Abstracts
The first cases of ornamental plants found infected by Brevipalpus transmitted viruses (BTV) were described in the 1990's from the region of Piracicaba, State of São Paulo, Brazil; subsequent cases were from other regions in the country and other American countries. Currently, 37 ornamental plant species (for the sake of simplicity, orchids being considered as a single species), belonging to 18 families of dicotyledons, have been reported hosting BTV. Because of the non systemic type of infection of these viruses, the localized diseases they cause are unimportant usually, but they have the potential to cause economic losses if severe outbreaks of Brevipalpus mite populations occur. Some ornamentals may serve as reservoirs to BTV known to cause serious damage to food crops as Citrus leprosis virus- cytoplasmic type (CiLV-C), passion fruit green spot virus (PFGSV) and Coffee ringspot virus (CoRSV).
Acari; Tenuipalpidae; ornamental industry
Os primeiros casos de plantas ornamentais encontradas naturalmente infetadas por vírus transmitidos por Brevipalpus (Acari: Tenuipalpidae) (VTB) foram registrados nos anos 1990 na região de Piracicaba, Estado de São Paulo, e ocorrências subseqüentes foram observadas em várias outras regiões do país e de outros países das Américas. Atualmente acham-se relatadas 37 espécies de ornamentais (para efeito de simplificação, orquídeas foram consideradas como única espécie) pertencentes a 18 famílias botânicas. Pelo fato de causarem apenas infecções localizadas, geralmente nas folhas, VTB em ornamentais não causam preocupações aos produtores, mas potencialmente podem causar perdas econômicas se ocorrerem explosões populacionais do ácaro vetor. Plantas ornamentais podem servir de reservatório de VTB de importância econômica como os vírus da leprose dos citros-tipo citoplasmático (CiLV-C), da mancha verde do maracujá (PFGSV) e da mancha anular do cafeeiro (CoRSV).
Acari; Tenuipalpidae; indústria de ornamentais
REWIEW
An annotated list of ornamentals naturally found infected by Brevipalpus mite-transmitted viruses
Lista comentada de plantas ornamentais naturalmente infectadas por vírus transmitidos por ácaros Brevipalpus
Elliot Watanabe KitajimaI,* * Corresponding author < ewkitaji@esalq.usp.br> ; José Carlos Verle RodriguesII,III; Juliana Freitas-AstuaIV,V
IUSP/ESALQ - Depto. de Fitopatologia e Nematologia, C.P. 9 - 13418-900 Piracicaba, SP - Brasil
IIUniversity of Puerto Rico, Jardim Botanico Sur, San Juan, PR 00926 USA
IIICentro de Energia, Meio Ambiente e Biodiversidade, Av. Carvalho Leal, 1777 69065 - Manaus, AM Brasil
IVEmbrapa Mandioca e Fruticultura Tropical, C.P. 7 - 44370-000 Cruz das Almas, BA - Brasil
VCentro APTA Citros Sylvio Moreira, C.P. 4 13490-970 Cordeirópolis, SP - Brasil
ABSTRACT
The first cases of ornamental plants found infected by Brevipalpus transmitted viruses (BTV) were described in the 1990's from the region of Piracicaba, State of São Paulo, Brazil; subsequent cases were from other regions in the country and other American countries. Currently, 37 ornamental plant species (for the sake of simplicity, orchids being considered as a single species), belonging to 18 families of dicotyledons, have been reported hosting BTV. Because of the non systemic type of infection of these viruses, the localized diseases they cause are unimportant usually, but they have the potential to cause economic losses if severe outbreaks of Brevipalpus mite populations occur. Some ornamentals may serve as reservoirs to BTV known to cause serious damage to food crops as Citrus leprosis virus- cytoplasmic type (CiLV-C), passion fruit green spot virus (PFGSV) and Coffee ringspot virus (CoRSV).
Key words: Acari, Tenuipalpidae, ornamental industry
RESUMO
Os primeiros casos de plantas ornamentais encontradas naturalmente infetadas por vírus transmitidos por Brevipalpus (Acari: Tenuipalpidae) (VTB) foram registrados nos anos 1990 na região de Piracicaba, Estado de São Paulo, e ocorrências subseqüentes foram observadas em várias outras regiões do país e de outros países das Américas. Atualmente acham-se relatadas 37 espécies de ornamentais (para efeito de simplificação, orquídeas foram consideradas como única espécie) pertencentes a 18 famílias botânicas. Pelo fato de causarem apenas infecções localizadas, geralmente nas folhas, VTB em ornamentais não causam preocupações aos produtores, mas potencialmente podem causar perdas econômicas se ocorrerem explosões populacionais do ácaro vetor. Plantas ornamentais podem servir de reservatório de VTB de importância econômica como os vírus da leprose dos citros-tipo citoplasmático (CiLV-C), da mancha verde do maracujá (PFGSV) e da mancha anular do cafeeiro (CoRSV).
Palavras-chave: Acari, Tenuipalpidae, indústria de ornamentais
BREVIPALPUS MITES
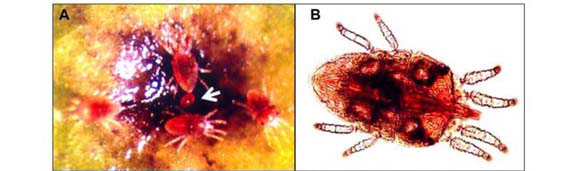
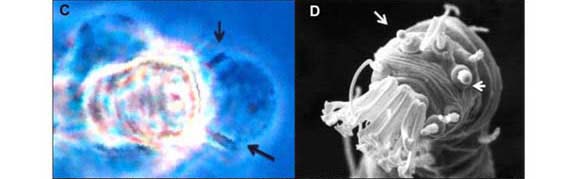
Brevipalpus (Figure 1 A-E) is a genus of Tenuipalpidae (Acari: Prostigmata), family whose members are usually referred to as false spider mites. They are ubiquitous in tropical and subtropical regions around the world. This genus includes more than 200 species (Mesa et al., 2009), but only three are known to be involved in the transmission of plant viruses, namely B. californicus Banks, B. obovatus Donnadieu and B. phoenicis (Geijskes) (Childers et al., 2003a), which have been found naturally infesting nearly 1000 different plant species belonging to more than 100 botanical families throughout the world (Childers et al., 2003b). Brevipalpus mites have a peculiar biology: most of the individuals in a colony are clones, generated by haploid females reproducing by thelytokous parthenogenesis (i.e, females begetting females). Only rarely are males produced (Childers et al., 2003a). It has been reported that these haploid mites remain as females because of the infection by bacterial symbionts of the genus Cardinium (Weeks et al., 2001) present in most of the organs of the mite (Kitajima et al., 2007). Non-infected eggs generate males. The reproductive role of the male is unknown. Although it copulates, it apparently does not fertilize females (Pijnacker et al., 1981). The three vector Brevipalpus species are distinguished based on few external characteristics, as the number of distal solenidia on tarsus leg II (Figure 1 C, D) and the number of dorsolateral body setae (Figure 1 E) (Wellbourn et al., 2003). Genetically distinct genotypes were reported and cryptic species occur among the three major recognized morphospecies (Rodrigues et al., 2004; Groot, 2006).
Substantial new information regarding mite/virus relationship based on ultrastructural and molecular data have been published. Transmission electron microscopy (TEM) associated with immunogold labeling and qRT-PCR studies strongly suggest that the Citrus leprosis virus, cytoplasmic type (CiLV-C) circulates but does not replicate in B. phoenicis tissues (Kitajima et al., 2009). Conversely, there are evidences that some nuclear type of the viruses transmitted by Brevipalpus (N-BTV) as Orchid fleck virus (OFV), Coffee ringspot virus (CoRSV), Clerodendrum chlorotic spot virus (ClCSV) and Citrus leprosis virus, nuclear type (CiLV-N) multiply in the mite vector's body (Kitajima et al., 2009).
