Abstracts
Direct reactions between [Ru(NH3)5(H2O)](PF6) 2 (1) and P(m-tol)3 (a), P(p-tol)3 (b) and P(OC5H11)3 (c) lead the complexes [Ru(NH3)5P(m-tol)3](PF 6)2 (2a), trans-[Ru(NH3)4{P(p -tol)3}2](PF6)2 (3b) and trans-[Ru(NH3)4{P(OC5H 11)3}2](PF6)2 (3c) respectively. These complexes have been isolated as hexafluorophosphate salts and characterized by elemental analysis, cyclic voltammetry and UV-VIS spectra measurements. One ammonia ligand in 2a is displaced at the a specific rate of 4.3 x 10-3 s-1 (25 ºC, μ = 0.10 mol L-1). The aquo species trans-[Ru(NH3)4(L)H2O]2+ (L = P(p-tol)3, 4b; L = P(OC5H11)3, 4c) are generated by the substitution of one L ligand with H2O, from trans-[Ru(NH3)4(L)2]2+ with k-1 = 2.7 x 10-3 s-1 [L = P(p-tol)3, 3b] and 3.5 x 10-3 s-1 [L = P(OC5H11)3 3c] (25 ºC, μ = 0.10 mol L-1) (NaCF3COO/CF3COOH; C H+ = 1.0 x 10-3 mol L-1). The formal reduction potentials for the species 3b, 3c and 2a are 0.50 ± 0.01V, 0.48 ± 0.01V and 0.50 ± 0.02V, respectively (vs SCE) [μ = 0.10 mol L-1 ; NaCF3COO; C H+ = 1.0 x 10-3 CF3COOH]. The following reactions: <IMG SRC="http:/img/fbpe/jbchs/v13n5/12819x2.gif"> [L = P(m-tol)3, P(p-tol)3 and P(OC5H11)3] were studied and the calculated values for k1, k-1 and Keq are 0.28 mol-1 L s-1, 0.06 s-1, 4.67 mol-1 L [L = P(m-tol)3, 4a]; 0.25 mol-1 L s-1, 0.04 s-1, 6.25 mol-1 L [L = P(p-tol)3, 4b]; 12.4 mol-1 L s-1, 5.8x10-1 s-1, and 21 mol-1 L [L = P(OC5H11)3, 4c], [25.0 ± 0.2 ºC; m = 0.10 mol L-1; NaCF3COO; C H+ = 1.0 x 10-3 CF3COOH].
ruthenium(II); ammine complexes; phosphine; phosphite ligands
As reações diretas entre [Ru(NH3)5(H2O)](PF6) 2 (1) e P(m-tol)3 (a), P(p-tol)3 (b) e P(OC5H11)3 (c) forneceram os complexos [Ru(NH3)5P(m-tol)3](PF 6)2 (2a), trans-[Ru(NH3)4{P(p- tol)3}2](PF6)2 (3b) e trans-[Ru(NH3)4{P(OC5H 11)3}2](PF6)2 (3c), respectivamente. A constante de velocidade específica de substituição da amônia em (2a) foi de 4,3 x 10-3 s-1 (25 ºC, μ = 0,10 mol L-1). Os aquo complexos trans-[Ru(NH3)4(L)(H2O)]2+ (L = P(p-tol)3, 4b; L = P(OC5H11)3, 4c) foram gerados pela aquação térmica de uma das moléculas de L, dos respectivos complexos trans-[Ru(NH3)4L2 ]2+ (L = P(p-tol)3, 3b; L = P(OC5H11)3, 3c) e os valores das constantes foram: k-1 = 2,7 x 10-3 s-1 e k-1 = 3,5 x 10-3 s-1, respectivamente. Os valores dos potenciais formais de redução para 3b, 3c e 2a foram 0,50 ± 0,01V; 0,48 ± 0,01V e 0,50 ± 0,02V, respectivamente (vs SCE) [μ = 0,10 mol L-1; NaCF3COO; C H+ = 1,0 x 10-3 CF3COOH]. As reações: <IMG SRC="http:/img/fbpe/jbchs/v13n5/12819x1.gif" > foram estudadas e os valores de k1, k-1 e Keq foram 0,28 mol-1 L s-1, 0,06 s-1, 4,67 mol-1 L [para L = P(m-tol)3, 4a]; 0,25 mol-1 L s-1, 0,04 s-1, 6,25 mol-1 L [para L = P(p-tol)3, 4b] e 12,4 mol-1 L s-1, 5,8 x 10-1 s-1, 21 mol-1 L [para L = P(OC5H11)3, 4c] [25ºC; μ = 0,10 mol L-1; NaCF3COO; C H+ = 1,0 x 10-3 CF3COOH].
