Abstracts
The solubility and availability of silicon can be influenced by soil reaction. A pot experiment with a clayey textured Rhodic Acrustox was conducted under greenhouse conditions to evaluate the effect of soil reaction on silicon availability to rice plants. The experiment was set up in a completely randomized design, using a factorial scheme (4 x 4) with four materials (calcitic lime, calcium and magnesium silicate, pure silicic acid, and wollastonite), four rates (0, 2500, 5000 and 7500 mg per 5 kg-pot) and four replicates. After 60 days, dry matter yield and silicon absorption by the rice shoot plants, pH CaCl2, and soluble silicon (0.5 mol L-1 acetic acid and 0.01 mol L-1CaCl2) in the soil were evaluated. The materials increased soil pH as the applied rates increased, except silicic acid. Soluble silicon extracted by 0.5 mol L-1 acetic acid also increased with applied rates. For calcium chloride, soluble silicon increased in the soil only with wollastonite and calcium and magnesium silicate, agreeing with its total content. Silicon absorption by the above-ground part of the rice plants was linearly correlated with rates of wollastonite, followed by calcium and magnesium silicate, silicic acid and calcitic lime. Soil pH increase with lime was not sufficient to provide silicon to the rice. The 0.01 mol L-1 CaCl2 soluble silicon had the best correlation with silicon absorption by plants. More studies are necessary under field conditions and other soils to corroborate the presented results.
Oriza sativa; soil; calcitic lime; silicate
A solubilidade e disponibilidade de silício podem ser influenciadas pela reação do solo. Com o objetivo de estudar o efeito da reação do solo sobre a disponibilidade de silício para a cultura do arroz, foi conduzido experimento em Latossolo Vermelho álico textura argilosa em casa-de-vegetação. O experimento foi conduzido em fatorial 4 x 4, delineamento em blocos inteiramente casualizados e quatro repetições. Quatro materiais (calcário, silicato de cálcio e magnésio, ácido silícico puríssimo e wollastonita) foram aplicados em doses equivalentes a 0, 2500, 5000, 7500 mg por vaso de 5 kg. Após 60 dias, foram avaliadas produção de matéria seca, absorção de silício da parte aérea do arroz, pH CaCl2, silício extraído em ácido acético (0,5 mol L-1) e CaCl2 (0,01 mol L-1) no solo. Os materiais aumentaram o pH do solo com as doses, exceto o ácido silícico. O silício solúvel em ácido acético foi crescente com as doses dos materiais. Para o CaCl2, apenas houve aumento de silício solúvel no solo com as doses de wollastonita e silicato de cálcio e magnésio, concordando com seu teor total. A absorção de silício pela parte aérea do arroz foi linearmente crescente com as doses da wollastonita, seguida do silicato de cálcio e magnésio, ácido silícico e calcário. O aumento do pH do solo com calcário não foi suficiente para fornecer silício ao arroz. O silício extraído com CaCl2 0,01 mol L-1 apresentou maior correlação com a absorção de silício pelas plantas. Mais estudos são necessários em condições de campo e outros solos para corroborar os resultados apresentados.
