Abstract
Purpose:
To evaluate the fibrosis induced by four different meshes: Marlex®, Parietex Composite®, Vicryl® and Ultrapro®.
Methods:
Histological cutouts of abdominal wall were analyzed with polarized light 28 days after the meshes implants and colorized by picrosirius to identify the intensity of collagen types I and III, and their maturation index.
Results:
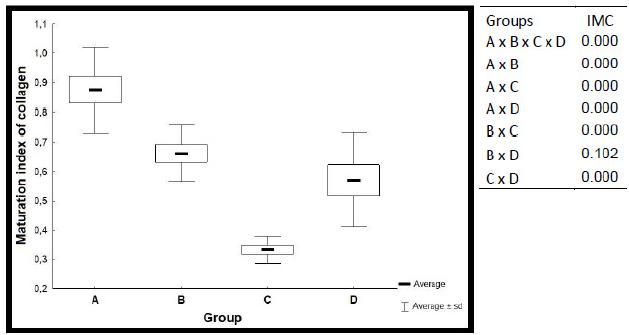
When the four groups were compared, the total collagen area analyzed was bigger in groups A and D, with no difference between them. The collagen type I density was bigger in group A, with an average of 9.62 ± 1.0, and smaller in group C, with an average of 3.86 ± 0.59. The collagen type III density was similar in groups A, B and C, and bigger in group D. The collagen maturation index was different in each of the four groups, bigger in group A with 0.87, group B with 0.66, group D with 0.57 and group C with 0.33 (p = 0.0000).
Conclusion:
The most prominent fibrosis promotion in the given meshes was found on Marlex® (polypropylene mesh) and the Parietex Composite® (non-biodegradable polyester); the collagen maturation index was higher in the Marlex® mesh, followed by Ultrapro®, Parietex Composite® and Vicryl® meshes.
Key words:
Abdominal Wall; Hernia; Fibrosis; Collagen; Surgical Mesh
Introduction
Incisional hernia, also called ventral hernia or eventration, consists in the protrusion of viscera through orifices or abdominal wall areas weakened due to trauma or surgical incisions11 de Castro Brás LE, Shurey S, Sibbons PD. Evaluation of crosslinked and non-crosslinked biologic prostheses for abdominal hernia repair. Hernia. 2012;16(1):77-89. doi: 10.1007/s10029-011-0859-0.
https://doi.org/10.1007/s10029-011-0859-...
. It is considered that one out of ten patients who had a laparotomy will develop incisional hernia22 Schreinemacher MH, Emans PJ, Gijbels MJ, Greve JW, Beets GL, Bouvy ND. Degradation of mesh coatings and intraperitoneal adhesion formation in an experimental model. Br J Surg. 2009;96(3):305-13. doi: 10.3109/13814788.2015.1055466.
https://doi.org/10.3109/13814788.2015.10...
. It is also estimated that approximately 50% of those hernias take place in the first couple of years after the surgery and up until 74% throughout the first three years33 Anthony T, Bergen PC, Kim LT, Henderson M, Fahey T, Rege RV, Turnage RH. Factors affecting recurrence following incisional herniorrhaphy. World J Surg. 2000;24(1):95-100. PMID: 10594211.,44 Rudmik LR, Schieman C, Dixon E, Debru E. Laparoscopic incisional hernia repair: a review of the literature. Hernia. 2006;10(2):110-9. PMID: 16453075.. From 1950 forth, after the introduction of the meshes usage by Usher and Wallace, this practice was intensified, which represented a great step for the ultimate treatment and the recurrence rate decrease55 Usher FC. Hernia repair with knitted polypropylene mesh. Surg Gynecol Obstet. 1963;117(2):239-40. PMID: 14048019..
The mesh works as a prop and the fibrosis formation around its lines and pores guarantees the incorporation into the tissues. The fibroplasia process consists in a harmonious and coordinated sequence of cell and molecular events which interact in order to promote the damaged tissue repair and reconstruction.
