Abstracts
Pterodon pubescens Benth. (Pp) seeds, commercially available in Brazil, are used in folk medicine in anti-inflammatory, analgesic, and anti-rheumatic preparations. The present study demonstrated the antinociceptive properties of isomers 6α-hydroxy-7β-acetoxy-vouacapan-17β-oate methyl ester and 6α-acetoxy-7β-hydroxy-vouacapan-17β-oate methyl ester (C1), isolated from (Pp), employing different experimental models. A stability study was performed to investigate the relationship of microencapsulation by spray-drying on the maintenance of antinociceptive action. Therefore, C1 and Pp extract samples were monitored in accelerated stability study, evaluating both microencapsulated and non-microencapsulated samples. It was observed that sample C1 possess antinociceptive activity revealed by writhing and formalin tests; C1 showed significantly anti-allodynic, but not antihyperalgesic effect; the microencapsulation maintained the activity and integrity of both, sample C1 and Pp crude extract; microencapsulation by spray drying is a useful alternative to increase shelf life.
microcapsules; stability; spray-drying; antinociceptive; Pterodon pubescens
As sementes de Pterodon pubescens Benth. (Pp), disponíveis comercialmente no Brasil, são utilizadas na medicina popular em preparações anti-inflamatórias, analgésicas e anti-reumáticas. O presente estudo demonstrou as propriedades antinociceptivas da mistura de isômeros 6α-hidroxi-7β-acetoxi-vouacapano-17β-oato de metila e 6α-acetoxi-7β-hidroxi-vouacapano-17β-oato de metila (C1) isolada de Pp, empregando modelos experimentais diferentes. Um estudo de estabilidade foi realizado para investigar a relação da microencapsulação por "spray-drying" na manutenção do efeito antinociceptivo. Para isso, amostras de C1 e extrato de Pp foram monitorados no estudo de estabilidade acelerada, avaliando tanto amostras microencapsuladas como não microencapsuladas. Foi observado que a amostra C1 possui atividade antinociceptiva, revelada pelos ensaios das contorções abdominais e de formalina; foi observada também atividade significativa antialodínica, mas não efeito anti-hiperalgésico. A microencapsulação manteve a atividade e integridade de C1 e do extrato bruto de Pp, demonstrando que a microencapsulação por "spray drying" é uma alternativa útil para aumentar o tempo de prateleira.
ARTICLE
Pterodon pubescens benth: stability study of microencapsulated extract and isolated compounds monitored by antinociceptive assays
Leila ServatI,II; Humberto M. SpindolaI,II; Rodney A. F. RodriguesI; Ilza M. O. SousaI; Ana L. T. G. RuizI; João E. de CarvalhoI,II; Mary A. FoglioI,II,* * e-mail: foglioma@cpqba.unicamp.br
ICentro Pluridisciplinar de Pesquisas Químicas, Biológicas e Agrícolas (CPQBA), Universidade Estadual de Campinas, CP 6171, 13083-970 Campinas-SP, Brazil
IIÁrea de Farmacologia, Anestesiologia e Terapêutica, Departamento de Ciências Fisiológicas, Faculdade de Odontologia de Piracicaba, Universidade Estadual de Campinas, CP 52, 13414-903 Piracicaba-SP, Brazil
ABSTRACT
Pterodon pubescens Benth. (Pp) seeds, commercially available in Brazil, are used in folk medicine in anti-inflammatory, analgesic, and anti-rheumatic preparations. The present study demonstrated the antinociceptive properties of isomers 6α-hydroxy-7β-acetoxy-vouacapan-17β-oate methyl ester and 6α-acetoxy-7β-hydroxy-vouacapan-17β-oate methyl ester (C1), isolated from (Pp), employing different experimental models. A stability study was performed to investigate the relationship of microencapsulation by spray-drying on the maintenance of antinociceptive action. Therefore, C1 and Pp extract samples were monitored in accelerated stability study, evaluating both microencapsulated and non-microencapsulated samples. It was observed that sample C1 possess antinociceptive activity revealed by writhing and formalin tests; C1 showed significantly anti-allodynic, but not antihyperalgesic effect; the microencapsulation maintained the activity and integrity of both, sample C1 and Pp crude extract; microencapsulation by spray drying is a useful alternative to increase shelf life.
