ABSTRACT:
Canine brucellosisis an infectious disease caused by bacteria of the genus Brucella, with world wide distribution and zoonotic impact, and in humans and animals is a neglected disease. In the present study, the sero prevalence of B. canis and B. abortus were determined in a probabilistic sample of housed dogs from the Atlantic Rainforest area of the state of Paraíba, Brazil, and the factors associated with sero positivity. A total of 386 dogs over three months of age were used. For the search for anti-B.canis antibodies the agar gel immune diffusion test (IDGA) was used as a screening and IDGA+2ME as confirmatory test, and to search for anti-B. abortus antibodies the Rose Bengal test (RBT) test was used. Apparent and real prevalences were calculated, and robust Poisson regression was used to identify factors associated with prevalence. The real prevalence fB. Canis was 12.6% and of B. abortus was 22.8%. The factors associated with sero positivity for B. canis were age greater than 10 years (prevalence ratio; PR = 6.38; P = 0.024) and dogs reared in they ard (PR = 5.20; P = 0.035) and for B. abortus was no treplacement of water of animals everyday (PR = 1.48; P = 0.033). It can be concluded that the prevalence of B. canis and B. Abortus in the region is high, which warns to the adopting of control and prevention measures, as well as greater care in the management of animals, especially for elderly dogs.
Key words:
Brucella canis; Brucella abortus; public health; epidemiology; dogs
RESUMO:
A brucelose canina é uma doença infecciosa causada por bactérias do gênero Brucella, com distribuição mundial e de caráter zoonótico, e em humanos e animais é uma doença negligenciada. No presente estudo foram determinados as soroprevalências de B. canis e B. abortusem uma amostra probabilística de cães domiciliados da área urbana de oito municípios localizados na região da Mata Atlântica do Estado da Paraíba, Brasil, e os fatores associados com a soropositividade. Foram utilizados 386 cães com mais de três meses de idade. Para a pesquisa de anticorpos anti-B. canis foi utilizado o teste de imunodifusão em gel de ágar (IDGA) como triagem e IDGA+2ME como confirmatório, e para a pesquisa de anticorpos anti-B. abortusfoi utilizado o teste do antígeno acidificado tamponado (AAT). Foram calculadas as prevalências aparente e real, e para a identificação de fatores associados com a prevalência foi empregada regressão robusta de Poisson. A prevalência real de B. canis foi de 12,6% e de B. abortusfoi 22,8%. Os fatores associados com a soropositividade para B. canis foram idade maior que 10 anos (razão de prevalência; RP = 6,38; P = 0,024) e cães criados presos no quintal (RP = 5,20; P = 0,035) e para B. abortus foi não trocar a água dos animais todos os dias (RP = 1,48; P = 0,033). Conclui-se que a prevalência de B. canis e B. abortusem cães da região é alta, o que alerta para a necessidade de adoção de medidas de controle e prevenção, bem como são sugeridos maiores cuidados no manejo dos animais, sobretudo cães idosos.
Palavras-chave:
Brucellacanis; Brucella abortus; saúde pública; epidemiologia; cães
INTRODUCTION:
Canine brucellosis is an infectious zoonotic disease prevalent worldwide and is caused by bacteria of the genus Brucella. These bacteria affect mainly the reproductive tract of animals, causing infertility, abortion, placenta retention, weak offspring, and stillbirths in females, while causing orchitis and epididymitis in males (MÉLO et al., 2013MÉLO, S. K. M. et al. Brucelose canina: revisão de literatura. Ciência Veterinária nos Trópicos, v.16, n.1/2/3, p.7-17, 2013. Available from: <Available from: http://www.rcvt.org.br/volume16/rcvt_16_pg7.pdf >. Accessed: Jun. 06, 2020.
http://www.rcvt.org.br/volume16/rcvt_16_...
; RODRIGUES et al., 2017RODRIGUES, R. T. G. A. et al. Canine brucellosis: a pratical review for the clinical veterinarian of small animals. Revista Brasileira de Higiene e Sanidade Animal, v.11, n.2, p.216-232, 2017. Available from: <Available from: https://www.researchgate.net/publication/318871002_Canine_brucellosis_a_pratical_review_for_the_clinical_veterinarian_of_small_animals >. Accessed: Jun. 06, 2020. doi: 10.5935/1981-2965.20170023.
https://www.researchgate.net/publication...
). Its transmission in dogs occurs by direct contact with vaginal fluids or infected semen (RODRIGUES et al., 2017), while in humans, the great majority of cases occur by consumption of contaminated dairy products or exposure to fluids and animal tissues of infected animals (ARENAS-GAMBOA et al., 2016ARENAS-GAMBOA, A. M. et al.Human brucellosis and adverse pregnancy outcomes.Current Tropical Medicine Reports, v.3, n.4, p.164-72, 2016. Available from: <Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5720386/ >. Accessed: Mar. 17, 2020. doi: 10.1007/s40475-016-0092-0. 2016.
https://www.ncbi.nlm.nih.gov/pmc/article...
).