Oriza sativa; solo; calcário; silicato
SOILS AND PLANT NUTRITION
Soil reaction and absorption of silicon by rice
Reação do solo e absorção de silício pelo arroz
Mônica Sartori de CamargoI,* * Corresponding author < mscamarg@yahoo.com.br> ; Hamilton Seron PereiraII; Gaspar Henrique KorndörferIII; Angélica Araújo QueirozIV; Caroline Borges dos ReisV
IAPTA- Pólo Regional Centro Sul, Unidade de Pesquisa e Desenvolvimento de Tietê, Rod. SP 127, km 69, C.P. 18 - 18300-000 - Tietê, SP - Brasil
IIUFG - Centro de Ciências Agrárias e Biológicas Campus de Jataí - Rod. BR 364 - km 192 - Zona Rural - 75800-000 - Jataí, GO - Brasil
IIIUFU - Instituto de Ciências Agrárias, C. P. 593 - 38400-902 - Uberlândia, MG - Brazil
IVUFU - Programa de Pós-Graduação em Agronomia
VGraduate student University of Florida - Soil and Water Science Department - 1061 NW 122nd terrace - Newberry, FL 32669 - United States of America
ABSTRACT
The solubility and availability of silicon can be influenced by soil reaction. A pot experiment with a clayey textured Rhodic Acrustox was conducted under greenhouse conditions to evaluate the effect of soil reaction on silicon availability to rice plants. The experiment was set up in a completely randomized design, using a factorial scheme (4 x 4) with four materials (calcitic lime, calcium and magnesium silicate, pure silicic acid, and wollastonite), four rates (0, 2500, 5000 and 7500 mg per 5 kg-pot) and four replicates. After 60 days, dry matter yield and silicon absorption by the rice shoot plants, pHCaCl2, and soluble silicon (0.5 mol L-1 acetic acid and 0.01 mol L-1CaCl2) in the soil were evaluated. The materials increased soil pH as the applied rates increased, except silicic acid. Soluble silicon extracted by 0.5 mol L-1 acetic acid also increased with applied rates. For calcium chloride, soluble silicon increased in the soil only with wollastonite and calcium and magnesium silicate, agreeing with its total content. Silicon absorption by the above-ground part of the rice plants was linearly correlated with rates of wollastonite, followed by calcium and magnesium silicate, silicic acid and calcitic lime. Soil pH increase with lime was not sufficient to provide silicon to the rice. The 0.01 mol L-1 CaCl2 soluble silicon had the best correlation with silicon absorption by plants. More studies are necessary under field conditions and other soils to corroborate the presented results.
Key words:Oriza sativa, soil, calcitic lime, silicate
RESUMO
A solubilidade e disponibilidade de silício podem ser influenciadas pela reação do solo. Com o objetivo de estudar o efeito da reação do solo sobre a disponibilidade de silício para a cultura do arroz, foi conduzido experimento em Latossolo Vermelho álico textura argilosa em casa-de-vegetação. O experimento foi conduzido em fatorial 4 x 4, delineamento em blocos inteiramente casualizados e quatro repetições. Quatro materiais (calcário, silicato de cálcio e magnésio, ácido silícico puríssimo e wollastonita) foram aplicados em doses equivalentes a 0, 2500, 5000, 7500 mg por vaso de 5 kg. Após 60 dias, foram avaliadas produção de matéria seca, absorção de silício da parte aérea do arroz, pHCaCl2, silício extraído em ácido acético (0,5 mol L-1) e CaCl2 (0,01 mol L-1) no solo. Os materiais aumentaram o pH do solo com as doses, exceto o ácido silícico. O silício solúvel em ácido acético foi crescente com as doses dos materiais. Para o CaCl2, apenas houve aumento de silício solúvel no solo com as doses de wollastonita e silicato de cálcio e magnésio, concordando com seu teor total. A absorção de silício pela parte aérea do arroz foi linearmente crescente com as doses da wollastonita, seguida do silicato de cálcio e magnésio, ácido silícico e calcário. O aumento do pH do solo com calcário não foi suficiente para fornecer silício ao arroz. O silício extraído com CaCl2 0,01 mol L-1 apresentou maior correlação com a absorção de silício pelas plantas. Mais estudos são necessários em condições de campo e outros solos para corroborar os resultados apresentados.
Palavras-chave:Oriza sativa, solo, calcário, silicato
INTRODUCTION
Silicon is considered a beneficial element for higher plants (Marschner, 1995), being fundamental for rice production. Silicon fertilization has been proclaimed for highly weathered soils used for Brazilian agriculture in the last years, even though it is commonly used as a lime in Europe and as a silicate fertilizer in Japan, Korea, Hawaii and China since 1955 (Takahashi et al., 1990; Nayar et al., 1977).
Silicon absorption by plants occurs as monosilicic acid (H4SiO4), being proportional to the Si concentration in soil solution (Jones & Handreck, 1967; Fox et al., 1967) for monocotyledons. It behaves as a very weak acid, and even in pH 7.0, only 2 mg kg-1 are ionized (H3SiO4-); as pH increases the degree of ionization increases (Jones & Handreck, 1965). Monosilicic acid concentration in soil solution is variable, from 1 to 100 mg dm-3 SiO2 (Raven, 1983), related with clay content (Raij & Camargo, 1973), iron and aluminum oxides (Freitas et al., 1977) and pH (Beckwith & Reeve, 1963; Jones & Handreck, 1965). Among these factors, soil pH can be altered with liming materials, modifying the solubility of the monosilicic acid. This fact is important for rice, once maximum silicon absorption occurs within the 4.7-7.6 pH range (Cheong, 1982).