Keywords: microcapsules, stability, spray-drying, antinociceptive, Pterodon pubescens
ABSTRACT
As sementes de Pterodon pubescens Benth. (Pp), disponíveis comercialmente no Brasil, são utilizadas na medicina popular em preparações anti-inflamatórias, analgésicas e anti-reumáticas. O presente estudo demonstrou as propriedades antinociceptivas da mistura de isômeros 6α-hidroxi-7β-acetoxi-vouacapano-17β-oato de metila e 6α-acetoxi-7β-hidroxi-vouacapano-17β-oato de metila (C1) isolada de Pp, empregando modelos experimentais diferentes. Um estudo de estabilidade foi realizado para investigar a relação da microencapsulação por "spray-drying" na manutenção do efeito antinociceptivo. Para isso, amostras de C1 e extrato de Pp foram monitorados no estudo de estabilidade acelerada, avaliando tanto amostras microencapsuladas como não microencapsuladas. Foi observado que a amostra C1 possui atividade antinociceptiva, revelada pelos ensaios das contorções abdominais e de formalina; foi observada também atividade significativa antialodínica, mas não efeito anti-hiperalgésico. A microencapsulação manteve a atividade e integridade de C1 e do extrato bruto de Pp, demonstrando que a microencapsulação por "spray drying" é uma alternativa útil para aumentar o tempo de prateleira.
Introduction
Pterodon pubescens Benth. (Leguminosae) seeds are commercially available in Brazil. The plant's crude alcoholic extract is used in folk medicine in anti-inflammatory, analgesic, and anti-rheumatic preparations.1,2 Phytochemical studies of Pterodon genus have revealed the presence of alkaloids, isoflavones and diterpenes. Furan diterpenes were identified and isolated from Pterodon species.3-7Authors have reported that furanoditerpenes possessing vouacapan skeleton are involved with the anti-inflammatory, antinociceptive and antiproliferative properties of Pterodon pubescens seed crude extract.7-12
We previously reported the antinociceptive properties of geranylgeraniol and 6α,7β-dihydroxy-vouacapan-17β-oate methyl ester, compounds isolated from Pterodon pubescens Benth seeds.12 This report gives sequence to that study, evaluating the antinociceptive properties of isomers 6α-hydroxy-7β-acetoxy-vouacapan-17β-oate methyl ester and 6α-acetoxy-7β-hydroxy-vouacapan-17β-oate methyl ester (C1) isolated from P. pubescens crude extract, as well as the influence of the microencapsulation process as a tool for maintaining stability of both the crystals and extracts. Therefore, we evaluated microencapsualed and free samples of C1 and the crude extract on the antinociceptive assays.
Experimental
General experimental procedures
IR spectra were recorded on a JASCO-FT/IR-410 spectrometer. 1H, 13C NMR and 2D experiments were conducted on a Varian Inova-500 spectrometer (11 T). Chemical shifts were recorded in CDCl3 solutions and quoted relative to TMS (δH 0.0, 1H NMR) and CHCl3 (δH 77.0, 13C NMR). High-resolution electron inpact ionization mass spectrometry (HREIMS) was recorded on a VG-AutoSpec high resolution mass spectrometer (70 eV) using direct probe. GC/MS was carried out using a HP-6890/5970 system equipped with a J&W Scientific DB-5 fused capillary column (25 m x 0.2 mm x 0.33 µ). Temperature program was 40 (5 oC min-1) - 300 ºC (10 min). Injector 250 ºC, detector 300 ºC. Helium was used as carrier gas (0.7 bar, 1 mL min-1). The MS were taken at 70 eV. Scanning speed was 0.84 scans s-1 from 40 to 550. Sample volume was 1 µL. Split: 1:40. Column chromatography (CC): silica gel (0.063 x 0.200 mm, Merck®). TLC (thin layer chromatography): precoated plates (775554 Merck®), UV detection and anisaldehyde solution.
Phytochemistry
Plant material
Pterodon pubescens (Pp) seeds were previously collected in Minas Gerais state (Ponto Chique city), in March 2008 and identified by Prof. Dr. Jorge Yoshio Tamashiro from IB-Unicamp (Department of Botany), who identified the plant species. A voucher specimen was deposited at the University of Campinas (Unicamp) Herbarium, under number 1402.
Extracts production and compounds isolation
Freeze-dried seeds (1000 g) were ground in mill (POLI model LS-06) and extracted with dichloromethane three times during 2 h periods, with 5:1 solvent/plant ratio, at room temperature. The extract was dried over anhydrous Na2SO4 to remove residual water, filtered and concentrated under vacuum (BUCHI model RE 215), providing the crude extract in 40% yield.
The crude extract (240 g) was chromatographed over silica gel (1900 g) and eluted sequentially with n-hexane/ethyl acetate gradients. The fractions were monitored by thin layer chromatography (TLC). The TLC plates were sprayed with p-anisaldehyde reagent (50 mL acetic acid, 0.5 mL sulfuric acid and 0.5 mL p-anisaldehyde) followed by heating at 100 ºC. Similar fractions were grouped according to their TLC profile. The fractions that were eluted with hexane:ethyl acetate (75:25) providing fraction E, gave crystals (8.4 g; 3.5%). The crystals were analysed by gas chromatography (GC-MS) with identification of an isomers mixture. Comparison of the physical and spectral data (mass,1H NMR, 13C NMR) of sample C1 with reported data4-6 provided identification of the isomer mixture as a 1:1 ratio of (i) 6α-hydroxy-7β-acetoxy-vouacapan-17β-oate methyl ester and (ii) 6α-acetoxy-7β-hydroxy-vouacapan-17β-oate methyl ester. The isomers mixture showed a melting point in the range of 178-182 ºC.
