Abstract
Purpose:
To determine whether the absence of transglutaminase 2 enzyme (TG2) in TG2 knockout mice (TG2-/-) protect them against early age-related functional and histological arterial changes.
Methods:
Pulse wave velocity (PWV) was measured using non-invasive Doppler and mean arterial pressure (MAP) was measured in awake mice using tail-cuff system. Thoracic aortas were excised for evaluation of endothelial dependent vasodilation (EDV) by wire myography, as well as histological analyses.
Results:
PWV and MAP were similar in TG2-/-mice to age-matched wild type (WT) control mice. Old WT mice exhibited a markedly attenuated EDV as compared to young WT animals. The TG2-/-young and old mice had enhanced EDV responses (p<0.01) as compared to WT mice. There was a significant increase in TG2 crosslinks by IHC in WT old group compared to Young, with no stain in the TG2-/-animals. Optical microscopy examination of Old WT mice aorta showed thinning and fragmentation of elastic laminae. Young WT mice, old and young TG2-/-mice presented regularly arranged and parallel elastic laminae of the tunica media.
Conclusion:
The genetic suppression of TG2 delays the age-induced endothelial dysfunction and histological modifications.
Key words:
Aging; Transglutaminases; Endothelium-Dependent Relaxing Factors; Mice
Introduction
Age is the most important predictor of cardiovascular disease, even in the absence of modifiable risk factors11 Najjar SS, Scuteri A, Lakatta EG. Arterial aging: is it an immutable cardiovascular risk factor? Hypertension. 2005;46(3):454-62. doi: 01.HYP.0000177474.06749.98.
https://doi.org/01.HYP.0000177474.06749....
. Vascular aging is associated with changes in arterial wall structure and function. The arterial ageing process results from alterations in the properties of all elements of the vascular wall, including endothelium, vascular smooth muscle, and extra-cellular matrix (ECM)22 Greenwald SE. Ageing of the conduit arteries. J Pathol. 2007;211(2):157-72. doi: 10.1002/path.2101.
https://doi.org/10.1002/path.2101...
,33 Wilkinson IB, Franklin SS, Cockcroft JR. Nitric oxide and the regulation of large artery stiffness: from physiology to pharmacology. Hypertension. 2004;44(2):112-6. doi: 10.1161/01.HYP.0000138068.03893.40.
https://doi.org/10.1161/01.HYP.000013806...
. These alterations contribute to the development of vascular stiffness and to an additional impairment of the endothelial function, both considered as independent risk factors for cardiovascular morbidity and mortality44 Sutton-Tyrrell K, Venkitachalam L, Kanaya AM, Boudreau R, Harris T, Thompson T, Mackey RH, Visser M, Vaidean GD, Newman AB. Relationship of ankle blood pressures to cardiovascular events in older adults. Stroke. 2008;39(3):863-9. doi: 10.1161/STROKEAHA.107.487439.
https://doi.org/10.1161/STROKEAHA.107.48...
5 Wallace SM, Yasmin, McEniery CM, Maki-Petaja KM, Booth AD, Cockcroft JR, Wilkinson IB. Isolated systolic hypertension is characterized by increased aortic stiffness and endothelial dysfunction. Hypertension. 2007;50(1):228-33. doi: 10.1161/HYPERTENSIONAHA.107.089391.
https://doi.org/10.1161/HYPERTENSIONAHA....
-66 Mitchell GF, Hwang SJ, Vasan RS, Larson MG, Pencina MJ, Hamburg NM, Vita JA, Levy D, Benjamin EJ. Arterial stiffness and cardiovascular events: the Framingham Heart Study. Circulation. 2010;121(4):505-11. doi: 10.1161/CIRCULATIONAHA.109.886655.
https://doi.org/10.1161/CIRCULATIONAHA.1...
.
TG2 is the most abundantly expressed member of the transglutaminase family of enzymes in the vasculature and catalyzes a transamidation reaction, producing the crosslinking of proteins through the formation of N-ε-(γ-glutamyl)lysine isopeptide bonds77 Bakker EN, Pistea A, VanBavel E. Transglutaminases in vascular biology: relevance for vascular remodeling and atherosclerosis. J Vasc Res. 2008;45(4):271-8. doi: 10.1159/000113599.
https://doi.org/10.1159/000113599...
. Importantly, TG2 crosslinking activity as well as TG2 subcellular distribution relates to endothelial NO bioavailability in endothelial cells88 Santhanam L, Tuday EC, Webb AK, Dowzicky P, Kim JH, Oh YJ, Sikka G, Kuo M, Halushka MK, Macgregor AM, Dunn J, Gutbrod S, Yin D, Shoukas A, Nyhan D, Flavahan NA, Belkin AM, Berkowitz DE. Decreased S-nitrosylation of tissue transglutaminase contributes to age-related increases in vascular stiffness. Circ Res. 2010;107(1):117-25. doi: 10.1161/CIRCRESAHA.109.215228.
https://doi.org/10.1161/CIRCRESAHA.109.2...
,99 Santhanam L, Berkowitz DE, Belkin AM. Nitric oxide regulates non-classical secretion of tissue transglutaminase. Commun Integr Biol. 2011;4(5):584-6. doi: 10.4161/cib.4.5.16512.
https://doi.org/10.4161/cib.4.5.16512...
