Abstract
Purpose:
To develop a new 24 hour extended liver ischemia and reperfusion (LIR) model analyzing the late biochemical and histopathological results of the isolated and combined application of recognized hepatoprotective mechanisms. In addition, we used a new stratification with zoning to classify the histological lesion.
Methods:
A modified animal model of severe hepatic damage produced through 90 minutes of segmental ischemia (70% of the organ) and posterior observation for 24 hours of reperfusion, submitted to ischemic preconditioning (IPC) and topical hypothermia (TH) at 26ºC, in isolation or in combination, during the procedure. Data from intraoperative biometric parameters, besides of late biochemical markers and histopathological findings, both at 24 hours evolution time, were compared with control (C) and normothermic ischemia (NI) groups.
Results:
All groups were homogeneous with respect to intraoperative physiological parameters. There were no losses once the model was stablished. Animals subjected to NI and IPC had worse biochemical (gamma-glutamyl transpeptidase, alkaline phosphatase, lactate dehydrogenase, aspartate aminotransferase, alanine aminotransferase, direct bilirubin, and total bilirubin) and histopathological scores (modified Suzuki score) compared to those of control groups and groups with isolated or associated TH (p < 0.05).
Conclusion:
The new extended model demonstrates liver ischemia and reperfusion at 24 hour of evolution and, in this extreme scenario, only the groups subjected to topical hypothermia, combined with ischemic preconditioning or alone, had better outcomes than those subjected to only ischemic preconditioning and normothermic ischemia, reaching similar biochemical and histopathological scores to those of the control group.
Key words:
Ischemic Preconditioning; Ischemia; Reperfusion; Liver; Hypothermia; Rats
Introduction
Liver ischemia and reperfusion (LIR) has been the subject of intense study in recent decades. LIR is involved in many healthcare scenarios, including trauma, hemorrhagic shock and resuscitation, septic shock, cardiac failure, respiratory insufficiency, hepatectomy and liver transplantation11 Abu-Amara M, Yang S, Tapuria N, Fuller B, Davidson B, Seifalian A. Liver ischemia/reperfusion injury: Processes in inflammatory networks-A review. Liver Transpl Surg. 2010;16(9):1016-32. doi: 10.1002/lt.22117.
https://doi.org/10.1002/lt.22117...
. With the introduction of surgical control techniques of the hepatic vascular pedicle, today eventually still necessary during hepatic surgical procedures22 Bismuth H, Eshkenazy R, Arish A. Milestones in the Evolution of Hepatic Surgery. Rambam Maimonides Med J. 2011;2(1):1-11. doi: 10.5041/RMMJ.10021.
https://doi.org/10.5041/RMMJ.10021...
, ischemia and reperfusion have been considered key elements in postoperative morbidity and mortality since severe complications such as liver dysfunction and/or failure can result from this reperfusion process and injure other organs and systems33 Jochmans I, Meurisse N, Neyrinck A, Verhaegen M, Monbaliu D, Pirenne J. Hepatic ischemia/reperfusion injury associates with acute kidney injury in liver transplantation: prospective cohort study. Liver Transpl. 2017;23(5):634-44. doi: 10.1002/lt.24728.
https://doi.org/10.1002/lt.24728...
. The major mechanisms of such injuries are the generation of reactive oxygen species (ROS) and reactive nitrogen species (RNS)44 Prieto I, Monsalve M. ROS homeostasis, a key determinant in liver ischemic-preconditioning. Redox Biol. 2017;12:1020-5. doi: 10.1016/j.redox.2017.04.036.
https://doi.org/10.1016/j.redox.2017.04....
together with the infiltration of polymorphonuclear leukocytes among other mechanisms of cellular injury55 Quesnelle K, Bystrom P, Toledo-Pereyra L. Molecular responses to ischemia and reperfusion in the liver. Arch Toxicol. 2015;89(5):651-7. doi: 10.1007/s00204-014-1437-x.
https://doi.org/10.1007/s00204-014-1437-...
. In normothermic conditions, the liver can tolerate clamping of the vessels of its pedicle for approximately 1 hour when the liver parenchyma is normal, whereas additional protection is required with diseased livers66 van Riel W, van Golen R, Reiniers M, Heger M, van Gulik T. How much ischemia can the liver tolerate during resection? Hepatobiliary Surg Nutr. 2016;5(1):58-71. doi: 10.3978/j.issn.2304-3881.2015.07.05.
https://doi.org/10.3978/j.issn.2304-3881...
.
To minimize the effects of LIR, several methods have been studied77 Patel R, Lang J, Smith A, Crawford J, J. Redox therapeutics in hepatic ischemia reperfusion injury. World J Hepatol. 2014;6(1):1-8. doi: 10.4254/wjh.v6.i1.1.
https://doi.org/10.4254/wjh.v6.i1.1...
8 Cannistra M, Ruggiero M, Zullo A, Gallelli G, Serafini S, Maria M, Naso A, Grande R, Serra R, Nardo B. Hepatic ischemia reperfusion injury: a systematic review of literature and the role of current drugs and biomarkers. Int J Surg. 2016;33 Suppl 1:57-70. doi: 10.1016/j.ijsu.2016.05.050.
https://doi.org/10.1016/j.ijsu.2016.05.0...
-99 Talebi Z, Peyvandi H, Dabbagh A. Cellular and molecular mechanisms in perioperative hepatic protection: a review of current interventions. J Cell Mol Anesth. 2017;2(2):82-93. doi: 10.22037/jcma.v2i2.14839.
https://doi.org/10.22037/jcma.v2i2.14839...
, including ischemic preconditioning (IPC)1010 Jaeschke H, Woolbright B. Current strategies to minimize hepatic ischemia-reperfusion injury by targeting reactive oxygen species. Transplant Rev (Orlando). 2012;26(2):103-14. doi: 10.1016/j.trre.2011.10.006.
https://doi.org/10.1016/j.trre.2011.10.0...