Silicon availability to plants is evaluated by extracting solutions with acetic acid 0.5 mol L-1 (Snyder, 2001; Korndörfer et al., 2001) and 0.01mol L-1 CaCl2 solution (Berthelsen et al., 2002). The pH of the extractant is also fundamental for monosilicic acid solubility, just as soil pH is important for soil solution concentration and silicon absorption by plants. Not enough silicate and lime comparative studies are available to prove if just the pH increase is able to provide this element to rice growing in soils with low silicon concentration, causing doubts about the effectiveness of silicate fertilizations. The objective was to evaluate the effect of soil reaction on silicon availability to rice.
MATERIAL AND METHODS
The experiment was carried out in a greenhouse with potted rice (5 kg dry soil capacity) using surface samples (0-0.2 m) of a Rhodic Acrustox. The soil chemical characteristics were: pHCaCl2 4.4; P (mg dm-3) 1.6 (extracted by H2SO4 0.025 molc dm-3 + HCl 0.05 molc dm-3); Si - 0.5 mol L-1 acetic acid (mg dm-3) 7.0; Al (mmolc dm-3) 6; Ca (mmolc dm-3) 2; Mg (mmolc dm-3) 1; K (mg dm-3) 49,8, Sum of bases (mmolc dm-3) 4; effective CEC (mmolc dm-3) 10; CEC pH 7.0 (mmolc dm-3) 49.2; V (%) 8.0; m (%) 60; and organic matter (g kg-1) 36. The physical characteristics were: very coarse (g kg-1) 80; fine coarse (g kg-1) 38, loam (g kg-1) 124, clay (g kg-1) 758. The total contents of SiO2, Al2O3, Fe2O3 (g kg-1) and Ki (SiO2/Al2O3 x 1.7) were 178, 282, 108 and 1.8 respectively.
The experiment was set up in a completely randomized design, using a factorial scheme (4 x 4) with four materials (calcitic lime, calcium and magnesium silicate, pure silicic acid, and wollastonite), four rates (0, 2500, 5000 and 7500 mg per pot) and four replicates. Rates of the materials were not corrected by EqCaCO3. The maximum rate of materials was defined as a function of the need of liming for this soil (3 t ha-1), calculated using the aluminum neutralization method described by Ribeiro et al. (1999). Additionally, two lower rates equivalent to 1 and 2 t ha-1 were used in this experiment. The materials were characterized regarding their origin, total Si, Ca, and Mg contents, and Neutralizing Power (NP) calculated and determined (Table 1). They were dried and sift through a 0.297-mm screen.
The soil samples were air dried, sift through a 5-mm screen and homogenized. Each pot (5 kg of soil) was fertilized with 5.825 g of superphosphate, 2.625 g of ammonium sulfate, 1.3125 g of KCl, 0.2625 g of FTE e 1.595 g of MgCl at sowing time and incorporated with the treatments. The rice seeds (Formoso variety) were sown at the rate of 20 seeds per pot. Thinning was performed ten days after germination, leaving 10 plants per pot. The surface fertilization took place 30 days after sowing with 2.62 g of ammonium sulfate per pot. The soil moisture, after sowing, was kept close to field capacity, using deionized water. Field capacity was determined according to Korndörfer et al. (1999).
Rice shoot was harvested 60 days after sowing. Plant material was dried, weighed and ground; silicon determination was made as described by Elliot & Snyder (1991). The biomass was evaluated based on dry matter production and the accumulated Si (uptake) was determined multiplying Si concentration by dry matter weight.