Microcapsules production
Pp extract and C1 microencapsulation
A mini spray-dryer Buchi, model B-290, with inert loop B-295 (Flawil, Switzerland) was used for the microencapsulation process; operating conditions are shown in Table 1.
Samples were homogenized with maltodextrin/arabic gum (1:1) in a ratio of 2:8, using Tween 80® as emulsifier and IKA® T10Basic-Ultra Turrax®.
Particle morphology
The samples processed by spray-dryer were set at a double-sided adhesive tape in cylindrical aluminum (stubs) according to the procedure described by Rosemberg and Young.13 The cylinders were subjected to metallization by coating with a thin layer of gold using a current of 40 mA during 180 s under vacuum. This technique, known as sputtering, was performed in an evaporator (Balzer). The morphology of the samples was evaluated using a scanning electronic microscope (JEOL), operating at 10 kV.
Stability evaluation
Microcapsules produced were stored at climate chamber, with constant temperature (40 ºC) and humidity (70%). Samples of microencapsulated C1 were taken at 0, 30, 60, 150 and 180 days and of microencapsulated Pp extract, were taken at 0 and 180 days. The samples were submitted to antinociceptive activity assay. Pp extract and C1 without encapsulation were used as a control.
Pharmacology
Drugs
All drugs and compounds were diluted with Tween80 1% (Sigma-Aldrich, USA) in 0.9% saline solution (NaCl diluted in distilled water), C1 microencapsulated was diluted with saline solution. The following substances were used: pentobarbital, indomethacin, morphine hydrochloride (Cristália, Brazil), acetic acid, complete Freund's adjuvant (CFA), carrageenan (Sigma- Aldrich, USA) and formaldehyde (Chemco-Brasil).
Animals
Male Swiss mice (25-35 g) and Wistar rats (150-250 g) were kept at 25 ± 2 ºC exposed to 12 h light-dark cycles (the light phase starting at 7:00 am) and maintained in animal facilities (10 and 5 animals per cage, respectively) with water and food ad libitum, for at least 7 days prior to the assays. Separate groups of mice and rats were used only once for each experiment, except in the hyperalgesia and allodynia assays. The intraperitoneal (i.p.) route was used for all experiments, based on previous report.12 Studies were carried out in accordance with the current guidelines for veterinary care of laboratory animals14 and were performed under the consent and surveillance of Unicamp's Institute of Biology Ethics Committee for Animal Research (1951-1).
Evaluation of locomotor activity
The ambulatory behavior was assessed in an open-field test as described previously.12 The apparatus consisted of a plastic box measuring 45 x 45 x 20 cm, with the floor divided into 9 equal squares (15 x 15 cm). The number of squares crossed with all paws (crossing) was counted in a 3-min session. Mice were 30 min previously treated intraperitoneally (i.p.) with C1 (30 mg kg-1), pentobarbital (35 mg kg-1) or vehicle. Results were expressed as mean ± S.E.M. of 4 animals per group.
Abdominal constriction induced by acetic acid (writhing test)
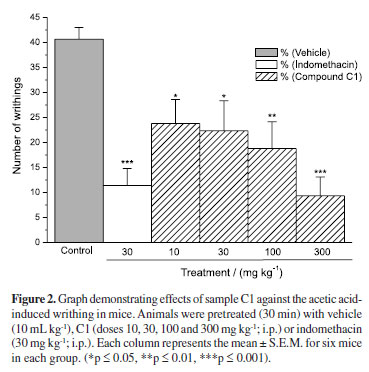
The writhing test was carried out as described previously.12 Groups of mice (n = 6) were treated i.p. with vehicle (10 mL kg-1), indomethacin (30 mg kg-1) or sample C1, in a dose-dependent manner: 10, 30, 100, and 300 mg kg-1, to determine the effective dose (ED50). Writhing was induced by an i.p. injection of 0.8% acetic acid solution (0.1 mL per 10 kg-1), 30 min after treatment. After injection of the acetic acid solution, the numbers of writhings (abdominal constrictions) were cumulatively counted over 15 min, for nociception measurement. Data represents the average of the total writhing observed per dose concentration.