Article
Synthesis and Characterization of New Ammine Ru(II) Complexes Containing P(m-tol)3, P(p-CH3)3 and P(OC5H11)3
Roberta Signinia and Selma E. Mazzetto* * e-mail: selma@ufc.br ,b
a Instituto de Química de São Carlos, Universidade de São Paulo, Av. Trabalhador São-Carlense, 400, Centro, 13566-590 São Carlos - SP, Brazil
b Departamento de Química Orgânica e Inorgânica, Universidade Federal do Ceará, Caixa Postal 12.200, 60455-760 Fortaleza - CE, Brazil
As reações diretas entre [Ru(NH3)5(H2O)](PF6) 2 (1) e P(m-tol)3 (a), P(p-tol)3 (b) e P(OC5H11)3 (c) forneceram os complexos [Ru(NH3)5P(m-tol)3](PF 6)2 (2a), trans-[Ru(NH3)4{P(p- tol)3}2](PF6)2 (3b) e trans-[Ru(NH3)4{P(OC5H 11)3}2](PF6)2 (3c), respectivamente. A constante de velocidade específica de substituição da amônia em (2a) foi de 4,3 x 10-3 s-1 (25 oC, μ = 0,10 mol L-1). Os aquo complexos trans-[Ru(NH3)4(L)(H2O)] 2+ (L = P(p-tol)3, 4b; L = P(OC5H11)3, 4c) foram gerados pela aquação térmica de uma das moléculas de L, dos respectivos complexos trans-[Ru(NH3)4L2 ]2+ (L = P(p-tol)3, 3b; L = P(OC5H11)3, 3c) e os valores das constantes foram: k-1 = 2,7 x 10-3 s-1 e k-1 = 3,5 x 10-3 s-1, respectivamente. Os valores dos potenciais formais de redução para 3b, 3c e 2a foram 0,50 ± 0,01V; 0,48 ± 0,01V e 0,50 ± 0,02V, respectivamente (vs SCE) [μ = 0,10 mol L-1; NaCF3COO; CH+ = 1,0 x 10-3 CF3COOH]. As reações:
foram estudadas e os valores de k1, k-1 e Keq foram 0,28 mol-1 L s-1, 0,06 s-1, 4,67 mol-1 L [para L = P(m-tol)3, 4a]; 0,25 mol-1 L s-1, 0,04 s-1, 6,25 mol-1 L [para L = P(p-tol)3, 4b] e 12,4 mol-1 L s-1, 5,8 x 10-1 s-1, 21 mol-1 L [para L = P(OC5H11)3, 4c] [25oC; μ = 0,10 mol L-1; NaCF3COO; CH+ = 1,0 x 10-3 CF3COOH].
Direct reactions between [Ru(NH3)5(H2O)](PF6) 2 (1) and P(m-tol)3 (a), P(p-tol)3 (b) and P(OC5H11)3 (c) lead the complexes [Ru(NH3)5P(m-tol)3](PF 6)2 (2a), trans-[Ru(NH3)4{P(p -tol)3}2](PF6)2 (3b) and trans-[Ru(NH3)4{P(OC5H 11)3}2](PF6)2 (3c) respectively. These complexes have been isolated as hexafluorophosphate salts and characterized by elemental analysis, cyclic voltammetry and UV-VIS spectra measurements. One ammonia ligand in 2a is displaced at the a specific rate of 4.3 x 10-3 s-1 (25 oC, μ = 0.10 mol L-1). The aquo species trans-[Ru(NH3)4(L)H2O] 2+ (L = P(p-tol)3, 4b; L = P(OC5H11)3, 4c) are generated by the substitution of one L ligand with H2O, from trans-[Ru(NH3)4(L)2]2+ with k-1 = 2.7 x 10-3 s-1 [L = P(p-tol)3, 3b] and 3.5 x 10-3 s-1 [L = P(OC5H11)33c] (25 oC, μ = 0.10 mol L-1) (NaCF3COO/CF3COOH; CH+ = 1.0 x 10-3 mol L-1). The formal reduction potentials for the species 3b, 3c and 2a are 0.50 ± 0.01V, 0.48 ± 0.01V and 0.50 ± 0.02V, respectively (vs SCE) [μ = 0.10 mol L-1 ; NaCF3COO; CH+ = 1.0 x 10-3 CF3COOH].
