Abstracts
Humidified fractions of organic matter and soil microorganism populations are used as environmental quality indicators. This work aimed to study the changes in chemical and microbiological soil attributes, as well as in the humidified fractions, of the organic matter in a substrate from a clay extraction area cropped with Brachiaria mutica, Acacia mangium and Eucalyptus. In the Eucalyptus area, the P contents increased linearly with planting time. However, only at the twelfth year, differences between Eucalyptus and B. mutica areas have occurred. In the A. mangium area, such differences in the P content occurred at the third year with increment of 43%, at the 0-10 cm layer, in relation to B. mutica. Also, at the 0-10 cm layer, the total carbon contents were 98%, 78%, 70% and 40% higher than those found in Eucalyptus with three, five, twelve years of age and in the B. mutica area, respectively. Such increments also occurred in the humidified fractions, especially in the fulvic acids (C FA). The population of microorganisms was higher in the A. mangium area, mainly in the summer, where it was observed a positively correlation with total carbon (total bacteria, r = 0.96**, total fungi, r = 0.91*, and phosphate solubilizer microorganisms, r = 0.98**) and with the C FA fraction (total bacteria r = 0.96**, total fungi, r = 0.90*, and phosphate solubilizer microorganisms, r = 0.98**). The use of A. mangium led to improvements in the chemical and microbiological soil attributes in the substrate
arborous legumes; mycorrhizas; rhizobium; humic substances; soil micro biota
Frações da matéria orgânica humificada e a população de microrganismos do solo são utilizadas como indicadores da qualidade ambiental. Objetivou-se verificar mudanças nos atributos químicos, microbiológicos e nas frações humificadas da matéria orgânica em substrato de cava de extração de argila com cobertura de Brachiaria mutica, Acacia mangium e Eucalyptus. Nas áreas de Eucalyptus os teores de P aumentaram linearmente em função do tempo de plantio, porém somente no décimo segundo ano diferiu da B. mutica. Na área com A. mangium tais diferenças ocorreram no terceiro ano, com incremento de 43% na camada de 0-10 cm em relação à B. mutica. Ainda, na camada de 0-10 cm os teores de carbono total foram maiores 98%, 78%, 70% e 40% em relação ao Eucalyptus com três, cinco, doze anos, e B. mutica, respectivamente. Tais incrementos também ocorreram nas frações humificadas, principalmente nos ácidos fúlvicos (C AF). A população de microrganismos foi maior na cobertura com A. mangium, acentuadamente no verão; correlacionando-se com o carbono total (bactérias totais, r = 0,96**; fungos totais, r = 0,91*; e solubilizadores de fosfato, r = 0,98**) e com a fração C AF (bactérias totais, r = 0,96**; fungos totais, r = 0,90*; e solubilizadores de fosfato, r = 0,98**). O uso da A. mangium proporcionou melhoria nos atributos químicos e microbiológicos do substrato da cava de extração de argila.
leguminosas arbóreas; micorrizas; rizóbio; substâncias húmicas; microbiota do solo
SOILS AND PLANT NUTRITION
Recovery of degraded areas revegeted with Acacia mangium and Eucalyptus with special reference to organic matter humification
Ecuperação de áreas degradadas revegetadas com acácia e eucalipto enfocando a humificação da matéria orgânica
Jolimar Antonio SchiavoI, * * Corresponding author < schiavo@uems.br> ; Jader Galba BusatoII; Marco Antonio MartinsII; Luciano Pasqualoto CanellasII
IUEMS - Depto. de Ciência do Solo, Rod. Aquidauana, km 12 - 79200-000 - Aquidauana, MS - Brasil
IIUENF - Depto de Ciência do Solo, Av. Alberto Lamego, 2000 - 28013-610 - Campos do Goytacazes, RJ - Brasil
ABSTRACT
Humidified fractions of organic matter and soil microorganism populations are used as environmental quality indicators. This work aimed to study the changes in chemical and microbiological soil attributes, as well as in the humidified fractions, of the organic matter in a substrate from a clay extraction area cropped with Brachiaria mutica, Acacia mangium and Eucalyptus. In the Eucalyptus area, the P contents increased linearly with planting time. However, only at the twelfth year, differences between Eucalyptus and B. mutica areas have occurred. In the A. mangium area, such differences in the P content occurred at the third year with increment of 43%, at the 0-10 cm layer, in relation to B. mutica. Also, at the 0-10 cm layer, the total carbon