The most deployed material is still polypropylene since it has been proven to be the responsible for the boost in the abdominal wall strength, however its high porosity leads to an intense inflammatory reaction with fibrosis ensued by elasticity loss66 Utrabo CAL, Czeczko NG, Busato CR, Montemor-Netto MR, Malafaia O, Dietz UA. Comparative study between polypropylene and polypropylene/poliglecaprone meshes used in the correction of abdominal wall defect in rats. Acta Cir Bras. 2012;27( 4):300-5. doi: 10.1590/S0102-86502012000400004.
https://doi.org/10.1590/S0102-8650201200...
. One major issue lies in the intraperitoneal adherences, which might be the reason for intra or extra-hospital attendance with situations ranging from chronic pelvic pain, intestinal obstruction and women’s infertility to intestinal strangling and necrosis (under high rates of morbidity and mortality). Furthermore, the difficulties and damage risks when eventual abdominal interventions are necessary are to be incurred77 van Goor H. Consequences and complications of peritoneal adhesions. Colorectal Dis. 2007;9(Suppl 2):25-34. PMID: 17824967.,88 Tingstedt B, Anderson E, Isaksson K, Andersson R. Clinical impact of abdominal adhesions: what is the magnitude of the problem? Scand J Gastroenterol. 2008;43(3):255-61. PMID: 18938657..
Hence, an ideal material for the procedure, which must result in fair resistance to traction with no carcinogenic potential and chemically inert (bereft of infection potential), being also capable of developing sufficient inflammatory reaction and not to cause rejection, is searched. Moreover, it is also highly important to ensure it won’t trigger any allergies or hypersensitivities besides being rather low-cost and able to resist to the mechanical stress, enabling the sterilization and, last but not least, its incorporation to the host99 Vaz M, Krebs RK, Trindade EN, Trindade MRM. Fibroplasia after polypropylene mesh implantation for abdominal wall hernia repair in rats. Acta Cir Bras. 2009;24(1):19-25. doi: 10.1590/S0102-86502009000100005.
https://doi.org/10.1590/S0102-8650200900...
-10 Jin J, Voskerician G, Hunter AS, McGee MF, Cavazzola LT, Schomish S, Harth K, Rosen MJ. Human peritoneal membrane controls adhesion formation and host tissue response following intra-abdominal placement in a porcine model. J Surg Res. 2009;156(2):297-304. doi: 10.1016/j.jss.2009.04.010.
https://doi.org/10.1016/j.jss.2009.04.01...
11 Pascual G, Rodríguez M, Sotomayor S, Pérez-Köhler B, Bellón JM. Inflammatory reaction and neotissue maturation in the early host tissue incorporation of polypropylene prostheses. Hernia. 2012;16(6):697-707. PMID: 22744412.
12 Grevious MA, Cohen M, Jean-Pierre F, Herrmann GE. The use of prosthetics in abdominal wall reconstruction. Clin Plast Surg. 2006;33(2):181-97. PMID: 16638462.
13 Gaertner WB, Bonsack ME, Delaney JP. Visceral adhesions to hernia prostheses. Hernia. 2010;14(4):375-81. doi: 10.007/S10029-10-0659-y.
https://doi.org/10.007/S10029-10-0659-y...
14 Mazzini DL, Mantovani M. Fechamento da parede abdominal com afastamento parcial das bordas da aponeurose utilizando sobreposição com telas de vicryl ou marlex em ratos. Acta Cir Bras.1999;14(1):10-8. doi: 10.1590/S0102-86501999000100006.
https://doi.org/10.1590/S0102-8650199900...
15 Junqueira LCU, Montes GS, Sanches EM. The influence of tissue section thickness on the study of collagen by the Picrosirius-Polarization Method. Histochemistry, 1982;74:153-6. PMID: 7085347.
1616 van Goor H. Consequences and complications of peritoneal adhesions. Colorectal Dis. 2007;9(Suppl 2):25-34. PMID: 17824967..