The following reactions:
[L = P(m-tol)3, P(p-tol)3 and P(OC5H11)3] were studied and the calculated values for k1, k-1 and Keq are 0.28 mol-1 L s-1, 0.06 s-1, 4.67 mol-1 L [L = P(m-tol)3, 4a]; 0.25 mol-1 L s-1, 0.04 s-1, 6.25 mol-1 L [L = P(p-tol)3, 4b]; 12.4 mol-1 L s-1, 5.8x10-1 s-1, and 21 mol-1 L [L = P(OC5H11)3, 4c], [25.0 ± 0.2 oC; m = 0.10 mol L-1; NaCF3COO; CH+ = 1.0 x 10-3 CF3COOH].
Keywords: ruthenium(II), ammine complexes, phosphine and phosphite ligands
Introduction
Phosphorus compounds are important ancillary ligands in the art of tailoring catalysts. Their presence in the metal coordination sphere can introduce significant changes in the kinetic and thermodynamic properties of metal complexes containing these ligands.
The chemistry of phosphine and phosphite complexes of Ru(II) and Ru(III) tetraammines has received considerable attention,1-11 efforts being directed to the understanding of the trans effect and trans influence of these ligands in the thermal1-11 and photochemical12-17 reactivities of the ruthenium center the complex itself. According to the accumulated data,1-11 for small phosphines and phosphites effect and trans influence are dictated mainly by electronic effects.1
In this work, the attention was focused on the reactions with the following bulky phosphorus ligands L = P(m-tol)3 (θ=170o), P(p-tol)3 (θ=145o) and P(OC5H11)3 (θ=170o). The basicity of these ligands does not change substantially from one another as do their sizes and therefore the influence of these ligands on the properties of [Ru(NH3)4(L)(H2O)]2+ complex ions would be primarily due to steric rather than electronic effects.
Experimental
Chemicals and reagents
The solvents employed were freshly distilled before use. Doubly distilled water was used throughout. All chemicals were of analytical grade purity. The syntheses of the ruthenium compounds, [Ru(NH3)5Cl]Cl2, [Ru(NH3)5(H2O)](PF6) 2 followed the procedures already described.18,19 Ruthenium trichloride (RuCl3.3H2O) was the starting material for the synthesis of the ruthenium complexes.
Apparatus and techniques
All manipulations were performed under argon due to the known sensitivity of Ru(II) complexes to air oxidation. The complexes were stored under vacuum and protected from light. Their purity was checked by means of elemental analysis, cyclic voltammetry and spectrophotometric measurements. Elemental analyses were performed by the staff of the Microanalytical Laboratory of Instituto de Química-USP.
The UV-VIS spectra were recorded on a HP 8451A-diode array spectrophotometer with 1.0 cm quartz cells at room temperature. Kinetic studies were carried out under pseudo-first-order conditions and were monitored by following the changes in absorbance at a selected wavelength.
Cyclic voltammetric measurements were performed in a multifunctional PARC system consisting of a model 175 Universal Programmer a model 173 potentiostat-galvanostat, a model 379 digital coulometer, a model 376 voltage-current conversor and a model Re 0074 X-Y recorder.
For cyclic voltammetric measurements a glassy carbon electrode, platinum wire and SCE were used as the working, auxiliary and reference electrodes, respectively. Formal reduction potentials for a number of substituted ruthenium ammine complexes were evaluated by cyclic voltammetry. The E1/2 values were taken as the average of the anodic and cathodic peak potentials.