Plant viruses transmitted by Brevipalpus mites
Until the beginning of 2000, only a few BTV were known. The economically most important and the first to be identified as such was the Citrus leprosis (CL), originally described in Florida at early 20th century (Hume, 1901; Fawcett, 1911). In the 1930's, it was found in Argentina, Uruguay, Paraguay and Brazil. Since then, it has been reported from other South American countries (Bolivia, Colombia and Venezuela) as well as from Central American countries (Panama, Costa Rica, Honduras, Guatemala, Mexico) (Rodrigues et al., 2003; Bastianel et al., 2006). The disease is of concern for citrus growers in the Caribbean region, where the virus is absent, and in the US, where it has not been found since the 1960's (Childers et al., 2003c). The transmission of CL virus by Brevipalpus mites was initially shown in Argentina by Frezzi (1940) and subsequently confirmed in Brazil and US by Musumeci and Rossetti (1963) and Knorr (1968), respectively. It was found that two distinct viruses, based on cytopathic effects, both transmitted by B. phoenicis, respectively cytoplasmic (CiLV-C) and nuclear (CiLV-N), cause leprosis symptoms, being the CiLV-C the prevalent (Rodrigues et al., 2003). The complete sequence of the genome of CiLV-C was obtained (Pascon et al., 2006) being a bipartite positive sense ssRNA distinct from known viruses. As a consequence, a new genus, Cilevirus, was proposed for this virus (Locali-Fabris et al., 2006).
Another well-known BTV is the Coffee ringspot virus (CoRSV) first described by Bitancourt (1938) in coffee (Coffea arabica L.) plantations of the state of São Paulo. This virus causes localized ringspots on leaves and berries. Ultrastructural studies demonstrated nuclear type of cytopathology in the tissues of the lesions (Kitajima and Costa, 1972). Its transmission by B. phoenicis and by mechanical means was demonstrated by Chagas (1973) and Chagas et al. (1961). Part of its genome has been sequenced, which allowed the design of primers that specifically amplify CoRSV by RT-PCR (Locali et al., 2005).
Passion fruit green spot virus (PFGSV) is another cytoplasmic type BTV that devastated passion fruit grooves at Vera Cruz, state of São Paulo, in the 1990s'. The virus is vectored by B. phoenicis and causes green spots on yellow fruits and in senescent yellowish leaves and necrotic lesions on stems. When infection is heavy, stem lesions fuse causing annealing and consequent death of the infected plant (Kitajima et al., 2003b). The genome of PFGSV has been partially sequenced and specific primers based on these sequences have been designed and used for its detection (Antonioli-Luizon et al., 2008). Because cytophatic effects caused by BTV-cytoplasmic type were only seen in these green areas of senescent leaves, it is possible that senescence is retarded in the virus-infected leaf tissues. This finding suggested that other plants could have senescent leaves with green spots, ringspots, brown spots, besides green leaves with chlorotic and/or necrotic ringspot, associated with Brevipalpus. Many suspected cases were found, mostly in ornamentals infested by Brevipalpus. Examination of the tissues of the lesion by TEM led to the discovery of BTV in almost 40 different plant species, mostly ornamentals (Kitajima et al., 2003a).
Symptoms caused by Brevipalpus-transmitted viruses
A common characteristic of the symptoms caused by BTV is that they are always localized: chlorotic and/or necrotic spots and ringspots on green leaves and green spots or ringspots on senescent leaves; chlorotic and/or necrotic spots on the stems, chlorotic or brown spots usually depressed on the fruits; brown spots on the flowers (Kitajima et al., 2003a). The process which keeps the infection by BTV restricted to the lesion is not understood yet. It may result from a tug-of-war between the viral replication and the cell defense mechanism and the inability of the BTV to infect the vascular region (Marques et al., 2007). This, however, does not always apply since systemic infection was observed when Chenopodium quinoa Wild. and C. amaraticolor Coste & Reyn. were mechanically inoculated with three viruses causing nuclear type of cytopathology (OFV, CoRSV and Clerodendrum chlorotic spot virus [ClCSV]) and kept at high temperatures (28-30ºC) for about two weeks (Kondo et al., 1995; Boari et al., 2004; Kitajima et al., 2008). Non-inoculated leaves of these plants exhibited chlorotic spots and chlorotic veins. In these plants, the mechanism which hinders the invasion of phloem vessels by the virus is somehow overcome and the pathogen spreads to the rest of the plant.
Cytopathology of Brevipalpus-transmitted viruses
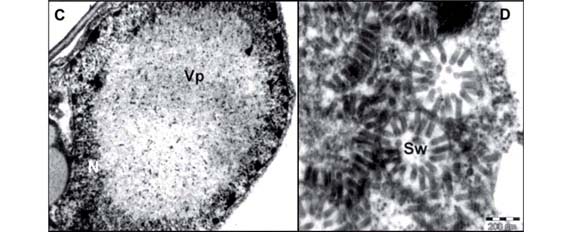
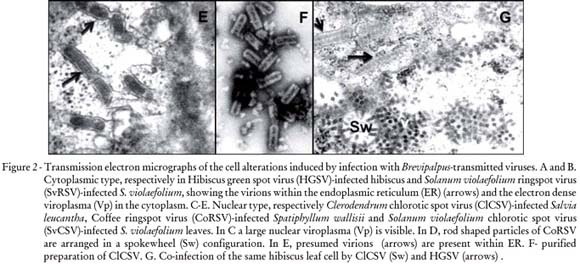
Another important feature of BTV is the alteration they induce in infected cells. Ultrastructural studies by TEM consistently revealed two types of cytopathology (Figure 2) (Kitajima et al., 2003a): Cytoplasmic type referred to now on as C-BTV and Nuclear type (N-BTV). C-BTV is characterized by the presence of short, bacilliform, membrane-bounded particle (60-70 nm × 110-120 nm) which occurs single or grouped within cisternae of the endoplasmic reticulum and by the appearance of an electron- dense, vacuolated inclusion, referred to as viroplasma, in the cytoplasm (Figure 2 A, B). Usually, there is a single, large viroplasma with elliptical or irregular profile. Immunogold labeling experiments with an antibody specific to the CiLV-C p29 (putative capsidal) protein produces heavy labeling of the viroplasma, in addition to the bacilliform particles, indicating that it is a site of accumulation of viral proteins. C-BTV was first described in cells infected by CiLV-C (Colariccio et al., 1995). In some instances, evidence of viral morphogenesis by budding process of viroplasma material into the endoplasmic reticulum has been observed (Kitajima et al., 2003a). Usually BTV causing green spots on senescent leaves are of cytoplasmic type.
The N-BTV viral or presumed viral particles are short and rodlike (40 nm × 100 nm). Some of them (OFV, CoRSV and ClCSV) were purified and their particles exhibit cross striation in negatively stained preparations (Figure 2 F). The particles are present in the nucleus or cytoplasm and are commonly arranged perpendicularly onto the membranes of the endoplasmic reticulum or nuclear envelope. Sometimes this membrane/virion complex produces cylindrical structures which in cross section exhibit radial arrangement of particles surrounded by a membrane ring, generating a configuration referred to as "spokewheel" (Figure 2 D). Also groups of viral particles may form a large, side-by-side, sheet-like, aggregate. An additional characteristic feature of N-BTV infection is the presence of an electron lucent mass, the viroplasma, in the nucleus (Figure 2 C). In the case of OFV, antibody produced against purified particles heavily impregnates the viroplasma, indicating that it may represent, as with the cytoplasmic viroplasma, a site of accumulation of viral materials. A possible variant of N-BTV seems to be the cases in which short, membrane bounded, rhabdoviruslike particles appear within endoplasmic reticulum with rare nuclear viroplasma as in the case of CoRSV-SP, Solanum violaefolium chlorotic spot virus (SvCSV) (Kitajima et al., 2004) (Figure 2 E) and Cestrum ringspot virus (CeRSV) (Kitajima et al., 2003a). In a few cases, as in Clerodendrum thomsoniae and Hibiscus rosa-sinensis, co-infection of the same cell by C-BTV and N-BTV has been observed (Figure 2 G).
Brevipalpus-transmitted viruses and ornamentals
As our knowledge about possible BTV advanced in the last decade, a large number of cases were detected, mostly in ornamentals, causing localized lesions. One reason why these viruses have been neglected is the lack of systemic infection. If the mite population declines, due to seasonal factors, and the infected organs fall off or die, the sources of inoculum are reduced, and the disease literally disappears. But they are potentially important in cases when favorable environmental conditions (climate, host species, lack of predators, etc.) result in outbreaks of the mite vector. In this scenario, a heavy infection may occur causing economic damage.