Many strategies have been applied to try and avoid the complications. One option would be narrowing down the usage of polypropylene in meshes or making use of absorbable materials, which would promote an initial tension and then would be absorbed, soothing the local inflammatory process and the foreign body reaction1616 van Goor H. Consequences and complications of peritoneal adhesions. Colorectal Dis. 2007;9(Suppl 2):25-34. PMID: 17824967.. It is worth bringing up that the polypropylene mesh associated with polyglecaprone 25 was created with the purpose of leading to a reaction 65% milder in the organism as compared with traditional meshes and offering a gain about four times bigger on the resistance to the abdominal pressure1717 Halm JA, de Wall LL, Steyerberg EW, Jeekel J, Lange JF. Intraperitoneal polypropylene mesh hernia repair complicates subsequent abdominal surgery. World J Surg. 2007;31(2):423-9. PMID: 17180562.. In regards of the mesh made of polyglactin 910 filaments, totally absorbable, Gaertner et al.1313 Gaertner WB, Bonsack ME, Delaney JP. Visceral adhesions to hernia prostheses. Hernia. 2010;14(4):375-81. doi: 10.007/S10029-10-0659-y.
https://doi.org/10.007/S10029-10-0659-y...
reckoned that its usage leads to the formation of adherences, although those had a smaller area as compared to synthetic prostheses. Other studies suggested that the eventual related complications could be avoided as per this selection1010 Jin J, Voskerician G, Hunter AS, McGee MF, Cavazzola LT, Schomish S, Harth K, Rosen MJ. Human peritoneal membrane controls adhesion formation and host tissue response following intra-abdominal placement in a porcine model. J Surg Res. 2009;156(2):297-304. doi: 10.1016/j.jss.2009.04.010.
https://doi.org/10.1016/j.jss.2009.04.01...
since, for such meshes, the inflammatory foreign body reaction has shown to be diminished as compared to other materials. In comparative studies on the collagen-coated, non-biodegradable polyester, it was demonstrated that the adherences promotion occurred more enduringly than on the polypropylene mesh. Nevertheless, concerning the involved surface, a smaller adherence area as related to polypropylene was found, whence the conclusion was that the collagen layer is more efficient on the adherence avoidance. Therefore, this work aspires to the histologic comparison of the intensity of the fibrosis induced by Marlex®, Parietex Composite®, Vicryl® an Ultrapro® meshes.
Methods
Ethical analysis
Histological cutouts obtained from abdominal walls with adherences, kept in paraffin blocks and brought from a previous work analyzed and approved by the Animal Ethics Committee/Biological Sciences Department, number 802 of June 12th, 2014, were scrutinized.
The meshes, with dimensions of 10 x 20 mm, were implanted in a standardized intraperitoneal fashion and secured on the corners of the meshes with polypropylene 5.0 and the knots were placed extraperitoneally.
Polypropylene (Marlex®), polypropylene associated with polyglecaprone 25 (Ultrapro®), collagen-coated polyester (Parietex Composite®) and polyglactin 910 (Vicryl®) meshes were attached to the abdominal wall in intraperitoneal position and evaluated after 28 days. On this evaluation, the abdominal walls which contained the mesh were dried and the adherences formed were kept in paraffin blocks. The walls that were given the polypropylene (Marlex®) mesh were branded as group A; the ones that were given the polypropylene associated with polyglecaprone 25 (Ultrapro®) mesh, group B; the ones that were given the polyglactin 910 (Vicryl®) mesh, group C; and the ones that were given the collagen-coated polyester (Parietex Composite®), group D. Among the blocks, the chosen were 5-micrometer-thick histological cutouts, which were displayed in slides. In order to recognize, classify and quantify the fibrosis, the Sirius Red F3AB dye, with acid, strong collagen-reagent features, was used. The examination under polarized light allows the verification of birefringence because its molecules mate to the collagen fibrils in a way that they remain parallel. The localization of different colors and the birefringence intensity with the biochemical distribution of collagen types I and III provide means of tissue differentiation. Collagen type I shows thick fibers, strongly birefringent, in shades of yellow or red; whereas type III shows thin fibers, weakly birefringent and in shades of green1515 Junqueira LCU, Montes GS, Sanches EM. The influence of tissue section thickness on the study of collagen by the Picrosirius-Polarization Method. Histochemistry, 1982;74:153-6. PMID: 7085347..
