/j.freeradbiomed....
. In our study, we also encountered reduced mitotic indices inversely proportional to the increased free oxygen radical levels in the fetal cardiac tissues of diabetic rats.
Conclusion
Increased levels of free oxygen radicals considered to be due to hyperglycemia may cause congenital anomalies, especially during organogenesis period, by disrupting cell homeostasis and adversely affecting mitosis.
References
-
1Yilmaz E, Celik O, Celik N, Celik E, Turkcuoglu I, Simsek Y, Minareci Y, Boz M, Aydin S. Maternal and fetal serum orexin-A levels in gestational diabetes mellitus. J Obstet Gynaecol Res. 2013;39:139-45. doi: 10.1111/j.1447-0756.2012.01955.x.
» https://doi.org/10.1111/j.1447-0756.2012.01955.x -
2Rodrigues F, de Lucca L, Neme WS, Goncalves TL. Influence of gestational diabetes on the activity of d-aminolevulinate dehydratase and oxidative stress biomarkers. Redox Rep. 2017;17:1-5. doi: 10.1080/13510002.2017.1402981.
» https://doi.org/10.1080/13510002.2017.1402981 -
3Baz B, Riveline JP, Gautier JF. Endocrinology of pregnancy. Gestational diabetes mellitus: definition, aetiological and clinical aspects. Eur J Endocrinol. 2016;174:43-51. doi: 10.1530/EJE-15-0378.
» https://doi.org/10.1530/EJE-15-0378 -
4Usluogullari B, Usluogullari CA, Balkan F, Orkmez M. Role of serum levels of irisin and oxidative stress markers in pregnant women with and without gestational diabetes. Gynecol Endocrinol. 2017;33:405-7. doi: 10.1080/09513590.2017.1284789.
» https://doi.org/10.1080/09513590.2017.1284789 -
5Thornberry NA, Rano TA, Peterson EP, Rasper DM, Timkey T, Garcia-Calvo M, Houtzager VM, Nordstrom PA, Roy S, Vaillancourt JP, Chapman KT, Nicholson DW. A combinatorial approach defines specificities of members of the caspase family and granzyme B. Functional relationships established for key mediators of apoptosis. J Biol Chem. 1997;272:17907-11. doi: 10.1074/jbc.272.29.17907.
» https://doi.org/10.1074/jbc.272.29.17907 -
6Simsek Y, Gul M, Yilmaz E, Ozerol IH, Ozerol E, Parlakpinar H. Atorvastatin exerts anti-nociceptive activity and decreases serum levels of high-sensitivity C-reactive protein and tumor necrosis factor-a in a rat endometriosis model. Arch Gynecol Obstet. 2014;290:999-1006. doi: 10.1007/s00404-014-3295-4.
» https://doi.org/10.1007/s00404-014-3295-4 -
7Karsen H, Binici I, Sunnetcioglu M, Baran AI, Ceylan MR, Selek S, Celik H. Association of paraoxonase activity and atherosclerosis in patients with chronic hepatitis B. Afr Health Sci. 2012;12:114-8. doi: 10.4314/ahs.v12i2.6.
» https://doi.org/10.4314/ahs.v12i2.6 -
8Levine AS, Sun L, Tan R, Gao Y, Yang L, Chen H, Teng Y, Lan L. The oxidative DNA damage response: A review of research undertaken with Tsinghua and Xiangya students at the University of Pittsburgh. Sci China Life Sci. 2017;60:1077-80. doi: 10.1007/s11427-017-9184-6.
» https://doi.org/10.1007/s11427-017-9184-6 -
9Gęgotek A, Nikliński J, Žarković N, Žarković K, Waeg G, Łuczaj W, Charkiewicz R, Skrzydlewska E. Lipid mediators involved in the oxidative stress and antioxidant defence of human lung cancer cells. Redox Biol. 2016;9:210-19. doi: 10.1016/j.redox.2016.08.010.
» https://doi.org/10.1016/j.redox.2016.08.010 -
10Pietryga M, Dydowicz P, Tobola K, Napierala M, Miechowicz I, Gasiorowska A, Brazert M, Florek E. Selected oxidative stress biomarkers in antenatal diagnosis as 11-14 gestational weeks. Free Radic Biol Med. 2017;108:517-23. doi: 10.1016/j.freeradbiomed.2017.04.020.
» https://doi.org/10.1016/j.freeradbiomed.2017.04.020 -
11Karacay O, Sepici-Dincel A, Karcaaltincaba D, Sahin D, Yalvac S, Akyol M, Kandemir O, Altan N. A quantitative evaluation of total antioxidant status and oxidative stress markers in preeclampsia and gestational diabetic patients in 24-36 weeks of gestation. Diabetes Res Clin Pract. 2010;89:231-8. doi: 10.1016/j.diabres.2010.04.015.
» https://doi.org/10.1016/j.diabres.2010.04.015 -
12Damasceno DC, Kiss AC, Sinzato YK, de Campos KE, Rudge MV, Calderon IM, Volpato GT. Maternal-fetal outcome, lipid profile and oxidative stress of diabetic rats neonatally exposed to streptozotocin. Exp Clin Endocrinol Diabetes. 2011;119:408-13. doi: 10.1055/s-0030-1269886.
» https://doi.org/10.1055/s-0030-1269886 -
13Zhao J, Hakvoort TB, Willemsen AM, Jongejan A, Sokolovic M, Bradley EJ, de Boer VC, Baas F, van Kampen AH, Lamers WH. Effect of hyperglycemia on gene expression during early organogenesis in mice. PLoS One. 2016;11(7):e0158035. doi: 10.1371/journal.pone.0158035.
» https://doi.org/10.1371/journal.pone.0158035 -
14Moazzen H, Lu X, Ma NL, Velenosi TJ, Urquhart BL, Wisse LJ, Gittenberger-de Groot AC, Feng Q. N-Acetylcysteine prevents congenital heart defects induced by pregestational diabetes. Cardiovasc Diabetol. 2014;13:1-13. doi: 10.1186/1475-2840-13-46.
» https://doi.org/10.1186/1475-2840-13-46 -
15Wang GF, Dong Q, Bai Y, Yuan J, Xu Q, Cao C, Liu X. Oxidative stress induces mitotic arrest by inhibiting Aurora A-involved mitotic spindle formation. Free Radic Biol Med. 2017;103:177-87. doi: 10.1016/j.freeradbiomed.2016.12.031.
» https://doi.org/10.1016/j.freeradbiomed.2016.12.031
-
Financial source:
none
-
1
Research performed at Animal Study Laboratory, Inonu University, Malatya, Turkey.
Publication Dates
-
Publication in this collection
Apr 2018
History
-
Received
15 Dec 2017 -
Reviewed
12 Feb 2018 -
Accepted
13 Mar 2018