Electrochemical reversibility was judged by (a) comparing the ratio of the peak current for the cathodic process relative to the peak current for the anodic process20,21 and (b) comparing under the same conditions the peak to peak separation with that of trans-[Ru(NH3)4P(OC2H 5)3H2O]2+ known to be reversible.1
The electrochemical process in acidic solution is reversible in the sense that the oxidation and reduction wave currents are equal.20-21 The peak to peak separation, 60±5 mV, is of the same magnitude observed for others ammine Ru(II) complexes systems1 which are also know to be reversible.
Kinetic measurements
The reaction shown in equation 1 was monitored both by potentiometric (pH) and spectrophotometric (λmax= 380 nm) measurements.
In the case of the complexes containing two L ligands, equation 2, the substitution reactions were followed spectrophotometrically at λmax= 374 nm for L = P(p-tol)3(3b) and λmax= 300 nm for L = P(OC5H11)3(3c). To avoid the back reaction, the complex concentration was kept around 5.0 x 10-5 mol L-1.
The reactions of trans-[Ru(NH3)4(L)(H2O)](PF 6)2 (L = P(m-tol)3, 4a; P(p-tol)3, 4b; P(OC5H11)3, 4c) shown in equation 3 were studied under pseudo-first-order conditions with excess pyrazine.
Known volumes of deaerated solutions of pyrazine with pH and ionic strength previously adjusted were prepared in a flask under argon. The solution was transferred rapidly through Teflon tubing by gas pressure to a sealed spectrophotometric cell sealed containing a constant and known volume of deaereted solutions of 4a, 4b or 4c.
The formation of trans-[Ru(NH3)4(L)pz](PF6) 2 (L = P(m-tol)3, 5a; P(p-tol)3, 5b; P(OC5H11)3, 5c) was monitored following the changes in absorbance at 402 nm, 400 nm and 390 nm for 4a, 4b and 4c respectively. Pyrazine was chosen as auxiliary ligand because the complexes 5a, 5b and 5c exhibit1 an intense metal-to-ligand charge transfer band (MLCT) in the visible region of the spectra and also because the ligand is very soluble in water and is very good p acceptor. Furthermore there is an extensive volume of data on the trans effect and trans influence of phosphines and phosphates using pyrazine as substituting ligand and trans-[Ru(NH3)4(L)H2O] 2+ type compounds as model.1-11
The observed pseudo-first-order rate constants (kobs) were determined graphically from plots of log (A∞ - At) versus time (where A∞ and At are the final absorbance value and that at time t, respectively). The plots were linear for at least three half-lives. For the substitution reactions, the specific rate constants k1 and k-1 were determined and the equilibrium constants Keq were calculated using the equation kobs = k1[pz]+k-1.
Preparation of the new complexes
In 100 mL of previously degassed acetone, P(p-tol)3 (0.46 g, 1.5 mmol) was dissolved. After 30 min stirring 0.2 g (0.4 mmol) of recently prepared [Ru(NH3)5H2O](PF6) 2 (1) were added. The solution was stirred for 3 h at room temperature, under argon and in the absence of light. The solution volume was then reduced to 10 mL under vacuum and the complex was precipitated upon the addition of 50 mL of degassed ether (peroxide free). The solid was collected by filtration, washed with ether and dried, producing a yield on the order of 50%. Elemental analysis: Found: C, 47.01; H, 4.90; N, 5.32; Calc. For C42H54N4P4 F12Ru1: C, 47.24; H, 5.10; N, 5.25%.
The procedure was the same as described for (3b). Yield 50%. The composition was confirmed by the analytical results (Found: C, 31.80; H, 4.60; N, 8.80; Calc. For C21H36N5P3 F12Ru1: C, 32.31; H, 4.65; N, 8.97%). The reaction of [Ru(NH3)5H2O](PF6) 2 with P(m-tol)3 leads to the formation of 2a. Ammonia trans to P(m-tol)3 is easily aquated, leading to trans-[Ru(NH3)4{P(m- tol)3}H2O]2+ (4a) in 90% yield.