The pictures were shot with a camera Sony® CCD101 and transmitted to the colored monitor Trinitron Sony®, then frozen and digitalized as per the lens TCX®. They were analyzed by the Image-Plus® 4.5 for Windows® program via Media Cybernetics® on a microcomputer.
From each cutout, ten fields were read and once the percentages of each collagen type were identified, they were averaged out. Afterwards, an average was also obtained from each one of the scrutinized walls. The relation collagen I/collagen III has enabled the acknowledgement of its maturation index.
Statistical analysis
The data were tabulated and submitted to statistical analysis. For the comparison amongst the groups about the collagen variables, the analysis of variance model (ANOVA) was made use of as a factor. For the comparison of the groups by two, the test used was the Least Significant Difference (LSD). It was established p≤0.05 or 5% as a level for the nullity hypothesis rejection. The data were analyzed with the computational program IBM® SPSS Statistics v.20.
Results
The abdominal walls which presented the greatest collagen concentration were the ones that were given the polypropylene meshes, followed by the ones that were given collagen-coated polyester: respectively groups A and D. The walls that received polypropylene associated with polyglecaprone 25 showed comparatively lower concentration, and the lowest concentration was found on the walls that were given polyglactin 910 (Figures 1 to 3).
Discussion
The classic incisional hernia treatment has been the correction of the fault without tension making use of meshes which, with the advent of the surgery by laparoscopic means, clung to the abdominal wall. Inasmuch as on one hand we have the perks of an earlier return to the activities and fewer abdominal wall infection occurrences, on the other hand new complications such as intraperitoneal adherences, fistulas, obstructions and infertility, besides the technical difficulties when new interventions are needed55 Usher FC. Hernia repair with knitted polypropylene mesh. Surg Gynecol Obstet. 1963;117(2):239-40. PMID: 14048019.-6 Utrabo CAL, Czeczko NG, Busato CR, Montemor-Netto MR, Malafaia O, Dietz UA. Comparative study between polypropylene and polypropylene/poliglecaprone meshes used in the correction of abdominal wall defect in rats. Acta Cir Bras. 2012;27( 4):300-5. doi: 10.1590/S0102-86502012000400004.
https://doi.org/10.1590/S0102-8650201200...
7 van Goor H. Consequences and complications of peritoneal adhesions. Colorectal Dis. 2007;9(Suppl 2):25-34. PMID: 17824967.
8 Tingstedt B, Anderson E, Isaksson K, Andersson R. Clinical impact of abdominal adhesions: what is the magnitude of the problem? Scand J Gastroenterol. 2008;43(3):255-61. PMID: 18938657.
9 Vaz M, Krebs RK, Trindade EN, Trindade MRM. Fibroplasia after polypropylene mesh implantation for abdominal wall hernia repair in rats. Acta Cir Bras. 2009;24(1):19-25. doi: 10.1590/S0102-86502009000100005.
https://doi.org/10.1590/S0102-8650200900...
10 Jin J, Voskerician G, Hunter AS, McGee MF, Cavazzola LT, Schomish S, Harth K, Rosen MJ. Human peritoneal membrane controls adhesion formation and host tissue response following intra-abdominal placement in a porcine model. J Surg Res. 2009;156(2):297-304. doi: 10.1016/j.jss.2009.04.010.
https://doi.org/10.1016/j.jss.2009.04.01...