contents were 98%, 78%, 70% and 40% higher than those found in Eucalyptus with three, five, twelve years of age and in the B. mutica area, respectively. Such increments also occurred in the humidified fractions, especially in the fulvic acids (CFA). The population of microorganisms was higher in the A. mangium area, mainly in the summer, where it was observed a positively correlation with total carbon (total bacteria, r = 0.96**, total fungi, r = 0.91*, and phosphate solubilizer microorganisms, r = 0.98**) and with the CFA fraction (total bacteria r = 0.96**, total fungi, r = 0.90*, and phosphate solubilizer microorganisms, r = 0.98**). The use of A. mangium led to improvements in the chemical and microbiological soil attributes in the substrate
Key words: arborous legumes, mycorrhizas, rhizobium, humic substances, soil micro biota
RESUMO
Frações da matéria orgânica humificada e a população de microrganismos do solo são utilizadas como indicadores da qualidade ambiental. Objetivou-se verificar mudanças nos atributos químicos, microbiológicos e nas frações humificadas da matéria orgânica em substrato de cava de extração de argila com cobertura de Brachiaria mutica, Acacia mangium e Eucalyptus. Nas áreas de Eucalyptus os teores de P aumentaram linearmente em função do tempo de plantio, porém somente no décimo segundo ano diferiu da B. mutica. Na área com A. mangium tais diferenças ocorreram no terceiro ano, com incremento de 43% na camada de 0-10 cm em relação à B. mutica. Ainda, na camada de 0-10 cm os teores de carbono total foram maiores 98%, 78%, 70% e 40% em relação ao Eucalyptus com três, cinco, doze anos, e B. mutica, respectivamente. Tais incrementos também ocorreram nas frações humificadas, principalmente nos ácidos fúlvicos (CAF). A população de microrganismos foi maior na cobertura com A. mangium, acentuadamente no verão; correlacionando-se com o carbono total (bactérias totais, r = 0,96**; fungos totais, r = 0,91*; e solubilizadores de fosfato, r = 0,98**) e com a fração CAF (bactérias totais, r = 0,96**; fungos totais, r = 0,90*; e solubilizadores de fosfato, r = 0,98**). O uso da A. mangium proporcionou melhoria nos atributos químicos e microbiológicos do substrato da cava de extração de argila.
Palavras-chave: leguminosas arbóreas, micorrizas, rizóbio, substâncias húmicas, microbiota do solo
INTRODUCTION
The production of bricks and clay roof tiles from clay materials mined in alluvial soils is of a great socioeconomic importance in Northern and Northeastern regions of the State of Rio de Janeiro. In the municipality of Campos dos Goytacazes, it is estimated that approximately 7,000 m3 of soil is daily removed, degrading an area of about 3,500 m2 day-1 (Ramos et al., 2003).
The tree-legume Acacia mangium Willd., due to its rusticity, adaptability to adverse soil and climate conditions, to its fast growth and high biomass production, associated to rhizobium and mycorrhizal fungi microsymbionts, is an important alternative for re-vegetation of these degraded areas (Franco & Faria, 1997). The annual deposition of biomass and nitrogen in areas cultivated with this legume species is about 9 t ha-1 (Andrade et al., 2000) and 100 kg of N ha-1 (Galiana et al., 2002), respectively. This high biomass deposition associated with high N contents promotes alterations on the organic matter contents, reflecting on soil fertility improvements.
The characteristics and content of the soil organic matter (SOM), due to its influence on the chemical, physical and biological soil attributes, can be used as indicators of environmental quality (Mielniczuk, 1999). The use of the relative distribution of organic matter factions as environmental quality evidence is described by works carried out by Kononova (1982) and Schnitzer (1991). The relationship between the carbon content in humic and fulvic acids (CHA/CFA) is related to the soil fertility (Kononova, 1982). Favorable conditions of pH, bases saturation and drainage, generally induces an increase in HA. This is probably due to the increment in microbial activity, which promotes the synthesis of more condensed humic substances (Orlov, 1998).
The SOM represents the main reservoir of energy for microorganisms and nutrients supply for plants. Microorganisms such as bacteria, fungi and other microfauna representatives are responsible for the energy and nutrients cycling and regulation of the soil organic matter transformations (Zech et al., 1997).