Except for OFV which has a world wide distribution (Kondo et al., 2003) because of the extensive international trade that spreads both the vector and the virus, other known BTV have been registered only in the American continent. However, growing international trade, including living plants, may help the dispersion of BTV and viruliferous mite vectors to other parts of the world. The situation becomes more complex because Brevipalpus mites have already been found in most of the tropical and subtropical regions of the world, infesting many plant species. Additionally, economically important BTV, such as CiLV-C and CoRSV are able to infect other plant species including some ornamentals (Bastianel et al., 2006; Novelli et al., 2008), under either natural or experimental conditions. Thus these viruses may accidentally be introduced into disease-free regions by infected ornamentals.
An annotated list of ornamental plants that can be infected by BTV is subsequentely presented, with comments on the corresponding diseases, known vectors and other known information including natural infection by other viruses (Albouy and Devergne, 1999; Alexandre et al., 2005). For this list, we considered BTV to have been identified, when at least experimental mite transmission has been reported. In other cases, we are referring to the presumed BTV found by TEM associated with the lesions as "unidentified BTV". Table 1 summarizes this annotated list. The description of the ornamental properties of the cited plant species were mostly based on Lorenzi and de Souza (2001), whereas plant names were based on the International Plant Name Index (www.inpi.org).
I. ACANTHACEAE
1. Thunbergia erecta (Benth.) T. Anderson - King's mantle (manto-do-rei)
King's mantle is a woody, erect shrub, 2-2.5 m tall, from Tropical Africa, with ovate, thick, brilliant leaves. The blue to purple flowers are tubular with yellow throat and they bloom nearly the entire year. Although grown in isolation, they are most commonly used as hedge. T. erecta plants with ringspot on leaves (Figure 3 A) and infested by B. phoenicis were found in a residential garden at Águas de São Pedro, SP. TEM of the lesions revealed the presence of cytopathic effect caused by C-BTV (Nogueira et al., 2003). Similar case was found with king's mantle being used as hedge in a hotel at Jaguariúna, SP (E.W. Kitajima, unpublished data). No further work to characterize and identify the virus was conducted.
No other virus has been reported infecting T. erecta in Brazil but Broad bean wilt virus (BBWV) and Datura yellow vein virus (DYVV) have been recorded elsewhere (Albouy and Devergne, 1999).
II. AGAVACEAE
2. Cordyline terminalis (L.) Kunth. - Ti-plant (dracena vermelha)
This plant was formerly classified as Liliaceae, but according to the International Plant Name Index (INPI) it is now considered a member of the family Agavaceae. Ti-plant is a semi-woody, erect shrub, which may reach 2 m in height, with terminal tufts of elongated, smooth, flexible white or colorful (green to red, sometimes striped) leaves. Ti-plants are planted in vases to be kept indoors or in outdoor rows. It is native to India, Malaysia and Polynesia. A few plants with pink spots and ringspots on red leaves (Figure 3 B) were found at the campus of Escola Superior de Agricultura "Luiz de Queiroz", Universidade de São Paulo (ESALQ/USP), Piracicaba, state of São Paulo (SP). Tissues of these lesions exhibited cytopathic effects typical of C-BTV (Ferreira et al., 2004a). The causal virus may be related to the Hibiscus green spot virus (HGSV) because these Ti-plants were growing next to affected hibiscus. A potyvirus has been reported in this plant associated with leaf discoloration and ringspots in Croacia and Italy (Albouy and Devergne, 1999).
III. APOCYNACEAE
3. Allamanda cathartica L. - Allamanda (alamanda)
Allamanda is a tropical climbing vine with milky sap, bright and thick leaves with trumpet shaped yellow flowers, native to the Brazilian coast. It is widely used as hedge or arbors. Plants with green spots and ringspots on senescent leaves (Figure 3 C) were found in residential garden in Manaus, AM, associated with infestation by B. phoenicis and cytopathic effects of N-BTV (Rodrigues et al., 2008). No additional work was carried out to identify the causal virus. Cucumber mosaic virus (CMV) has been found naturally infecting allamanda in Brazil (Alexandre et al., 2005).
4. Beaumontia grandiflora Wall. - Herald's trumpet (trombeta do arauto)
This is an evergreen, branched woody twining plant with large, thick and glossy leaves and large, white, fragrant, trumpet shaped flowers. It is native to tropical Himalaya and it is suitable for arbors or as isolated plants. Some plants growing in the campus of ESALQ, Piracicaba, SP, were found with green spots and ringspots on senescent leaves (Figure 3 D) in which cytopathology caused by C-BTV was observed (Kitajima et al., 2006). The causal virus remains unidentified.
5. Trachelospermum jasminoides Lem. - star jasmine (jasmim estrelado)
Star jasmine is an evergreen, branched, lactiferous, climbing vine, with small dark green leaves and abundant pure-white, intensely fragrant flowers with tubular corolla opening out into five petal-like twisted lobes. It is native to Eastern and Southeastern Asia, and it is widely used in arbors and as hedge. Star jasmine plants with chlorotic ringspots (Figure 3 E) were found in a residential garden in North Central Florida, USA, associated with B. phoenicis infestation and N-BTV cytopathology (Rodrigues et al., 2004b). The causal virus is still unidentified.
IV. ARACEAE
6. Anthurium sp. - Anthurium (antúrio)
The genus Anthurium contains 600-800 species with neotropical distribution mostly from Central and South America. They grow as twining epiphytes or terrestrial. Several species are used as ornamentals due to the inflorescence, which has a large spathe of varied colors and small flowers arranged on a fleshy axis, the spadix. Leaves are usually large, spatulate or round. Sample of anthurium leaves, possibly A. andraeanum Linden, showing chlorotic spots and ringspots (Figure 3 F) from a commercial nursery of Cruz das Almas, BA, associated with Brevipalpus mite infestation were examined by TEM and revealed C-BTV cytopathology (Ferreira et al., 2004a). The same finding was made in a plant growing in the campus of ESALQ at Piracicaba, SP (E.W. Kitajima, unpublished data).
7. Dieffenbachia amoena Hort. ex. Gentil - dumbcane (comigo-ninguém-pode)
Dumbcane is a perennial, herbaceous, erect plant reaching about 1.5 m, with very ornamental, dark green leaves with irregular white zones along the veins. It is widely used in interiorscape at half shade in vases or in gardens. The sap of dumbcane is poisonous. A dumbcane plant showing ringspots on the leaves (Figure 4 A), associated with infestation by B. phoenicis, was found in a residential garden in Piracicaba, SP. Cells from the lesions showed cyopathology of C-BTV (E.W. Kitajima, unpublished data). Because this plant was together with Salvia leucantha also infected by an unidentified C-BTV, it is possible that the same virus was infecting both species. A plant of dumbcane growing near ClCSV-infected bleeding heart, in the campus of ESALQ/USP developed chlorotic spots (Figure 4 B). The plant was infested by B. phoenicis and TEM examination of the lesion tissues revealed cell alterations induced by N-BTV. Further analysis revealed that this plant was infected by ClCSV, the bleeding heart acting as the virus source (Kitajima et al., 2008).
8. Monstera deliciosa Liebm. - widowleaf (costela-de-adão)
The widowleaf is an evergreen, vigorous liana, with large, coriaceous and perforated leaves and grows well in half shade. It usually grows over wall, moss pillar or tree. Plants of M. deliciosa showing ringspot on their leaves (Figure 4 C) were found in a residential garden in Manaus, AM, associated with infestation by B. phoenicis. TEM examination revealed cytopathology of N-BTV in the tissues of the lesion (Rodrigues et al., 2008). The virus remains unidentified.