1111 Pascual G, Rodríguez M, Sotomayor S, Pérez-Köhler B, Bellón JM. Inflammatory reaction and neotissue maturation in the early host tissue incorporation of polypropylene prostheses. Hernia. 2012;16(6):697-707. PMID: 22744412.were also found. These complications derive from the viscera rate of adherence to the wall, the foreign body reaction and the possibility of internal hernias formation. The physiological cicatrization reaction, mediated by inflammation and repair, is responsible for the prostheses integration, resulting in inflammatory cells infiltration and connective tissue deposition2525 Park JE, Barbul A. Understanding the role of immune regulation in wound healing. Am J Surg. 2004;187(5A):11S-6. doi: 10.1016/S0002-9610(03)00296-4.
https://doi.org/10.1016/S0002-9610(03)00...
.
The acute inflammatory reaction leads to an increase in the capillary permeability as well as in the leakage of exudate high in fibrinogen, whence fibrin deposition ensues2626 Hunt TK, Hopf HW. Wound healing and wound infection. What surgeons and anesthesiologists can do? Surg Clin North Am. 1997;77(3):587-606. PMID: 9194882. Klinge U, Klosterhalfen B, Müller M, Schumpelick V. Foreign body reaction to meshes used for the repair of abdominal wall hernias. Eur J Surg. 1999;165(7):665-73. PMID: 10452261.. The latter is the responsible for the signalization of inflammatory cells, culminating in the adhesion of the two of the serous surfaces, with fibroblasts infiltration, collagen deposition and neovascularization. The whole phenomenon begins in the surgical operation and ends five to seven weeks later1010 Jin J, Voskerician G, Hunter AS, McGee MF, Cavazzola LT, Schomish S, Harth K, Rosen MJ. Human peritoneal membrane controls adhesion formation and host tissue response following intra-abdominal placement in a porcine model. J Surg Res. 2009;156(2):297-304. doi: 10.1016/j.jss.2009.04.010.
https://doi.org/10.1016/j.jss.2009.04.01...
.
The technological enhancement has created prostheses with absorbable layers to inhibit the adherences. The polypropylene mesh is swathed by a dense fiber optic tissue. The fibrosis derives from the local reaction to the lesion itself and to the presence of the mesh2828 Klinge U, Klosterhalfen B, Müller M, Anurov M, Ottinger A, Schumpelick V. Influence of polyglactin-coating on functional and morphological parameters of polypropylene-mesh modifications for abdominal wall repair. Biomaterials. 1999;20(7):613-23. PMID: 10208403.. It is the outcome of a chain of cell and molecular events the tissue repair process consists of29-31 and its intensity depends on the inflammatory activity32,33. Another prosthesis, partially absorbable, is made of low-density filaments and polypropylene and polyglecaprone 25 (Ultrapro®). The polypropylene layer is incorporated and the polyglecaprone is reabsorbed within about 90 days. In a study carried out with rabbits, when polypropylene, Ultrapro® and Proceed® were applied in intraperitoneal position, there was no significant difference regarding the adherence area (p=0.134) and the inflammatory reaction, fibrosis and mesothelial reaction was similar in the three groups99 Vaz M, Krebs RK, Trindade EN, Trindade MRM. Fibroplasia after polypropylene mesh implantation for abdominal wall hernia repair in rats. Acta Cir Bras. 2009;24(1):19-25. doi: 10.1590/S0102-86502009000100005.
https://doi.org/10.1590/S0102-8650200900...
.