, fibroblasts1010 Telci D, Collighan RJ, Basaga H, Griffin M. Increased TG2 expression can result in induction of transforming growth factor beta1, causing increased synthesis and deposition of matrix proteins, which can be regulated by nitric oxide. J Biol Chem. 2009;284(43):29547-58. doi: 10.1074/jbc.M109.041806.
https://doi.org/10.1074/jbc.M109.041806...
and smooth muscle cells (SMC)1111 Jandu SK, Webb AK, Pak A, Sevinc B, Nyhan D, Belkin AM, Flavahan NA, Berkowitz DE, Santhanam L. Nitric oxide regulates tissue transglutaminase localization and function in the vasculature. Amino Acids. 2013;44(1):261-9. doi: 10.1007/s00726-011-1090-0.
https://doi.org/10.1007/s00726-011-1090-...
. More recently, TG2 expression has been involved in multiple vascular pathophysiological processes, including vascular remodeling1212 van den Akker J, VanBavel E, van Geel R, Matlung HL, Guvenc Tuna B, Janssen GM, van Veelen PA, Boelens WC, De Mey JG, Bakker EN. The redox state of transglutaminase 2 controls arterial remodeling. PloS one. 2011;6(8):e23067. doi: 10.1371/journal.pone.0023067.
https://doi.org/10.1371/journal.pone.002...
13 Pistea A, Bakker EN, Spaan JA, Hardeman MR, van Rooijen N, van Bavel E. Small artery remodeling and erythrocyte deformability in L-NAME-induced hypertension: role of transglutaminases. J Vasc Res. 2008;45(1):10-8. doi: 10.1159/000109073.
https://doi.org/10.1159/000109073...
-1414 Bakker EN, Pistea A, Spaan JA, Rolf T, de Vries CJ, van Rooijen N, Candi E, VanBavel E. Flow-dependent remodeling of small arteries in mice deficient for tissue-type transglutaminase: possible compensation by macrophage-derived factor XIII. Circ Res. 2006;99(1):86-92. doi: 10.1161/01.RES.0000229657.83816.a7.
https://doi.org/10.1161/01.RES.000022965...
, atherosclerosis1515 Matlung HL, Groen HC, de Vos J, van Walsum T, van der Lugt A, Niessen WJ, Wentzel JJ, Vanbavel E, Bakker EN. Calcification locates to transglutaminases in advanced human atherosclerotic lesions. Am J Pathol. 2009;175(4):1374-9. doi: 10.2353/ajpath.2009.090012.
https://doi.org/10.2353/ajpath.2009.0900...
, vascular calcification1616 Vanbavel E, Bakker EN. A vascular bone collector: arterial calcification requires tissue-type transglutaminase. Circ Res. 2008;102(5):507-9. doi: 10.1161/CIRCRESAHA.108.173013.
https://doi.org/10.1161/CIRCRESAHA.108.1...
and age-dependent aortic stiffening88 Santhanam L, Tuday EC, Webb AK, Dowzicky P, Kim JH, Oh YJ, Sikka G, Kuo M, Halushka MK, Macgregor AM, Dunn J, Gutbrod S, Yin D, Shoukas A, Nyhan D, Flavahan NA, Belkin AM, Berkowitz DE. Decreased S-nitrosylation of tissue transglutaminase contributes to age-related increases in vascular stiffness. Circ Res. 2010;107(1):117-25. doi: 10.1161/CIRCRESAHA.109.215228.
https://doi.org/10.1161/CIRCRESAHA.109.2...
,1717 Steppan J, Sikka G, Jandu S, Barodka V, Halushka MK, Flavahan NA, Belkin AM, Nyhan D, Butlin M, Avolio A, Berkowitz DE, Santhanam L. Exercise, vascular stiffness, and tissue transglutaminase. J Am Heart Assoc. 2014;3(2):e000599. doi: 10.1161/JAHA.113.000599.
https://doi.org/10.1161/JAHA.113.000599...
.
We conducted an experimental rodent model using a genetic approach to assess early age-related functional and microstructural arterial changes mediated by TG2. We therefore, hope to provide insights on future therapeutic targets to prevent subclinical vascular aging deleterious processes.
Methods
All protocols used in this study were approved by the Institutional Animal Care and Use Committee at Johns Hopkins University School of Medicine and ratified by the Committee of Ethics in Experimental Research of the Universidade Federal de São Paulo (protocol no 3516260515).
Male TG2 -/-mice were used in the study with their wild-type background BL6/129S as control. For the experimental model, 20 young mice (10 TG2-/-and 10 WT) and 20 old mice (10 TG2-/-and 10 WT) were used. To assess early age-related changes, we used young animals at 3 months of age and old animals at 8 months of age. All animals were fed and watered ad libitum. Thereafter, the animals were sacrificed and the thoracic aortae were removed for assessing endothelial function, morphological and immunohistochemical (IHC) studies.
Noninvasive mean arterial pressure and pulse-wave velocity
Blood pressure was measured concurrently in unanesthetized mice using an XBP1000 non-invasive tail-cuff blood pressure system (Kent Scientific Corporation). For analytic purposes, we computed noninvasive mean arterial pressure (MAP).
Noninvasive pulse-wave velocity (PWV) was assessed as a measure of the central aortic stiffness index. For this purpose, we used a high frequency, high resolution Doppler spectrum analyzer (DSPW), a real-time signal acquisition and spectrum analyzer system (Indus Instruments, Houston, Texas) as previously described1818 Soucy KG, Ryoo S, Benjo A, Lim HK, Gupta G, Sohi JS, Elser J, Aon MA, Nyhan D, Shoukas AA, Berkowitz DE. Impaired shear stress-induced nitric oxide production through decreased NOS phosphorylation contributes to age-related vascular stiffness. J Appl Physiol (1985). 2006;101(6):1751-59. doi: 10.1152/japplphysiol.00138.2006.
https://doi.org/10.1152/japplphysiol.001...