9. Spathiphyllum wallisii Regel - Peace lily (lírio-da-paz)
Vigorous, perennial, erect plant, 30-40 cm tall with bright, coriaceous leaves originated from Colombia and Venezuela. It produces inflorescence with characteristic white spathe that turns green as it ages, without fragrance. Peace lily is cultivated in flower beds or vases at half shade, and also in aquarium. Several plants cultivated in a vase in a bakery at Piracicaba, SP, exhibited ringspots on their leaves (Figure 4 D), associated with infestation with B. phoenicis, and the cells of the lesions showed cytopathic effects of C-BTV. The causal virus was not identified yet. As in the case of dumbcane, S. wallisii plants growing next to a ClCSV-infected bleeding heart in the campus of ESALQ/USP at Piracicaba, SP, were infected under natural conditions by this virus, developing chlorotic spots (Figure 4 E). These symptoms were reproduced by experimental transmission of the ClCSV by the mite B. phoenicis and the presence of the virus was confirmed by TEM, RT-PCR and serology (Kitajima et al., 2008). Peace lily plants with chlorotic spots were also found in a public park at Maringá, PR. Lesions contained unidentified N-BTV (E.W. Kitajima, unpublished data). A peace lily plant purchased in a nursery at Limeira, SP, developed chlorotic spots (Figure 4 F) initially taken as a result of infection by ClCSV and indeed showing cytopathology of N-BTV, but RT-PCR assays indicated that this particular plant was infected by an isolate of CoRSV (Novelli et al., 2008). Thus peace lily seems to be susceptible to several BTV. Another case of natural infection of peace lily with CoRSV was found in a plant collected in Araras, SP. This plant was co-infected by PFGSV as revealed by RT-PCR and TEM assays (V.M. Novelli, unpublished data).
Some ornamental araceae as Anthurium and Dieffenbachia have been found naturally infected by Dasheen mosaic virus (DsMV) in several parts of Brazil (Alexandre et al., 2005). Elsewhere, natural infection of ornamental araceae by viruses like Tomato spotted wilt virus (TSWV), Alfalfa mosaic virus (AMV), CMV, Arabis mosaic virus (ArMV) and Potato virus X (PVX), besides DsMV, are reported (Albouy and Devergne, 1999).
V. ARALIACEAE
10. Hedera canariensis Willd. - Algerian or Canary Island ivy (hera)
H. canariensis is a semi-herbaceous, clinging vine with ornamental, lobate leaves. Aerial roots permit the plant to cling on walls and trees. It is also used as ground cover. Algerian ivy, clinging to trees in the campus of ESALQ/USP, Piracicaba, was found showing large green patches on senescent leaves (Figure 5 A) associated with B. phoenicis. TEM examination revealed C-BTV cytopathology (Kitajima et al., 2003a) in the leaf tissues of the green areas. A similar case was observed in Algerian ivy plants growing in a residential garden at Maringá, PR (E.W. Kitajima, unpublished data). The causal virus is still unidentified. In a survey conducted at the northern and central Florida, USA, Algerian ivy plants with clear ringspots on leaves (Figure 5 B) were found, associated with B. phoenicis. Cell alterations caused by N-BTV were observed, but the virus is still unidentified (Kitajima et al., 2003). In Italy and Yugoslavia, an unassigned rhabdovirus, referred to as Ivy vein clearing virus, was found infecting this species (Albouy and Devergne, 1999).
11. Schefflera actinophylla (Endl.) Hams. - umbrella tree (árvore guarda-chuva)
The umbrella tree is an evergreen semi-woody tree, 5-7 m tall, single or multi-stemmed trunks native to Australia, with shiny green leaves held in groups of 7-9 compound leaves and terminal inflorescence. It may be cultivated indoors in vases or outdoors and is considered extremely invasive. Green spots and ringspots on senescent leaves of umbrella tree (Figure 5 C) were found on the campus of ESALQ/USP, Piracicaba, associated with B. phoenicis, with C-BTV cytopathology (Kitajima et al., 2003a). Because these plants were close to Algerian ivy with green spots, it is possible that the symptoms in these two species are caused by the same virus. A similar case was found at the Universidade Estadual de Campinas (Unicamp), at Campinas, SP (E.W. Kitajima, unpublished data).
Schefflera is also susceptible to Schefflera ringspot virus (SRV) found in Australia and Europe (Albouy and Devergne, 1999) and also in Brazil (Alexandre et al., 2005).
VI. ASTERACEAE
12. Unxia kubitzkii H. Robinson - (botão-de-ouro)
This is a perennial, erect, 30-50 cm tall, herbaceous and branched plant, with yellowish green, somewhat rough leaves with serrate margins. It produces small, golden yellow flowers, nearly the entire year. It is commonly cultivated at the border of gardens. Some plants of U. kubitzkii growing among Solanum violaefolium ringspot virus (SvRSV)-infected S. violaefolium plants in a public park at Piracicaba, SP, showed chlorotic spots (Figure 5 D), associated with infestation by B. obovatus. TEM revealed typical C-BTV cytopathology in the lesioned tissue. The causal virus was identified by mite transmission to S. violaefolium as SvRSV, thus being a case of natural infection of U. kubitzkii by SvRSV (E.W. Kitajima, unpublished data).
VII. DRACAENACEAE
13. Dracaena marginata Lam. - Madagascar dragon tree (dracena-de-Madagascar)
Madagascar dragon tree is a slow growing, erect shrub, 2-4 m tall, with large trunk, native to Madagascar. It produces several branches each terminating in a rosette of narrow ribbon like green leaves with purple margin. The curved branches produce a nice silhouette for ornamental purposes in vases or outside. D. marginata plants showing chlorotic spots and ringspots (Figure 5 E) were found in a residential garden at Águas de São Pedro, SP, and later at Piracicaba, SP, associated with B. phoenicis infestation. TEM revealed C-BTV cytopathology in lesioned tissue (Nogueira et al., 2004). This presumed C-BTV is still unidentified.
VIII. GERANIACEAE
14. Pelargonium hortorum L. H. Bailey - common geranium (gerânio)
Common geranium is a tender perennial, strongly scented, upright, semi woody plant with scalloped leaves exhibiting a concentric broad color zone. This plant produces erect inflorescences, each with a long stalk holding several white to pink single or folded flowers. It is native to South Africa and it has a large number of varieties. Common geranium is usually cultivated in vases held in windows and balconies as well as in gardens. Common geranium plants showing ringspots (Figure 5 F) were found in a residential garden at Águas de São Pedro, SP, associated with infestation by B. phoenicis. Cells from the lesioned tissue exhibited C-BTV cytopathology, but the virus is still unidentified (Nogueira et al., 2003). In the campus of ESALQ/USP, Piracicaba, some P. hortorum plants growing near to bleeding-heart infected by ClCSV were found with chlorotic spots on their leaves (Figure 5 G). These plants were infested by B. phoenicis and lesioned tissue showed N-BTV cytopathology. The causal virus was identified as ClCSV and it was possibly a case of natural transmission of ClCSV from the infected bleeding heart (Kitajima et al., 2008).
In Brazil no other virus has been registered naturally infecting common geranium, but elsewhere Pelargonium leaf curl virus (PLCV), CMV, several nepoviruses, Pelargonium ringspot virus (PRSV), Pelargonium flower breaking virus (PFBV) and Eggplant mottled dwarf virus (EMDV) have been found infecting this ornamental (Albouy and Devergne, 1999).
IX. LAMIACEAE
15. Clerodendrum x speciosum Tiej. & Binn. - bleeding heart (coração sangrento)
16. C. thomsoniae Balf. glory - bower (lágrima-de-cristo)
17. C. splendens G. Don. - flaming - bower (clerodendro vermelho)
18. C. speciosissimum Hort - Java glory - bower (clerodendro)
Clerodendrum is a very large genus containing about 400 species of trees, shrubs and vines from tropical and subtropical woodlands of Africa, America and Asia. Many are used as ornamentals. Taxonomy of this genus was uncertain and although it is considered belonging to the Verbenaceae, recent molecular data using nuclear ribosomal ITS and chloroplast ndhF sequence, suggested that the genus Clerodendrum should be included in the Lamiaceae (Steane et al., 2004).