,1111 Suyavaran A, Thirunavukkarasu C. Preconditioning methods in the management of hepatic ischemia reperfusion-induced injury: update on molecular and future perspectives. Hepatol Res. 2017;47(1):31-48. doi: 10.1111/hepr.12706.
https://doi.org/10.1111/hepr.12706...
and topical hypothermia (TH)1212 Abdo EE, Figueira ERR, Rocha-Filho JA, Chaib E, D'Albuquerque LAC, Bacchella T. Preliminary results of topical hepatic hypothermia in a model of liver ischemia/reperfusion injury in rats. Arq Gastroenterol. 2017;54(3):246-9. doi: 10.1590/s0004-2803.201700000-30.
https://doi.org/10.1590/s0004-2803.20170...
. The reduction of oxidative stress with the application of IPC is multifactorial1313 Khodosovsky MN. Effects of hypoxic preconditioning on the mechanisms of oxygen transport and oxidative damage during hepatic ischemia-reperfusion syndrome in rabbits. Int J Phys Pathophys. 2017;8(2):165-75. doi: 10.1615/IntJPhysPathophys.v8.i2.70.
https://doi.org/10.1615/IntJPhysPathophy...
,1414 Bystrom P, Foley N, Toledo-Pereyra L, Quesnelle K. Ischemic preconditioning modulates ROS to confer protection in liver ischemia and reperfusion. EXCLI J. 2017;16:483-96. doi: 10.17179/excli2017-166.
https://doi.org/10.17179/excli2017-166...
, with the activation of specific genes, production of antioxidant enzymes and production of nitric oxide, the latter being a potent vasodilator with important antagonist effects during the ischemia phase1515 Song X, Zhang N, Xu H, Cao L, Zhang H. Combined preconditioning and postconditioning provides synergistic protection against liver ischemic reperfusion injury. Int J Biol Sci. 2012;8(5):707-18. doi: 10.7150/ijbs.4231.
https://doi.org/10.7150/ijbs.4231...
,1616 Reyes-Gordillo K, Shah R, Muriel P. Oxidative stress and inflammation in hepatic diseases: Current and future therapy. Oxid Med Cell Longev. 2017;2017:1-2. doi: 10.1155/2017/3140673.
https://doi.org/10.1155/2017/3140673...
. Hypothermia achieves its objectives by inducing hypometabolism via suppressing mitochondrial activity, as an essential part of its cytoprotective effect1717 Olthof PB, Reiniers MJ, Dirkes MC, Van Gulik TM, Heger M, Van Golen RF. Protective mechanisms of hypothermia in liver surgery and transplantation. Mol Med. 2015;21:833-46. doi: 10.2119/molmed.2015.00158.
https://doi.org/10.2119/molmed.2015.0015...
,1818 Tapuria N, Junnarkar S, Abu-Amara M, Fuller B, Seifalian AM, Davidson BR. Haemoxygenase modulates cytokine induced neutrophil chemoattractant in hepatic ischemia reperfusion injury. World J Gastroenterol. 2016;22(33):7518-35. doi: 10.3748/wjg.v22.i33.7518.
https://doi.org/10.3748/wjg.v22.i33.7518...
.
The aim of this study was to compare the association of IPC with TH compared to either method alone in an LIR model with extreme injury, monitoring intraoperative physiological parameters1919 Lentsch AB. Regulatory mechanisms of injury and repair after hepatic ischemia/reperfusion. Scientifica (Cairo). 2012;2012:1-14. doi: 10.6064/2012/513192.
https://doi.org/10.6064/2012/513192...
and their possible histopathological2020 Peralta C, Jimenez-Castro MB, Gracia-Sancho J. Hepatic ischemia and reperfusion injury: effects on the liver sinusoidal milieu. J Hepatol. 2013;59(5):1094-106. doi: 10.1016/j.jhep.2013.06.017.
https://doi.org/10.1016/j.jhep.2013.06.0...
and biochemical2121 Alkozai E, Lisman T, Porte R, Nijsten M. Early elevated serum gamma glutamyl transpeptidase after liver transplantation is associated with better survival. F1000Res. 2014;3(85):1-11. doi: 10.12688/f1000research.3557.
https://doi.org/10.12688/f1000research.3...
consequences measured at 24 hours of reperfusion after ischemic insult. The application of TH, alone or in conjunction with IPC, has already been evaluated by our group in an acute animal model with partial 90-minute liver ischemia followed by 120 minutes of reperfusion and immediate euthanasia, with the demonstration of its protective effects through the reduction of inflammatory markers such as tumor necrosis factor-α (TNF-α) and interleukin (IL)-1, IL-6, and IL-12; an increase in anti-inflammatory cytokines such as IL-10; increased levels of nitric oxide; and elevated expression of Nrf2, Keap1, and NQO1 genes; all are related to antioxidant effects2222 Grezzana Filho Tde J, Mendonca TB, Gabiatti G, Kruel CD, Corso CO. Topic liver hypothermia and ischemic preconditioning: a new model of ischemia and reperfusion in rats. Acta Cir Bras. 2009;24(4):262-6. doi: 10.1590/S0102-86502009000400004.
https://doi.org/10.1590/S0102-8650200900...
23 Grezzana Filho Tde J, Mendonca TB, Gabiatti G, Rodrigues G, Marroni NA, Treis L, De Rossi SD, Corso CO. Topical hepatic hypothermia plus ischemic preconditioning: analysis of bile flow and ischemic injuries after initial reperfusion in rats. Acta Cir Bras. 2011;26(3):194-201. doi: 10.1590/S0102-86502011000300007.
https://doi.org/10.1590/S0102-8650201100...
-2424 Longo L, Sinigaglia-Fratta LX, Weber GR, Janz-Moreira A, Kretzmann NA, Grezzana-Filho Tde J, Possa-Marroni N, Corso CO, Schmidt-Cerski CT, Reverbel-da-Silveira T, Alvares-da-Silva MR, dos-Santos JL. Hypothermia is better than ischemic preconditioning for preventing early hepatic ischemia/reperfusion in rats. Ann Hepatol. 2016;15(1):110-20. doi: 10.5604/16652681.1184285.
https://doi.org/10.5604/16652681.1184285...
.