After harvest, soil samples from the pots were air dried and analyzed for pH(CaCl2) (EMBRAPA, 1999), soluble silicon concentration using 0.5 mol L-1 acetic acid (Korndorfer et al., 1999) and 0.01 mol L-1 CaCl2. Ten grams of soil were shaken for 1 hour with 100 mL of acetic acid or CaCl2. After that, the solution was decanted after a 15- minute waiting period; the solution was filtrated, and then left resting for 12 hours at room temperature. Si determination in different soil extracts was performed by beta molybdosilicic complex formation (Kilmer, 1965), using a spectrophotometer at 660 nm. The statistical analyses for dry matter production, soil pHCaCl2, soluble Si extracted by 0.5 mol L-1 acetic acid and 0.01 mol L-1 CaCl2 were made using the SAS (Statistical Analysis System, 1996). The analyses of variance were made applying the F test. The materials were compared by the Tukey test at 5%. A polynomial regression was used to analyze the effects of rates of materials.
RESULTS AND DISCUSSION
Soil pH increased to 5.9; 6.6 and 6.3 at the rates equivalent to 7500 mg per pot of wollastonite, calcium and magnesium silicate and lime, respectively. These pH values were important to provide an adequate range (4.7-7.6) to maximum silicon absorption by rice (Cheong, 1982). The pH increase is explained by enhanced hydroxyl (OH-) concentration from the materials (Alcarde, 1992). These results are in agreement with those found for wollastonite (Korndörfer et al., 1999), lime (Alcarde, 1992), and calcium and magnesium silicate (Dalto, 2003; Korndörfer et al., 2002; Pereira et al., 2004), since these materials are formed basically by CaSiO3 (Coelho, 1998) or CaCO3 (Alcarde, 1992). On the other hand, silicic acid did not promote changes on pH values (mean soil pH = 3.93), also observed by Benedito (2004) and Chagas (2004) growing rice in medium clayey textured soils.
The materials presented increases in acetic acid extracted silicon, except silic acid (Figure 1). There is a strong correlation between soluble silicon in acetic acid and soil pH (Figure 2). Dalto (2003), using lime on sugarcane straw, observed high silicon concentrations in 0.5 mol L-1 acetic acid than water extracted from soil after soybean harvest. It was also observed by Camargo et al. (2005), working with different soils treated with lime after 90 days of incubation. These data are in agreement with other articles that already showed the efficiency of the acid solutions compared to neutral-pH extracting solutions (Berthelsen et al., 2002; Sumida, 2002).
The higher extraction power of acetic acid is explained by the lowest pH (1.0-2.0) necessary for molybdosilicic complex formation (Kilmer, 1965). The change in soil pH contributed to a higher silicon extraction by the acetic acid, once this difference was higher with rates of materials. This could explain the high silicon concentrations in lime, wollastonite and calcium and magnesium silicate. Additionally, the acetic acid is able to extract non available silicon present in some materials such as calcium and magnesium silicate, but it is not true for wollastonite (Pereira et al., 2004). Xu et al. (2001) attributed to calcium silicate dissolution the high silicon extracted with acid extracting solution (pH 4) in calcareous soils. According to Brown & Mahler (1987), acidity and anions could additively impact Si release from soils, as showed by Wang et al. (2004).
The soluble silicon concentration extracted by 0.01 mol L-1 CaCl2 increased in soils treated with wollastonite and calcium and magnesium silicate. Silicic acid and lime did not increase soluble silicon concentration extracted by CaCl2 in the soil (Figure 3). Results are in agreement with total silicon concentration in those materials (Table 1). The pH changes did not show marked interference as observed with the acetic acid extractant (Figure 2). Once again, these results showed low solubility of silicon form silicic acid to the soil solution, agreeing with Chagas (2004).
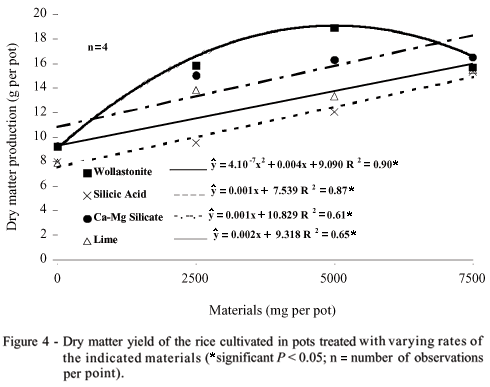
The dry matter production increased with rates of wollastonite, followed by calcium and magnesium silicate, lime, and silicic acid (Figure 4), reaching similar results at equivalent rates to 7500 mg per 5 kg-pot. Otherwise, silicon absorption by the rice plants (Figure 5) was a good indicator of total silicon of materials (Table 1). Silicic acid provided low silicon uptake as a consequence of its low silicon content, as already shown by Benedito (2004) and Chagas (2004).