In terms of public health, the close relationship between man and dog may lead to brucellosis transmission (DENTINGER et al., 2015DENTINGER, C. M. et al. Human Brucella canis infection and subsequent laboratory exposures associated with a puppy, New York City, 2012. Zoonoses and Public Health, v.62, n.5, p.407-14, 2015. Available from: <Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4639931/ >. Accessed: Dec. 12, 2019. doi: 10.1111/zph.12163.
https://www.ncbi.nlm.nih.gov/pmc/article...
). In man, the infection is usually neglected and underdiagnosed because of its diverse and unspecific symptoms, which make its diagnosis difficult (ROUSHAN et al., 2016ROUSHAN, M. R. H. et al. Different clinical presentations of Brucellosis. Jundishapur Journal of Microbiology, v.9, n.4, article e33765, 2016. Available from: <Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4897599/ >. Accessed: Jan. 13, 2020. doi: 10.5812/jjm.33765.
https://www.ncbi.nlm.nih.gov/pmc/article...
).
In Brazil, the values of seroprevalence for B. canis in dogs are widely disparate depending on the locality, varying from 2 to 28% (RODRIGUES et al., 2017RODRIGUES, R. T. G. A. et al. Canine brucellosis: a pratical review for the clinical veterinarian of small animals. Revista Brasileira de Higiene e Sanidade Animal, v.11, n.2, p.216-232, 2017. Available from: <Available from: https://www.researchgate.net/publication/318871002_Canine_brucellosis_a_pratical_review_for_the_clinical_veterinarian_of_small_animals >. Accessed: Jun. 06, 2020. doi: 10.5935/1981-2965.20170023.
https://www.researchgate.net/publication...
). Conversely, the serological surveys for B. abortus are rare. In the state of Paraíba, a few studies investigating the prevalence of B. canis in dogs have been conducted (ALVES et al., 2003ALVES, C. J., et al. Aspectos epidemiológicos de Brucella canis em Patos, Paraíba, Brasil. Ciência Animal, v.13, n.1, p.45-49, 2003. Available from: <Available from: http://www.uece.br/cienciaanimal/dmdocuments/Comunicacao1.2003.1.pdf >. Accessed: Jun. 07, 2020.
http://www.uece.br/cienciaanimal/dmdocum...
; VASCONCELOS et al., 2008VASCONCELOS, R. T. J. et al. Soroprevalência e fatores de risco associados à infecção por Brucella canis em cães da cidade de Campina Grande, estado da Paraíba. Revista Brasileira de Saúde e Produção Animal, v.9. n.3, p.436-42, 2008. Available from: <Available from: http://revistas.ufba.br/index.php/rbspa/article/view/1003/664 >. Accessed: Jun. 07, 2020.
http://revistas.ufba.br/index.php/rbspa/...
; FERNANDES et al., 2011FERNANDES, A. R. F. et al. Brucella canisinfection in dogs attended in veterinary clinics from patos, Paraíba state, Brazil. Brazilian Journal of Microbiology, v.42, n.4, p.1405-8, 2011. Available from: <Available from: http://www.scielo.br/scielo.php?script=sci_arttext&pid=S1517-83822011000400023&lng=en&nrm=iso&tlng=en >. Accessed: Nov. 11, 2019. doi: 10.1590/S1517-83822011000400023.
http://www.scielo.br/scielo.php?script=s...
), but only one study was conducted on B. abortus (AZEVEDO et al., 2003AZEVEDO, S. S. et al. Ocorrência de anticorpos contra Brucellaabortus em cães errantes da cidade de Patos, estado da Paraíba, Brasil. Arquivos do Instituto Biológico, v.70, n.4, p.499-500, 2003. Available from: <Available from: http://www.biologico.sp.gov.br/uploads/docs/arq/V70_4/azevedo.pdf >. Accessed: Jun. 07, 2020.
http://www.biologico.sp.gov.br/uploads/d...
). In addition, there are no studies that show the occurrence of canine brucellosis in the Brejo Paraibano microregion, a place which has characteristics that may influence the occurrence of infection such as the presence of fragments of the Atlantic Forest, high rainfall, and a wide variety of fauna that may serve as natural reservoirs (ZHELUDKOV & TSIRELSON, 2010ZHELUDKOV, M. M.; TSIRELSON, L. E. Reservoirs of Brucella infection in nature. Biology Bulletin, v.37, n.7, p.709-15, 2010. Available from: <Available from: https://link.springer.com/article/10.1134/S106235901007006X >. Accessed: Jan. 22, 2020. doi: 10.1134/S106235901007006X
https://link.springer.com/article/10.113...
). Thus, this research aimed to determine the seroprevalence of B. canis and B. abortus in the canine population of the Brejo Paraibano microregion and identify factors associated with seropositivity.