Trials recorded on literature have shown controversial results. Bringman et al.2828 Klinge U, Klosterhalfen B, Müller M, Anurov M, Ottinger A, Schumpelick V. Influence of polyglactin-coating on functional and morphological parameters of polypropylene-mesh modifications for abdominal wall repair. Biomaterials. 1999;20(7):613-23. PMID: 10208403., working with rabbits which polypropylene, Ultrapro® and Proceed® meshes were applied on, documented that the presence of adherence, inflammatory reaction and fibrosis was similarly intense. Klinge et al.2727 Bringman S, Conze J, Cuccurullo D, Deprest J, Junge K, Klosterhalfen B, Parra-Davila E, Ramshaw B, Schumpelick V. Hernia repair: the search for ideal meshes. Hernia. 2010;14(1):81-7. doi: 10.1007/s10029-009-0587-x.
https://doi.org/10.1007/s10029-009-0587-...
set rats polypropylene and polyglactin meshes and reported that their association contributed to the fibrosis. Pereira-Lucena et al.3030 van Amerongen MJ, Harmsen MC, Petersen AH, Kors G, van Luyn MJ. The enzymatic degradation of scaffolds and their replacement by vascularized extracellular matrix in the murine myocardium. Biomaterials. 2006; 27(10):2247-57. PMID: 16310846.also set rats polypropylene and polyglactin meshes and stated that the latter resulted in lower collagen deposition as well as its lower maturation index. Gaertner et al.1313 Gaertner WB, Bonsack ME, Delaney JP. Visceral adhesions to hernia prostheses. Hernia. 2010;14(4):375-81. doi: 10.007/S10029-10-0659-y.
https://doi.org/10.007/S10029-10-0659-y...
studied, in rats, many types of meshes. According to them, the polyglactin mesh offered the lowest collagen concentration and the polypropylene mesh, the highest concentration, whereas Parietex Composite® resulted in an intermediate concentration.
In the present experiment it was noted that the Marlex® mesh developed the most fibrosis, followed by Parietex Composite®. This result might be explained by the fact that the Marlex® mesh consists of non-absorbable material and it maintains the foreign body reaction. The same is found on the Parietex Composite® mesh because it is composed by a layer of polyester, an unabsorbable material able to keep the foreign body reaction, although it contains a collagen coat which should be in contact with the viscera. This film should be absorbed and deteriorated via neutrophil collagenase within approximately 30 days31. This way, lesser fibrosis intensity and adherences formation would show. Throughout the study hereby presented, it was observed that the polypropylene (Marlex®) mesh developed the most fibrosis and showed a maturation index higher than the Ultrapro® mesh (p =0.0000). This might have been led by the fact that the Ultrapro® mesh presents an absorbable component, polyglecaprone 25, all over its weft. The polyglactin was the one to show the least collagen density and the lowest maturation index, very likely due to its absorbable composition. These results undergird the ones previously documented by Gaertner et al.1313 Gaertner WB, Bonsack ME, Delaney JP. Visceral adhesions to hernia prostheses. Hernia. 2010;14(4):375-81. doi: 10.007/S10029-10-0659-y.
https://doi.org/10.007/S10029-10-0659-y...
.
Conclusion
Amongst the analyzed meshes, the ones that promoted most fibrosis were the polypropylene (Marlex®) and non-biodegradable polyester (Parietex Composite®); the maturation index was higher on the polypropylene (Marlex®) mesh, followed by polypropylene together with polyglecaprone 25 (Ultrapro®), non-biodegradable polyester (Parietex Composite®) and polyglactin 910 (Vicryl®).
References
-
1de Castro Brás LE, Shurey S, Sibbons PD. Evaluation of crosslinked and non-crosslinked biologic prostheses for abdominal hernia repair. Hernia. 2012;16(1):77-89. doi: 10.1007/s10029-011-0859-0.
» https://doi.org/10.1007/s10029-011-0859-0 -
2Schreinemacher MH, Emans PJ, Gijbels MJ, Greve JW, Beets GL, Bouvy ND. Degradation of mesh coatings and intraperitoneal adhesion formation in an experimental model. Br J Surg. 2009;96(3):305-13. doi: 10.3109/13814788.2015.1055466.
» https://doi.org/10.3109/13814788.2015.1055466 -
3Anthony T, Bergen PC, Kim LT, Henderson M, Fahey T, Rege RV, Turnage RH. Factors affecting recurrence following incisional herniorrhaphy. World J Surg. 2000;24(1):95-100. PMID: 10594211.