The procedure was the same as described for (3b), except that the phosphite complex 3c was precipitated by adding a mixture of ethanol-ether (1:2) and produced in approximately 25% yield. Elemental analysis: Found: C, 34.30; H, 8.00; N, 5.60; Calc. For C30H78N4O6 P4F12Ru1: C, 34.52; H, 7.53; N, 5.37%.
Results and Discussion
The product from the reaction of [Ru(NH3)5(H2O)(PF6) 2 and P(m-tol)3 was isolated in the form of the pentaammine complex, trans-[Ru(NH3)5{P(m- tol)3}](PF6)2, (2a). The trans complexes containing P(p-tol)3 and P(OC5H11)3 were obtained as tetraammine species trans-[Ru(NH3)4L2 ](PF6)2, L = P(p-tol)3, 3b; P(OC5H11)3, 3c. Compounds 3b and 3c exhibit a fast L substitution, to form trans-[Ru(NH3)4(L)H2O](PF 6)2, L = P(p-tol)3, 4b; P(OC5H11)3, 4c. Complex 2a generates trans-[Ru(NH3)4{P(m- tol)3}H2O](PF6)2, 4a, through a fast trans NH3 ligand substitution.
For 3b and 3c the measured rate constants k-1, for the trans L aquation, are 3.5 x 10-3 s-1 and 2.7 x 10-3 s-1 respectively. For 2a the rate constant for ammonia dissociation is 4.3 x 10-3 s-1, and is independent of [H+] concentration in the 10-1-10-5 mol L-1 range. In acidic solution, the free phosphite ligand hydrolyzes and the product of the hydrolyses has no affinity22-25 for 4c.
In aqueous solutions, the UV spectra of 4a, 4b and 4c exhibit absorption bands at λ = 370 nm (ε = 7.9 x 102 mol L-1 cm-1), λ = 300 nm (ε = 10 x 102 mol L-1 cm-1) and λ = 380 nm (ε = 8.2 x 102 mol L-1 cm-1), respectively. The molar absortivities for these transitions are in the range of those usually observed for the LF (ligand field) transitions in ruthenium(II) ammines.1,26-28 Therefore these bands have been attributed to the symmetry and spin allowed 1A1→1E(1) transition, considering the C4v microssimetry in spite of the phosphane ligands, Figure 1.
The colorless aqueous solutions containing the aquo species 4a-4c rapidly turn pale yellow upon addition of pyrazine. This color is due to the MLCT band, observed in the visible region of the spectrum, 402 nm (ε = 4.8 x 103 mol L-1 cm-1), 400 nm (ε = 5.2 x 103 mol L-1 cm-1) and 390 nm (ε = 7.3 x 103 mol L-1 cm-1) for the P(m-tol)3, (5a); P(p-tol)3, (5b) and P(OC5H11)3 (5c) pyrazine complexes, respectively.
The cyclic voltammograms show single, well defined waves for the Ru(III)/Ru(II) couple for all complexes, displaying reversible behavior at sweep rates between 20-200 mV s-1, see Figure 2. The formal reduction potential for the Ru(III)/Ru(II) couple of the 4a, 4b and 4c ions are, respectively, 0.50 ± 0.01V; 0.48 ± 0.01V and 0.50 ± 0.02V (vs SCE), [25 oC; μ = 0.10 mol L-1; NaCF3COO; CH+ = 1.0 x 10-3 CF3COOH]. The small variation in the formal reduction potential data is expected since the electronic properties of these ligands are quite similar.1,8,10
The lability of the coordinated water molecule in 4a-4c was assessed by measuring the rate at which it is substituted by pyrazine. The plots of kobs as a function of [pz] lead to calculated values of 0.28 mol-1 L s-1, 0.06 s-1 , 4.67 mol-1 L for k1, k-1 and Keq respectively (25.0 ± 0.2oC) when L = (4a); 0.25 mol-1 L s-1, 0.04 s-1 , 6.25 mol-1 L = (4b) and 12.4 mol-1 L s-1, 5.8 x 10-1 s-1 , 21 mol-1 L = (4c).