Several modifications have been made to the original partial ischemia model, especially related to the anesthesia method, surgical incision, and intra- and post-operative care, thus allowing an experimental model with extended observation time of 24 hours of reperfusion, enabling the analysis of the late effects of different cytoprotective methods (IPC and TH). The combined application of IPC and TH in a model with this time duration, both with ischemia and reperfusion, has not been described to date; however, it is fully justified considering the therapeutic plateau reached regarding the pursuit of proven effective options related to the control of experimental LIR. It should be noted that it carries a greater likelihood of clinical applicability, possibly the critical point of the current studies related to LIR, namely the application and reproduction of experimental studies in clinical care models2525 McCafferty K, Forbes S, Thiemermann C, Yaqoob MM. The challenge of translating ischemic conditioning from animal models to humans: the role of comorbidities. Dis Model Mech. 2014;7(12):1321-33. doi: 10.1242/dmm.016741.
https://doi.org/10.1242/dmm.016741...
26 van Golen RF, Reiniers MJ, Heger M, Verheij J. Solutions to the discrepancies in the extent of liver damage following ischemia/reperfusion in standard mouse models. J Hepatol. 2015;62(4):975-7. doi: 10.1016/j.jhep.2014.12.014.
https://doi.org/10.1016/j.jhep.2014.12.0...
-2727 Nadarajah L, Yaqoob MM, McCafferty K. Ischemic conditioning in solid organ transplantation: is it worth giving your right arm for? Curr Opin Nephrol Hypertens. 2017;26(6):467-76. doi: 10.1097/mnh.0000000000000367.
https://doi.org/10.1097/mnh.000000000000...
.
Methods
This study, identified by number 08486, was approved by the Bioethical Committee and funded by the Post-Graduation Group of the HCPA-UFRGS, which adheres to the rules of the Council for International Organization of Medical Sciences (CIOMS).
Thirty-three male Wistar rats (mean weight, 279g; mean age, 65 days) were divided into five groups: control (C; n = 5), normothermic ischemia (NI = 7), topical hypothermia at 26ºC (TH; n = 7), ischemic preconditioning (IPC; n = 7), and ischemic preconditioning + topical hypothermia at 26ºC (IPC+TH; n = 7).
Anesthesia protocol
The animals were subjected to inhalation anesthesia administered through a facial cannula connected to a respirator, which maintained a constant oxygen flow of 0.5 L per minute and a mixture of anesthesia with isoflurane 1-2%. The upper portion of the abdomen and the right cervical region were trichotomized and prepared with aqueous iodine solution.
Monitoring was performed through a catheter installed in the right carotid artery (PE-50; Portex, Kent, England) to control the mean blood pressure (MBP) during the entire procedure. In addition, oximetry was performed through an external sensor applied to the animal`s tail, that enabled also the recording of heart rate (HR). Saline solution (NaCl 0.9%, 2 mL/kg/h) was continuously infused into the carotid artery to replace the basal needs, compensate for hydroelectrolytic losses and maintain catheter patency. The body temperature was monitored by a rectal digital thermometer and maintained at 36-37.5ºC with the aid of a heating device of the surgical table and by external lamp heating.
Surgical procedure
Access to the abdominal cavity was attained through a median laparotomy. In group C, the hepatic lobes were isolated without any additional procedure. In the groups with ischemia (NI, IPC, TH, and IPC+TH), the liver was isolated in the operative field and an atraumatic neurosurgical clip was used to interrupt the portal and arterial blood flow to the left and middle lobes (70% of the hepatic mass) for 90 minutes, while the right and caudate lateral lobes remained uninterrupted. In the groups with IPC, the left and median lobes of the liver were isolated in the operative field and submitted to 10 minutes of ischemia, followed by reperfusion for another 10 minutes before the final clamping for 90 minutes.
In the hypothermia groups, the hepatic lobes were isolated in the operative field using a concha-like thermo-insulating styrofoam device and cooled with cold physiological solution dripped onto the surface of the ischemic lobes during the 90-minute ischemia period. The excess liquid was removed by aspirators, preventing the freezing liquid from penetrating the abdominal cavity and causing systemic hypothermia in the animals, similar to the model developed by Biberthaler et al.2828 Biberthaler P, Luchting B, Massberg S, Teupser D, Langer S, Leiderer R, Messmer K, Krombach F. The influence of organ temperature on hepatic ischemia-reperfusion injury: a systematic analysis. Transplantation. 2001;72(9):1486-90. doi: 10.1097/00007890-200111150-00003.
https://doi.org/10.1097/00007890-2001111...
. A flexible microprobe (TI-23; Braintree Scientifics) was implanted into the left lobe and the intrahepatic temperature (IHT) was monitored continuously to reach 26ºC locally (Thermalert Monitoring Thermometer TH-5; Boston Scientifics). In the other groups, IHT was also recorded at the same insertion point and with the same measurement duration.
During the entire procedure, at 15-minute intervals, vital physiological parameters such as HR, MBP, respiratory rate (RR), systemic temperature (ST), and intrahepatic temperature (IHT) were recorded. In all groups, the same parameters were recorded 1 minute after declamping of the pedicle (91-minute procedure). The percentage of isoflurane in the inhalational anesthetic mixture was also recorded during all procedures, as was the temperature of the surgical room.
Post-anesthesia procedures and sampling
After 90 minutes of ischemia, the metal clips of the pedicle were removed, and the organ was reperfused. The abdominal wall was closed, the surgical incision infiltrated with 0.1 mL of 0.5% bupivacaine solution in 0.9 mL of saline solution, and the animal was transferred to an incubator for anesthesia recovery.
At the end of 24 hours, the rats were anesthetized again with isoflurane inhalation as described in the previous procedure. The abdomen was opened and the animals were sacrificed by cardiac puncture and exsanguinating blood aspiration. The liver was removed and stored; a fraction was fixed in formalin and another fraction was frozen in nitrogen and kept in a freezer at -80ºC. The blood was immediately centrifuged and the plasma obtained frozen in liquid nitrogen and stored in a freezer at -80ºC. The liver samples were identified from the area in which they originated in each experiment: ischemic (IL), belonging to the intervention zone (left lobe and median), and non-ischemic (NIL), belonging to the hepatic segment that was protected against ischemia by pedicular clamping and the application of hypothermia (right lobe).