Soil microorganisms represent important component in the evaluation of the soil quality and may be used as biological indicators or as sustainability index for production systems (Franchini et al., 2007), and generally present strong correlation with the soil organic matter, i.e., they reflect changes on the organic matter concentration.
The objective of the present study was to evaluate changes in chemical and microbiological soil attributes in samples of substrates from clay extraction degraded areas re-vegeted with Acacia mangium and Eucalyptus. The humidified fractions of the organic matter were also evaluated.
MATERIAL AND METHODS
The field experiment was conducted in the municipality of Campos dos Goytacazes, state of Rio de Janeiro (21°25', 22°15'S and 40°55', 41°50'W). The predominant climate is classified as Aw (according to the Köppen classification), with average annual precipitation of 1,080 mm and average annual temperature ranging from 24°C to 25°C. The soil is classified as Fulvic Cambisol (Embrapa, 2006), formed in the Paraíba do Sul river delta.
In the clay extraction area, the soil superficial layer was removed, and the clay was collected untill the depth of 3 m. After extraction, the superficial layer was returned to the digged site. Samples of this substrate were collected at the 0 - 10 cm layer, and presented the following chemical characteristics: 5.5 of pH H2O; 7.0 mg kg-1 of P; 0.7, 35.0, 20.0 and 1.5 mmolc dm-3 of Al3+, Ca2+, Mg2+ and K+, respectively. The sand, silt and clay contents were of 480, 230 and 290 g kg-1, respectively. In the clay extraction area, brachiaria [Brachiaria mutica (Forsk.) Stapf] emerged as spontaneous vegetation.
One year after the clay extraction, the eucalyptus planting started being performed at different seasons, as well as the acacia legume species (Acacia mangium Willd), with the following treatment: eucalyptus (3, 5 and 12 years after planting), acacia (three years after planting) and brachiaria (control). Each plant coverage was formed by a planted area of 1,200 m2, using spacing of 3 × 2 between plants.
The acacia seedlings were inoculated with both a mixed inoculum of mycorrhizal fungi (Glomus macrocarpum, Glomus etunicatum and Entrophospora colombiana) and with specific rhyzobium strains BR 3609 and BR 6009. The rhyzobium strains were provided by Embrapa Agrobiologia and mycorrhizas by Norte Fluminense State University Soils Laboratory Collection.
Substrate samples were randomly collected from two soil layers (0 - 10 and 10 - 20 cm. Thereafter, the samples were dried at room temperature and sieved (2 mm mesh size). The pH was measured in water (soil:solution relationship equal to 1:2.5); the P and K contents were extracted by Mehlich-1 solution; Ca, Mg and Al by KCl 1 mol L-1; and the total N was determined through Kjeldahl method.
The organic matter fractioning was performed according to Guerra & Santos (1999). The substrate samples were submitted to pre-treatment with 2 mol L-1 orthophosphoric acid solution which separated the light fraction through density and also solubilized the free fulvic acids (FFA) fractions. Thereafter, a 0.1 mol L-1 NaOH + 0.1 mol L-1 Na4P2O7 solution was added to the same substrate sample at proportion of 1:10 (v v-1), which solubilized the fulvic acids (FA) and humic acids (HA). The insoluble solid residue resulting from the fractioning process was called as humine (H). The HA were separated from the FA through centrifugation after precipitation in acid medium obtained with the addition of concentrated H2SO4 up to pH 1-1.5. The carbon content in each fraction was determined by means of dichromatometry.
For the microorganisms quantification, the sampling was similar to that used in the chemical analyses, being collected in two seasons of the 2004 year [winter (July) and summer (January)] at the 0 - 10 cm layer. After collection, the soil samples were placed in trays for the removal of gross material (roots, pieces of wood and leaves), and then they were sieved in a 2mm sieve and stored in a freezer for one week, at 4°C. The microorganisms were counted using the series dilution procedure, Bunt & Rovira medium (1955), pH 6.5 - 7.0 added of cyclohexamide (10 μg mL-1) for bacteria counting; and Martin medium (1950), pH 5.6 added of streptomycin (100 μg mL-1) and rose bengal (70 μg mL-1) for fungi counting. The phosphate solubilizer microorganisms were performed according procedures adopted by Nahas et al. (1994).