MATERIALS AND METHODS:
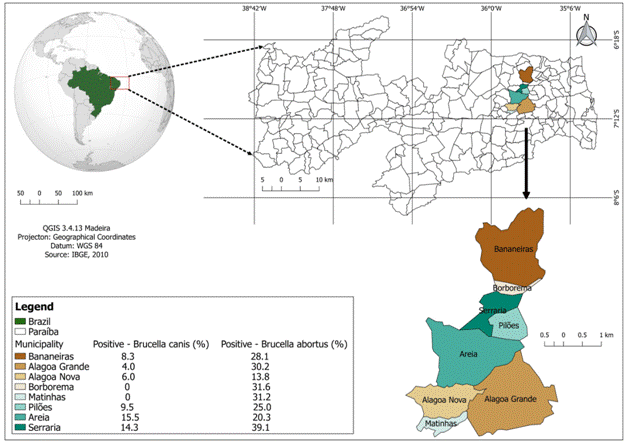
This study was conducted on dogs in the urban area of the Brejo Paraibano microregion, State of Paraíba, Brazilian Northeast. The region has a territorial area of 840.452 km2, a population of 116,488 inhabitants, and is composed of eight municipalities: Alagoa Grande, Alagoa Nova, Areia, Bananeiras, Borborema, Matinhas, Pilões and Serraria.
To define the minimum number of animals to be sampled, a formula was used for simple random samples (THRUSFIELD, 2007THRUSFIELD, M. Veterinary epidemiology. Oxford: Blackwell Science, 2007. 610p.) which considered the following parameters: an expected prevalence of 50% (maximization of sample), a confidence level of 95% (z = 1.96), and a sample error of 5%, resulting in a minimum sample size of 384 dogs. However, 386 dogs more than three months old, of both sexes, and varied breeds were used. The minimum number of dogs to be sampled in each municipality of the microregion was defined proportionally according to the number of inhabitants (Table 1).
The field work research conducted by a trained team by visiting the homes of the residents of the microregion. The dog owners were made aware of the study’s objectives and, after agreeing to participate, signed informed consent forms. Blood collection was conducted between May to October 2017. The owners were made to answer a 35-item questionnaire in order to obtain data for analysis of factors associated with seroprevalence. Variables considered on the questionnaire were related to the dogs’ information, owner, residence, and characteristics of animal management.
Research of anti-B.canis antibodies was conducted using the agar gel immunodiffusion (AGID) technique through kits produced by the Institute of Technology of Paraná (TECPAR). The technique was conducted according to the manufacturer’s recommendations using lipopolysaccharides and proteins antigens of Brucella ovis, sample Reo 198. To confirm, the same technique was applied in serum treated by 2-mercaptoetanol (AGID+2-ME) according to AZEVEDO et al. (2004AZEVEDO, S. S. et al. Comparação de três testes sorológicos aplicados ao diagnóstico da infecção de caninos por Brucella canis. Brazilian Journal of Veterinary Research and Animal Science, v.41, n.2, p.106-112, 2004. Available from: <Available from: http://www.scielo.br/scielo.php?script=sci_arttext&pid=S1413-95962004000200005 >. Accessed: Mar. 10, 2020. doi: 10.1590/S1413-95962004000200005.
http://www.scielo.br/scielo.php?script=s...
). For the anti-B.abortus sp. antibodies research, the Buffered Acidified Antigen (BAA) technique was performed through the use of kits produced by TECPAR. The antigen consisted of an inactivated suspension of B. abortus sample 1119-3, stained by Rose Bengal, and diluted 8.0% in buffer solution pH 3.65 ± 0.05. Due to the mixture’s pH, some unspecific agglutinins are inhibited, thus allowing an increase in test specificity.
Apparent and real prevalences were calculated according to NOORDHUIZEN et al. (1997NOORDHUIZEN, J. P. T. M. et al. Application of quantitative methods in Veterinary Epidemiology. Wageningen: Wageningen Press, 1997. 445p.). Apparent prevalence was obtained by dividing the number of seroreactive animals by the total number of animals, while real prevalence was estimated by adjusting the apparent prevalence considering sensitivity (52.94%) and specificity (98.7%) of the diagnosis using the B. canis protocol (KEID et al., 2009KEID, L. B. et al. Comparison of agar gel immunodiffusion test, rapid slide agglutination test, microbiological culture and PCR for the diagnosis of canine brucellosis. Research in Veterinary Science, v.86, n.1, p.22-26, 2009.Available from: <Available from: https://www.sciencedirect.com/science/article/abs/pii/S0034528808001161?via%3Dihub >. Accessed: Nov. 13, 2019. doi: 10.1016/j.rvsc.2008.05.012.
https://www.sciencedirect.com/science/ar...
), as well as the diagnosis sensitivity (100%) and specificity (96.29%) for infection by B. abortus (TALUKDER et al., 2011TALUKDER, B. C. et al. Comparative evaluation of commercial serodiagnostic tests for the seroprevalence study of brucellosis in stray dogs in Bangladesh. Bangladesh Journal of Veterinary Medicine, v.9, n.1, p.79-83, 2011. Available from: <Available from: https://www.banglajol.info/index.php/BJVM/article/view/11217 >. Accessed: Feb. 16, 2020. doi: 10.3329/bjvm.v9i1.11217.
https://www.banglajol.info/index.php/BJV...