Plasma analysis
Serum biochemistry of the stored material was performed by the certified and specialized laboratory of the Faculty of Veterinary Medicine of the UFRGS using a WIENER-CM200 with imported reagents specific for the animal used (WIENER and RANDOX). The levels of aspartate aminotransferase (AST), alanine aminotransferase (ALT), ammonia (AMO), total bilirubin (TB) and direct bilirubin (DB), alkaline phosphatase (AP), gamma-glutamyl transpeptidase (GGT), glucose (GLU), lactate (LAC), and lactate dehydrogenase (LDH) were assessed.
Histopathological analysis
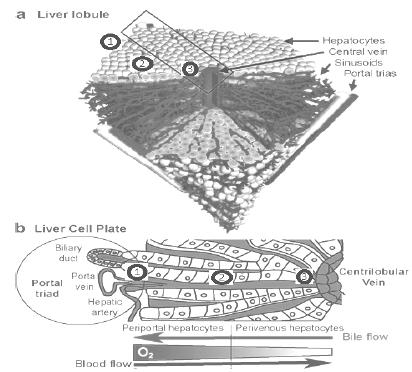
The liver samples, divided into IL and NIL from the same animal, were fixed in 10% formalin and stained with hematoxylin-eosin. The blinded evaluation of each specimen was performed by the pathology service at the Hospital de Clínicas de Porto Alegre using an optical microscope and assigning a value to the morphological changes observed following the original model proposed by Suzuki et al.2929 Suzuki S, Toledo-Pereyra LH, Rodriguez FJ, Cejalvo D. Neutrophil infiltration as an important factor in liver ischemia and reperfusion injury. Modulating effects of FK506 and cyclosporine. Transplantation. 1993;55(6):1265-72. PMID: 7685932. to examine the findings of sinusoidal congestion (SC), hydropic degeneration (HD), neutrophilic leukocyte infiltrate (NLI), and hepatocellular necrosis. The rating of steatosis (S) was added to these parameters and the analysis in the necrosis area was improved (Modified Histopathological Classification of Suzuki [MHCS]), performed in a stratified way using liver zoning (Figure 1)3030 Colnot S, Perret C. Liver zonation. In: Monga SPS, editor. Molecular pathology of liver diseases. Boston, MA: Springer US; 2011. p.7-16. doi: 10.1007/978-1-4419-7107-4_2.
https://doi.org/10.1007/978-1-4419-7107-...
, in which each lobe is divided into three zones: zone 1 (NZ1), starting from the area next to the triad portal; Zone 2 (NZ2), passing by the sinusoidal intermediate region; and Zone 3 (NZ3), in the region near the central lobular vein (Table 1).
Schematic representation of three-dimensional liver zoning. Zone 1, next to the portal triad; Zone 2, intermediate sinusoidal region; Zone 3, proximal central lobular vein. Modified from Colnot S., 2011.
The final histological score (Modified Suzuki Score [MSS] was calculated using Formula 1:
All data are presented as mean and standard deviation and were analyzed statistically using the Statistical Package for Social Sciences (SPSS) version 17 and Statistical Analysis System (SAS) version 9.4 assuming a significance of 0.05 for the tests employed (mixed-model repeated-measures analysis of variance [ANOVA], generalized linear model with a gamma distribution to evaluate the effect of groups and Kruskal-Wallis test for independent samples).
Results
Intraoperative biometric data
The analysis of intraoperative vital signs (mixed-model repeated-measures ANOVA) confirmed the homogeneity of the parameters in the groups. At 91 minutes, 1 minute after declamping of the hepatic pedicle, a uniform decrease occurred in the MBP in all groups, while a rapid elevation of the IHT in the groups submitted to hypothermia. There was no statistically significant difference in ST or other parameters between the intraoperative physiological data of the different groups. The percentage of isoflurane anesthetic mixture remained stable between groups in the different assessed moments without intergroup differences. The variables of weight, age, and surgical room temperature also showed no significant intergroup differences.
Biochemical analysis of plasma after 90 minutes of ischemia and 24 hours of reperfusion
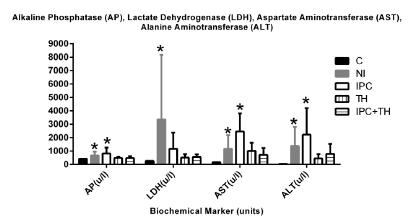
The analysis of biochemical data performed through the adjustment of a generalized linear model with a gamma distribution to evaluate the effect of groups (for asymmetric data) revealed significant differences (p<0.05) in serum GGT, AP, LDH, AST, ALT, DB, and TB (Figures 2 and 3) for the IPC and NI groups. AMO, LAC, and GLU levels were not significantly different between groups.
Levels of DB, TB and GGT in 24-hour plasma for all groups: Control (C), Normothermic Ischemia (NI), Ischemic Preconditioning (IPC), Topical Hypothermia (TH), and Ischemic Preconditioning + Topical Hypothermia (IPC+TH). Data are presented as mean and standard deviation converted to log 10. *Generalized linear model with gamma distribution to assess the group effect revealed p<0.05 for NI and IPC groups.
Levels of AP, LDH, AST and ALT in 24-hour plasma for all groups: Control (C), Normothermic Ischemia (NI), Ischemic Preconditioning (IPC), Topical Hypothermia (TH), and Ischemic Preconditioning + Topical Hypothermia (IPC+TH). Data are presented as mean and standard deviation. *Generalized linear model with gamma distribution to assess the group effect revealed p <0.05 for NI and IPC groups.
Histological analysis after 90 minutes of ischemia and 24 hours of reperfusion
The analysis of the histopathological data (through adjustment of the generalized linear model with a gamma distribution to evaluate the effect of groups) revealed significant differences (p<0.05) for the MSS in IL samples of the NI and IPC groups (Figure 4) compared to those of the other groups. Comparing individual items of the MSS among the groups, using the Kruskal-Wallis test for independent samples, significant differences were observed (p<0.05) for items NZ2 and NZ3 and for NLI in IL samples of the NI group.