. Mice were lightly anesthetized with 1.5% isoflurane, blood pressures and heart rates were allowed to stabilize into the physiologic range prior to study. 10 and 20 MHz probes were used to measure the descending aortic and abdominal aortic flow velocity.
Aorta preparation
Aorta was used to assess vascular elasticity and endothelial dysfunction. Mice were anesthetized in a closed chamber with isoflurane. Anesthesia was maintained by mask ventilation of 1.5% isoflurane with a coupled charcoal scavenging system. Animals were positioned supine with limbs taped to the surgical table. Midline laparotomy was performed towards caudal-cranial, with extension into the chest after opening the diaphragm. The animals were administered an intracardiac injection of sodium heparin (1,00 IU), 1 min prior the aorta dissection. The mice were euthanized and a maximum length of aorta was isolated and removed. The dissected vessel was immediately placed in a culture dish with iced Krebs buffer (containing [mM] 118 NaCl, 4.7 KCl, 25.0 NaHCO3, 1.16 MgSO4, 1.18 KH2PO4, 11.1 D-(+)- glucose, 3.24 CaCl2). The aorta was cleaned of connective tissue ex-vivo. The aorta was segmented in 2 mm rings at the level of descending thoracic aorta for assessment of vasoreactivity as well as histological analyzes.
Endothelial function assessment
We characterized endothelial function ex vivo assessing endothelial dependent vasodilation (EDV) using vascular tension experiments. In brief, aortic rings were suspended from strain gauges in 25-mL organ chambers filled with oxygenated (95/5% O2/ CO2) Krebs buffer maintained at pH 7.4 and 37°C. The rings were stretched at 10-min intervals in 100-mg increments to reach optimal tone (500mg). Indomethacin (10-5 M) was added to the aortic rings to prevent any generation of endothelium-derived contracting prostanoids for NO-dependent relaxation responses. Vessels were preconstricted with phenylephrine (PE, 10-6 M) (Sigma) for 15 min. Cumulative dose-responses to acetylcholine (ACh, 10-9-10-5 M) (Sigma) were obtained to determine relaxation activity. Furthermore, cumulative dose-responses to sodium nitroprusside (SNP, 10-9-10-5 M) (Sigma) were obtained to characterize endothelial independent vasorelaxation. Vasorelaxation is expressed as percent relaxation, as calculated by the percent decrease in tension from the PE-induced preconstriction.
Tissue preparation and staining techniques
Mice aortic tissue segments were fixed for 24 hours in 10% neutral formaldehyde. The tissue segments were embedded in paraffin, and 5-7mm transverse sections were cut using a Reichert-Jung 2030 biocut rotary microtome. The tissue sections were mounted on positively charged aminopropyl- triethoxysilane-coated glass slides and deparaffinized using successive washes with xylene/ethanol/water. Briefly, after deparaffinization and rehydration, slides were incubated with 3% H2O2 to block endogenous peroxidase activity. Antigen retrieval was performed using 0.01 mol/L of sodium citrate buffer in a microwave oven for 10 minutes. Before IHC staining, the sections were blocked with 10% goat serum for 30 minutes. To localize the TG2-generated cross-links, the sections were incubated with 10 l g/mL of mAb 81D4 specific
for Nε-(γ -glutamyl)-lysine isodipeptide cross-links (Covalab) for 18 hours at 4°C. The slides were washed and incubated with anti-mouse IgM (for cross-links) conjugated with HRP for 30 minutes at room temperature. The slides were washed and incubated with secondary goat anti-mouse immunoglobulin (Ig)G conjugated with HRP for 30 minutes at room temperature. HRP activity was developed with 3,30-diaminobenzidine chromagenic substrate (Vector Laboratories, Burlingame, CA) for 5 minutes per the manufacturer’s instructions. The sections were counterstained with Harris Hematoxylin and Eosin (H&E) for histological examination.
IHC scoring
Digital images were taken using an Olympus DP20 camera system (Olympus, Tokyo, Japan) and white balanced using Photoshop CS2 (Adobe). An open-source image analysis software program (FrIDA) was used to score individual images. Regions of interest (ROIs) were separately created for the media and adventitia of each vessel. Brown pixels were identified using a color-picking algorithm that captured a user- defined region of hue, saturation, and luminosity, and the percent of brown pixels to total pixels within each ROI was determined. For TG2, the media ROI was evaluated and for the cross-links the adventitia ROI was used.
Data analysis
All statistical analyses were performed using Prism 5 for Mac by GraphPad Software Inc. and Microsoft Excel version 14.1.3 statistical analysis software. The results were expressed as mean and standard error (mean ± SEM). One-way analysis of ANOVA and the Bonferroni post hoc test for multiple-comparison were used for comparing all groups and pairs of groups respectively. A value of p<0.05 was considered statistically significant.