The changes in humic substance fractions and soil microorganisms were analyzed by means of a random design (with treatments for brachiaria, acacia and eucalyptus with 3, 5 and 12 years after planting). The analysis of variance was performed using the F test, and data were compared through the Tukey test (p < 0.05). In addition, the Pearson correlation analysis was performed between microorganism population and total carbon and organic matter humidified fractions.
RESULTS AND DISCUSSION
The re-vegetation with arborous species affected the soil reactions and the nutrient contents (Table 1). An increase in the soil acidity was observed in areas covered with eucalyptus or acacia in all planting ages. This behavior, probably, is due to the higher organic matter level; and in areas covered with arborous legume species, probably due to the biological fixation of atmospheric N2. To maintain the intracellular pH, the legume species perform protons extrusion with consequent rhizosfera acidification (Marschner & Römheld, 1983).
A linear increase in P substrate contents was observed in areas covered with eucalyptus with increasing planting time (Table 1). However, such increases were significant in relation to brachiaria, only after the twelfth year after planting. On the other hand, when compared to acacia, the P content increased already at the third year after planting. The turnover of the phosphorus forms accumulated in the vegetal biomass produced in higher amounts by arborous legume species could have contributed for this increment. Busato et al. (2005) showed that the maintenance of the sugar cane straw for long periods provided increases on the soil P compartments with important impacts to the P content of humic acids.
At the 0 - 10 cm layer, Ca2+ and Mg2+ contents under eucalyptus coverage with five years of planting were 63% and 172% higher, respectively, than those in the grass spontaneous vegetation (Table 1). At the 10 - 20 cm layer, the Mg2+ contents presented behavior similar to that observed for the superficial layer. On the other hand, the Ca2+ contents were higher for the three-year-old eucalyptus coverage, with increment of 49% in relation to grass. Eucalyptus generally produces relatively poor burlaps due its efficient biochemical cycling, thus its contribution to the soil nutrients reposition is poor (Costa et al., 2005). However, Cunha et al. (2005) evaluating the biochemical and biogeochemical cycling in Eucalyptus grandis areas evidenced higher amounts of nutrients in the soil than those absorbed by plants. In a study comparing Acacia mangium and Eucalyptus grandis, Garay et al. (2003) verified that, seven years after planting, arborous legumes provided higher nutrient contents in the superficial horizon of the recomposed soil.
In general, the Na+ contents were high (Table 1). The soils formation environment and the delta sedimentary basin of the Paraíba do Sul River, which composes the landscape where this study was conducted tend to provide high Na+ contents. According to the Brazilian Soil Classification System (Embrapa, 2006), the sodium feature is acquired with sodium saturation higher than 20% as observed in the 10 - 20 cm layer under five-years-old eucalyptus coverage.
At the 0 - 10 cm layer, both the effective CEC and CEC at pH 7.0, under five-year-old eucalyptus coverage, were 109% and 74% higher, respectively, than those of grass coverage (Table 1). At the 10 - 20 cm layer, the effective CEC was higher under five-year-old eucalyptus coverage, while CEC at pH 7.0 was higher under three-year-old eucalyptus coverage, when compared with grass coverage. Values of both effective CEC and CEC at pH 7.0 were originated from high Ca2+, Mg2+, Na+ and H+ values found in the substrate under five-years-old eucalyptus coverage. A contribution of H+ to the CEC values at pH 7.0 in relation to Al3+ was observed, since its contents were low in substrate (Table 1). Also, CEC presented high bases saturation values (V% value), which was intensified in the five-years-old eucalyptus coverage. However, these high V% values are a result of the high Na+ saturation, which contributed to increase the V% values.
Regarding the coverage of the clay extraction site, the three-years-old acacia coverage, at the 0 - 10 cm layer, provided increases of 43% and 41% in the P and K contents, respectively, in relation to grass coverage. In addition, these values were higher than those observed in substrate under eucalyptus coverage, except for that with twelve years. This arborous legume can present biomass deposition of 9 t ha-1 year-1 (Andrade et al., 2000), with N, P, K+, Ca2+ and Mg2+ contents ranging from 130 to 170, 4.9 to 7.9, 24 to 31, 150 to 190 and 28.6 to 40 kg ha-1 year-1, respectively (Costa et al., 2004), which can improve the soil fertility (Garay et al., 2003).