) according to this formula:
Where,
RP = Real prevalence
AP = Apparent prevalence
ESP = Specificity
SEN = Sensitivity
To identify factors associated with seropositivity, a univariable analysis was initially conducted in which each independent variable was submitted to an association analysis with a dependent variable (seropositivity in the serological tests). Variables with P≤0.2 in the chi-square test were selected for multivariable analysis using robust Poisson regression. The collinearity between independent variables was verified by a correlation analysis. For the variables with strong collinearity (correlation coefficient > 0.9), one of two variables was excluded in the multiple agreement analysis with biological plausibility. To assess the adjustment of the regression model, the Pearson chi-square test was used. The level of significance adopted in the multiple analysis was 5%, and the analyses were conducted in R (R CORE TEAM, 2019R CORE TEAM (2019). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. Available from: <Available from: https://www.R-project.org/ >. Accessed: Jan. 13, 2020.
https://www.R-project.org/...
) using the “Epi” (CARSTENSEN et al., 2019CARSTENSEN, B. et al. Epi: A Package for Statistical Analysis in Epidemiology. R package version 2.40, 2019. Available from: <Available from: https://CRAN.R-project.org/package=Epi >. Accessed: Dec. 10, 2019.
https://CRAN.R-project.org/package=Epi...
), “Sandwich” (BERGER et al., 2017BERGER, S. et al. “Various versatile variances: An Object-Oriented Implementation of Clustered Covariances in R”. 2017. Technical Report 2017-12, Working Papers in Economics and Statistics, Research Platform Empirical and Experimental Economics, Universität Innsbruck. Available from: <Available from: http://EconPapers.RePEc.org/RePEc:inn:wpaper:2017-12 >. Accessed: Dec. 10, 2019.
http://EconPapers.RePEc.org/RePEc:inn:wp...
), and “lmtest” packages (ZEILEIS &HOTHORN, 2002ZEILEIS, A.; HOTHORN, T. Diagnostic checking in regression relationships. R News 2(3), 7-10, 2002. Available from: <https://CRAN.R-project.org/doc/Rnews/>. Accessed: Jan. 20, 2020.
https://CRAN.R-project.org/doc/Rnews/...
).
RESULTS AND DISCUSSION:
Of the 386 examined samples for B. canis, 99 were reactive in AGID, of which 30 were further positive in AGID+ME. This confirmed an apparent prevalence of 7.8% and real prevalence of 12.6%. Of the 366 examined samples for B. abortus, 94 were reactive in BAA, with anapparent prevalence of 25.7% and a real prevalence of 22.8%. Depending on locality, the prevalence of B. canis varied from 0 to 15.5%, while it varied from 13.8-39.1% in B. abortus (Table 1 and Figure 1).
Geographic representation of the municipalities of the BrejoParaibanomicroregion, Brazil that had their dogs sampled between May and October 2017 for determination of the respective frequencies of seropositive dogs for Brucella canisandBrucella abortus.
The high real prevalence of B. canisand B. abortus in dogs of the Brejo Paraibano microregion is alarming due to the health risks posed to the public (DENTINGER et al., 2015DENTINGER, C. M. et al. Human Brucella canis infection and subsequent laboratory exposures associated with a puppy, New York City, 2012. Zoonoses and Public Health, v.62, n.5, p.407-14, 2015. Available from: <Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4639931/ >. Accessed: Dec. 12, 2019. doi: 10.1111/zph.12163.
https://www.ncbi.nlm.nih.gov/pmc/article...
). All of the cities in the microregion had seropositive dogs for B. abortus, showing that the agent is disseminated all over the region. These data should encourage adoption of control measures for preventing occurrence of the disease in dogs, slowing its propagation, and preventing it from being transmitted to human beings and other animal species. It should be highlighted that the characteristics of the studied region, including its high humidity and warm temperature for most of the year, may have facilitated the maintenance of bacteria in the environment.
In this study, BAA was used as an option to identify seroreactive dogs for B. abortus due to the technique’s high sensitivity and specificity (TALUKDER et al., 2011TALUKDER, B. C. et al. Comparative evaluation of commercial serodiagnostic tests for the seroprevalence study of brucellosis in stray dogs in Bangladesh. Bangladesh Journal of Veterinary Medicine, v.9, n.1, p.79-83, 2011. Available from: <Available from: https://www.banglajol.info/index.php/BJVM/article/view/11217 >. Accessed: Feb. 16, 2020. doi: 10.3329/bjvm.v9i1.11217.
https://www.banglajol.info/index.php/BJV...
). In addition, in bovines and small ruminants, BAA showed better performance than the tube agglutination test (CHACHRA et al., 2009CHACHRA, D. et al. Comparative efficacy of Rose Bengal plate test, standard tube agglutination test and Dot ELISA in immunological detection of antibodies to Brucella abortus in sera.Journal of Bacteriology Research, v.1, n.3, p.30-33, 2009. Available from: <Available from: https://www.researchgate.net/publication/228767138_Comparative_efficacy_of_Rose_Bengal_plate_test_standard_tube_agglutination_test_and_Dot_ELISA_in_immunological_detection_of_antibodies_to_Brucella_abortus_in >. Accessed: Feb. 12, 2020.
https://www.researchgate.net/publication...
; SADHU et al., 2015SADHU, D. B. et al. Seroprevalence and comparison of different serological tests for brucellosis detection in small ruminants. Veterinary World, v.8, n.5, p.561-566, 2015. Available from: <Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4774713/ >. Accessed: Jan. 17, 2020. doi: 10.14202/vetworld.2015.561-566.
https://www.ncbi.nlm.nih.gov/pmc/article...