Results
PWV and MAP of TG2-/-mice were similar to those of age-matched WT mice
In Figure 1A, PWV did not have a statistically significant difference in TG2-/-mice compared to that of WT age-matched controls (Old TG2-/- 4.44 ± 0.20m/s vs. Old WT 3.85 ± 0.2m/s, n = 10, p = 0.06). There was also no statistically significant difference between young and old animals within the same groups (Young TG2-/- 4.39 ± 0.22m/s vs. Old TG2-/- 4.44 ± 0.20m/s; n = 10, p = 0.88 / Young WT 3.71 ± 0.18m/s vs. Old WT 3.85 ± 0.21m/s; n = 10, p = 0.62).
Comparisons for pulse-wave velocity (PWV; A) and mean arterial pressure (MAP; B) within study groups (n = 10; all p values between groups > 0.05). TG2-/- - mice knockout to Transglutaminase 2 enzyme; WT - wild type mice.
Also shown in Figure 1B, MAP was not statistically different between age-matched WT and TG2-/- mice (Old TG2-/- 78.23 ± 4.77mmHg vs. old WT 83.69 ± 5.37mmHg; p=0.45; n=10 / young TG2-/- 85.99 ± 3.45mmHg vs. young WT 87.90 ± 3.7mmHg; n = 10, p = 0.71). There was no statistically significant difference between young and old animals within the same groups (young TG2-/- 85.99±3.45mmHg vs. old TG2-/- 78.23±4.7mmHg; n=10, p=0.20/young WT 87.90±3.79mmHg vs. old WT 83.69±5.37mmHg; n=10, p=0.53).
Endothelial nitric oxide-dependent aortic relaxation was increased in TG2 knockout mice.
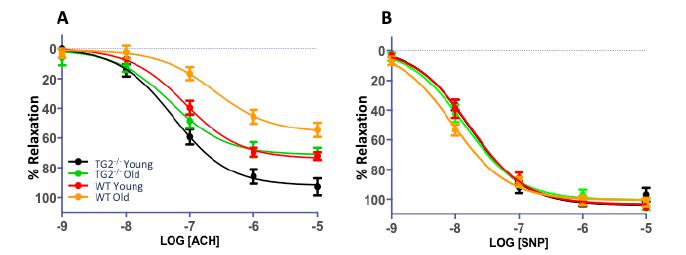
The EDV responses to acetylcholine in aged mice were significantly attenuated compared to those from the young animals (old WT 54.48±5% vs. young WT 72.11±2%, p<0.001, n=10; old TG2-/- 70.55±4% vs. young TG2-/- 92.87±6%, p = 0.001, n=10). The young and old TG2-/-mice had enhanced EDV responses as compared to control mice, as shown in Figure 2. The response to the endothelium-independent vasodilator SNP was not significantly different in any of the groups.
(A) Old WT mice exhibited a markedly attenuated vascular relaxation as compared to Young WT animals. The TG2-/- young and old mice had enhanced vasorelaxation responses (**p<0.001) as compared to control mice. (B) SNP dose response curve demonstrates no significant differences in endothelial independent responses to SNP. p≤0.001 WT young vs. WT old; TG2-/- young vs. TG2-/- old; WT old vs. TG2-/- old and WT young vs. TG2-/- young. Abbreviations as in Figure 1.
TG2-generated cross-links is increased in old WT mice compared to young ones.
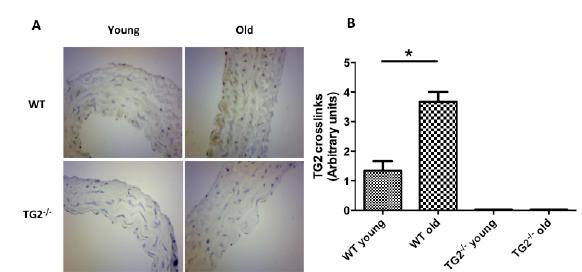
TG2-generated cross-links was determined by IHC (Figure 3). There was a significant increase in old WT compared to young WT (3.66 ± 0.33 vs. 1.33 ± 0.33, p=0.007, n=3), with no stain in the TG2-/- animals.
A. Immunohistochemical analysis of TG2 presence (immune staining with anti-TG2 monoclonal antibody, x400) in wildtype young and old groups compared to TG2-/- young and old groups. B. IHC scores are represented in the bar graphs (n=3 in each group). There was no statistically significant difference between the WT young vs. TG2-/- young and TG2-/- young vs. old TG2-/- groups. * represents values of p<0.05. Abbreviations as in Figure 1.
Aorta arteries of old WT mice present ageing histological modifications, not observed in the old TG2-/-mice.
Examination of old WT mice aortas with optical microscopy (H&E, x400) showed reduced elastic laminae, with areas of fragmentation compared to old TG2-/-mice. Also, old TG2-/- mice maintain tissue structure similar to young groups (Figure 4). In addition, cellular infiltrate in media tunica was found in old WT mice.
Optical microscopy (H&E, x400) of aortics rings from wildtype old and young groups compared to TG2-/- old and young groups. Aorta of old wildtype animals show reduced elastic laminae (green arrow) compared to old TG2-/- animals (yellow arrow), which maintains tissue structure similar to young groups. Abbreviations as in Figure 1.
Discussion
We show that young WT mice had a similar endothelial function response when compared to 8-months old TG2-/- mice. In our findings, old WT mice had the worse endothelial response among all groups. Furthermore, we show histological modification in aorta arteries media layers as related to the TG2-mediated early vascular ageing process.