The pH increase propitiated higher silicon uptake with rates of wollastonite and calcium and magnesium silicates (Figure 5), once they are silicon sources and liming materials. On the contrary, the lime treatment provided low increase of silicon absorption. It is necessary to choose adequate extracting solutions to measure available silicon to plants without overestimating results.
The 0.01 mol L-1 CaCl2 extractant presented the best correlation between soluble silicon concentration and plant silicon concentration (Figure 6), agreeing with Pereira et al. (2004) for the rice crops. These results are also in agreement with silicon total concentration of the studied materials (Table 1).
Considering soil and plant data of this experiment and the lowest influence by soil pH compared with acetic acid, the CaCl2 0.01 mol L-1 must be included in future research to indicate silicon availability to rice plants. Additionally, the pH increase with lime is not sufficient to provide highest silicon uptake by rice, comparing Wollastonite and Ca-Mg silicate.
Received March 20, 2006
Accepted March 02, 2007
- ALCARDE, J.C. Corretivos da acidez dos solos: características e interpretações técnicas. São Paulo: ANDA. 1992. (Boletim Técnico, 6).
- BECKWITH, R.S.; REEVE, R. Studies on soluble silica in soils. 1-The sorption of silicon acid by soils and minerals. Australian Journal Soil Research, v.1, p.157-168, 1963.
- BENEDITO, D.S. Interação boro x silício na nutrição, crescimento e produção do arroz. Lavras: UFLA, 2004. 62p. (Dissertação - Mestrado).
- BERTHELSEN, S.; NOBLE, A.; KINGSTON, G.; HURNEY, A.; RUDD, A. Effect of Ca-silicate amendments on soil chemical properties under a sugarcane cropping system. In: SILICON IN AGRICULTURE CONFERENCE, 2., Tsuruoka, Japão, 2002. Proceedings Tsuruoka: Japanese Society of Soil Science and Plant Nutrition, 2002. p.57.
- BROWN, T.H.; MAHLER, R.L. Effects of phosphorus and acidity levels on levels of silica extracted from a Palouse silt loam. Soil Science Society of America Journal, v.59, p.39-45, 1987.
- CAMARGO, M.S. de; KORNDÖRFER, G.H.; CORRÊA, G.F.; PEREIRA, H.S.; SCALIA, D.; RESENDE, R.H. Extratores de silício em solos: influência do calcário e fósforo. Bioscience Journal, v.21, p.9-19, 2005.
- CHAGAS, R.S.C. Avaliação de fontes de silício para as culturas do arroz e milheto. Piracicaba: USP/CENA, 2004. 100p. (Tese - Doutorado).
- CHEONG, B.T. Some significant functions of silicon to higher plants. Journal Plant Nutrition, v. 5, p.1345-1353, 1982.
- COELHO, P.E. Da escória ao vidro. Revista Limpeza Pública, v.49, p.36-45, 1998.
- DALTO, G. Manejo de silicato e calcário em soja cultivada sobre palhada de cana-de-açúcar Uberlândia: UFU, 2003. 90p. (Dissertação - Mestrado).
- Elliott, C.L.; Snyder, G.H. Autoclave-induced digestion for the colorimetric determination of silicon in rice straw. Journal of Agricultural and Food Chemistry, v.39, p.1118-1119, 1991.
- EMPRESA BRASILEIRA DE PESQUISA AGROPECUÁRIA. Manual de análises químicas de solos, plantas e fertilizantes Brasília: Embrapa Comunicação para Transferência de Tecnologia, 1999. 370p.
- FOX, R.L.; LEVA, J.A.; YOUNG, O.P.; PLUNCKNETT, U.L.; HERMAW, G.D. Soil and plant silicon and silicate response by sugarcane. Soil Science Society of America Proceedings, v.31, p.775-779, 1967.
- FREITAS, L.C.; COSTA FILHO, J.F. da; ALOISI, R.R.; MELO, W.J. Contribuição ao estudo da sílica solúvel em alguns perfis de solos. Científica, v.5, p.296-305, 1977.