). Moreover, due to its excellent sensitivity and specificity in detecting IgM, IgG, and IgA of infected patients with brucellosis, it has good applicability in humans (DÍAZ et al., 2011DÍAZ, R. et al. The Rose Bengal Test in human brucellosis: A neglected Test for the Diagnosis of a Neglected Disease. Plos Neglected Tropical Diseases, v.5, n.4, e950, 2011. Available from: <Available from: https://journals.plos.org/plosntds/article?id=10.1371/journal.pntd.0000950 >. Accessed: Nov 11, 2019.doi: 10.1371/journal.pntd.0000950.
https://journals.plos.org/plosntds/artic...
).
It is possible that the high prevalence of B. abortus observed in dogs of the Brejo Paraibano microregionis also related to the presence of infected bovines in the region. In an official epidemiological study for bovine brucellosis in the state of Paraíba (CLEMENTINO et al., 2016CLEMENTINO, I. J. et al. Epidemiological situation of bovine brucellosis in the state of Paraiba, Brazil. Semina: Ciências Agrárias, v.37, n.5, p.3403-3412, 2016. Available from: <Available from: http://www.uel.br/revistas/uel/index.php/semagrarias/article/view/27230 >. Accessed: Dec. 10, 2019. doi: 10.5433/1679-0359.2016v37n5Supl2p3403.
http://www.uel.br/revistas/uel/index.php...
), it was shown that the mesoregion of Agreste and Mata Paraibana, which included the study area, had the greatest prevalence of seroreactive bovines for B. abortus. Hence, taking into consideration the mode of transmission of the disease, it is possible that dogs in the region may have comein contact with fetal or placenta remains of infected bovines, or may have consumed milk products or raw meat of bovines infected by the pathogen.
Note that the region has several Atlantic Forest fragments near urban areas and is composed of great fauna and flora biodiversity, increasing the chances of domestic dogs having contact with wild animals that could act as a bacterial reservoir. In a study conducted by AZEVEDO et al. (2010AZEVEDO, S. S. et al. Anticorpos antiBrucellaabortus, antiBrucella canis e antiLeptospira spp. em raposas (Pseudalopexvetulus) do semiárido paraibano, Nordeste do Brasil.Ciência Rural, v.40, n.1, p.1-3, 2010. Available from: <Available from: http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0103-84782010000100031&lng=en&nrm=iso&tlng=pt >. Accessed: Mar. 15, 2020. doi: 10.1590/S0103-84782009005000232.
http://www.scielo.br/scielo.php?script=s...
), 26.6% of hoary foxes (Pseudalopexvetulus) were seroreactive for B. abortus in BAA in the semiarid region of Paraíba.
In table 2, the factors associated with prevalence of B. canis and B. abortus that were selected in the univariable analysis (P≤0.2) are shown. For B. canis, the following variables were selected: type of rearing (P = 0.006), age (P = 0.042), animal baths (P = 0.096) and severe infestation by fleas (P = 0.052). For B. abortus, breed (P = 0.105), age (P = 0.194) and frequency of water exchange in the animal’s bowel (P = 0.123) were selected. After robust Poisson regression analysis, two factors were confirmed to be associated with serore activity for B. canis and one for B. abortus. These were an age greater than 10 years old ( prevalence ratio - PR = 6.38; CI 95% = 1.28 - 31.86; P = 0.024) and dogs raised in the backyard (PR = 5.20; CI 95% = 1.12 - 24.12; P = 0.035) for B. canis, while it was not changing the animal’s water every day (PR = 1.48; CI 95% = 1.03 - 2.13; P = 0.033) for B. abortus. Both models for B. canis (Pearson’s chi-squares = 333.23; degrees of freedom (df) = 377; value/df =0.884) and B. abortus (Pearson’s chi-squared = 274.48; df = 359; value/df =0.765) had a good fit.
Univariable analysis of factors associated (P<0.20) with seroprevalence of Brucella canis and Brucella abortus in dogs of the Brejo Paraibano micro region.
In the present research, robust Poisson regression was used to estimate PR, and it was observed that dogs older than 10 years old had more chances of being seropositive (PR = 6.38). This finding may be related to greater exposure time to the agent when compared to younger animals. However, it is noteworthy that 87% of the sampled adult animals in this study were not neutered, which could be an associated factor. Thus, venereal disease transmission remains to be an important mode of transmission due to contact with vaginal fluids (KEID et al., 2007KEID, L. B. et al.A polymerase chain reaction for detection of Brucella canis in vaginal swabs of naturally infected bitches. Theriogenology, v.68, n.9, p.1260-70, 2007. Available from: <Available from: https://www.sciencedirect.com/science/article/abs/pii/S0093691X07005298 >. Accessed: Nov. 11, 2019. doi: 10.1016/j.theriogenology.2007.08.021.
https://www.sciencedirect.com/science/ar...
), urine and semen of infected animals (VOLKWEIS et al., 2018VOLKWEIS, F. S. et al. Detection of Brucella canis in blood, urine and seminal fluid of a naturally infected dog by PCR. Brazilian Journal of Veterinary Medicine, v.40, article e002718, 2018. Available from: <Available from: http://rbmv.org/index.php/BJVM/article/view/27/789 >. Accessed: Feb. 13, 2020. doi: 10.29374/2527-2179.bjvm002718.
http://rbmv.org/index.php/BJVM/article/v...
