Abstract
Successful Heck-Matsuda arylations of non-activated olefins applying microwave irradiation as an alternative heating mode are presented. Cleaner reactions were performed with arenediazonium tetrafluoroborates bearing electron-donating and electron-withdrawing groups in good to excellent yields with reaction times reduced from 3.5-18 h to 12-30 min.
microwave-assisted organic synthesis; Heck-Matsuda; non-activated olefins; arenediazonium salts; palladium
Introduction
The carbon-carbon bond forming process is one of the most important transformations in organic synthesis aimed at the construction of complex molecules. In this regard, the Heck reaction poses as a pivotal synthetic methodology widely used for the formation of carbon-carbon bonds starting from olefins and an electrophile.11 The Mizoroki-Heck Reaction; Oestreich, M., ed.; Wiley: Chichester, 2009.
2 Wu, X.-F.; Anbarasan, P.; Neumann, H.; Beller, M.; Angew. Chem., Int. Ed. 2010, 49, 9047.
3 McCartney, D.; Guiry, P. J.; Chem. Soc. Rev. 2011, 40, 5122.-44 Seechurn, C. C. C. J.; Kitching, M. O.; Colacot, T. J.; Snieckus, V.; Angew. Chem., Int. Ed. 2012, 51, 5062. An alternative of growing importance to the conventional Heck reaction was developed by Matsuda in 1977, using arenediazonium salts as arylating agents. The use of arenediazonium salts makes the arylation method significantly easier to carry out in laboratories, greener (phosphine-free), and faster. The higher reactivity of diazonium salts allows the efficient arylation of several olefin types under milder and simpler conditions. Notably, the arenediazonium tetrafluoroborates are crystalline and thermostable compounds, which can be easily prepared in several g scales from anilines, and stored for reasonably long periods.55 Taylor, J. G.; Moro, A. V.; Correia, C. R. D.; Eur. J. Org. Chem. 2011, 1403.
6 Felpin, F.-X.; Nassar-Hardy, L.; Le Callonnec, F.; Fouquet, E.; Tetrahedron 2011, 67, 2815.
7 Schwalm, C. S.; Correia, C. R. D.; Tetrahedron Lett. 2012, 53, 4836.
8 Schwalm, C. S.; de Castro, I. B. D.; Ferrari, J.; de Oliveira, F. L.; Aparício, R.; Correia, C. R. D.; Tetrahedron Lett. 2012, 53, 1660.
9 Correia, C. R. D.; Oliveira, C. C.; Salles Jr, A. G.; Santos, E. A. F.; Tetrahedron Lett. 2012, 53, 3325.
10 Roglans, A.; Pla-Quintana, A.; Moreno-Mañas, M.; Chem. Rev. 2006, 106, 4622.
11 Bonin, H.; Fouquet, E.; Felpin, F.-X.; Adv. Synth. Catal. 2011, 353, 3063.
12 Mo, F.; Dong, G.; Zhang, Y.; Wang, J.; Org. Biomol. Chem. 2013, 11, 1582.-1313 Biajoli, A. F. P.; Schwalm, C. S.; Limberger, J.; Claudino, T. S.; Monteiro, A. L.; J. Braz. Chem. Soc. 2014, 25, 2186.