Endothelial dysfunction appears to be one of the main markers of aging-related vascular changes, as a reduction in the endothelium-dependent vasodilatation is strongly associated to age-related endothelial dysfunction1919 Brandes RP, Fleming I, Busse R. Endothelial aging. Cardiovasc Res. 2005;66(2):286-94. doi: 10.1016/j.cardiores.2004.12.027.
https://doi.org/10.1016/j.cardiores.2004...
,2020 Toda N. Age-related changes in endothelial function and blood flow regulation. Pharmacol Ther. 2012;133(2):159-76. doi: 10.1016/j.pharmthera.2011.10.004.
https://doi.org/10.1016/j.pharmthera.201...
. In this regard, preventive efforts have been focusing on new therapeutic targets that may delay the vascular ageing process.
In our study, TG2 has an important role in early phases of vascular ageing, as young WT animals had similar degrees of endothelial response when compared to TG2-/- old mice. In addition, we found early aortic histological abnormalities in old WT animals that are known to relate to arterial aging process2121 Abu-Dief EE, Abdelrahim EA, Abdelrahim KM. Histological modifications aging aorta in male albino rat. J Cytol Histol. 2016;7(2):6.,2222 Gupta SD, Gupta SK, Pal DK, Sarawagi R, Gupta P. Microscopic study of aorta in relation of different age groups: an observational study. Int J Biol Med Res. 2011;2(1):6.. Interestingly, noninvasive measures of PWV and MAP were not significantly different among groups at this 8-month follow-up assessment.
Our results show TG2-mediated aortic microscopic changes as early as at 8 months of age. In accordance with our findings, previous studies on microscopic abnormalities in the aortic elastic laminae have shown relation to the vascular ageing process2121 Abu-Dief EE, Abdelrahim EA, Abdelrahim KM. Histological modifications aging aorta in male albino rat. J Cytol Histol. 2016;7(2):6.
22 Gupta SD, Gupta SK, Pal DK, Sarawagi R, Gupta P. Microscopic study of aorta in relation of different age groups: an observational study. Int J Biol Med Res. 2011;2(1):6.-2323 Fritze O, Romero B, Schleicher M, Jacob MP, Oh DY, Starcher B, Schenke-Layland K, Bujan J, Stock UA. Age-related changes in the elastic tissue of the human aorta. J Vasc Res. 2012;49(1):77-86. doi: 10.1159/000331278.
https://doi.org/10.1159/000331278...
. Histological examination stained with H&E and IHC are validated methods to assess age-related microstructural early arterial changes88 Santhanam L, Tuday EC, Webb AK, Dowzicky P, Kim JH, Oh YJ, Sikka G, Kuo M, Halushka MK, Macgregor AM, Dunn J, Gutbrod S, Yin D, Shoukas A, Nyhan D, Flavahan NA, Belkin AM, Berkowitz DE. Decreased S-nitrosylation of tissue transglutaminase contributes to age-related increases in vascular stiffness. Circ Res. 2010;107(1):117-25. doi: 10.1161/CIRCRESAHA.109.215228.
https://doi.org/10.1161/CIRCRESAHA.109.2...
,2121 Abu-Dief EE, Abdelrahim EA, Abdelrahim KM. Histological modifications aging aorta in male albino rat. J Cytol Histol. 2016;7(2):6..
TG2 has consistently shown an important role in age-dependent vascular stiffness88 Santhanam L, Tuday EC, Webb AK, Dowzicky P, Kim JH, Oh YJ, Sikka G, Kuo M, Halushka MK, Macgregor AM, Dunn J, Gutbrod S, Yin D, Shoukas A, Nyhan D, Flavahan NA, Belkin AM, Berkowitz DE. Decreased S-nitrosylation of tissue transglutaminase contributes to age-related increases in vascular stiffness. Circ Res. 2010;107(1):117-25. doi: 10.1161/CIRCRESAHA.109.215228.
https://doi.org/10.1161/CIRCRESAHA.109.2...
,99 Santhanam L, Berkowitz DE, Belkin AM. Nitric oxide regulates non-classical secretion of tissue transglutaminase. Commun Integr Biol. 2011;4(5):584-6. doi: 10.4161/cib.4.5.16512.
https://doi.org/10.4161/cib.4.5.16512...
,1111 Jandu SK, Webb AK, Pak A, Sevinc B, Nyhan D, Belkin AM, Flavahan NA, Berkowitz DE, Santhanam L. Nitric oxide regulates tissue transglutaminase localization and function in the vasculature. Amino Acids. 2013;44(1):261-9. doi: 10.1007/s00726-011-1090-0.
https://doi.org/10.1007/s00726-011-1090-...
,2424 Jung SM, Jandu S, Steppan J, Belkin A, An SS, Pak A, Choi EY, Nyhan D, Butlin M, Viegas K, Avolio A, Berkowitz DE, Santhanam L. Increased tissue transglutaminase activity contributes to central vascular stiffness in eNOS knockout mice. Am J Physiol Heart Circ Physiol. 2013;305(6):H803-10. doi: 10.1152/ajpheart.00103.2013.
https://doi.org/10.1152/ajpheart.00103.2...
,2525 Steppan J, Bergman Y, Viegas K, Armstrong D, Tan S, Wang H, Melucci S, Hori D, Park SY, Barreto SF, Isak A, Jandu S, Flavahan N, Butlin M, An SS, Avolio A, Berkowitz DE, Halushka MK, Santhanam L. Tissue transglutaminase modulates vascular stiffness and function through crosslinking-dependent and crosslinking-independent functions. J Am Heart Assoc. 2017;6(2). doi: 10.1161/JAHA.116.004161.
https://doi.org/10.1161/JAHA.116.004161...