MSS for ischemic liver samples of all groups: Control (C), Normothermic Ischemia (NI), Ischemic Preconditioning (IPC), Topical Hypothermia (TH), and Ischemic Preconditioning + Topical Hypothermia (IPC+TH). Data are presented as mean and standard deviation. *Generalized linear model with a gamma distribution to assess the group effect revealed p<0.05 for NI and IPC groups.
The other individual aspects of histopathological scores evaluated (SC, HD, NZ1, and S) showed no significant differences between the IL specimens. Under the point of view of analysis of the method applied in the experiment, the comparative analysis of the NIL of all groups showed no statistically significant differences in any comparisons.
When analyzed pairwise, the MSS between samples of the IL and NIL zone within the same groups demonstrated marked statistical significance for groups NI (p = 0.028) and IPC (p = 0.046), but in groups C (p = 1.0), TH (p = 0.75), and IPC+TH (p = 0.470) there were no significant differences between the IL and NIL specimens.
Discussion
Here we evaluated the effects of LIR monitored during its implementation through intraoperative physiological parameters and 24 hours after the initial insult with assessments of biochemical and histopathological parameters using different combinations of protective mechanisms (TH, IPC, IPC+TH), associating the principle of zoning in hepatic histopathological analysis (Formula 1, Table 1). Given the already expected serious injury produced by 90 minutes of liver ischemia in this type of model, the obtained biochemical and histopathological findings indicated that, in this extreme scenario, only the animals subjected to TH, with or without the application of IPC, displayed histopathological findings with obvious cytoprotection compared to those in the other groups (Figure 4). Animals subjected to NI with or without IPC displayed worse results in the MSS compared to the animals subjected to TH, with or without IPC. Considering the findings of biochemical analyses in relation to the findings of the MSS in each group, the ALT, AST, DB, TB, LDH, AP, and GGT levels were different in groups C, TH, and IPC+TH compared to those in groups NI and/or IPC, i.e., higher MSS scores were associated with higher biochemical alterations of these markers.
No references were found in the literature with an LIR model that combined 90 minutes of ischemia associated with the application of hypothermia and/or preconditioning and subsequent evolution of 24 hours during the reperfusion phase. So we can only infer some considerations of our findings and those available in other LIR models in which the ischemia phase and/or the reperfusion phase and/or the protective mechanisms tested were distinct and/or occurred at different times. In an IPC model associated with 90 minutes of IR associated with the combined prior infusion of several substances, Peralta et al.3131 Peralta C, Prats N, Xaus C, Gelpi E, Rosello-Catafau J. Protective effect of liver ischemic preconditioning on liver and lung injury induced by hepatic ischemia-reperfusion in the rat. Hepatology. 1999;30(6):1481-9. doi: 10.1002/hep.510300622.
https://doi.org/10.1002/hep.510300622...
reported a protective effect of IPC in the liver and the lungs. In an IPC model consisting of 60 minutes of ischemia and up to 24 hours of reperfusion, Jin et al.3232 Jin LM, Liu YX, Zhou L, Xie HY, Feng XW, Li H, Zheng SS. Ischemic preconditioning attenuates morphological and biochemical changes in hepatic ischemia/reperfusion in rats. Pathobiology. 2010;77(3):136-46. doi: 10.1159/000292647.
https://doi.org/10.1159/000292647...
showed a protective effect of IPC with reduced enzyme levels, a reduction in apoptosis, and improved NLI. In an IPC model consisting of 60 minutes of ischemia and 3 hours of reperfusion, Adam et al.3333 Adam AN. Some mechanisms of the protective effect of ischemic preconditioning on rat liver ischemia-reperfusion injury. Int J Gen Med. 2014;7:483-9. doi: 10.2147/IJGM.S66766.
https://doi.org/10.2147/IJGM.S66766...
also demonstrated a beneficial effect of IPC with elevated levels of hepatic glycogen compared to the group without IPC and concluded that the levels of glycogen would be inversely related to ischemia duration. In a model of pharmacological IPC (minocycline) with 90 minutes of ischemia and up to 24 hours of reperfusion, Li et al.3434 Li Y, Li T, Qi H, Yuan F. Minocycline protects against hepatic ischemia/reperfusion injury in a rat model. Biomed Rep. 2015;3(1):19-24. doi: 10.3892/br.2014.381.
https://doi.org/10.3892/br.2014.381...
showed a peak of enzymatic activity (AST, ALT, LDH) at 6 hours of reperfusion, marked histopathological changes from 6 hours of reperfusion, a progressive decline in these markers after 24 hours of evolution and a statistically significant cytoprotective effect in the group subjected to IPC. In our study, biochemical markers including bilirubin (DB and TB), transaminases (ALT and AST), alkaline phosphatase (AP), and GGT indicated significant organ lesions in the normothermic groups (NI and IPC) compared to those of the hypothermic groups (TH and IPC+TH) (Figures 2 and 3).
When analyzed separately, in this specific model, the mechanism of IPC was ineffective at promoting hepatic cytoprotection since the scores of animals subjected to IPC were similar to those of the NI group, through the histopathological assessment and the intraoperative biochemical and physiological effects. Similar findings were described by Steenks et al.3535 Steenks M, van Baal MC, Nieuwenhuijs VB, de Bruijn MT, Schiesser M, Teo MH, Callahan T, Padbury RT, Barritt GJ. Intermittent ischaemia maintains function after ischaemia reperfusion in steatotic livers. HPB (Oxford). 2010;12(4):250-61. doi: 10.1111/j.1477-2574.2010.00160.x.
https://doi.org/10.1111/j.1477-2574.2010...
in an animal model comparing IPC with intermittent ischemia in steatotic livers, in which the authors described a smaller protective effect of IPC compared to that of intermittent or continued ischemia and argued that such findings might be explained by the negative effects of cytokines and other inflammatory molecules released by the ischemic lobe subjected to IPC onto the untreated lobes. Chu et al.3636 Chu MJ, Vather R, Hickey AJ, Phillips AR, Bartlett AS. Impact of ischaemic preconditioning on experimental steatotic livers following hepatic ischaemia-reperfusion injury: a systematic review. HPB (Oxford). 2015;17(1):1-10. doi: 10.1111/hpb.12258.
https://doi.org/10.1111/hpb.12258...