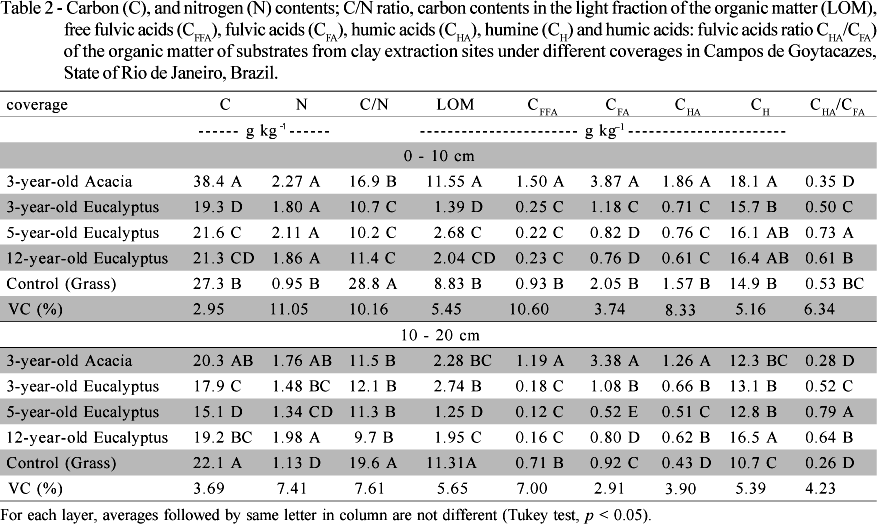
The carbon content in coverage with acacia was higher than the other treatments in the superficial substrate layer (Table 2). Increments of 98%, 78%, 80% and 40% were observed in relation to eucalyptus with three years, five years and twelve years and grass, respectively. At the 10 - 20 cm layer, the total C content was similar in acacia and grass coverages and higher than contents observed in eucalyptus areas. The nitrogen content, at the 0 - 10 cm layer, was not different between coverages, except for grass, which presented the lowest value (Table 2). At the 10 - 20 cm layer, the substrate under twelve-year-old eucalyptus presented the highest nitrogen content (1.98 g kg-1), followed by acacia (1.76 g kg-1).
The highest C/N ratio values were observed in area under grass coverage (Table 2). At the 0 - 10 cm layer, this value was 28.82, while at the 10 - 20 cm layer, it was 19.63. The decomposition rate is regulated by many factors, among them, the chemical quality of the material supplied, which may be expressed through the C/N ratio (Swift et al., 1979). In other words, low nitrogen contents are associated with lower decomposition rate, as observed in the grass coverage.
The organic carbon accumulation in acacia coverage area at the surface layer (0 - 10 cm) increased the C contents in all organic matter fractions (Table 2). At the 0 - 10 cm layer, the light fraction of the organic matter (LOM) under acacia coverage presented increments of 730, 330, 466 and 30% in relation to eucalyptus coverage with three, five and twelve years of age and to grass, respectively. However, at the 10 - 20 cm layer, higher LOM contents were observed in the grass coverage. The free fulvic acids fraction (CFFA) represented the smallest portion of the carbon content in all areas studied. In the fulvic acids fraction (CFA) in acacia coverage, at the 0 - 10 cm layer, increments of 228%, 372%, 409% and 88% were observed when compared to eucalyptus coverage (three, five and twelve years of age) and grass, respectively. The decrease observed for the CFFA contents and the increase in the CFA fraction may indicate that either the chemical evolution of the organic compounds with humification or the transportation of this fraction to deeper soil layers has occurred. Canellas et al. (2000) verified the CFA fraction mobility along the soil profile. This may be due to the fact that FA are soluble in both alkaline and acid mediums, which may facilitate their transportation through the soil profile.
The humic acids fraction (CHA) presented the lowest carbon content of the substrate. Among treatments evaluated, at the 0 - 10 cm layer, the highest CHA values were observed in the substrate covered with acacia, with increments of 18%, 162%, 145% and 205% in relation to substrate covered with grass or eucalyptus with three, five or twelve years of age, respectively. Similarly, at the 10 - 20 cm layer, these increments were 193%, 90%, 147% and 103%.