Clerodendrum x speciosum known as bleeding heart, is a hybrid of C. splendens and C. thomsoniae. It is a semi-herbaceous climbing, everblooming bush, with red flowers, of persistent calyx produced in profusion at the tip of the vine. Chlorotic to necrotic spots (Figure 6 A) on bleeding heart leaves were first observed in a residential garden at Piracicaba, SP, and later in many spots of the city and also at Campinas and Araras, SP, Manaus, AM and Brasília, DF, associated with infestation by B. phoenicis. The lesioned tissue exhibited N-BTV cytopathology. The symptoms were reproduced by infestation of mites collected from symptomatic plants revealing that a N-BTV was the causal agent, which was named Clerodendrum chlorotic spot virus (ClCSV). Several other plant species of the same genus (C. thomsoniae, C. splendens, C. speciosissimum) and others of different families (Hibiscus rosa-sinensis, H. coccineus, H. cannabinus, Malvaviscus arboreus, Salvia leucantha, Annona muricata, Spathiphyllum wallisii, Dendrobium sp., Chenopodium quinoa, C. amaranticolor, Tetragonia expansa, etc.) were demonstrated to be susceptible to ClCSV either by natural or experimental transmission by B. phoenicis and also by mechanical transmission. Plants of C. quinoa and C. amaranticolor developed chlorotic spots on the leaves after mechanical inoculation, and if the plants are kept under high temperatures (28-30ºC) for about two weeks, systemic infection developed and uninoculated leaves developed chlorotic spots and yellow veins. These systemically infected leaved had a high virus content. Using these plants, it was possible to purify the virus and produce a specific antiserum (Kitajima et al., 2008).
The purified virus permitted sequencing of part of its genome, and to design pair of primers that can be used for specific molecular detection of ClCSV by RT-PCR (Kubo et al., 2007). ClCSV was also found naturally infecting glory - bower (C. thomsoniae) at Piracicaba, SP and Belém, state of Pará, causing chlorotic/necrotic spots on the leaves (Figure 6 B). At least in one instance, brownish spots were observed in the white sepal (Figure 6 C) in which virus was present (Kitajima et al., 2008). This plant, native to West Africa, is similar to bleeding heart but the flowers have a white and inflated sepal and an expanded red corolla.
Flaming bower (C. splendens) plants growing in residential gardens at Piracicaba and Holambra, SP, were also found naturally infected by ClCSV, showing chlorotic spots (Figure 6 D) (Kitajima et al., 2008). This species native to tropical Africa has oval and wrinkled leaves, and scarlet flowers arranged in dense terminal clusters.
During a survey of plant viruses in the Amazon basin, a plant of Java glory - bower (C. speciosissimum) was found infected by ClCSV. Affected leaves showed dark green spots and ringspots (Figure 6 E) on their leaves (Rodrigues et al., 2008). This Clerodendrum species is native to Sri Lanka and Java and has broad dark green leaves and a large upright panicle of fiery scarlet flowers produced practically all over the year. Glory - bower (C. thomsoniae) plants were found at Piracicaba, SP, showing green spots on senescent leaves (Figure 6 F) in which C-BTV cytopathology was observed. These symptoms were reproduced by infestation of healthy glory - bower with mites collected from affected leaves, and the virus was named Clerodendrum green spot virus (ClGSV) (Kitajima et al., 2003a). In a few instances, these lesions also contained N-BTV and co-infection in the same leaf parenchymal cell by both N- and C- BTV were observed (Kitajima et al., 2003a). This is considered strong evidence that C- and N-BTV are unrelated because they do not interfere with the replication of each other.
Ringspots on leaves of C. x speciosum were found at Cordeirópolis, SP, associated with the infection of an unidentified tospovirus (E.W. Kitajima, unpublished data). Elsewhere there is a report of the infection of Clerodendrum by TSWV (Aubouy and Devergne, 1999).
19. Salvia leucantha Cav. - Mexican bush sage (salvia branca)
S. leucantha is a perennial, erect, bushy evergreen shrub, 50-90 cm tall, with long branches and lanceolate leaves, with white and woolly underneath. Its flowers are white with lavender-blue calyces, blooming nearly the entire year. Mexican bush sage is native to Mexico. It is planted mostly at the edges of gardens. Mites can transmit ClCSV to S. leucantha producing chlorotic spots (Figure 7 A) (Kitajima et al., 2008). S. leucantha was also found infected by an unidentified C-BTV in a residential garden at Piracicaba, SP, showing green spots on senescent leaves (Figure 7 B) (Kitajima et al., 2003a).
X. MALVACEAE
20. Hibiscus rosa sinensis L. - Chinese hibiscus (hibisco, mimo-de-Venus)
21. H. schizopetalus Hook. f. - Japanese lantern (hibisco crespo)
22. H. syriacus L. - rose of Sharom (rosa-de-sarom, hibisco-da-Síria)
23. H. coccineus Walter- scarlet hibiscus
24. H. cannabinus L. - kenaf (kenaf)
The genus Hibiscus comprises more than 200 species native to tropical and subtropical regions, and includes annual and perennial herbaceous plants as well as woody shrubs and small trees. Flowers in this genus are characteristically trumpet shaped with five or more colorful petals. Many Hibiscus species are ornamental, but there are fibrous plants like kenaf (H. cannnabinus) and others are edible (H. sabdariffa).
Chinese hibiscus is probably the most widely cultivated Hibiscus species in tropical and subtropical regions. It is an evergreen shrub native to tropical Asia, 3-5 m tall, and presents a large number of varieties. Isolated flowers are large without scent and colors vary from red, white, yellow, orange and pink with single or double sets of petals, blooming nearly the entire year usually attracting hummingbirds. Chinese hibiscus may be cultivated as isolated plants or in rows, forming living fences. H. schizopetalus is native to eastern Africa, 3-4 m tall with long arching branches and narrow lanceolate leaves. Solitary flowers hang at the end of the stems with a long pendunculus with fringed and lacy petals, usually red or pink. H. syriacus, originated in Asia, is a woody shrub with upright habit, reaching 2-3 m in height with oval shaped leaves on open and loose branches. Flowers, smaller than those of Chine hibiscus, are of varied colors (white, pink, red, lavender) and produced during the entire year.
A C-BTV was found in the tissues of green or brown spots (Figure 7 C) or green ringspots observed in senescent leaves of H. rosa-sinensis for the first time at Piracicaba, SP (Kitajima et al., 2003a) associated with B. phoenicis infestation. These spots were reproduced by transferring mites found in affected plants to non symptomatic H. rosa sinensis plants. The presumed virus was named Hibiscus green spot virus (HGSV). Later Chinese hibiscus infected by HGSV were found in several other places in the state of São Paulo (Campinas, Cordeirópolis, Águas de São Pedro, São Pedro, Araras, Atibaia, Jaboticabal, etc.) as well as in Brasília, DF, and other states as Rio de Janeiro, Minas Gerais, Paraná, Pará, Mato Grosso, Mato Grosso do Sul (E.W. Kitajima, unpublished data) and Amazonas (Rodrigues et al., 2008), and in other countries as Bolivia (Santa Cruz de la Sierra), Panama (Panama City), Cuba (Havana), Argentina (Córdoba), Paraguay (Assunción) (E.W. Kitajima, unpublished data), Puerto Rico (San Juan), and the USA (Florida) (J.C.V. Rodrigues, unpublished data), revealing that this virus has a wide distribution in the Americas. A quick survey in several parts of Europe, South Africa, Thailand, Singapore, Hong Kong, Japan and Australia did not detect this disease on hibiscus but these observations were restricted to few places and it is not possible to discard the possibility that HGSV may occur outside the American continent. Similar symptoms were also found in plants of H. schizopetalus (Figure 7 D) and H. syriacus (Figure 7 E) associated with B. phoenicis infestation and cytopathology of C-BTV and believed to be caused by the HGSV. These cases were registered in residential gardens and public parks at Piracicaba and Campinas, SP, and Brasília, DF. While searching for HGSV in Chinese hibiscus plants, another type of symptom was found, represented by chlorotic spots with irregular borders (Figure 8 A). The symptoms were also associated with B. phoenicis infestation and electron microscopy revealed cytopathic effect of N-BTV (Kitajima et al., 2003a). These plants were found at Piracicaba and Campinas, SP and also at Rio de Janeiro and Campos dos Goytacazes, RJ, and Argentina (La Plata). Later it was found that this N-BTV causing chlorotic spots, initially referred to as Hibiscus chlorotic spot virus (HCSV) was probably ClCSV (Kitajima et al., 2008). Japanese lantern plants were naturally infected by mite-transmitted ClCSV, resulting in chlorotic spots on their leaves (Figure 8 B). As observed in Clerodendrum x speciosum some few cases of co-infection of HGSV and ClCSV in the same cell were observed (Kitajima et al., 2003a). H. coccineus (scarlet hibiscus), native to southern USA, is a slender, upright shrubby herbaceous ornamental which may reach 2 m tall. Leaves are palmate with jagged teeth along their margins and the flowers have five brilliant, crimson petals about 15 cm in diameter. Some plants growing near ClCSV-infected bleeding heart became naturally infected by mite-transmitted ClCSV, resulting in chlorotic spots on their leaves (Figure 8 C) (Kitajima et al., 2008). Kenaf is a Persian name for this fibrous Hibiscus species. With unbranched stems 2-3 cm wide, the plant may become 2-3 m tall with lobed leaves. The fibers extracted from the stems are used for ropes, textiles, insulation and more recently in paper. Though not used as an ornamental, it produces white, cream yellow or dark trumpet-shaped flowers. Kenaf plants cultivated near ClCSV-infected bleeding heart at Piracicaba, SP, became naturally infected by this mite-transmitted virus, which produced chlorotic spots on the leaves (Figure 8 D). Also conspicuous brown lesions were produced in the stems (Figure 8 E). Experimentally, B. phoenicis transmitted HGSV to kenaf, which produced green spots on senescent leaves (Ferreira et al., 2004b) (Figure 7 F). Chinese hibiscus has been experimentally infected by CiLV-C (Bastianel et al., 2006).