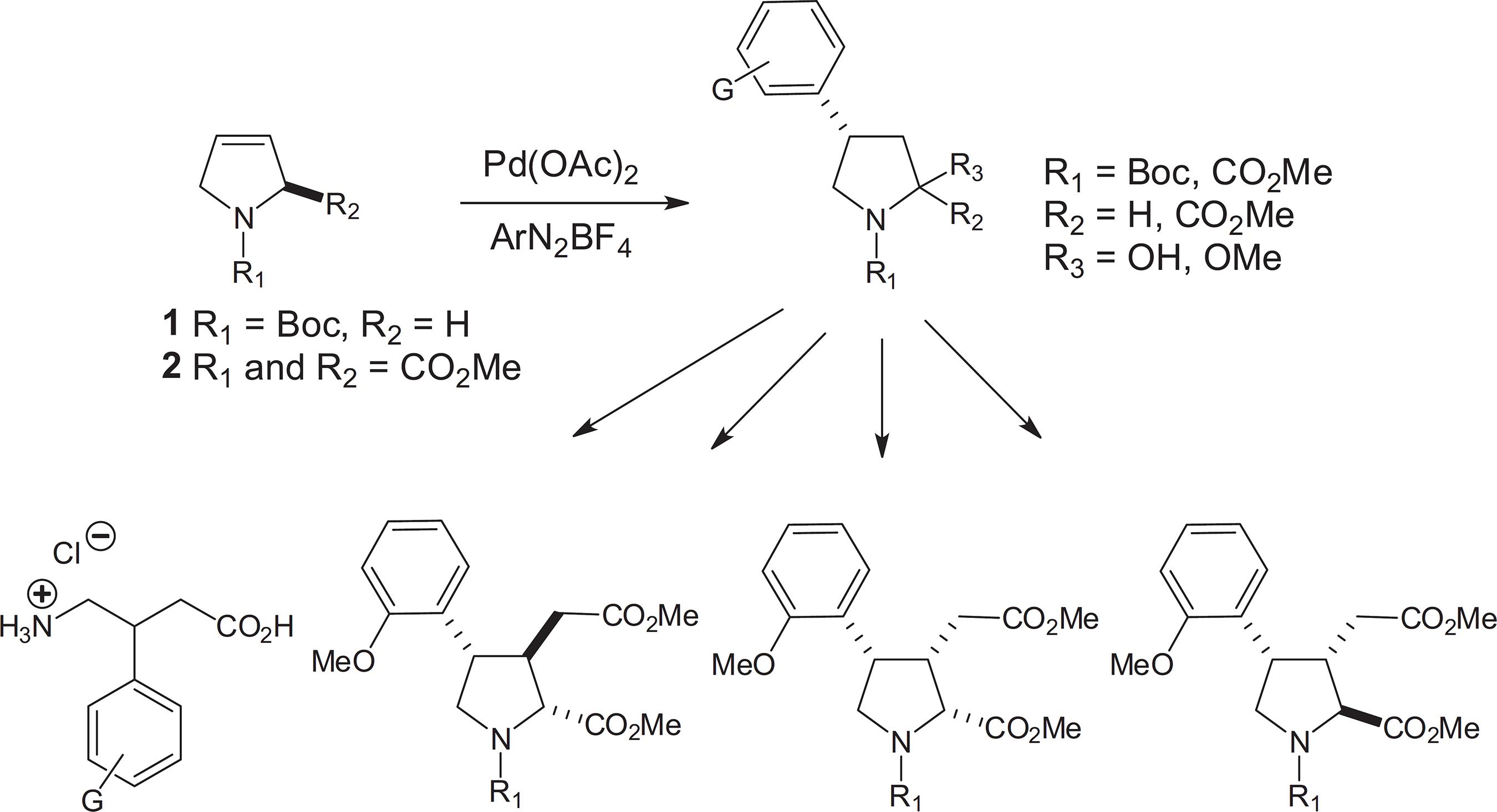
In the recent past, our group reported the use of Heck-Matsuda (HM) arylations of non-activated N-acyl-3-pyrrolines as a critical step in the synthesis of several pharmacological active compounds as a series of arylated γ-aminobutyric acid (GABA) derivatives, including baclofen, and some neuroexcitatory aryl kainoids (Scheme 1).1414 Carpes, M. J. S.; Correia, C. R. D.; Tetrahedron Lett. 2002, 43, 741.,1515 da Silva, K. P.; Godoi, M. N.; Correia, C. R. D.; Org. Lett. 2007, 9, 2815. Very often, these transition metal catalyzed coupling reactions are performed under conventional heating. However, this heating technique has in many cases some disadvantages, such as longer reaction times, resulting in side products, which reduce the reaction efficiency. In contrast, microwave-assisted organic synthesis (MAOS) has appeared as a viable heating alternative in the last decades, leading in several occasions to shorter reaction times and cleaner reactions. The value of this technique has been recognized both in the academic and industrial settings, and has been successfully applied to several organic transformations including transition metal catalyzed couplings.1616 Hayes, B. L.; Aldrichimica Acta 2004, 37, 66.