, in a systematic review, described conflicting findings of some studies that evaluated the effect of IPC on steatotic livers submitted to LIR, suggesting that future experimental studies should attempt to better emulate the usual clinical scenario to provide an accurate understanding of the mechanisms and effects of IPC and identify a particular group of patients that can benefit from IPC. The scenario in which this method is most effective, likely does not include ischemia periods as long as the one used in this study. It must be also highlighted that randomized studies in humans have failed to demonstrate these beneficial effects of IPC identified in experimental models3737 O'Neill S, Leuschner S, McNally SJ, Garden OJ, Wigmore SJ, Harrison EM. Meta-analysis of ischaemic preconditioning for liver resections. Br J Surg. 2013;100(13):1689-700. doi: 10.1002/bjs.9277.
https://doi.org/10.1002/bjs.9277...
,3838 Selzner N, Boehnert M, Selzner M. Preconditioning, postconditioning, and remote conditioning in solid organ transplantation: basic mechanisms and translational applications. Transplant Rev (Orlando). 2012;26(2):115-24. doi: 10.1016/j.trre.2011.07.003.
https://doi.org/10.1016/j.trre.2011.07.0...
.
The MHCS and the MSS, variants developed from the initial proposal of their authors, were used in this project to grade and compare the histopathological findings, permitting, with this expansion of its original indicators, especially those related to the division of necrotic areas in zones from the prior knowledge concerning the functional zoning of this organ, the obtaining of more accurate data in relation to the effects of the LIR and each protective mechanism tested, thus calculating a more comprehensive and more detailed injury score upon separate analysis of the different histological areas of necrosis. Originally, the table of Suzuki et al.2929 Suzuki S, Toledo-Pereyra LH, Rodriguez FJ, Cejalvo D. Neutrophil infiltration as an important factor in liver ischemia and reperfusion injury. Modulating effects of FK506 and cyclosporine. Transplantation. 1993;55(6):1265-72. PMID: 7685932. considered a single value for the item of necrosis and did not include the degree of S. There was no significant finding for NZ1 in all groups, but the findings for NZ2 and NZ3 in isolation are significant for group NI. The same significant finding alone in Group NI is repeated with the NLI item of the score. It is speculated that the lack of significance for the findings of NZ1 in this study was due to the proximity of this zone with the triad portal, a zone with greater tissue perfusion.
The complementary analyses of the intraoperative physiological parameters of different groups (p > 0.05) between the NIL samples of different groups (p > 0.05) and the paired analysis between the IL and NIL samples within each group (p < 0.05) using the MHCS and MSS scores, making each animal as a control of oneself and the others, highlight the methodological rigor employed since the different groups have similar intraoperative physiological parameters. They also show the effectiveness of the experimental model modified from the acute original model and used in this project. The modifications developed from the original technique of the LIR model2828 Biberthaler P, Luchting B, Massberg S, Teupser D, Langer S, Leiderer R, Messmer K, Krombach F. The influence of organ temperature on hepatic ischemia-reperfusion injury: a systematic analysis. Transplantation. 2001;72(9):1486-90. doi: 10.1097/00007890-200111150-00003.
https://doi.org/10.1097/00007890-2001111...
, especially related to the anesthetic method, as already reported by Steenks et al.3535 Steenks M, van Baal MC, Nieuwenhuijs VB, de Bruijn MT, Schiesser M, Teo MH, Callahan T, Padbury RT, Barritt GJ. Intermittent ischaemia maintains function after ischaemia reperfusion in steatotic livers. HPB (Oxford). 2010;12(4):250-61. doi: 10.1111/j.1477-2574.2010.00160.x.
https://doi.org/10.1111/j.1477-2574.2010...
, in addition to the surgical incision and intra- and postoperative care, allowed for a viable extended LIR experimental model with 24 hours of evolution after severe original ischemic insult, thus enabling the analysis of the effects of different methods of cytoprotection cited previously compared between themselves and jointly in this scientific project.
Conclusions
In this extreme scenario of LIR of this new model, evaluated with 24 hours of evolution after reperfusion, we concluded that only the groups submitted to intraoperative topical hypothermia demonstrated an expected hepatoprotective effect. This LIR model performs well and opens possibilities to test other tools to get better understanding and control of this important pathological process.
Acknowledgements
All members of this research group, in addition to the personnel of the Animal Experimentation Unit of the HCPA, for their support during the study execution and data analysis.
References
-
1Abu-Amara M, Yang S, Tapuria N, Fuller B, Davidson B, Seifalian A. Liver ischemia/reperfusion injury: Processes in inflammatory networks-A review. Liver Transpl Surg. 2010;16(9):1016-32. doi: 10.1002/lt.22117.
» https://doi.org/10.1002/lt.22117 -
2Bismuth H, Eshkenazy R, Arish A. Milestones in the Evolution of Hepatic Surgery. Rambam Maimonides Med J. 2011;2(1):1-11. doi: 10.5041/RMMJ.10021.
» https://doi.org/10.5041/RMMJ.10021 -
3Jochmans I, Meurisse N, Neyrinck A, Verhaegen M, Monbaliu D, Pirenne J. Hepatic ischemia/reperfusion injury associates with acute kidney injury in liver transplantation: prospective cohort study. Liver Transpl. 2017;23(5):634-44. doi: 10.1002/lt.24728.
» https://doi.org/10.1002/lt.24728 -
4Prieto I, Monsalve M. ROS homeostasis, a key determinant in liver ischemic-preconditioning. Redox Biol. 2017;12:1020-5. doi: 10.1016/j.redox.2017.04.036.
» https://doi.org/10.1016/j.redox.2017.04.036 -
5Quesnelle K, Bystrom P, Toledo-Pereyra L. Molecular responses to ischemia and reperfusion in the liver. Arch Toxicol. 2015;89(5):651-7. doi: 10.1007/s00204-014-1437-x.