A decrease in the CHA/CFA ratio in treatments under acacia coverage in both soil layers was observed. The decrease is due to the increase on the fulvic acids fraction, granting a fulvic feature to the soluble alkaline fraction of humic substances in the substrate under arborous legume coverage. Canellas et al. (2004) evaluated herbaceous legumes and observed increases in the fulvic acids fraction contents and decreases on the CHA/CFA ratio.
The humine fraction (CH) represented the most abundant fraction of the humified carbon, and the highest contents were observed in substrates under acacia, 0 - 10 cm layer, and twelve-year-old eucalyptus, at 10 - 20 cm layers. The relative comparison of the humified organic matter by means of the participation of each fraction in the humified carbon total distribution (CFFA + CFA + CHA + CH = 100%) is presented in Figure 1. In general, the alkaline-soluble fractions (CFFA + CFA + CHA) were lower, reaching no more than 32% in the substrate under acacia coverage at the 10 - 20 cm layer. At the 0 - 10 cm layer, the humine fraction represented between 71% and 91% of the carbon content; and at the 10 - 20 cm layer, these values ranged from 68% to 91%. The majority of the humus present in tropical soils is composed of the humine fraction, which is formed by the insolubilization of vegetal residues that reach the soil (Andreux, 1996). In both soil layers, the lowest and highest values of CFFA and CH fractions were observed in substrates under eucalyptus coverage. In substrates under acacia coverage, the CFA fraction was the main constituent of alkaline-soluble fractions, corresponding to 15% and 18% at 0 - 10 cm and 10 - 20 cm layers, respectively. Changes in distribution of humidified fractions express the humus quality and the dynamics of the humification process. In area covered with acacia with accumulation of organic matter, an increase on the CFA fraction and decrease on the CHA/CFA ratio was observed.
The soil microorganisms were influenced by the type of coverage and season (Table 3). The number of bacteria, in both winter and summer seasons, was higher than fungi population in all coverages. During the summer, the coverage with acacia promoted the bacterial and fungal growth. The increases in the number of colony-forming units (CFU) of bacteria in acacia coverage were 1,126, 1,125, 1,582 and 425% higher than those observed in the coverage with eucalyptus (three, five and twelve years old) and grass, respectively. For the CFU of fungi, the increments were 495, 350, 626 and 660% higher than coverage with eucalyptus (three, five and twelve years old) and grass, respectively. Several factors influence the quantity and activity of the soil microbiota. A high number of bacteria and microbial activity in soils under eucalyptus and pinus occurred during the summer due to the higher rain precipitation (> humidity), higher temperatures and higher substrate supply as biomass deposited on the soil surface (Rigobelo & Nahas, 2004). Vieira & Nahas (2000) verified that the high CFU and number of total bacteria present in a Red Latosol was obtained at a temperature of 30°C.
During the winter, the increase in the phosphate-solubilizing microorganism population occurred in substrate under grass coverage. During the summer, the population remained unchanged, except for the coverage with acacia, which presented increments of 927, 787, 1,747 and 227% in relation to eucalyptus coverage (three, five and twelve years of age) and grass, respectively. Barroti & Nahas (2000) verified increases on the phosphate-solubilizing microorganism population in the soil under "guandu" (Cajanus cajan), coverage associated with rock phosphate fertilization. In the present study, the increase in population of this group of microorganisms in the acacia covered area could be due to the fact that at the seedling production phase, inoculation with rhizobium and mycorrhizal fungi were performed. Symbiosis between legumes and rhizobium require higher amounts of phosphorus, which may be obtained via phosphate solubilization through acid phosphatases produced by mycorrhizal fungi.
A significant correlation between microorganisms (total bacteria, total fungi and phosphate solubilizers) and the soil organic carbon content was observed (Table 4), indicating that the variation of microorganism population is due to the organic matter content. For the light fraction of the organic matter (LOM), this correlation occurred only for the phosphate-solubilizing microorganisms (r = 0.90*). In relation to organic matter humidified fractions, total bacteria presented significant and positive correlation with the free fulvic acids fractions (CFFA, r = 0.91*) and with the fulvic acids (CFA, r = 0.96**); total fungi with fulvic acids fractions (CFA, r = 0.90*) and humine (CH, r = 0.89*) phosphate solubilizers with all fractions (CFFA, r = 0.95*; CFA, r = 0.98**; CHA, r = 0.88*), except for humine.