Formalin test
The procedure was the same as described by Hunskaar and Hole,15 with few changes. The nociceptive stimulus is a subplantar (s.pl.) injection of formalin solution (2% formaldehyde in PBS; 20 µL per paw) into the dorsal surface of the right hind paw of the mice. Groups of animals (n = 6) were treated with sample C1 (3, 10, 30 and 100 mg kg-1, i.p.) 30 min before the formalin injection. Indomethacin (30 mg kg-1, i.p.) and morphine (10 mg kg-1, i.p.) were used as reference drugs. Control animals received only the vehicle. The animals were placed individually in an observation chamber made of transparent acrylic to allow clear observation of the animal's paws. The amount of time the animal spent licking the injected paw, indicative of pain, was recorded during 45 min after formalin injection. The initial nociceptive scores peaked 5 min after formalin injection (early phase) and 25-45 min after formalin injection (late phase), representing the neurogenic and inflammatory pain responses, respectively.
Mechanical allodynia induced by complete Freund adjuvant (CFA)
The procedures were developed and standardized in our laboratory based on the method previously described16 with changes in protocol and data analysis. Different groups of rats (n = 5) were used, and inflammation was induced with a solution of CFA (1 mg mL-1 of heat killed Mycobacterium tuberculosis in 85% paraffin oil and 15% mannide monoleate) injected (0.1 mL) into the plantar surface of the right hind paw. The left hind paw received the same volume of saline solution (NaCl 0.9% diluted in distilled water) in order to equalize the sensibility of animals caused by the injection. Mechanical allodynia was assessed using the dynamic plantar anaestesiomether apparatus (Ugo Basile, model 37450, Italy), which consisted of an elevated wire mesh platform to allow access to the ventral surface of the hind paws. A steel rod (diameter 0.5 mm) was pushed against the hind paw with ascending force (touch stimulator). The force ranged from 0 to 35 g over a 20 s period. When the animal withdrew the hind paw, the mechanical stimulus was automatically stopped, and the force applied by the animal to withdraw the paw was recorded to the nearest 0.1 g. An allodynia score was determined after four consecutive measurements using the touch stimulator sequentially on the left and right hind paw and calculated considering the formula below determined by the authors:
Left hind paw value / Right hind paw value = Allodynia score
The basal score was measured before CFA injection on day 0, and animals considered for testing were those with a mean value nearest to 1 (demonstrating no significant difference between both paw stimuli). After CFA injection, measurements were carried out considering three different phases, as follows: 4 h on day 0 (acute pain); 24 h on day 1 (sub-acute pain) and on day 14 (chronic pain). Vehicle (10 mL kg-1) or sample C1 were administered (30 mg kg-1, i.p.) 30 min prior to touch stimulation, in order to evaluate the possible anti-allodynic activity observed for each phase. A positive control was not employed as the aim of this test was to evaluate the activity of compounds using doses equal to those used in the tests described below for evaluating potential antinociceptive mechanisms.
Mechanical hyperalgesia induced by carrageenan
The procedures used for this study were similar to those described previously17with some changes in the protocol and data analysis. Different groups of rats (n = 6) were submitted to pressure stimulus (0 to 500 g) on the right hind paw using an analgesy-meter (Ugo Basile, model 37215/372116, Italy) prior to carrageenan injection, in order to determine the basal value. The hypernociceptive response was considered when animals vocalized or withdrew the paw from the equipment, demonstrating pain. After this first measurement, animals received a carrageenan (0.1 mL) intraplantar (i.pl.) injection (2.5% in saline) into the right hind paw surface. After 2.5 h, they were submitted to pressure again, to evaluate whether the hypernociceptive state was reached (40% decrease). Animals were than treated with vehicle (10 mL kg-1, i.p. - negative control), indomethacin (30 mg kg-1, i.p. - positive control) and sample C1 (30 mg kg-1, i.p.), and the mechanical hyperalgesia was evaluated after 30 min, 1 h, 2 h and 3 h. The value demonstrating mechanical hyperalgesia was obtained after each measurement, and the results were shown as decreased percentage compared to the pressure tolerated in the basal (100%) value for each group.
Statistical analysis
All results were submitted to one way analysis of variance (ANOVA) with repeated measurements, p < 0.05 was considered the critical level for evaluating significant difference between the control and treated groups, followed by Duncan´s Test, using StatSoft® software. Graphs were designed using the Origin® software.
Results and Discussion
In the present study, we investigated the antinociceptive activity of vouacapans sample C1 (Figure 1) isolated from P. pubescens (Pp), employing different experimental models. A stability study was performed to investigate the influence of microencapsulation by spray-drying on maintenance of antinociceptive action. For this purpose, were monitored sample C1 and Pp crude extract activity in acelerated stability study (180 days period), comparing both microencapsulated and non-microencapsulated samples.