. In particular, Santhanam et al.88 Santhanam L, Tuday EC, Webb AK, Dowzicky P, Kim JH, Oh YJ, Sikka G, Kuo M, Halushka MK, Macgregor AM, Dunn J, Gutbrod S, Yin D, Shoukas A, Nyhan D, Flavahan NA, Belkin AM, Berkowitz DE. Decreased S-nitrosylation of tissue transglutaminase contributes to age-related increases in vascular stiffness. Circ Res. 2010;107(1):117-25. doi: 10.1161/CIRCRESAHA.109.215228.
https://doi.org/10.1161/CIRCRESAHA.109.2...
showed that PWV measures of TG2-/- mice were similar to age-matched WT controls, but those TG2-/- were protected from the increase in PWV resulting from NOS inhibition using L-NAME88 Santhanam L, Tuday EC, Webb AK, Dowzicky P, Kim JH, Oh YJ, Sikka G, Kuo M, Halushka MK, Macgregor AM, Dunn J, Gutbrod S, Yin D, Shoukas A, Nyhan D, Flavahan NA, Belkin AM, Berkowitz DE. Decreased S-nitrosylation of tissue transglutaminase contributes to age-related increases in vascular stiffness. Circ Res. 2010;107(1):117-25. doi: 10.1161/CIRCRESAHA.109.215228.
https://doi.org/10.1161/CIRCRESAHA.109.2...
. In addition, elevated arterial pressure is known to relate to older ages in clinical and experimental settings2626 Piper MA, Evans CV, Burda BU, Margolis KL, O'Connor E, Smith N, Webber E, Perdue LA, Bigler KD, Whitlock EP. Screening for high blood pressure in adults: a systematic evidence review for the U.S. Preventive Services Task Force. Rockville (MD) 2014. PMID: 25632496.. Other aging studies, however, were performed on older animals when compared to our study, with ages ranging from 13 to 32 months2727 Choi K, Yoon J, Lim HK, Ryoo S. Korean red ginseng water extract restores impaired endothelial function by inhibiting arginase activity in aged mice. Korean J Physiol Pharmacol. 2014;18(2):95-101. doi: 10.4196/kjpp.2014.18.2.95.
https://doi.org/10.4196/kjpp.2014.18.2.9...
28 Chen K, Zhou X, Sun Z. Haplodeficiency of klotho gene causes arterial stiffening via upregulation of scleraxis expression and induction of autophagy. Hypertension. 2015;66(5):1006-13. doi: 10.1161/HYPERTENSIONAHA.115.06033.
https://doi.org/10.1161/HYPERTENSIONAHA....
29 Rammos C, Hendgen-Cotta UB, Deenen R, Pohl J, Stock P, Hinzmann C, Kelm M, Rassaf T. Age-related vascular gene expression profiling in mice. Mech Ageing Dev. 2014;135:15-23. doi: 10.1016/j.mad.2014.01.001.
https://doi.org/10.1016/j.mad.2014.01.00...
-3030 Gano LB, Donato AJ, Pasha HM, Hearon CM, Jr., Sindler AL, Seals DR. The SIRT1 activator SRT1720 reverses vascular endothelial dysfunction, excessive superoxide production, and inflammation with aging in mice. Am J Physiol Heart Circ Physiol. 2014;307(12):H1754-63. doi: 10.1152/ajpheart.00377.2014
https://doi.org/10.1152/ajpheart.00377.2...
.
Our findings suggest that, even in mice ages as young as 8-months, TG2 relates to an early degree of endothelial dysfunction. In the vascular ageing process, endothelial dysfunction and vascular histological abnormalities seem to precede changes detected by standard noninvasive methods widely used in clinical settings, such as PWV and MAP. As the animal ages, the endothelial dysfunction possibly progresses to a threshold point in which subclinical abnormalities may appear as clinically identifiable vascular diseases. Therefore, our study indicates that a comprehensive understanding of the role of TG2 in age-related vascular changes may help in the preventive efforts of the early subclinical vascular disease.
Conclusions
Genetic suppression of TG2 in mice delays early age-induced endothelial dysfunction and aortic microscopic abnormalities. Furthermore, TG2 inhibition augments vasorelaxation in an endothelial-dependent manner. Endothelial dysfunction and microstructural abnormalities were found as early age-related changes mediated by TG2, in the absence of significant alterations in noninvasive standard parameters such as pulse-wave velocity and blood pressure. These findings suggest that TG2 inhibition rises as a promising therapeutic target for future drug development aiming to prevent early subclinical age-related vascular abnormalities.
Acknowledgement
To Koenraad Vandergaer for his technical assistance.
References
-
1Najjar SS, Scuteri A, Lakatta EG. Arterial aging: is it an immutable cardiovascular risk factor? Hypertension. 2005;46(3):454-62. doi: 01.HYP.0000177474.06749.98.
» https://doi.org/01.HYP.0000177474.06749.98 -
2Greenwald SE. Ageing of the conduit arteries. J Pathol. 2007;211(2):157-72. doi: 10.1002/path.2101.
» https://doi.org/10.1002/path.2101 -
3Wilkinson IB, Franklin SS, Cockcroft JR. Nitric oxide and the regulation of large artery stiffness: from physiology to pharmacology. Hypertension. 2004;44(2):112-6. doi: 10.1161/01.HYP.0000138068.03893.40.