17 Kappe, C. O.; Angew. Chem., Int. Ed. 2004, 43, 6250.
18 Singh, B. K.; Kaval, N.; Tomar, S.; Van der Eycken, E.; Parmar, V. S.; Org. Process Res. Dev. 2008, 12, 468.
19 Moro, A. V.; Cardoso, F. S. P.; Correia, C. R. D.; Org. Lett. 2009, 11, 3642.
20 Mehta, V. P.; Van der Eycken, E. V.; Chem. Soc. Rev. 2011, 40, 4925.-2121 Sharma, A.; Appukkuttan, P.; Van der Eycken, E.; Chem. Commun. 2012, 48, 1623.
Based on the potential benefits of microwave irradiation as an alternative heating mode and on the synthetic advantages of Heck-Matsuda reaction, we present herein our results regarding the optimization of the Heck-Matsuda arylations of non-activated N-acyl-3-pirrolines 1 and 2 under conventional heating and microwave conditions employing several arenediazonium tetrafluoroborates.
Results and Discussion
Our studies started with the Heck-Matsuda reactions of olefin 1, prepared through the ring-closing metathesis of N-Boc-bisallyl amine in 93% yield.2222 Fu, G. C.; Grubbs, R. H.; J. Am. Chem. Soc. 1993, 115, 9856.,2323 Schumalz, H.-G.; Angew. Chem., Int. Ed. 1995, 34, 1833. Previous studies for this key step were performed employing 2-4 mol% of Pd(OAc)2, in the biphasic system CCl4:H2O or in CH3CN:H2O, at room temperature, for 2-4 h. After oxidation, the desired lactams were obtained in 70-90% yields.1414 Carpes, M. J. S.; Correia, C. R. D.; Tetrahedron Lett. 2002, 43, 741.These Heck arylations were re-investigated employing 1.5 eq of olefin 1, 0.5-4 mol% of Pd(OAc)2 in CH3CN:H2O (1:1), with 1 eq of the arenediazonium tetrafluoroborates, under microwave irradiation, followed by oxidation with pyridinium chlorochromate (PCC) of the crude lactamols (Table 1).
The best results were obtained when the microwave-assisted Heck-Matsuda reactions were performed at 60 °C (100 W power) for 15 min (Table 1, entries 1-5). In all cases, the reaction times were measured after complete consumption of the arenediazonium tetrafluoroborates. It is worth mentioning that under conventional heating (60 °C), we observed a significant decrease in yields for those reactions carried out in CH3CN:H2O (for example: a maximum of 11% yield for compound 5 30 min after complete consumption of the arenediazonium tetrafluoroborate). This reaction was also tested under a lower catalyst loading (1 to 0.5 mol%), but lactam 5 was isolated in lower yields (33-18% yield; Table 1, entries 6-8). All reactions were carried out in a 0.5 to 1 mmol scale in closed vessels. It should be emphasized that, in spite of the pressure increase due to nitrogen release during the Heck arylations, we did not register any incidents using the appropriate pressure-resistant vials (all other safety precautions were in place during these experiments: they were carried out in a well-ventilated hood under the protection of a safety shield).
It is also important to highlight that the order of addition of the reagents is critical for the success of these arylation reactions. The palladium catalyst is first added to the olefin in aqueous acetonitrile, resulting in a dark black reaction mixture, followed by the addition of the arenediazonium tetrafluoroborate.