The most relevant findings of this paper were that sample C1 (i) possess antinociceptive activity revealed by writhing test; (ii) is more effective on the first phase (neurogenic pain) then second phase (inflammatory pain) of formalin test; (iii) showed significantly anti-allodynic, but not antihyperalgesic effect; (iv) the microencapsulation kept the activity and integrity of both, sample C1 and Pp crude extract; (v) microencapsulation by spray drying is a useful alternative to increase shelf life time.
Evaluation of C1 antinociceptive, anti-hypernociceptive and anti-allodynic activities
Initially, the open field test was performed in order to exclude the possibility of the sample C1 action could be related to non-specific disturbances in the locomotor activity of the animals.12 The treatment with sample C1 (30 mg kg-1, i.p.) did not cause any significant change in the ambulation of mice when tested in the open field. However, pentobarbital (35 mg kg-1, i.p.) significantly (p < 0.001) reduced the locomotor activity of animals in this test. The mean number of crossings was 48.2 ± 2.2, 13.7 ± 6.1, 40 ± 9 for vehicle, pentobarbital and sample C1, respectively.
With this result, the antinociceptive properties of sample C1 were evaluated using the writhing test. Acetic-acid induced pain is a screening tool for the assessment of analgesic or anti-inflammatory properties of natural or synthetic compounds. Inflammatory pain transmission primarily involves peripheral polymodal receptors around small vessels which signal to the CNS via sensory afferent C-fibers entering the dorsal horn. Acetic acid may act indirectly by inducing the release of endogenous mediators, which stimulate the nociceptive neurons and can be blocked by nonsteroidal anti-inflammatory drugs (NSAIDs) and opioids.18 This method has good sensitivity but poor specificity allowing misinterpretation of the results. This problem can be avoided by complementation with other nociception models, as described here.
The results presented in Figure 2 showed that sample C1 produced a significant effect, reducing writhings of the mice in a dose-dependent manner, compared to the control group (vehicle only), presenting ED50 = 35.6 mg kg-1.
In order to confirm this property, and distinguish this action between inflammatory and neurogenic pain, the formalin test was performed. The intraplantar injection of formalin produces a biphasic response characterized by an immediate and intense increase in the spontaneous activity followed by a quiescent phase and then a more prolonged increase in cell firing of both primary afferents as well as dorsal horn neurons.19 Neurogenic pain is caused by direct activation of nociceptive nerve terminals, while inflammatory pain is mediated by a combination of peripheral input and spinal cord sensitization.15 Intraplantar injection of formalin in rodents produces significant increases in spinal levels of different mediators, such as excitatory amino acids, PGE2, and nitric oxide, as well as kinins and other peptides.20 Furthermore, systemic spinal and supraspinal administration of tachykinin receptor antagonists, NOS inhibitors, NMDA receptor antagonists, opioids,α2-adrenoceptor agonist, and NSAIDs can all antagonize formalin induced nociception.21
As shown in Figure 3, treatment with sample C1 produced a marked reduction of the licking time in the first phase. This result suggests that the antinociceptive action may be related to both central and peripheral mechanisms. In the second phase (inflammatory pain), sample C1 was effective only at the highest doses (100 mg kg-1). The reference drug indomethacin (30 mg kg-1) inhibited pain induced by formalin in the second phase, whereas morphine (10 mg kg-1) significantly attenuated the pain responses of both phases.
Next, we investigated the anti-hypernociceptive effect of sample C1 induced by carrageenan in rats. Carrageenan-induced hind paw inflammation is a neutrophil-mediated acute inflammatory response that produces hind paw swelling, erythematic and localized hyperthermia.22 This inflammatory method has become a widely used model for studying acute inflammation, involving macrophages, mast cells, and endothelial cells.23 The results presented in Figure 4 showed that indomethacin (30 mg kg-1, i.p.) produced significant anti-hypernociception activity increasing the percentage of stimulus compared to the control group (vehicle), whereas sample C1 (30 mg kg-1, i.p.) did not present any effect 30 min, 1 h, 2 h and 3 h after carrageenan injection. In this assay, which has essentially inflammatory components, sample C1 was not effective, suggesting that C1 has a higher affinity for neurogenic components, corroborating the results observed in the second phase of the formalin test, in which there was a need to increase dosage to have activity in inflammatory pain.
Finally, the anti-allodynic effect of sample C1 was evaluated throughout a persistent pain model caused by complete Freund adjuvant (CFA). Allodynia (pain-like responses to normally innocuous stimuli) was induced by CFA intraplantar injection,24 which involves central sensitization due to the release of multiple inflammatory and pain mediators that account for sensitivity increase of both peripheral sensory afferents at the injury site, and in the central nervous system.25
The results presented in Figure 5 showed that sample C1 (30 mg kg-1, i.p.) produced a significant anti-allodynic effect reducing the allodynic score compared to the control group during the acute (4 h post CFA) and sub acute phases (24 h post CFA) of the test. Sample C1 exerted no activity during the chronic phase (14 days post CFA). These results demonstrated the activity of sample C1 during the acute and sub acute phases of allodynic response, suggesting activity related to central and/or peripheral pathway.