The plots of kobs versus [pz] for the substitution of the water molecule in complexes 4a-4c have been studied using a number of different phosphines and phosphates L1-11 and exhibit different profiles according to the nature of the entering ligand L. For the complexes reported herein the plots of kobs versus [pz], were linear until [pz] < 0.10 mol L-1. However if [pz] > 0.10 mol L-1, the rates become independent of the substituting ligand concentration.
The trans effect and trans influence of phosphane ligands L have been evaluated1-11 using the kinetic parameters k1, k-1 and Keq for the reaction shown in equation 4.
Based on this approach, the specific rate constant k1, at which the water molecule is substituted by pz, (related to ΔG#1 ), is proposed as an indicator of the relative trans effect of L.1 The Keq, (related to ΔGeq), was proposed as a relative measure of the trans influence of L.1
According to this approach, the positions of the P(m-tol)3, P(p-tol)3 and P(OC5H11)3 ligands in the series of increasing trans effect are as follows: P(OPh)3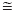 ETPB < P(p-tol)3
ETPB < P(p-tol)3 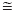 P(m-tol)3 < PTA < P(OC2H4Cl)3 ≤ PPh3 < dppe
P(m-tol)3 < PTA < P(OC2H4Cl)3 ≤ PPh3 < dppe 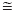 H2Pcy < P(OMe)3 < P(OEt)3 < DMPP < P(tBu)3
H2Pcy < P(OMe)3 < P(OEt)3 < DMPP < P(tBu)3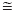 P(OiPr)3 < P(OnBu)3 < P(OC5H11)3 < P(OH)(OEt)2
P(OiPr)3 < P(OnBu)3 < P(OC5H11)3 < P(OH)(OEt)2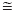 P(OH)3 < P(nBu)3 < P(Et)3 < P(OH)2O- < P(OEt)2O -; and the increasing trans influence series can be written as: P(Et)3
P(OH)3 < P(nBu)3 < P(Et)3 < P(OH)2O- < P(OEt)2O -; and the increasing trans influence series can be written as: P(Et)3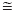 DMPP
DMPP 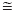 H2PCy
H2PCy 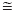 P(nBu)3 < P(OEt)2O -
P(nBu)3 < P(OEt)2O -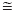 P(OH)2O- < dppe < PPh3
P(OH)2O- < dppe < PPh3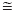 P(OnBu)3 < P(tBu)3
P(OnBu)3 < P(tBu)3
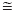 P(OiPr)3
P(OiPr)3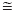 P(OEt)3 < P(OH)3
P(OEt)3 < P(OH)3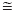 P(OH)(OEt)2 < P(OMe)3 < PTA < P(OC2H4Cl)3
P(OH)(OEt)2 < P(OMe)3 < PTA < P(OC2H4Cl)3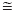 ETPB < P(p-tol)3 < P(m-tol)3 < P(OPh)3.
ETPB < P(p-tol)3 < P(m-tol)3 < P(OPh)3.
Figure 3 shows the plot of log k1 versus Eo,Ru(III)/Ru(II) for the trans-[Ru(NH3)4(L)H2O] 3+/2+ species, which displayed a good linear fit. In this figure the trend can be interpreted in the following way: the more positive the Eo,Ru(III)/Ru(II), the more stable is the complex containing the Ru(II) metal center in comparison with Ru(III) and the slower will be the rate of the water substitution by pz, since the electronic stabilization of the Ru is basically due to the Ru(II)→L back bonding intensity.1,2,26,29-31 It is reasonable to assume for the series that the strongest back bonding interaction occurs when L = P(OPh)3 and the weakest, when L = P(OH)2O-. The plot still exhibits a linear tendency after the inclusion of the new ligands.
Acknowledgments
The authors thank the Brazilian agencies FAPESP, CNPq for financial support.
9. Caetano, W.; Alves, J.J.F.; Lima Neto, B.S.; Franco, D.W.; Polyhedron 1994, 14, 1295.
11. Mazzetto, S.E.; Gambardella, M.T.P.; Santos, R.H.A.; Lopes, L.G.F.; Franco, D.W.; Polyhedron 1999, 18, 979.
16. Borges, S.S.S.; Franco, D.W.; unpublished results.
17. Gomes, M.G.; Franco, D.W.; unpublished results.
Received: April 23, 2001
Published on the web: August 21, 2002
FAPESP helped in meeting the publication costs of this article.