» https://doi.org/10.1161/01.HYP.0000138068.03893.40 -
4Sutton-Tyrrell K, Venkitachalam L, Kanaya AM, Boudreau R, Harris T, Thompson T, Mackey RH, Visser M, Vaidean GD, Newman AB. Relationship of ankle blood pressures to cardiovascular events in older adults. Stroke. 2008;39(3):863-9. doi: 10.1161/STROKEAHA.107.487439.
» https://doi.org/10.1161/STROKEAHA.107.487439 -
5Wallace SM, Yasmin, McEniery CM, Maki-Petaja KM, Booth AD, Cockcroft JR, Wilkinson IB. Isolated systolic hypertension is characterized by increased aortic stiffness and endothelial dysfunction. Hypertension. 2007;50(1):228-33. doi: 10.1161/HYPERTENSIONAHA.107.089391.
» https://doi.org/10.1161/HYPERTENSIONAHA.107.089391 -
6Mitchell GF, Hwang SJ, Vasan RS, Larson MG, Pencina MJ, Hamburg NM, Vita JA, Levy D, Benjamin EJ. Arterial stiffness and cardiovascular events: the Framingham Heart Study. Circulation. 2010;121(4):505-11. doi: 10.1161/CIRCULATIONAHA.109.886655.
» https://doi.org/10.1161/CIRCULATIONAHA.109.886655 -
7Bakker EN, Pistea A, VanBavel E. Transglutaminases in vascular biology: relevance for vascular remodeling and atherosclerosis. J Vasc Res. 2008;45(4):271-8. doi: 10.1159/000113599.
» https://doi.org/10.1159/000113599 -
8Santhanam L, Tuday EC, Webb AK, Dowzicky P, Kim JH, Oh YJ, Sikka G, Kuo M, Halushka MK, Macgregor AM, Dunn J, Gutbrod S, Yin D, Shoukas A, Nyhan D, Flavahan NA, Belkin AM, Berkowitz DE. Decreased S-nitrosylation of tissue transglutaminase contributes to age-related increases in vascular stiffness. Circ Res. 2010;107(1):117-25. doi: 10.1161/CIRCRESAHA.109.215228.
» https://doi.org/10.1161/CIRCRESAHA.109.215228 -
9Santhanam L, Berkowitz DE, Belkin AM. Nitric oxide regulates non-classical secretion of tissue transglutaminase. Commun Integr Biol. 2011;4(5):584-6. doi: 10.4161/cib.4.5.16512.
» https://doi.org/10.4161/cib.4.5.16512 -
10Telci D, Collighan RJ, Basaga H, Griffin M. Increased TG2 expression can result in induction of transforming growth factor beta1, causing increased synthesis and deposition of matrix proteins, which can be regulated by nitric oxide. J Biol Chem. 2009;284(43):29547-58. doi: 10.1074/jbc.M109.041806.
» https://doi.org/10.1074/jbc.M109.041806 -
11Jandu SK, Webb AK, Pak A, Sevinc B, Nyhan D, Belkin AM, Flavahan NA, Berkowitz DE, Santhanam L. Nitric oxide regulates tissue transglutaminase localization and function in the vasculature. Amino Acids. 2013;44(1):261-9. doi: 10.1007/s00726-011-1090-0.
» https://doi.org/10.1007/s00726-011-1090-0 -
12van den Akker J, VanBavel E, van Geel R, Matlung HL, Guvenc Tuna B, Janssen GM, van Veelen PA, Boelens WC, De Mey JG, Bakker EN. The redox state of transglutaminase 2 controls arterial remodeling. PloS one. 2011;6(8):e23067. doi: 10.1371/journal.pone.0023067.
» https://doi.org/10.1371/journal.pone.0023067 -
13Pistea A, Bakker EN, Spaan JA, Hardeman MR, van Rooijen N, van Bavel E. Small artery remodeling and erythrocyte deformability in L-NAME-induced hypertension: role of transglutaminases. J Vasc Res. 2008;45(1):10-8. doi: 10.1159/000109073.
» https://doi.org/10.1159/000109073 -
14Bakker EN, Pistea A, Spaan JA, Rolf T, de Vries CJ, van Rooijen N, Candi E, VanBavel E. Flow-dependent remodeling of small arteries in mice deficient for tissue-type transglutaminase: possible compensation by macrophage-derived factor XIII. Circ Res. 2006;99(1):86-92. doi: 10.1161/01.RES.0000229657.83816.a7.
» https://doi.org/10.1161/01.RES.0000229657.83816.a7 -
15Matlung HL, Groen HC, de Vos J, van Walsum T, van der Lugt A, Niessen WJ, Wentzel JJ, Vanbavel E, Bakker EN. Calcification locates to transglutaminases in advanced human atherosclerotic lesions. Am J Pathol. 2009;175(4):1374-9. doi: 10.2353/ajpath.2009.090012.
» https://doi.org/10.2353/ajpath.2009.090012 -
16Vanbavel E, Bakker EN. A vascular bone collector: arterial calcification requires tissue-type transglutaminase. Circ Res. 2008;102(5):507-9. doi: 10.1161/CIRCRESAHA.108.173013.
» https://doi.org/10.1161/CIRCRESAHA.108.173013 -
17Steppan J, Sikka G, Jandu S, Barodka V, Halushka MK, Flavahan NA, Belkin AM, Nyhan D, Butlin M, Avolio A, Berkowitz DE, Santhanam L. Exercise, vascular stiffness, and tissue transglutaminase. J Am Heart Assoc. 2014;3(2):e000599. doi: 10.1161/JAHA.113.000599.