In general, treatments with eucalyptus (between five and twelve years) and acacia presented the best results for the chemical and microbiological attributes of the substrate. However, the use of the arborous legume Acacia mangium in the revegetation of the clay extraction site provided increase in the following contents: organic carbon, total nitrogen, alkaline-soluble fractions of the organic matter and microbiota. The results suggest a great potential of the use of legume species in degraded areas.
CONCLUSIONS
The planting of Eucalyptus between five and twelve years of age increase the P, K+, Ca++ and Mg++ contents in the substrate.
A. mangium with only three years of age, at the superficial layer of the substrate, increases the P, K+, N and total carbon contents in humidified fractions of organic matter, remarkably fulvic acids, and in the summer, it increases the number of total bacteria and fungi and phosphate solubilizer microorganisms.
The microorganism population in the substrate presented correlation with organic carbon content and with the fulvic acids fraction.
Received June 15, 2007
Accepted September 12, 2008
- ANDRADE, A.G.; COSTA, G.S.; FARIA, S.M. Deposição e decomposição da serapilheira em povoamentos de Mimosa caesalpiniifolia, Acacia mangium e Acacia holosericea com quatro anos de idade em Planossolo. Revista Brasileira de Ciência do Solo, v.24, p.777-785, 2000.
- ANDREUX, F. Humus in world soils. In: PICCOLO, A. (Ed.) Humic substances in terrestrial ecosystems Amsterdam: Elsevier, 1996. p.45-100.
- BARROTI, G.; NAHAS, E. População microbiana total e solubilizadora de fosfato em solo submetido a diferentes sistemas de cultivo. Pesquisa Agropecuária Brasileira, v.35, p.2034-2050, 2000.
- BUNT, J.S.; ROVIRA, A.D. Microbiological studies of some subantartic soils. Journal of Soil Science, v.6, p.119-128, 1955.
- BUSATO, J.G.; CANELLAS, L.P.; VELLOSO, A.C.X. Fósforo num Cambissolo cultivado com cana-de-açúcar por longo tempo. I. Fracionamento seqüencial. Revista Brasileira de Ciência do Solo, v.29, p.935-944, 2005.
- CANELLAS, L.P.; BERNER, P.G.; SILVA, S.G.; SILVA, M.B.; SANTOS, G.A. Frações da matéria orgânica em seis solos de uma toposseqüência no Estado do Rio de Janeiro. Pesquisa Agropecuária Brasileira, v.35, p.133-143, 2000.
- CANELLAS, L.P.; ESPINDOLA, J.A.A.; REZENDE, C.E.; DE CAMARGO, P.B.; ZANDONADI, D.B.; RUMJANEK, V.M.; GUERRA, J.G.M.; TEIXEIRA, M.G.; BRAZ-FILHO, R. Organic matter quality in a soil cultivated with perennial herbaceous legumes. Scientia Agricola, v.61, p.53-61, 2004.
- COSTA, G.S.; FRANCO, A.A.; DAMASCENO, R.N.; FARIA, S.M. Aporte de nutrientes pela serapilheira em uma área degradada e revegetada com leguminosas arbóreas. Revista Brasileira de Ciência do Solo, v.28, p.919-927, 2004.
- COSTA, G.S.; GAMA-RODRIGUES, A.C.; CUNHA, G.M. Decomposição e liberação de nutrientes da serapilheira foliar em povoamentos de Eucalyptus grandis no norte Fluminense. Revista Árvore, v.29, p.563-570, 2005.
- CUNHA, G.M.; GAMA-RODRIGUES, A.C.; COSTA, G.S. Ciclagem de nutrientes em Eucalyptus grandis W. Hill ex Maiden no Norte Fluminense. Revista Árvore, v.29, p.353-363, 2005.
- EMBRAPA. Centro Nacional de Pesquisa de Solos. Manual de métodos de análises de solo 2. ed. Rio de Janeiro: Embrapa/CNPS, 1997. 212p.
- EMBRAPA. Centro Nacional de Pesquisa de Solos. Sistema Brasileiro de Classificação de Solos Rio de Janeiro: Embrapa/CNPS, 2006. 412p.