Evaluation stability study
Spray-drying is the most commonly used method in the herbal processing industries, but there are few reports on the influence of adjuvants and technological processing on the herbal extracts or herbal substances.26 In the development of herbal medicines, bioguided techniques have been the methods of choice among the technological processes. This approach allows optimizing formulations with a view toward maximizing the therapeutic effect.27 This study evaluated for the first time the contribution of microencapsulation by spray drying in stability of crude extract and isolated compounds, employing antinociceptive activity assays.
Once we demonstrated the potential use of C1 for pain relief, we proceed to evaluate the microencapsulation by spray-drying processes as a tool for maintaining P. pubescens extracts and sample C1 integrity.
The microcapsules of Pp crude extract or sample C1 were produced by a spray-drying technique using maltodextrin and arabic gum (1:1) as wall material. The excellence properties of arabic gum as emulsifier, good volatile retention and low viscosity added to low cost of maltodextrin, makes this blend a great choice for wall material in the production of microcapsules.28 Images obtained by scanning electron microscopy (images not show) demonstrated that the microencapsulated material has predominantly rough surface, which is very common for this type of technique due to contraction of the particles during the fast drying.29
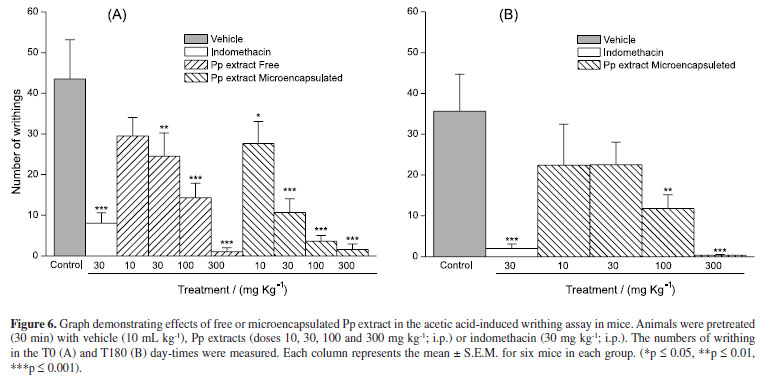
Initially, we evaluated the Pp crude extract stability, employing writhing test for the antinociceptive activity evaluation. This assay was performed with free and microencapsulated Pp extract samples, representative of 0 (T0) and 180 (T180)-day times of the stability study. The results are shown in Figure 6.
The free and microencapsulated extracts demonstrated activity in all the tested dosages (10, 30, 100 and 300 mg kg-1, i.p.). The free and microencapsulated extracts gave ED50 values of 32.3 mg kg-1 and 12.8 mg kg-1 respectively, showing that microencapsulation processes maintained the in vivo extract activity. An assay with the extract´s microcapsules was performed after 180 day time, demonstrating that the effect was preserved (ED50 32 mg kg-1). The free extract was not evaluated in this time due to the glassy appearance of the sample, which is not adequate for the antinociceptive assay.
The results showed that spray-drying microencapsulation enabled the use of de Pp extract after stocking, besides preserving the in vivo effect. This technique showed to be a useful alternative to increase shelf life.