» https://doi.org/10.1161/JAHA.113.000599 -
18Soucy KG, Ryoo S, Benjo A, Lim HK, Gupta G, Sohi JS, Elser J, Aon MA, Nyhan D, Shoukas AA, Berkowitz DE. Impaired shear stress-induced nitric oxide production through decreased NOS phosphorylation contributes to age-related vascular stiffness. J Appl Physiol (1985). 2006;101(6):1751-59. doi: 10.1152/japplphysiol.00138.2006.
» https://doi.org/10.1152/japplphysiol.00138.2006 -
19Brandes RP, Fleming I, Busse R. Endothelial aging. Cardiovasc Res. 2005;66(2):286-94. doi: 10.1016/j.cardiores.2004.12.027.
» https://doi.org/10.1016/j.cardiores.2004.12.027 -
20Toda N. Age-related changes in endothelial function and blood flow regulation. Pharmacol Ther. 2012;133(2):159-76. doi: 10.1016/j.pharmthera.2011.10.004.
» https://doi.org/10.1016/j.pharmthera.2011.10.004 -
21Abu-Dief EE, Abdelrahim EA, Abdelrahim KM. Histological modifications aging aorta in male albino rat. J Cytol Histol. 2016;7(2):6.
-
22Gupta SD, Gupta SK, Pal DK, Sarawagi R, Gupta P. Microscopic study of aorta in relation of different age groups: an observational study. Int J Biol Med Res. 2011;2(1):6.
-
23Fritze O, Romero B, Schleicher M, Jacob MP, Oh DY, Starcher B, Schenke-Layland K, Bujan J, Stock UA. Age-related changes in the elastic tissue of the human aorta. J Vasc Res. 2012;49(1):77-86. doi: 10.1159/000331278.
» https://doi.org/10.1159/000331278 -
24Jung SM, Jandu S, Steppan J, Belkin A, An SS, Pak A, Choi EY, Nyhan D, Butlin M, Viegas K, Avolio A, Berkowitz DE, Santhanam L. Increased tissue transglutaminase activity contributes to central vascular stiffness in eNOS knockout mice. Am J Physiol Heart Circ Physiol. 2013;305(6):H803-10. doi: 10.1152/ajpheart.00103.2013.
» https://doi.org/10.1152/ajpheart.00103.2013 -
25Steppan J, Bergman Y, Viegas K, Armstrong D, Tan S, Wang H, Melucci S, Hori D, Park SY, Barreto SF, Isak A, Jandu S, Flavahan N, Butlin M, An SS, Avolio A, Berkowitz DE, Halushka MK, Santhanam L. Tissue transglutaminase modulates vascular stiffness and function through crosslinking-dependent and crosslinking-independent functions. J Am Heart Assoc. 2017;6(2). doi: 10.1161/JAHA.116.004161.
» https://doi.org/10.1161/JAHA.116.004161 -
26Piper MA, Evans CV, Burda BU, Margolis KL, O'Connor E, Smith N, Webber E, Perdue LA, Bigler KD, Whitlock EP. Screening for high blood pressure in adults: a systematic evidence review for the U.S. Preventive Services Task Force. Rockville (MD) 2014. PMID: 25632496.
-
27Choi K, Yoon J, Lim HK, Ryoo S. Korean red ginseng water extract restores impaired endothelial function by inhibiting arginase activity in aged mice. Korean J Physiol Pharmacol. 2014;18(2):95-101. doi: 10.4196/kjpp.2014.18.2.95.
» https://doi.org/10.4196/kjpp.2014.18.2.95 -
28Chen K, Zhou X, Sun Z. Haplodeficiency of klotho gene causes arterial stiffening via upregulation of scleraxis expression and induction of autophagy. Hypertension. 2015;66(5):1006-13. doi: 10.1161/HYPERTENSIONAHA.115.06033.
» https://doi.org/10.1161/HYPERTENSIONAHA.115.06033 -
29Rammos C, Hendgen-Cotta UB, Deenen R, Pohl J, Stock P, Hinzmann C, Kelm M, Rassaf T. Age-related vascular gene expression profiling in mice. Mech Ageing Dev. 2014;135:15-23. doi: 10.1016/j.mad.2014.01.001.
» https://doi.org/10.1016/j.mad.2014.01.001 -
30Gano LB, Donato AJ, Pasha HM, Hearon CM, Jr., Sindler AL, Seals DR. The SIRT1 activator SRT1720 reverses vascular endothelial dysfunction, excessive superoxide production, and inflammation with aging in mice. Am J Physiol Heart Circ Physiol. 2014;307(12):H1754-63. doi: 10.1152/ajpheart.00377.2014
» https://doi.org/10.1152/ajpheart.00377.2014
-
Financial source:
American Heart Association (Grants 09BGIA2220181)
-
1
Research performed at Department of Anesthesiology and Critical Care Medicine, Johns Hopkins University, School of Medicine, Baltimore, MD, USA. Part of PhD degree thesis, Postgraduate Program in Translational Medicine, UNIFESP. Tutor: Dr. Murched Omar Taha.
Publication Dates
-
Publication in this collection
Nov 2018
History
-
Received
02 July 2018 -
Reviewed
09 Sept 2018 -
Accepted
05 Oct 2018