» https://doi.org/10.1007/s00204-014-1437-x -
6van Riel W, van Golen R, Reiniers M, Heger M, van Gulik T. How much ischemia can the liver tolerate during resection? Hepatobiliary Surg Nutr. 2016;5(1):58-71. doi: 10.3978/j.issn.2304-3881.2015.07.05.
» https://doi.org/10.3978/j.issn.2304-3881.2015.07.05 -
7Patel R, Lang J, Smith A, Crawford J, J. Redox therapeutics in hepatic ischemia reperfusion injury. World J Hepatol. 2014;6(1):1-8. doi: 10.4254/wjh.v6.i1.1.
» https://doi.org/10.4254/wjh.v6.i1.1 -
8Cannistra M, Ruggiero M, Zullo A, Gallelli G, Serafini S, Maria M, Naso A, Grande R, Serra R, Nardo B. Hepatic ischemia reperfusion injury: a systematic review of literature and the role of current drugs and biomarkers. Int J Surg. 2016;33 Suppl 1:57-70. doi: 10.1016/j.ijsu.2016.05.050.
» https://doi.org/10.1016/j.ijsu.2016.05.050 -
9Talebi Z, Peyvandi H, Dabbagh A. Cellular and molecular mechanisms in perioperative hepatic protection: a review of current interventions. J Cell Mol Anesth. 2017;2(2):82-93. doi: 10.22037/jcma.v2i2.14839.
» https://doi.org/10.22037/jcma.v2i2.14839 -
10Jaeschke H, Woolbright B. Current strategies to minimize hepatic ischemia-reperfusion injury by targeting reactive oxygen species. Transplant Rev (Orlando). 2012;26(2):103-14. doi: 10.1016/j.trre.2011.10.006.
» https://doi.org/10.1016/j.trre.2011.10.006 -
11Suyavaran A, Thirunavukkarasu C. Preconditioning methods in the management of hepatic ischemia reperfusion-induced injury: update on molecular and future perspectives. Hepatol Res. 2017;47(1):31-48. doi: 10.1111/hepr.12706.
» https://doi.org/10.1111/hepr.12706 -
12Abdo EE, Figueira ERR, Rocha-Filho JA, Chaib E, D'Albuquerque LAC, Bacchella T. Preliminary results of topical hepatic hypothermia in a model of liver ischemia/reperfusion injury in rats. Arq Gastroenterol. 2017;54(3):246-9. doi: 10.1590/s0004-2803.201700000-30.
» https://doi.org/10.1590/s0004-2803.201700000-30 -
13Khodosovsky MN. Effects of hypoxic preconditioning on the mechanisms of oxygen transport and oxidative damage during hepatic ischemia-reperfusion syndrome in rabbits. Int J Phys Pathophys. 2017;8(2):165-75. doi: 10.1615/IntJPhysPathophys.v8.i2.70.
» https://doi.org/10.1615/IntJPhysPathophys.v8.i2.70 -
14Bystrom P, Foley N, Toledo-Pereyra L, Quesnelle K. Ischemic preconditioning modulates ROS to confer protection in liver ischemia and reperfusion. EXCLI J. 2017;16:483-96. doi: 10.17179/excli2017-166.
» https://doi.org/10.17179/excli2017-166 -
15Song X, Zhang N, Xu H, Cao L, Zhang H. Combined preconditioning and postconditioning provides synergistic protection against liver ischemic reperfusion injury. Int J Biol Sci. 2012;8(5):707-18. doi: 10.7150/ijbs.4231.
» https://doi.org/10.7150/ijbs.4231 -
16Reyes-Gordillo K, Shah R, Muriel P. Oxidative stress and inflammation in hepatic diseases: Current and future therapy. Oxid Med Cell Longev. 2017;2017:1-2. doi: 10.1155/2017/3140673.
» https://doi.org/10.1155/2017/3140673 -
17Olthof PB, Reiniers MJ, Dirkes MC, Van Gulik TM, Heger M, Van Golen RF. Protective mechanisms of hypothermia in liver surgery and transplantation. Mol Med. 2015;21:833-46. doi: 10.2119/molmed.2015.00158.
» https://doi.org/10.2119/molmed.2015.00158 -
18Tapuria N, Junnarkar S, Abu-Amara M, Fuller B, Seifalian AM, Davidson BR. Haemoxygenase modulates cytokine induced neutrophil chemoattractant in hepatic ischemia reperfusion injury. World J Gastroenterol. 2016;22(33):7518-35. doi: 10.3748/wjg.v22.i33.7518.
» https://doi.org/10.3748/wjg.v22.i33.7518 -
19Lentsch AB. Regulatory mechanisms of injury and repair after hepatic ischemia/reperfusion. Scientifica (Cairo). 2012;2012:1-14. doi: 10.6064/2012/513192.
» https://doi.org/10.6064/2012/513192 -
20Peralta C, Jimenez-Castro MB, Gracia-Sancho J. Hepatic ischemia and reperfusion injury: effects on the liver sinusoidal milieu. J Hepatol. 2013;59(5):1094-106. doi: 10.1016/j.jhep.2013.06.017.
» https://doi.org/10.1016/j.jhep.2013.06.017 -
21Alkozai E, Lisman T, Porte R, Nijsten M. Early elevated serum gamma glutamyl transpeptidase after liver transplantation is associated with better survival. F1000Res. 2014;3(85):1-11. doi: 10.12688/f1000research.3557.
» https://doi.org/10.12688/f1000research.3557 -
22Grezzana Filho Tde J, Mendonca TB, Gabiatti G, Kruel CD, Corso CO. Topic liver hypothermia and ischemic preconditioning: a new model of ischemia and reperfusion in rats. Acta Cir Bras. 2009;24(4):262-6. doi: 10.1590/S0102-86502009000400004.