Because ornamental Hibiscus species are usually propagated clonally they tend to accumulate several viruses. In Brazil, Hibiscus chlorotic ringspot virus (HCRSV) and Hibiscus latent ringspot virus (HLRSV), reported abroad (Albouy and Devergne, 1999) are commonly found (Alexandre et al., 2005). Also a nucleorhabdovirus was found together with a phytoplasma in Chinese hibiscus plants with witches× broom symptom (Alexandre et al., 2005), which may be the Eggplant mottled dwarf virus (EMDV) found in hibiscus abroad (Albouy and Devergne, 1999). Additionally an unidentified caulimovirus has been found in hibiscus from Brazil, Panama and Paraguay (E.W. Kitajima, unpublished data).
25. Malvaviscus arboreus Cav. - Turk's hat (malvavisco)
Turk's hat is a sprawling evergreen woody shrub, native to Mexico, 3-4 m tall with dark green oval leaves and pendant, isolated, bell shaped flowers, which remain semi-closed blooming year round. Flowers are usually red, but there are pink varieties. It is frequently used as a living fence. Plants with chlorotic spots on their leaves (Figure 8 F) were found in residential gardens at Piracicaba, SP, associated with B. phoenicis infestation and cytopathic effects of N-BTV in the cells of the lesions (Kitajima et al., 2003a). Later it was found that these lesions were caused by ClCSV (Kitajima et al., 2008). Other cases of ClCSV-infected M. arboreus were registered in Campinas, SP, Rio de Janeiro, RJ, and Brasília, DF. Under experimental conditions B. phoenicis transmitted CiLV-C to M. arboreus resulting in chlorotic lesions (Bastianel et al., 2006) and indicating that Turk's hat may serve as an alternative host for this virus.
Bright, yellow mosaic in Turk's hat is occasionally found in Brazil, caused by a still unidentified begomovirus (E.W. Kitajima and J.A.M. Rezende, unpublished data).
XI. OLEACEAE
26. Ligustrum lucidum W.T. Aiton- tree privet (alfeneiro)
27. L. sinensis Lour. - chinese privet (alfeneiro-da-China)
The genus Ligustrum has 40-50 species of evergreen shrubs and small trees, native to Europe, Asia, Australasia and Africa. They are used for privacy hedging. They have small fragrant flowers, borne in panicles. The tree privet (L. lucidum) is the largest species in the genus and may reach up to 20 m tall. It is used for shade in the streets and also as living fence. A disease similar to citrus leprosis, referred to as "lepra explosiva" and transmitted by Brevipalpus mites was first reported in Concórdia, Argentina in L. sinensis (Vergani, 1942). L. lucidum with ringspots (Figure 8 G) was found in Curitiba, PR, and Piracicaba, SP, exhibiting cytopathic effects of what now we consider C-BTV in the cells of the lesion. The disease was transmitted by B. phoenicis (Kitajima et al., 2003a). L. sinensis is smaller, bushy, with smaller leaves and commonly used as living fences and for bonsai. There is a variegated form widely used in gardens. Chlorotic spots on the leaves of the Chinese privet (Figure 8 H) were found associated with cytopathic effect of C-BTV and infestation by B. phoenicis at Águas de São Pedro and Piracicaba, SP (Nogueira et al., 2004). Similar occurrence was observed at Monte Alegre, SP, and Brasília, DF (E.W. Kitajima and J.C.V. Rodrigues, unpublished data).
No other virus was found naturally infecting Ligustrum in Brazil, but in Europe several soil-borne viruses as nepoviruses, Tobacco rattle virus (TRV), Tomato bushy stunt virus (TBSV), Tobacco necrosis virus (TNV), Tobacco mosaic virus (TMV) and CMV have been described infecting plants of the genus Ligustrum (Albouy and Devergne, 1999).
XII. ORCHIDACEAE
28. Orchids
Orchidaceae is the largest plant family comprising more than 800 genera and 22,000 accepted species. Orchids are found all around the world, mostly in tropical areas of Asia, Africa, Central and South America, though they have been found in sub-Antarctic and sub-Arctic regions. Most orchids are epiphytes but there are lithophytes and terrestrial species. They are perennial herbs lacking woody structure with simple parallel innervated leaves with variable shape and size, and their structure depend on the plant habitat. Terrestrial orchids are provided with rhizomes or form tubers while the epiphytic ones have modified aerial roots. In several epiphytical species the base of the stem is thickened to form a reservoir structure referred to as pseudobulb. Orchid flowers present many structural variations, some producing single flowers while most produce racemose inflorescence. These flowers usually have three sepals, and three petals, one of them is modified and enlarged and called labellum. Some orchids such as Vanilla are economically important as foodstuff flavoring while others are important in perfume industry. But most of the orchids are appreciated for ornamental purpose all around the globe attracting collectors and associations and business resulting in intense international movement.
Orchid fleck is a viral disease resulting in chlorotic spots and ringspots on the leaves (Figure 9 A). It was first described on Cymbidium sp. in Japan and soon after in several parts of the world in different orchid species and genera (Doi et al., 1977; Freitas-Astúa et al., 1999; Kitajima et al., 2001; Kondo et al., 2003). The viral nature was suggested by the first reported case of N-BTV cytopathology and later by the mechanical infection of some herbaceous species (Doi et al., 1977). The virus was named Orchid fleck virus (OFV). Kondo et al. (1995) first purified the virus and produced a specific antiserum and subsequently managed to sequence the viral genome revealing that it is bipartite (ca. 6 kb each) negative sense ssRNA with genomic organization similar to that of rhabdoviruses and they proposed a new genus Dichorhabdovirus in this family to accommodate OFV (Kondo et al., 2006). Primers are also available to detect OFV by RT-PCR (Blanchfield et al., 2001; Kubo et al., 2009a). The vector was identified as B. californicus and the virus/vector relationship is of circulative/propagative type (Kondo et al., 2003). An in situ immunocytochemical study demonsrated that the rodlike particles seen in the tissues represent the OFV, that the nuclear viroplasm contain OFV structural proteins and that the antiserum produced against a Japanese isolate of OFV recognizes OFV isolates from Brazil and Australia (Kitajima et al., 2001). Some genomic variation among OFV isolates was detected using single strand conformation polymorphism technique (Kubo et al., 2009b) confirming previous works by Blanchfield et al. (2001). OFV has been registered in several orchid genera throughout the world: Angraecum (AUS), Aspasia (BR), Baptistonia (AUS), Bifrenaria (BR), Brassia (BR, USA), Bulbophyllm (AUS), Calanthe (JP), Cattleya (AUS, KOR), Coelogyne (BR), Colmanara (JP), Cymbidium (AUS, JP, KOR, USA), Dendrobium (AUS, BR, DEN, GE, JP, KOR), Diplocaulobium (AUS), Dockrillia (AUS) , Encyclia (BR), Flickingeria (AUS), Hormidium (AUS, BR), Liparia (AUS), Masdevallia (AUS), Maxillaria (AUS, BR), Miltonia (AUS, BR, GE), Odontoglossum (AUS, BR, GE, JP), Oncidium (AUS, BR, GE, JP, KOR), Oncidium x Odontoglossum (BR), Paphiopedilum (BR, GE), Pascatorea (JP), Phaius (BR), Phalaenopsis (DEN), Polystachya (AUS), Renanthera (GE), Stanhopea (AUS, GE), Stenia (AUS), Trigonidium (BR), Vanda (GE), Zygopetalum (KOR) (compilation made by Kitajima et al., 2001). After this literature survey was made, new observations made at ESALQ, included some new cases: Eria (AUS), Oncidum (CO, CR), Trichopilia (CR), Xylobium (BR) (Freitas-Astúa et al., 2002; E.W. Kitajima, unpublished data) [Key for country abbreviation-AUS- Australia, BR- Brazil, CO- Colombia, CR- Costa Rica, DEN- Denmark, GE- Germany, JP- Japan, KOR- Korea, USA - United States of America].