- FRANCHINI, J.C; CRISPINO, C.C; SOUZA, R.A; TORRES, E.; HUNGRIA, M. Microbiological parameters as indicators of soil quality under various soil management and crop rotation systems in southern Brazil. Soil and Tillage Research, v.92, p.18-29, 2007
- FRANCO, A.A.; FARIA, S.M. The contribution of N2-fixing tree legumes to land reclamation and sustainability in the tropics. Soil Biology and Biochemistry, v.29, p.897-903, 1997.
- GALIANA, A.; BALLE, P.; GUESSAN KANGA, A.N.; DOMENACH, A.M. Nitrogen fixation estimated by the 15N natural abundance method in Acacia mangium Willd. inoculated with Bradyrhizobium sp. and grown in silvicultural conditions. Soil Biology and Biochemistry, v.34, p.251-262, 2002.
- GARAY, I.; KINDEL, A.; CARNEIRO, R.; FRANCO, A.A.; BARROS, E.; ABBADIE, L. Comparação da matéria orgânica e de outros atributos do solo entre plantações de Acacia mangium e Eucalyptus grandis Revista Brasileira de Ciência do Solo, v.27, p.705-712, 2003.
- GUERRA, J.G.M.; SANTOS G.A. Métodos químicos e físicos. In: SANTOS, G.A.; CAMARGO, F.A.O. (Ed.) Fundamentos da matéria orgânica do solo: ecossistemas tropicais e subtropicais. Porto Alegre: Gênesis, 1999. p.267-291.
- KONONOVA, M.M. Materia orgánica del suelo: su naturaleza, propiedades y métodos de investigación. Barcelona: Oikostau, 1982. 364p.
- MARTIN, J.P. Use of acid, rose bengal, and streptomycin in the plate method for estimating soil fungi. Soil Science, v.69, p.215-232, 1950.
- MARSCHNER, H.; RÖMHELD, V. In vivo measurement of root-induced pH changes at the soil-root interface. Effect of plant species and nitrogen source. Zeitschrift für Pflanzenphysiologie, v.111, p.241-251, 1983.
- MIELNICZUK, J. Matéria orgânica e sustentabilidade de sistemas agrícolas. In: SANTOS, G.A.; CAMARGO, F.A.O. (Ed.) Fundamentos da matéria orgânica do solo: ecossistemas tropicais e subtropicais. Porto Alegre: Gênesis, 1999. p.1-7.
- NAHAS, E.; CENTURION, J.F.; ASSIS, L.C. Microrganismos solubilizadores de fosfato e produtores de fosfatases de vários solos. Revista Brasileira de Ciência do Solo, v.18, p.43-48, 1994.
- ORLOV, D.S. Organic substances of Russian soils. Eurasian Soil Science, v.31, p.946-953, 1998.
- RAMOS, I.S.; ALEXANDRE, J.; ALVES, M.G.; BARROSO, J.A.; TEIXEIRA, L.S.; CORREA, F.P. Dimensionamento da indústria cerâmica em Campos dos Goytacazes, RJ. In: CONGRESSO BRASILEIRO DE CERÂMICA , 47., João Pessoa, 2003. Anais CD-ROM,
- RIGOBELO, E.C.; NAHAS, E. Seasonal fluctuations of bacterial population and microbial actvity in soil cultivated with eucayptus and pinus. Scientia Agricola, v.61, p.88-93, 2004.
- SCHNITZER, M. Soil organic matter: the next 75 years. Soil Science, v.151, p.41-58, 1991.
- SWIFT, M.J.; HEAL, O.W.; ANDERSON, J.M. Decomposition in terrestrial ecosystems Oxford: Blackwell, 1979. 372p.
- VIEIRA, F.C.S.; NAHAS, E. Quantificação de bactérias totais e esporuladas no solo. Scientia Agricola, v.57, p.539-545, 2000.
- ZECH, W.; SENESI, N.; GUGGENBERGER, G.; KAISER, K.; LEHMANN, J.; MIAN, T.M.; MILTNER, A.; SCHROTH, G. Factors controlling humification and mineralization of soil organic matter in the tropics. Geoderma, v.79, p.117-161, 1997.
Publication Dates
-
Publication in this collection
29 June 2009 -
Date of issue
June 2009
History
-
Received
15 June 2007 -
Accepted
12 Sept 2008