). The older the animal, the greater the chances of mating. and wider the variety of sexual partners; which consequently, leads to more chances of being possibly contaminated with the etiological agent.
Regarding the way the animals were raised, it was observed that dogs raised stuck in a backyard had more chances of being seropositive for B. canis (PR = 5.20). However, it must be highlighted that these animals did not have access to the street and had little contact with humans due to high aggressiveness, which may be an associated factor. Since a large portion of backyards in the houses of the region had only fences, these animals may have been able to have sexual contact with other dogs possibly entering these backyards, thus exposing them to a contracting a variety of infectious diseases.
Not changing the animals’ water daily also increased the chance of seropositivity for B. abortus (PR = 1.48). FERNANDES et al. (2011FERNANDES, A. R. F. et al. Brucella canisinfection in dogs attended in veterinary clinics from patos, Paraíba state, Brazil. Brazilian Journal of Microbiology, v.42, n.4, p.1405-8, 2011. Available from: <Available from: http://www.scielo.br/scielo.php?script=sci_arttext&pid=S1517-83822011000400023&lng=en&nrm=iso&tlng=en >. Accessed: Nov. 11, 2019. doi: 10.1590/S1517-83822011000400023.
http://www.scielo.br/scielo.php?script=s...
) verified that failure to adequately clean the environment of the animals was associated with seropositivity. This may be related to contamination of water by fluids of the reproductive tract possessing the infectious agent, which consequently leads to maintenance of bacteria after failing to change the water. Since these bacteria are known to survive for months in potable water, this reinforces the importance of changing the water in animals’ bowls daily.
CONCLUSION:
The current study’s results indicated a high prevalence of dogs seropositive for B. canis and B. abortus in the Brejo Paraibano microregion, which should encourage urgent implementation of control and prevention measures for this species. Based on the association analysis, proper management of animals, especially of older dogs, must be encouraged.
REFERENCES
- ALVES, C. J., et al. Aspectos epidemiológicos de Brucella canis em Patos, Paraíba, Brasil. Ciência Animal, v.13, n.1, p.45-49, 2003. Available from: <Available from: http://www.uece.br/cienciaanimal/dmdocuments/Comunicacao1.2003.1.pdf >. Accessed: Jun. 07, 2020.
» http://www.uece.br/cienciaanimal/dmdocuments/Comunicacao1.2003.1.pdf - ARENAS-GAMBOA, A. M. et al.Human brucellosis and adverse pregnancy outcomes.Current Tropical Medicine Reports, v.3, n.4, p.164-72, 2016. Available from: <Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5720386/ >. Accessed: Mar. 17, 2020. doi: 10.1007/s40475-016-0092-0. 2016.
» https://doi.org/10.1007/s40475-016-0092-0. 2016.» https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5720386/ - AZEVEDO, S. S. et al. Anticorpos antiBrucellaabortus, antiBrucella canis e antiLeptospira spp. em raposas (Pseudalopexvetulus) do semiárido paraibano, Nordeste do Brasil.Ciência Rural, v.40, n.1, p.1-3, 2010. Available from: <Available from: http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0103-84782010000100031&lng=en&nrm=iso&tlng=pt >. Accessed: Mar. 15, 2020. doi: 10.1590/S0103-84782009005000232.
» https://doi.org/10.1590/S0103-84782009005000232» http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0103-84782010000100031&lng=en&nrm=iso&tlng=pt - AZEVEDO, S. S. et al. Comparação de três testes sorológicos aplicados ao diagnóstico da infecção de caninos por Brucella canis Brazilian Journal of Veterinary Research and Animal Science, v.41, n.2, p.106-112, 2004. Available from: <Available from: http://www.scielo.br/scielo.php?script=sci_arttext&pid=S1413-95962004000200005 >. Accessed: Mar. 10, 2020. doi: 10.1590/S1413-95962004000200005.
» https://doi.org/10.1590/S1413-95962004000200005.» http://www.scielo.br/scielo.php?script=sci_arttext&pid=S1413-95962004000200005 - AZEVEDO, S. S. et al. Ocorrência de anticorpos contra Brucellaabortus em cães errantes da cidade de Patos, estado da Paraíba, Brasil. Arquivos do Instituto Biológico, v.70, n.4, p.499-500, 2003. Available from: <Available from: http://www.biologico.sp.gov.br/uploads/docs/arq/V70_4/azevedo.pdf >. Accessed: Jun. 07, 2020.
» http://www.biologico.sp.gov.br/uploads/docs/arq/V70_4/azevedo.pdf - BERGER, S. et al. “Various versatile variances: An Object-Oriented Implementation of Clustered Covariances in R”. 2017. Technical Report 2017-12, Working Papers in Economics and Statistics, Research Platform Empirical and Experimental Economics, Universität Innsbruck. Available from: <Available from: http://EconPapers.RePEc.org/RePEc:inn:wpaper:2017-12 >. Accessed: Dec. 10, 2019.