During a survey made on viruses occurring in orchids in Brazil, several isolates of OFV were found infecting different orchid genera. However in two of them (Phaius, and Jummelia- Figure 9 B), with symptoms undistinguishable from those of OFV, cytopathology revealed the presence of C-BTV (Freitas-Astúa et al., 1999). Later other cases of the presence of C-BTV on orchids were observed in Brassia, Coelogyne, Hormidium, Xylobium and Arundina, in the state of São Paulo (Kubo et al., 2009a). Another case on Arundina (Figure 9 C) was found in the Amazon Basin (Rodrigues et al., 2008) and Bragança Paulista (E.W. Kitajima and R. Gioria unpublished data), and also in Epidendrum at Primavera do Leste, MT (Figure 9 D) and Piracicaba, SP (E.W. Kitajima, unpublished data). The identity of the C-BTV infecting orchid is not known yet, but certainly is distinct from CiLV-C and SvRSV (Kubo et al., 2009a), since it is not recognized by primers specific to these viruses (Locali et al., 2003; Ferreira et al., 2007). On the other hand, it should be mentioned that a plant of Dendrobium sp. (Figure 9 E) growing next to ClCSV-infected bleeding heart became infected by this virus through mite transmission (Kitajima et al., 2008). This also happened with one Oncidium sp. (Figure 9 F) growing near to SvRSV-infected Solanum violaefolium which was infected with SvRSV. The virus was transmitted by viruliferous B. obovatus (Ferreira et al., 2007). In both cases the symptoms were essentially similar to those caused by OFV. This means that orchids may be susceptible to several BTV, both N or C type, and not only to OFV.
In Brazil several orchid species have been found infected naturally by Cymbidium mosaic virus (CyMV) and Odontoglossum ringspot virus (ORSV). A case of infection of Dendrobium by CMV is also registered (Alexandre et al., 2005). Elsewhere, beside these viruses, several others have been found infecting orchids: 13 potyviruses as well as nepovirus, nucleorhabdovirus, tombusvirus and tospovirus (Albouy and Devergne, 1999; Gibbs et al., 2000).
XIII. PITTOSPORACEAE
29. Pittosporum tobira [Dryand.] - Japanese pittosporum/mock orange (pitósporo japonês)
Japanese pittosporum is a broad-leafed, aromatic, evergreen shrub, 2-3 m tall originally from China. Leaves are thick and concentrated at the end of the branches. There is a variegated form with yellowish border on the leaves. When blooming, the small flowers are held at the branch tips. They are commonly used for screens and hedges. P. tobira with chlorotic ringspots on the leaves (Figure 10 A) were found in Florida, USA, associated with infestation by Brevipalpus mites. Electron microscopy revealed cell alterations of the N-BTV, but the presumed virus could not be identified (Kitajima et al., 2003a).
An isolate of EMDV, a nucleorhabdovirus, was found causing vein clearing in Pittosporum in Europe and Africa, and also tospoviruses and CMV were described in this ornamental (Albouy and Devergne, 1999).
XIV. PLUMBAGINACEAE
30. Plumbago auriculata Lam. - Leadwort (Bela Emília)
Leadwort is a semi woody, erect, evergreen shrub, intensely branched, native from South Africa with intense blooming. It can be grown as isolated plants or as a vine, forming living fences. Leaves are oblong and tender. Sky blue tubular flowers are borne in rounded terminal clusters and bloom during the entire year. In a residential garden at Atibaia, SP, leaves of leadwort showing chlorotic spots with necrotic centers (Figure 10 B) were observed associated with infestation by Brevipalpus mites and the tissues of the lesion had cytopathic effect of the C-BTV (Freitas-Astúa et al., 2004). The presumed virus remains still unidentified.
XV. PRIMULACEAE
31. Lysimachia congestiflora Hemsl.- creeping Jenny (lisimaquia)
Creeping Jenny is a mat forming perennial, 5-7 cm tall, native to China. It has opposed dark green, elliptical leaves and terminal or axilar inflorescences with many cup-shaped yellow flowers. L. congestiflora plants with chlorotic spots on the leaves (Figure 10 C) were found in a residential garden at Águas de São Pedro, SP, associated with infestation of Brevipalpus mites. Cells of the lesions showed cytopathic effect of C-BTV (Nogueira and Rossi, 2005). The presumed virus was not identified.
There is a report of infection of Lysimachia by CMV elsewhere (Albouy and Devergne, 1999).
XVI. RUBIACEAE
32. Gardenia jasminoides Ellis - gardenia (gardenia)
Gardenia is a fragrant flowering evergreen tropical, semi woody bushy plant, native to China and cultivated worldwide. It may become 1 to 1.5 m tall with dark green, glossy leaves. Flowers are small and white, extremely fragrant becoming yellowish as they age. It may be planted isolated or in rows. During a survey for BTV in the Amazon basin at Urucu, AM, an isolated base for oil prospection, gardenia plants growing in a residential garden were found with chlorotic spots (Figure 10 D) associated with Brevipalpus mite infestation. Cells from the lesions exhibited alterations of the N-BTV (Rodrigues et al., 2008). The virus could not be identified. Gardenia is also susceptible to CMV (Albouy and Devergne, 1999).
33. Mussaenda erythrophylla Schumach. & Thonn. - ashanti blood (mussaenda vermelha)
This is a semi-deciduous rambler with multiple stems that may grow 3 m or more, native to East and Central Africa. Opposite leaves are round to ovate, pubescent at underside and strongly veined. M. erythrophylla produces dense inflorescence with several flowers borne in branching terminal panicles. Flowers are small with creamy funnel shaped corolla and a red felt center. Ashanti blood may be cultivated isolated or in rows in parks and gardens. A few plants in a small nursery at Manaus, AM, were found with diffuse chlorotic spots on the leaves (Figure 10 E) associated with Brevipalpus mite infestation. Electron microscopy of the tissues from the lesions revealed cytopathic effect of the N-BTV (Rodrigues et al., 2008). The presumptive BTV could not be identified.
XVII. SOLANACEAE
34. Brunfelsia uniflora D. Don. Yesterday today - tomorrow (manacá)
35. B. pauciflora Benth -Yesterday today - tomorrow (jasmim paraguayo)
Brunfelsia is a genus with about 40 species of neotropical shrubs and small trees. Both B. uniflora and B. pauciflora are very similar, native to Brazil. They are woody shrubs with several stems which may reach 1-3 m tall, with oval, smooth and dark green leaves. While B. uniflora produces solitary flowers at the tip of the stems, in B. pauciflora they are produced in clusters. Flowers are fragrant and produced in the spring and summer. They open purple, then turn pale lavender and finally white. They are cultivated isolated or in clusters, forming hedges. B. uniflora plants with green spots and ringspot on senescent leaves (Figure 10 F) were found in a residential garden at Águas de São Pedro (Nogueira et al., 2003) associated with Brevipalpus mite infestation and cytopathic effect of C-BTV in the lesions. Later similar symptoms were found in several plants growing in parks and residential gardens at Piracicaba, Araras and Atibaia, SP. RT-PCR using primers for CiLV-C, SvRSV and PFGSV did not result in amplification of the viral genome, suggesting that the virus causing the green spots on B. uniflora leaves is different from these viruses. In a survey carried out in several flower nurseries at La Plata, Argentina, B. pauciflora plants were found with chlorotic spots (Figure 10 G) associated with Brevipalpus mite infestation. The lesion cells exhibited alterations of N-BTV (Dal Bo et al., 2007). These viruses are still unidentified.