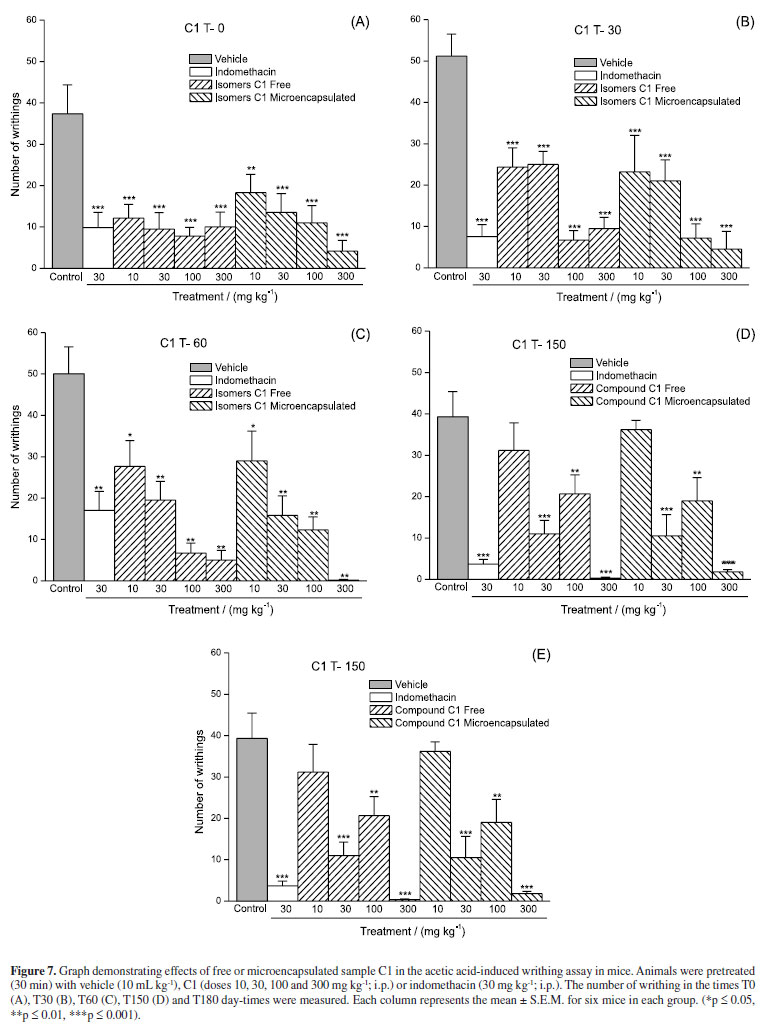
To determine the stability of sample C1, writhing test were performed with free and microencapsulated C1 at T0, T30, T60, T150 and T180 day-times of stability study. The results of dose-response curves are represented in Figure 7.
The ED50 values, calculated from the results of dose-response curves, were 10, 10.5, 13, 32 and 55 mg kg-1 for the free and 10, 8, 14, 39 and 44 mg kg-1 for the microencapsulated samples (Figure 8).
In the in vivo assays, using the writhing test in mice, both the free and the microencapsulated sample C1, were effective in the dosages tested, maintaining the original characteristics.
At lower dosages both free and microencapsulated sample C1, decreased activity as stability study time lapsed (Figure 7), resulting in need of higher dosages necessary to reduce the number of contortions to 50% (ED50) (Figure 8). A gradual decrease in both samples C1 effectiveness was observed, mainly after 60 days, preserving however their activity until the end of the study (T180-day time).
The free and microencapsulated sample C1 maintained stability even when stored in drastic temperature and humidity conditions. The microencapsulation process, not only preserves the content, but also solves the inconvenience of low aqueous solubility.
Conclusions
The most relevant findings of this paper were that sample C1 (i) possess antinociceptive activity revealed by writhing test; (ii) is more effective on the first phase (neurogenic pain) then second one (inflammatory pain) of formalin test; (iii) showed significantly anti-allodynic, but not antihyperalgesic effect; (iv) the microencapsulation kept the activity and integrity of both sample C1 and Pp crude extract; (v) microencapsulation by spray drying is a useful alternative to increase shelf life time.
The free and microencapsulated sample C1 maintained stability even when stored in drastic temperature and humidity conditions. Whereas the crude Pp extract only maintained stability with the microencapsulation process, not only preserving the content, but also solving the inconvenience of low aqueous solubility.
Acknowledgments
The authors thank CAPES, FAPESP and CNPq for financial support and research fellowships.
Submitted: May 9, 2011
Published online: June 14, 2012
FAPESP has sponsored the publication of this article.
- 1. Pio Correa, M.; Dicionário das Plantas úteis do Brasil e das Exóticas Cultivadas, vol. I, Inst. Bras. Des. Flor.: Rio de Janeiro, 1975.
- 2. Lorenzi, H.; Árvores Brasileiras. Manual de Identificação e Cultivo de Plantas Arbóreas Nativas do Brasil, vol. 1, 2nd ed., Instituto Plantarum de Estudos da Flora: São Paulo, 1998.
- 3. Mahjan, J. R.; Monteiro, M. B.; J. Chem. Soc. Perkin Trans. 1973, 1, 520.
- 4. Fascio, M.; Mors, W. B.; Gilbert, B.; Mahajan, J. R.; Monteiro, M. B.; dos Santos Filho, D.; Vichnewski, W.; Phytochemistry 1975, 15, 201.
- 5. Campos, A. M.; Silveira, E. R.; Braz-Filho, R.; Teixeira, T. C.; Phytochemistry 1994, 36, 403.
- 6. Arriaga, A. M.; Gomes, G. A.; Braz-Filho, R.; Fitoterapia 2000, 71, 211.
- 7. Spindola, H. M.; Carvalho, J. E.; Ruiz, A. L. T. G.; Rodrigues, R. A. F.; Denny, C.; Sousa, I. M. O.; Tamashiro, J. Y.; Foglio, M. A.; J. Braz. Chem. Soc. 2009, 20, 569.