In parallel to these studies, attempts were made to optimize the Heck-Matsuda reactions of the chiral, non-activated, olefin 2. This olefin was prepared in multigram scale from the commercially available L-4-hydroxyproline (55% overall yield over 4 steps).2424 Lin, C.-C.; Weitz-Schmidt, G.; Wong, C.-H.; J. Am. Chem. Soc. 1996, 118, 6826. In our previous studies with this olefin, the arylation reactions were carried out employing 10 mol% of Pd(OAc)2 in MeOH at 60 °C under conventional heating (oil bath), for 3.5-18 hours. These reactions provided the corresponding 4-aryl-prolines in moderate to good yield in high stereo- and regioselectivities.1515 da Silva, K. P.; Godoi, M. N.; Correia, C. R. D.; Org. Lett. 2007, 9, 2815. However, only moderate yields were obtained with arenediazonium salts bearing strong electron-withdrawing groups, thus limiting the scope of the method. Therefore, we considered that this Heck arylation could be considerably improved, mainly regarding the exceptional long reaction times required for the Heck arylation under conventional heating.
Arylations with arenediazonium salts bearing electron-donating substituents, in MeOH under conventional heating, provided the desired Heck products in good to excellent yields, but only after long periods of heating (Table 2, entries 1, 2 and 11). Aiming at optimizing these reactions, we decided to evaluate the use of co-solvents and additives (Table 2). The 2-OMe and 4-OMe arenediazonium tetrafluoroborates were chosen as arylating agents for these experiments because of their higher thermal stabilities.
Experiments using dimethyl sulfoxide (DMSO) as co-solvent in MeOH in ratios varying from 1:1 to 1:9 provide total consumption of the starting olefin and considerable reductions in the reaction times (Table 2, entries 3-7). Unfortunately, the yields were only moderate in these cases (ca. 50% yield) with formation of many by-products, which complicated the purification of Heck products. At higher temperatures, a significant fraction of olefin 2 was recovered and the product 4-aryl proline 8 was obtained in only 38% yield (Table 2, entry 6), probably due to decomposition of the diazonium salt at that temperature.
Attempts to use less nucleophilic solvents such as acetonitrile or mixtures of acetonitrile, benzonitrile and methanol were also made, but these reactions only provided low yields of the Heck products (Table 2, entries 8 and 9) together with side products arising probably from the decomposition of the diazonium salts and the in situ aromatization of olefin 2 (formation of pyrrole derivatives).
Aiming at a better performance of the Heck arylation, reaction of olefin 2was also evaluated in water in the presence of sodium dodecyl sulfate to promote olefin solubilization. However, lactamol 10 was isolated in 43% yield without total consumption of the starting olefin (Table 2, entry 10). Overall, the use of co-solvents reduced the reaction time, but they also decreased the efficiency of arylation reaction.
In view of the successful Heck-Matsuda arylation of olefin 1 under microwave irradiation, we decided to evaluate its application in the Heck arylations of the non-activated dehydroproline 2. It was then submitted to microwave-assisted Heck arylation with different arenediazonium tetrafluoroborates in MeOH as solvent, in the presence of Pd(OAc)2 as catalyst, and the results obtained are shown in Table 3.
To our satisfaction, all reactions were completed in 12-30 min with yields varying from good to excellent (62 to 83%, with the exception of entry 3) when compared with the reactions carried out under conventional heating. In all cases the reaction times were determined after complete consumption of the olefin by thin-layer chromatography (TLC) analysis. Similar to what was done with olefin 1, reactions were carried out in closed vessels and occurred without any incident.
In all cases examined, arylations of dihydroproline 2 with arenediazonium salts under microwave irradiation were cleaner than those carried out under conventional heating, making purification of these products straightforward. Moreover, arenediazonium tetrafluoroborates bearing electron-withdrawing groups (EWG) or electron-donating groups (EDG) provided the Heck products in comparable yields. All reactions proceeded in a highly regioselective manner to furnish products 8, 9, 11-16 as a mixture of diastereomeric compounds.