- 1. Franco, D.W.; Coord. Chem. Rev 1992, 119, 199.
- 2. Nascimento Filho, J.C.; Lima, J.B.; Lima Neto, B.S.; Franco, D.W.; J. Mol. Cat 1994, 90, 257.
- 3. Santos, L.S.; Araujo, S.B.; Franco, D.W.; J. Braz. Chem. Soc 1994, 5, 117.
- 4. Mazzetto, S.E.; Rodrigues, E.; Franco, D.W.; Polyhedron 1993, 12, 971.
- 5. Rezende, N.M.S.; Martins, S.C.; Marinho, L.A.; Santos, J.A.V.; Tabak, M.; Perussi, J.R.; Franco, D.W.; Inorg. Chem 1991,182, 87.
- 6. Neiva, S.M.C.; Santos, L.S.; Araujo, S.B.; Nascimento Filho, J.C.; Franco, D.W.; Can. J. Chem 1987, 65, 372.
- 7. Frugeri, P.M.; Vasconcellos, L.C.G.; Mazzetto, S.E.; Franco, D.W.; New J. Chem 1997, 21, 349.
- 8. Lima Neto, B.S.; Nascimento Filho, J.C.; Franco, D.W.; Polyhedron 1995, 15, 1965.
- 10. Lima Neto, B.S.; Franco, D.W.; Eldik, R.; J. Chem. Soc., Dalton Trans 1995, 463.
- 12. Mazzetto, S.E.; Franco, D.W.; Plicas, L.M A.; Tfouni, E.; Inorg. Chem 1992, 31, 516.
- 13. Mazzetto, S.E.; Tfouni, E.; Franco, D.W.; Inorg. Chem 1996, 35, 3509.
- 14. Mazzetto, S.E.; Gehlen, M.H.; Franco, D.W.; Inorg. Chim. Acta 1997, 254, 79.
- 15. Mazzetto, S.E.; Gehlen, M.H.; Neumann, M.G.; Franco, D.W.; J. Photochem. Photobiol 1997, 104, 39.
- 18. Franco, D.W.; Taube, H.; Inorg. Chem 1978, 17, 571.
- 19. Isied, S.S.; Taube, H.; Inorg. Chem 1973, 13, 1545 and references therein.
- 20. Nicholson, R.S.; Anal. Chem 1966, 38, 1406.
- 21. Headridge, J.B.; In Electrochemical Techniques for Inorganic Chemists; Academic Press: London, 1969.
- 22. Gerrard, W.; Hudson, G.; Gillard, R.D.; Mc Cleverty, J.A.;In Comprehensive Coordination Chemistry; Pergamon Press: Oxford, 1987, vol 2.
- 23. Corbridge, D.E.C.; In Phosphorus; Elsevier: Amsterdam, 1978.
- 24. Nascimento Filho, J.C.; Rezende, J.M.; Lima Neto, B.S.; Franco, D.W.; Inorg. Chim. Acta 1988, 145, 111.
- 25. Rezende, J.M.; Franco, D.W.; Trans. Met. Chem 1987, 12, 267.
- 26. Taube, H.; Com. Inorg. Chem 1981, 17, 1 and references therein.
- 27. Lever, A.B.P.; In Inorganic Electronic Spectroscopy; Elsevier: Amsterdam, 1984.
- 28. Mazzetto, S.E.; Tfouni, E.; Franco, D.W.; Inorg. Chem 1996, 35, 3509.
- 29. Tolman, C.A.; Chem. Rev 1977, 77, 313.
- 30. Mc Auliffe, C.A., In Comprehensive Coordination Chemistry; Wilkinson, G.; Gillard, R.D.; Mc Cleverty, J.A.; Pergamon: Oxford, 1987, vol 2.
- 31. Isied, S.S.; Taube, H.; Inorg. Chem. 1976, 15, 3070.
Publication Dates
-
Publication in this collection
08 Oct 2008 -
Date of issue
Sept 2002
History
-
Received
23 Apr 2001