- 8. Nunan, E. A.; Carvalho, M. G.; Piloveloso, D.; Braz. J. Med. Biol. Res.1982, 15, 450.
- 9. Carvalho, J. C. T.; Sertié, J. A. A.; Barbosa, M. V. J.; Patrício, K. C. M.; Caputo, L. R. G.; Sarti, S. J.; Ferreira, L. P.; Bastos, J. K.; J. Ethnopharmacol. 1999, 64, 127.
- 10. Silva, M. C. C.; Gayer, C. R. M.; Lopes, C. S.; Calixto, N. O.; Reis, P. A.; Passeas, C. P. B.; Paes, M. C.; Dalmau, S. R.; Sabino, K. C. C.; Todeschini, A. R.; Coelho, M. G. P.; J. Pharm. Pharmacol 2004, 55, 135.
- 11. Vieira, C. R.; Marques, M. F.; Soares, P. R.; Matuda, L.; De Oliveira, C. M. A. P.; Kato, L.; Da Silva, C. C.; Guillo, L. A.; Phytomedicine 2008, 15, 528.
- 12. Spindola, H. M.; Servat, L.; Denny, C.; Rodrigues, R. A. F.; Eberlin, M. N.; Cabral, E.; Sousa, I. M. O.; Carvalho, J. E.; Tamashiro, J. Y.; Foglio, M. A.; BMC Pharmacol. 2010, 10, 1.
- 13. Rosemberg, M.; Young, S. L.; Food Struct.1993, 12, 31.
- 14. Voipio, H. M.; Baneux, P.; Gomez de Segura, I. A.; Hau, J.; Wolfensohn, S.; Lab. Anim. 2008, 42, 1.
- 15. Hunskaar, S.; Hole, K.; Pain 1987, 30, 103.
- 16. Villeti, G.; Bergamarchi, M.; Bassani, F.; Bolzoni, P. T.; Maiorino, M.; Pietra, C.; Rondelli, I.; Chamiot-Clark, P.; Simonato, M.; Barbieri, M.; J. Pharmacol. Exp. Ther 2003, 306, 804.
- 17. Randall, R. O.; Selitto, J. J.; Arch. Int. Pharmacodyn 1957, 111, 409.
- 18. Collier, H. O.; Dinneen, L. C.; Johnson, C. A.; Schneider. C.; Br. J. Pharmacol. Chemother.1968, 32, 295.
- 19. Shannon, H. E.; Jones, C. K.; Li, D. L.; Peters, S. C.; Simmons, R. M. A.; Iyengar, S.; Pain 2001, 93, 221.
- 20. Santos, A. R. S.; Gadotti, V. M.; Oliveira, G. L.; Tibola, D.; Paszuck, A. P.; Neto, A.; Spindola, H. M.; Souza, M. M.; Rodrigues, A. L. S.; Calixto, J. B.; Neuropharmacology 2005, 48, 1021.
- 21. Santos, A. R. S.; Calixto, J. B.; Neurosci. Lett. 1997, 235, 73.
- 22. Cunha, M. T.; Verri Jr, W. A.; Silva, J. S.; Poole, S.; Cunha, F. Q.; Ferreira, S. H.; Proc. Natl. Acad. Sci. U. S. A. 2005, 102, 1755.
- 23. Winter, C. A.; Risley, E. A.; Nuss, G. W.; Proc. Soc. Exp. Biol. Med. 1962, 111, 544.
- 24. Milano, J.; Rossato, M. F.; Oliveira, S. M.; Drewes, C.; Machado, P.; Beck, P.; Zanatta, N.; Martins, M. A. P.; Mello, C. F.; Rubin, M. A.; Ferreira, J.; Bonacorso, H. G.; Life Sci. 2008, 83, 739.
- 25. Samad, T. A.; Moore, K. A.; Sapirstein, A.; Billet, S.; Allchorne, A.; Poole, S.; Bonventre, J. V.; Woolf, C. J.; Nature 2001, 410, 471.
- 26. Cunha, A. M.; Menon, S.; Menon, R.; Couto, A. G.; Burger, C.; Biavatti, M. W.; Phytomedicine 2010, 17, 37.
- 27. De Souza, K. C. B.; Bassani, V. L.; Schapoval, E. E. S.; Phytomedicine 2007, 14, 102.
- 28. Fernandes, L. P.; Turatti, I. C. C.; Lopes, N. P.; Ferreira, J. C.; Candido, R. C.; Oliveira, W. P.; Drying Technol. 2008, 26, 1534.
- 29. Rosemberg, M.; Kopelman, I. J.; Talmon, Y.; J. Agric. Food Chem. 1990, 38, 1288.
Publication Dates
-
Publication in this collection
03 Aug 2012 -
Date of issue
July 2012
History
-
Received
09 May 2011 -
Accepted
14 July 2012