-
4Rudmik LR, Schieman C, Dixon E, Debru E. Laparoscopic incisional hernia repair: a review of the literature. Hernia. 2006;10(2):110-9. PMID: 16453075.
-
5Usher FC. Hernia repair with knitted polypropylene mesh. Surg Gynecol Obstet. 1963;117(2):239-40. PMID: 14048019.
-
6Utrabo CAL, Czeczko NG, Busato CR, Montemor-Netto MR, Malafaia O, Dietz UA. Comparative study between polypropylene and polypropylene/poliglecaprone meshes used in the correction of abdominal wall defect in rats. Acta Cir Bras. 2012;27( 4):300-5. doi: 10.1590/S0102-86502012000400004.
» https://doi.org/10.1590/S0102-86502012000400004 -
7van Goor H. Consequences and complications of peritoneal adhesions. Colorectal Dis. 2007;9(Suppl 2):25-34. PMID: 17824967.
-
8Tingstedt B, Anderson E, Isaksson K, Andersson R. Clinical impact of abdominal adhesions: what is the magnitude of the problem? Scand J Gastroenterol. 2008;43(3):255-61. PMID: 18938657.
-
9Vaz M, Krebs RK, Trindade EN, Trindade MRM. Fibroplasia after polypropylene mesh implantation for abdominal wall hernia repair in rats. Acta Cir Bras. 2009;24(1):19-25. doi: 10.1590/S0102-86502009000100005.
» https://doi.org/10.1590/S0102-86502009000100005 -
10Jin J, Voskerician G, Hunter AS, McGee MF, Cavazzola LT, Schomish S, Harth K, Rosen MJ. Human peritoneal membrane controls adhesion formation and host tissue response following intra-abdominal placement in a porcine model. J Surg Res. 2009;156(2):297-304. doi: 10.1016/j.jss.2009.04.010.
» https://doi.org/10.1016/j.jss.2009.04.010 -
11Pascual G, Rodríguez M, Sotomayor S, Pérez-Köhler B, Bellón JM. Inflammatory reaction and neotissue maturation in the early host tissue incorporation of polypropylene prostheses. Hernia. 2012;16(6):697-707. PMID: 22744412.
-
12Grevious MA, Cohen M, Jean-Pierre F, Herrmann GE. The use of prosthetics in abdominal wall reconstruction. Clin Plast Surg. 2006;33(2):181-97. PMID: 16638462.
-
13Gaertner WB, Bonsack ME, Delaney JP. Visceral adhesions to hernia prostheses. Hernia. 2010;14(4):375-81. doi: 10.007/S10029-10-0659-y.
» https://doi.org/10.007/S10029-10-0659-y -
14Mazzini DL, Mantovani M. Fechamento da parede abdominal com afastamento parcial das bordas da aponeurose utilizando sobreposição com telas de vicryl ou marlex em ratos. Acta Cir Bras.1999;14(1):10-8. doi: 10.1590/S0102-86501999000100006.
» https://doi.org/10.1590/S0102-86501999000100006 -
15Junqueira LCU, Montes GS, Sanches EM. The influence of tissue section thickness on the study of collagen by the Picrosirius-Polarization Method. Histochemistry, 1982;74:153-6. PMID: 7085347.
-
16van Goor H. Consequences and complications of peritoneal adhesions. Colorectal Dis. 2007;9(Suppl 2):25-34. PMID: 17824967.
-
17Halm JA, de Wall LL, Steyerberg EW, Jeekel J, Lange JF. Intraperitoneal polypropylene mesh hernia repair complicates subsequent abdominal surgery. World J Surg. 2007;31(2):423-9. PMID: 17180562.
-
18LeBlanc KA, Elieson MJ, Corder JM. Enterotomy and mortality rates of laparoscopic incisional and ventral hernia repair: a review of the literature. JSLS. 2007;11(4):408-14. PMID: 18237502.