- JONES, J.H.; HANDRECK, K.A. Silica in soils, plants, and animals. Advances in Agronomy, v.19, p.107-149, 1967
- JONES, J.H.; HANDRECK, K.A. Studies of silica in the oat plant. III-Uptake of silica from soils by the plant. Plant and Soil, v.23, p.79-96, 1965.
- KILMER, V.J. Silicon. In: Black, C.A. (Ed.). Methods of soil analysis Madison: ASA, 1965. pt1, p.959-962. (Agronomy, 9).
- KORNDÖRFER, C.M.; KORNDÖRFER, G.H.; CARDOSO, K. Aplicação do silicato de cálcio na recuperação de pastagem degradada de Brachiaria decumbens In: REUNIÃO BRASILEIRA DE FERTILIDADE DO SOLO E NUTRIÇÃO DE PLANTAS, 25., Rio de Janeiro. Proceedings. Rio de Janeiro: SBCS, 2002. p.8-11.
- KORNDÖRFER, G.H.; COELHO, N.M.; SNYDER, G.H.; MIZUTANI, C.T. Avaliação de métodos de extração de silício para solos cultivados com arroz de sequeiro. Revista Brasileira de Ciência do Solo, v.23, p.101-106, 1999.
- KORNDÖRFER, G.H.; SNYDER, G.H.; ULLOA, M.; POWELL, G.; DATNOFF, L.E. Calibration of soil and plant silicon analysis for rice production. Journal of Plant Nutrition, v.24, p.1071-1084, 2001.
- MARSCHNER, H. Mineral nutrition of higher plants 2.ed. London: Academic Press, 1995. 889p.
- NAYAR, P.K.; MISRA, A.K.; PATNAIK, S. Evaluation of silica-supplying power of soils for growing rice. Food and Fertilizer Technology Center, v.23, p.197-221, 1977.
- PEREIRA, H.S.; KORNDÖFER, G.H.; VIDAL, A.A.; CAMARGO, M.S. Silicon sources for the rice plants. Scientia Agricola, v.61, p.522-528, 2004.
- RAIJ, B. van; CAMARGO, O.A. Sílica solúvel em solos. Bragantia, v.32, p.223-236, 1973.
- RAVEN, J.A. The transport and function of silicon in plants. Biological Reviews of Cambridge Philosophical Society, v.58, p.179-207, 1983.
- RIBEIRO, A.C.; GUIMARÃES, P.T.G.; ALVAREZ V., V.H.; Recomendações para o uso de corretivos e fertilizantes em Minas Gerais. 5Ş Aproximação. Viçosa: UFV, 1999. 359p.
- SAS Institute INC. The SAS-system for windows: release 6.11 (software). Cary: SAS Institute, 1996.
- SNYDER, G.H. Methods for silicon analysis in plants, soils and fertilizers. In: DATNOFF, L.E.; SNYDER, G.H.; KORNDÖRFER, G.H. Silicon in agriculture Amsterdam: Elsevier, 2001. p.185-196.
- SUMIDA, H. Plant available silicon in paddy soils. In: SILICON IN AGRICULTURE CONFERENCE, 2., Tsuruoka, Japão, 2002. Proceedings Tsuruoka: Japanese Society of Soil Science and Plant Nutrition, 2002. p.43-49.
- TAKAHASHI, E.; MA, J.F.; MIYAKE, Y. The possibility of silicon as an essential element for higher plants. Comments in Agriculture and Food Chemistry, v.2, p.99-122, 1990.
- XU, G.; ZHAN, X.; CHUNHUA, L.; BAO, S.; LIU, X.; CHU, T. Assessing methods of available silicon in calcareous soils. Communication in Soil Science and Plant Analysis, v.32, p.787-801, 2001.
- WANG, J.J.; DODLA, S.K.; HENDERSON, R.E. Soil silicon extractability with seven selected extractants in relation to colorimetric and ICP determination. Soil Science, v.169, p.861-870, 2004.
Publication Dates
-
Publication in this collection
09 May 2007 -
Date of issue
2007
History
-
Received
20 Mar 2006 -
Accepted
02 Mar 2007