The proposed catalytic cycle of the Heck-Matsuda arylation of the non-activated olefins is shown in Scheme 2. The proposed catalytic cycle involves a highly reactive cationic palladium species, operating under a polar mechanism.2525 Sabino, A. A.; Machado, A. H. L.; Correia, C. R. D.; Eberlin, M. N.; Angew. Chem., Int. Ed. 2004, 43, 2514; Sabino, A. A.; Machado, A. H. L.; Correia, C. R. D.; Eberlin, M. N.; Angew. Chem., Int. Ed. 2004, 43, 4389. The Heck-Matsuda products obtained in this study are derived from solvolysis of the corresponding endocyclic enecarbamates, which are the primary products of these reactions. As all the reactions were carried out without a base, the slightly acidic medium promotes this side reaction efficiently. The solvolysis step has the advantage of avoiding a second Heck-Matsuda arylation of the rather reactive enecarbamate, thus preventing the formation of secondary and undesired products.
Conclusions
In summary, the microwave-assisted Heck-Matsuda arylation of the non-activated olefins N-acyl pyrrolidine 1 and dehydroproline 2with arenediazonium salts bearing EWG or EDG can be performed efficiently and cleanly, in good to excellent yields and in very short reaction times. Despite the fact that the Heck-Matsuda reactions release one eq of nitrogen, they were carried out under pressure in appropriate vessels without any incident. These results show the broad scope and applicability of the Heck-Matsuda reactions, making them good alternatives to the conventional Heck arylations in organic synthesis.
Experimental
Typical experimental procedure for the microwave-assisted Heck-Matsuda arylation of olefin 1
To a solution of 1.5 eq of olefin 1 (114.22 mg, 0.675 mmol) in CH3CN:H2O (1:1) it was added Pd(OAc)2 (2 mol%). After 5 min, 1 eq of arenediazonium salt was added to the reaction mixture and submitted to microwave (MW) irradiation at 60 °C and power of 100 W for 15 min. Upon completion, the mixture was cooled at r.t., diluted with AcOEt, and washed with saturated NaHCO3 and NaCl solutions. The organic layers were separated, combined, and dried over anhydrous Na2SO4. After filtration, the solvent was evaporated in vacuo. To a solution of the crude material in CH2Cl2 previously dried was added 1.2 eq of pyridinium chlorochromate (PCC) (174.60 mg, 0.810 mmol). After 4 h under stirring at r.t. the crude material was filtered through a plug of silica gel and washed with AcOEt. This material was evaporated and purified by flash chromatography on silica gel (AcOEt:Hex, 2:3).
Typical experimental procedure for the microwave-assisted Heck-Matsuda arylation of olefin 2
To a solution of 1.0 eq of olefin 2 (50.0 mg, 0.27 mmol) in CH3OH (1.0 mL) it was added Pd(OAc)2 (10 mol%) followed by the addition of 2 eq of the corresponding arenediazonium salt. The reaction mixture under stirring was submitted to MW irradiation at 100 °C and power of 100 W for 12-30 min. Upon completion, the reaction mixture was cooled at r.t., diluted with EtOAc, and washed with saturated NaHCO3 and NaCl solutions. The organic layers were separated, combined, and dried over anhydrous Na2SO4. After filtration, the solvent was evaporated in vacuo and the crude material purified by flash chromatography on silica gel.
-
Supplementary InformationSupplementary information (experimental procedures and spectral data) is available free of charge at http://jbcs.sbq.org.br as PDF file.
-
FAPESP has sponsored the publication of this article.
Acknowledgments
The authors thank São Paulo Research Foundation (FAPESP research grant 2011/2383-6, and postdoctoral fellowships to F. G. F. and M. N. G.), CNPq and CAPES for financial support and fellowships.