-
19Castro PM, Rabelato JT, Monteiro GG, del Guerra GC, Mazzurana M, Alvarez GA. Laparoscopy versus laparotomy in the repair of ventral hernias: systematic review and meta-analysis. Arq Gastroenterol. 2014;51(3):205-11. PMID: 25296080.
-
20Anderson JM, Rodriguez A, Chang DT. Foreign body reaction to biomaterials. Semin Immunol. 2008;20(2):86-100. doi: 10.1016/j.smim.2007.11.004.
» https://doi.org/10.1016/j.smim.2007.11.004 -
21Otcu S, Ozturk H, Aldemir M, Kilinc N, Dokucu AI. Influence of the plateletactivating factor receptor antagonist BB-882 on intra-abdominal adhesion formation in rats. Eur Surg Res. 2003;35(1):41-5, 2003. PMID: 12566786.
-
22D'acampora AJ, Joli FS, Tramonte R.Expanded polytetrafluoroethylene and polypropylene in the repairing of abdominal wall defects in Wistar rats: comparative study. Acta Cir Bras. 2006;21(6):409-15. doi: 10.1590/S0102-86502006000600010.
» https://doi.org/10.1590/S0102-86502006000600010 -
23Witte MB, Barbul A. General principles of wound healing. Surg Clin North Am. 1997;77(3):509-28. PMID: 91944878.
-
24Efron DT, Most D, Shi HP, Tantry US, Barbul A. A novel method of studying wound healing. J Surg Res. 2001;98(1):16-20. PMID: 11368532.
-
25Park JE, Barbul A. Understanding the role of immune regulation in wound healing. Am J Surg. 2004;187(5A):11S-6. doi: 10.1016/S0002-9610(03)00296-4.
» https://doi.org/10.1016/S0002-9610(03)00296-4 -
26Hunt TK, Hopf HW. Wound healing and wound infection. What surgeons and anesthesiologists can do? Surg Clin North Am. 1997;77(3):587-606. PMID: 9194882. Klinge U, Klosterhalfen B, Müller M, Schumpelick V. Foreign body reaction to meshes used for the repair of abdominal wall hernias. Eur J Surg. 1999;165(7):665-73. PMID: 10452261.
-
27Bringman S, Conze J, Cuccurullo D, Deprest J, Junge K, Klosterhalfen B, Parra-Davila E, Ramshaw B, Schumpelick V. Hernia repair: the search for ideal meshes. Hernia. 2010;14(1):81-7. doi: 10.1007/s10029-009-0587-x.
» https://doi.org/10.1007/s10029-009-0587-x -
28Klinge U, Klosterhalfen B, Müller M, Anurov M, Ottinger A, Schumpelick V. Influence of polyglactin-coating on functional and morphological parameters of polypropylene-mesh modifications for abdominal wall repair. Biomaterials. 1999;20(7):613-23. PMID: 10208403.
-
29Pereira-Lucena CG, Artigiani-Neto R, Lopes Filho GJ, Frazão CV, Goldenberg A, Matos D, Linhares MM. Experimental study comparing meshes made of polypropylene, polypropylene +polyglactin and polypropylene+titanium: inflamatory cytokines, histological changes and morphometric analysis of collagen. Hernia. 2010;14(3):299-304. PMID: 20072792.
-
30van Amerongen MJ, Harmsen MC, Petersen AH, Kors G, van Luyn MJ. The enzymatic degradation of scaffolds and their replacement by vascularized extracellular matrix in the murine myocardium. Biomaterials. 2006; 27(10):2247-57. PMID: 16310846.
-
Financial source:
none
-
1
Research performed at Department of Surgical Technique and Experimental Surgery, Universidade Federal do Paraná (UFPR), Curitiba-PR, Brazil.
Publication Dates
-
Publication in this collection
Sept 2018
History
-
Received
15 May 2018 -
Reviewed
11 July 2018 -
Accepted
13 Aug 2018