» http://EconPapers.RePEc.org/RePEc:inn:wpaper:2017-12 - CARSTENSEN, B. et al. Epi: A Package for Statistical Analysis in Epidemiology R package version 2.40, 2019. Available from: <Available from: https://CRAN.R-project.org/package=Epi >. Accessed: Dec. 10, 2019.
» https://CRAN.R-project.org/package=Epi - CHACHRA, D. et al. Comparative efficacy of Rose Bengal plate test, standard tube agglutination test and Dot ELISA in immunological detection of antibodies to Brucella abortus in sera.Journal of Bacteriology Research, v.1, n.3, p.30-33, 2009. Available from: <Available from: https://www.researchgate.net/publication/228767138_Comparative_efficacy_of_Rose_Bengal_plate_test_standard_tube_agglutination_test_and_Dot_ELISA_in_immunological_detection_of_antibodies_to_Brucella_abortus_in >. Accessed: Feb. 12, 2020.
» https://www.researchgate.net/publication/228767138_Comparative_efficacy_of_Rose_Bengal_plate_test_standard_tube_agglutination_test_and_Dot_ELISA_in_immunological_detection_of_antibodies_to_Brucella_abortus_in - CLEMENTINO, I. J. et al. Epidemiological situation of bovine brucellosis in the state of Paraiba, Brazil. Semina: Ciências Agrárias, v.37, n.5, p.3403-3412, 2016. Available from: <Available from: http://www.uel.br/revistas/uel/index.php/semagrarias/article/view/27230 >. Accessed: Dec. 10, 2019. doi: 10.5433/1679-0359.2016v37n5Supl2p3403.
» https://doi.org/10.5433/1679-0359.2016v37n5Supl2p3403.» http://www.uel.br/revistas/uel/index.php/semagrarias/article/view/27230 - DENTINGER, C. M. et al. Human Brucella canis infection and subsequent laboratory exposures associated with a puppy, New York City, 2012. Zoonoses and Public Health, v.62, n.5, p.407-14, 2015. Available from: <Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4639931/ >. Accessed: Dec. 12, 2019. doi: 10.1111/zph.12163.
» https://doi.org/10.1111/zph.12163.» https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4639931/ - DÍAZ, R. et al. The Rose Bengal Test in human brucellosis: A neglected Test for the Diagnosis of a Neglected Disease. Plos Neglected Tropical Diseases, v.5, n.4, e950, 2011. Available from: <Available from: https://journals.plos.org/plosntds/article?id=10.1371/journal.pntd.0000950 >. Accessed: Nov 11, 2019.doi: 10.1371/journal.pntd.0000950.
» https://doi.org/10.1371/journal.pntd.0000950.» https://journals.plos.org/plosntds/article?id=10.1371/journal.pntd.0000950 - FERNANDES, A. R. F. et al. Brucella canisinfection in dogs attended in veterinary clinics from patos, Paraíba state, Brazil. Brazilian Journal of Microbiology, v.42, n.4, p.1405-8, 2011. Available from: <Available from: http://www.scielo.br/scielo.php?script=sci_arttext&pid=S1517-83822011000400023&lng=en&nrm=iso&tlng=en >. Accessed: Nov. 11, 2019. doi: 10.1590/S1517-83822011000400023.
» https://doi.org/10.1590/S1517-83822011000400023.» http://www.scielo.br/scielo.php?script=sci_arttext&pid=S1517-83822011000400023&lng=en&nrm=iso&tlng=en - KEID, L. B. et al.A polymerase chain reaction for detection of Brucella canis in vaginal swabs of naturally infected bitches. Theriogenology, v.68, n.9, p.1260-70, 2007. Available from: <Available from: https://www.sciencedirect.com/science/article/abs/pii/S0093691X07005298 >. Accessed: Nov. 11, 2019. doi: 10.1016/j.theriogenology.2007.08.021.
» https://doi.org/10.1016/j.theriogenology.2007.08.021.» https://www.sciencedirect.com/science/article/abs/pii/S0093691X07005298 - KEID, L. B. et al. Comparison of agar gel immunodiffusion test, rapid slide agglutination test, microbiological culture and PCR for the diagnosis of canine brucellosis. Research in Veterinary Science, v.86, n.1, p.22-26, 2009.Available from: <Available from: https://www.sciencedirect.com/science/article/abs/pii/S0034528808001161?via%3Dihub >. Accessed: Nov. 13, 2019. doi: 10.1016/j.rvsc.2008.05.012.
» https://doi.org/10.1016/j.rvsc.2008.05.012.» https://www.sciencedirect.com/science/article/abs/pii/S0034528808001161?via%3Dihub - MARTINEZ, B. A. F. et al. Odds ratio or prevalence ratio? Anoverview of Reported Statistical Methods and Appropriateness of Interpretations in Cross-sectional Studies with Dichotomous Outcomes in Veterinary Medicine. Frontiers in Veterinary Science, v.4, article 193, 2017. Available from: <Available from: https://www.frontiersin.org/articles/10.3389/fvets.2017.00193/full >. Accessed: Dec. 13, 2019. doi: 10.3389/fvets.2017.00193.