References
-
1The Mizoroki-Heck Reaction; Oestreich, M., ed.; Wiley: Chichester, 2009.
-
2Wu, X.-F.; Anbarasan, P.; Neumann, H.; Beller, M.; Angew. Chem., Int. Ed. 2010, 49, 9047.
-
3McCartney, D.; Guiry, P. J.; Chem. Soc. Rev. 2011, 40, 5122.
-
4Seechurn, C. C. C. J.; Kitching, M. O.; Colacot, T. J.; Snieckus, V.; Angew. Chem., Int. Ed. 2012, 51, 5062.
-
5Taylor, J. G.; Moro, A. V.; Correia, C. R. D.; Eur. J. Org. Chem. 2011, 1403.
-
6Felpin, F.-X.; Nassar-Hardy, L.; Le Callonnec, F.; Fouquet, E.; Tetrahedron 2011, 67, 2815.
-
7Schwalm, C. S.; Correia, C. R. D.; Tetrahedron Lett. 2012, 53, 4836.
-
8Schwalm, C. S.; de Castro, I. B. D.; Ferrari, J.; de Oliveira, F. L.; Aparício, R.; Correia, C. R. D.; Tetrahedron Lett. 2012, 53, 1660.
-
9Correia, C. R. D.; Oliveira, C. C.; Salles Jr, A. G.; Santos, E. A. F.; Tetrahedron Lett. 2012, 53, 3325.
-
10Roglans, A.; Pla-Quintana, A.; Moreno-Mañas, M.; Chem. Rev. 2006, 106, 4622.
-
11Bonin, H.; Fouquet, E.; Felpin, F.-X.; Adv. Synth. Catal. 2011, 353, 3063.
-
12Mo, F.; Dong, G.; Zhang, Y.; Wang, J.; Org. Biomol. Chem. 2013, 11, 1582.
-
13Biajoli, A. F. P.; Schwalm, C. S.; Limberger, J.; Claudino, T. S.; Monteiro, A. L.; J. Braz. Chem. Soc. 2014, 25, 2186.
-
14Carpes, M. J. S.; Correia, C. R. D.; Tetrahedron Lett. 2002, 43, 741.
-
15da Silva, K. P.; Godoi, M. N.; Correia, C. R. D.; Org. Lett. 2007, 9, 2815.
-
16Hayes, B. L.; Aldrichimica Acta 2004, 37, 66.
-
17Kappe, C. O.; Angew. Chem., Int. Ed. 2004, 43, 6250.
-
18Singh, B. K.; Kaval, N.; Tomar, S.; Van der Eycken, E.; Parmar, V. S.; Org. Process Res. Dev. 2008, 12, 468.
-
19Moro, A. V.; Cardoso, F. S. P.; Correia, C. R. D.; Org. Lett. 2009, 11, 3642.
-
20Mehta, V. P.; Van der Eycken, E. V.; Chem. Soc. Rev. 2011, 40, 4925.
-
21Sharma, A.; Appukkuttan, P.; Van der Eycken, E.; Chem. Commun 2012, 48, 1623.
-
22Fu, G. C.; Grubbs, R. H.; J. Am. Chem. Soc 1993, 115, 9856.
-
23Schumalz, H.-G.; Angew. Chem., Int. Ed 1995, 34, 1833.
-
24Lin, C.-C.; Weitz-Schmidt, G.; Wong, C.-H.; J. Am. Chem. Soc. 1996, 118, 6826.
-
25Sabino, A. A.; Machado, A. H. L.; Correia, C. R. D.; Eberlin, M. N.; Angew. Chem., Int. Ed. 2004, 43, 2514; Sabino, A. A.; Machado, A. H. L.; Correia, C. R. D.; Eberlin, M. N.; Angew. Chem., Int. Ed. 2004, 43, 4389.
Publication Dates
-
Publication in this collection
May 2015
History
-
Received
20 Nov 2014 -
Accepted
03 Mar 2015