» https://doi.org/10.1590/S0102-86502009000400004 -
23Grezzana Filho Tde J, Mendonca TB, Gabiatti G, Rodrigues G, Marroni NA, Treis L, De Rossi SD, Corso CO. Topical hepatic hypothermia plus ischemic preconditioning: analysis of bile flow and ischemic injuries after initial reperfusion in rats. Acta Cir Bras. 2011;26(3):194-201. doi: 10.1590/S0102-86502011000300007.
» https://doi.org/10.1590/S0102-86502011000300007 -
24Longo L, Sinigaglia-Fratta LX, Weber GR, Janz-Moreira A, Kretzmann NA, Grezzana-Filho Tde J, Possa-Marroni N, Corso CO, Schmidt-Cerski CT, Reverbel-da-Silveira T, Alvares-da-Silva MR, dos-Santos JL. Hypothermia is better than ischemic preconditioning for preventing early hepatic ischemia/reperfusion in rats. Ann Hepatol. 2016;15(1):110-20. doi: 10.5604/16652681.1184285.
» https://doi.org/10.5604/16652681.1184285 -
25McCafferty K, Forbes S, Thiemermann C, Yaqoob MM. The challenge of translating ischemic conditioning from animal models to humans: the role of comorbidities. Dis Model Mech. 2014;7(12):1321-33. doi: 10.1242/dmm.016741.
» https://doi.org/10.1242/dmm.016741 -
26van Golen RF, Reiniers MJ, Heger M, Verheij J. Solutions to the discrepancies in the extent of liver damage following ischemia/reperfusion in standard mouse models. J Hepatol. 2015;62(4):975-7. doi: 10.1016/j.jhep.2014.12.014.
» https://doi.org/10.1016/j.jhep.2014.12.014 -
27Nadarajah L, Yaqoob MM, McCafferty K. Ischemic conditioning in solid organ transplantation: is it worth giving your right arm for? Curr Opin Nephrol Hypertens. 2017;26(6):467-76. doi: 10.1097/mnh.0000000000000367.
» https://doi.org/10.1097/mnh.0000000000000367 -
28Biberthaler P, Luchting B, Massberg S, Teupser D, Langer S, Leiderer R, Messmer K, Krombach F. The influence of organ temperature on hepatic ischemia-reperfusion injury: a systematic analysis. Transplantation. 2001;72(9):1486-90. doi: 10.1097/00007890-200111150-00003.
» https://doi.org/10.1097/00007890-200111150-00003 -
29Suzuki S, Toledo-Pereyra LH, Rodriguez FJ, Cejalvo D. Neutrophil infiltration as an important factor in liver ischemia and reperfusion injury. Modulating effects of FK506 and cyclosporine. Transplantation. 1993;55(6):1265-72. PMID: 7685932.
-
30Colnot S, Perret C. Liver zonation. In: Monga SPS, editor. Molecular pathology of liver diseases. Boston, MA: Springer US; 2011. p.7-16. doi: 10.1007/978-1-4419-7107-4_2.
» https://doi.org/10.1007/978-1-4419-7107-4_2 -
31Peralta C, Prats N, Xaus C, Gelpi E, Rosello-Catafau J. Protective effect of liver ischemic preconditioning on liver and lung injury induced by hepatic ischemia-reperfusion in the rat. Hepatology. 1999;30(6):1481-9. doi: 10.1002/hep.510300622.
» https://doi.org/10.1002/hep.510300622 -
32Jin LM, Liu YX, Zhou L, Xie HY, Feng XW, Li H, Zheng SS. Ischemic preconditioning attenuates morphological and biochemical changes in hepatic ischemia/reperfusion in rats. Pathobiology. 2010;77(3):136-46. doi: 10.1159/000292647.
» https://doi.org/10.1159/000292647 -
33Adam AN. Some mechanisms of the protective effect of ischemic preconditioning on rat liver ischemia-reperfusion injury. Int J Gen Med. 2014;7:483-9. doi: 10.2147/IJGM.S66766.
» https://doi.org/10.2147/IJGM.S66766 -
34Li Y, Li T, Qi H, Yuan F. Minocycline protects against hepatic ischemia/reperfusion injury in a rat model. Biomed Rep. 2015;3(1):19-24. doi: 10.3892/br.2014.381.
» https://doi.org/10.3892/br.2014.381 -
35Steenks M, van Baal MC, Nieuwenhuijs VB, de Bruijn MT, Schiesser M, Teo MH, Callahan T, Padbury RT, Barritt GJ. Intermittent ischaemia maintains function after ischaemia reperfusion in steatotic livers. HPB (Oxford). 2010;12(4):250-61. doi: 10.1111/j.1477-2574.2010.00160.x.
» https://doi.org/10.1111/j.1477-2574.2010.00160.x -
36Chu MJ, Vather R, Hickey AJ, Phillips AR, Bartlett AS. Impact of ischaemic preconditioning on experimental steatotic livers following hepatic ischaemia-reperfusion injury: a systematic review. HPB (Oxford). 2015;17(1):1-10. doi: 10.1111/hpb.12258.
» https://doi.org/10.1111/hpb.12258 -
37O'Neill S, Leuschner S, McNally SJ, Garden OJ, Wigmore SJ, Harrison EM. Meta-analysis of ischaemic preconditioning for liver resections. Br J Surg. 2013;100(13):1689-700. doi: 10.1002/bjs.9277.
» https://doi.org/10.1002/bjs.9277 -
38Selzner N, Boehnert M, Selzner M. Preconditioning, postconditioning, and remote conditioning in solid organ transplantation: basic mechanisms and translational applications. Transplant Rev (Orlando). 2012;26(2):115-24. doi: 10.1016/j.trre.2011.07.003.
» https://doi.org/10.1016/j.trre.2011.07.003
-
Financial source:
none
-
1
Research performed at Experimental Surgery Unit, Hospital de Clinicas de Porto Alegre (HCPA), Universidade Federal do Rio Grande do Sul (UFRGS), Porto Alegre-RS, Brazil. Part of PhD degree thesis, Postgraduate Program in Surgery. Tutor: Prof. Dr. Carlos Otávio Corso.
Publication Dates
-
Publication in this collection
Oct 2018
History
-
Received
07 June 2018 -
Reviewed
10 Aug 2018 -
Accepted
09 Sept 2018