» https://doi.org/10.3389/fvets.2017.00193.» https://www.frontiersin.org/articles/10.3389/fvets.2017.00193/full - MÉLO, S. K. M. et al. Brucelose canina: revisão de literatura. Ciência Veterinária nos Trópicos, v.16, n.1/2/3, p.7-17, 2013. Available from: <Available from: http://www.rcvt.org.br/volume16/rcvt_16_pg7.pdf >. Accessed: Jun. 06, 2020.
» http://www.rcvt.org.br/volume16/rcvt_16_pg7.pdf - NOORDHUIZEN, J. P. T. M. et al. Application of quantitative methods in Veterinary Epidemiology. Wageningen: Wageningen Press, 1997. 445p.
- R CORE TEAM (2019). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. Available from: <Available from: https://www.R-project.org/ >. Accessed: Jan. 13, 2020.
» https://www.R-project.org/ - RODRIGUES, R. T. G. A. et al. Canine brucellosis: a pratical review for the clinical veterinarian of small animals. Revista Brasileira de Higiene e Sanidade Animal, v.11, n.2, p.216-232, 2017. Available from: <Available from: https://www.researchgate.net/publication/318871002_Canine_brucellosis_a_pratical_review_for_the_clinical_veterinarian_of_small_animals >. Accessed: Jun. 06, 2020. doi: 10.5935/1981-2965.20170023.
» https://doi.org/10.5935/1981-2965.20170023.» https://www.researchgate.net/publication/318871002_Canine_brucellosis_a_pratical_review_for_the_clinical_veterinarian_of_small_animals - ROUSHAN, M. R. H. et al. Different clinical presentations of Brucellosis. Jundishapur Journal of Microbiology, v.9, n.4, article e33765, 2016. Available from: <Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4897599/ >. Accessed: Jan. 13, 2020. doi: 10.5812/jjm.33765.
» https://doi.org/10.5812/jjm.33765.» https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4897599/ - SADHU, D. B. et al. Seroprevalence and comparison of different serological tests for brucellosis detection in small ruminants. Veterinary World, v.8, n.5, p.561-566, 2015. Available from: <Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4774713/ >. Accessed: Jan. 17, 2020. doi: 10.14202/vetworld.2015.561-566.
» https://doi.org/10.14202/vetworld.2015.561-566.» https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4774713/ - TALUKDER, B. C. et al. Comparative evaluation of commercial serodiagnostic tests for the seroprevalence study of brucellosis in stray dogs in Bangladesh. Bangladesh Journal of Veterinary Medicine, v.9, n.1, p.79-83, 2011. Available from: <Available from: https://www.banglajol.info/index.php/BJVM/article/view/11217 >. Accessed: Feb. 16, 2020. doi: 10.3329/bjvm.v9i1.11217.
» https://doi.org/10.3329/bjvm.v9i1.11217.» https://www.banglajol.info/index.php/BJVM/article/view/11217 - THRUSFIELD, M. Veterinary epidemiology. Oxford: Blackwell Science, 2007. 610p.
- VASCONCELOS, R. T. J. et al. Soroprevalência e fatores de risco associados à infecção por Brucella canis em cães da cidade de Campina Grande, estado da Paraíba. Revista Brasileira de Saúde e Produção Animal, v.9. n.3, p.436-42, 2008. Available from: <Available from: http://revistas.ufba.br/index.php/rbspa/article/view/1003/664 >. Accessed: Jun. 07, 2020.
» http://revistas.ufba.br/index.php/rbspa/article/view/1003/664 - VOLKWEIS, F. S. et al. Detection of Brucella canis in blood, urine and seminal fluid of a naturally infected dog by PCR. Brazilian Journal of Veterinary Medicine, v.40, article e002718, 2018. Available from: <Available from: http://rbmv.org/index.php/BJVM/article/view/27/789 >. Accessed: Feb. 13, 2020. doi: 10.29374/2527-2179.bjvm002718.
» https://doi.org/10.29374/2527-2179.bjvm002718.» http://rbmv.org/index.php/BJVM/article/view/27/789 - ZEILEIS, A.; HOTHORN, T. Diagnostic checking in regression relationships. R News 2(3), 7-10, 2002. Available from: <https://CRAN.R-project.org/doc/Rnews/>. Accessed: Jan. 20, 2020.
- ZHELUDKOV, M. M.; TSIRELSON, L. E. Reservoirs of Brucella infection in nature. Biology Bulletin, v.37, n.7, p.709-15, 2010. Available from: <Available from: https://link.springer.com/article/10.1134/S106235901007006X >. Accessed: Jan. 22, 2020. doi: 10.1134/S106235901007006X
» https://doi.org/10.1134/S106235901007006X» https://link.springer.com/article/10.1134/S106235901007006X
-
CR-2020-0374.R1
BIOETHICS AND BIOSECURITY COMMITTEE APPROVAL
-
This study was approved by the Ethics Committee of the Center for Health and Rural Technology/Universidade Federal de Campina Grande under protocol of number 025/2017.
Publication Dates
-
Publication in this collection
11 Dec 2020 -
Date of issue
2021
History
-
Received
22 Apr 2020 -
Accepted
13 Aug 2020 -
Reviewed
18 Sept 2020