36. Cestrum nocturnum L. - night jasmine (dama-da-noite)
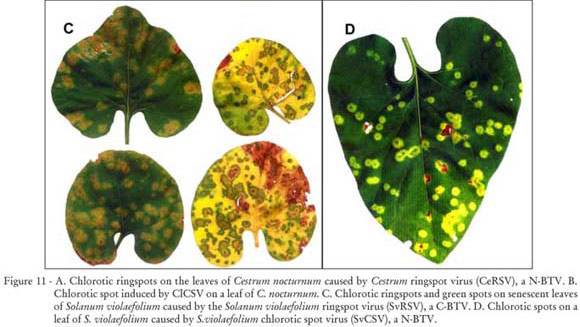
Night jasmine is a semi woody, evergreen shrub native to the Antilles, 1.5-3 m tall with narrow, lanceolate, bright green, glossy, coriaceous leaves. Flowers are small, tubular, strongly scented during the night, cream yellowish produced in numerous cymose inflorescences. Plants are usually cultivated isolated. Night jasmine plants showing chlorotic ringspots (Figure 11 A) on their leaves were found in a residential garden at Atibaia, SP, associated with infestation by B. obovatus (Kitajima et al., 2003a; Guidotti et al., 2006). Mites collected from affected plants reproduced the symptoms on healthy one indicating that the symptoms were due to a BTV, which was named Cestrum ringspot virus (CeRSV). This virus was also experimentally transmitted by B. phoenicis (J. Freitas-Astúa, unpublished data). Cytopathological observations on the tissues of the lesion revealed a pattern of N-BTV. However, instead the typical N-BTV, presumed virions were found within endoplasmic reticulum and nuclear viroplasma was rarely seen (Kitajima et al., 2003a). On the other hand, a plant of C. nocturnum growing near bleeding heart infected by ClCSV presenting chlorotic spots on their leaves (Figure 11 B) was found at Piracicaba, SP. Further serological and molecular studies indicated that this plant was naturally infected by ClCSV (Kitajima et al., 2008).
37. Solanum violaefolium (Schott.) (=Solanum asarifolium Kunth. & Bouch., Lycianthes asarifolia Bitter) - ginger leaf (solano violeta)
Ginger leaf has a prostrate, trailing growth habit with stolons that root and produce dark green, cordiform leaves at every node. Small, white flowers are formed between the leaves during the entire year. It may reach 10-15 cm tall. Stolons produce a mat near the surface of the soil and do not appear to maintain structures that become buried. This plant prefers wet and shady areas. S. violaefolium plants showing conspicuous chlorotic ringspots on the leaves (Figure 11 C) were found in several parks and gardens at Piracicaba, SP. When leaves become senescent, the affected area remained green. These lesions were associated with B. obovatus or B. phoenicis infestation. Ultrastructural studies on the tissues of the lesion revealed cytopathic effect of C-BTV (Figure 2 B). The causal agent was transmitted experimentally from ginger leaf to ginger leaf by these two mite species, and also to some other plant species as Datura stramonium, Catharanthus roseus and several Nicotiana species. Part of the viral genome was sequenced and specific primers were designed for the molecular detection of the virus, which was named Solanum violaefolium ringspot virus (SvRSV) (Ferreira et al., 2007). The genome of SvRSV, on the other hand, could not be amplified by primers which amplify CiLV-C and PFGSV, two other C-BTV (Antonioli-Luizon et al., 2009). Virions of SvRSV though similar to that of other C-BTV, frequently appear longer in sections. Another BTV was found in ginger leaf growing in the gardens at the Instituto Agronômico de Campinas, SP. These plants were infested by B. phoenicis and showed chlorotic spots on the leaves (Figure 11 D), and TEM revealed cytopathic effects of the N-BTV type similar to that found in night jasmine (Freitas-Astúa et al., 2002b) and the São Paulo isolate of CoRSV (Kitajima and Chagas, 2009) with presumed virions within the endoplasmic reticulum (Figure 2 E). Mites collected from infected plants reproduced the symptoms when transferred to healthy plants. The virus was named Solanum violaefolium chlorotic spot virus (SvCSV) (Kitajima et al., 2004).
CiLV-C was transmitted experimentally by B. phoenicis from sweet orange (Citrus sinensis Osbeck) to S. violaefolium and the mite was able to transmit the virus between S. violaefolium plants but not back to citrus (Rodrigues et al., 2005). Electron microscopy and RT-PCR with CiLV-C confirmed that the same virus was responsible for the infections. However, the dsRNA profile from infected S. violaefolium and sweet orange were different, indicating possible changes in the viral genome.
An isolate of Eggplant mosaic virus (EMV) was found at Piracicaba, SP, causing mosaic symptoms on S. violaefolium leaves (E.W. Kitajima, unpublished data). Also, an isolate of TRV was found co-infecting S. violaefolium plants with SvRSV inducing a larger chlorotic lesions (Alexandre et al., 2005).
XVIII. VIOLACEAE
38. Viola odorata L. - Sweet violet (violeta)
Besides orchids, this is the only case of the occurrence of BTV outside the American continent. Violet is an evergreen, perennial, herbaceous, woodland plant native to Europe, Africa and Asia. Plants have cordiform leaves and spreads with stolons. Dark blue flowers have long pedunculus and sweet scent. It makes excellent weed-excluding ground cover. In Nambour, Queensland, Australia, Gowanlock and Dietzegen (1995) reported the occurrence of chlorotic spots on the leaves of violet and TEM revealed cytopathic effects of N-BTV. It is likely that the causal agent may be OFV, since specific primers for this virus resulted in amplification of cDNA by RT-PCR (A. Gibbs, personal communication). There is no information regarding infestation by Brevipalpus mites.
Violet is also susceptible to other viruses as Viola mottle virus (VMoV) (Albouy and Devergne, 1999).
Acknowledgements
This work received financial support from FAPESP (20007/50809-0 and 2008/52691-9) and CNPq (47.1552/2007-0). The authors thanks to the criticisms and suggestions made by the two anonymous referees, which helped to improve considerably this text and Dr. Neusa de Lima Nogueira, for providing images of localized symptoms in some of the described ornamentals.
Received May 13, 2009
Accepted February 26, 2010
- Albouy, J.; Devergne, J.C. 1999. Diseases produced by viruses on ornamental plants. Ediciones Mundi-Prensa, Madrid, Spain. 480 p. (in Spanish).
- Alexandre, M.A.V.; Rivas, E.B.; Tozetto, A.R.P.; Duarte, L.M.L. 2005. An annotated list on the natural occurrence of viruses in ornamental plants in Brazil. Instituto Biológico, São Paulo, SP, Brazil. 54 p. (in Portuguese).
- Antonioli-Luizon, R.; Freitas-Astúa, J.; Locali-Fabris, E.C.; Machado, M.A.; Kitajima, E.W. 2009. Detection of the passion fruit green spot virus (PFGSV) by RT-PCR. Abstracts Brazilian Congress of Genetics. Sociedade Brasileira de Genética, Salvador, BA, Brazil, CDRom (in Portuguese).
- Bastianel, M.; Freitas-Astúa, J.; Kitajima, E.W.; Machado, M.A. 2006. The citrus leprosis pathosystem. Summa Phytopathologica 32: 211-220.
- Bitancourt, A.A. 1938. Ringspot, a new disease of the coffee. O Biológico 4: 404-405 (in Portuguese).
- Blanchfield, A.L.; Mackenzie, A.M.; Gibbs, A.; Kondo, H.; Tamada, T.; Wilson, C.R. 2001. Identification of orchid fleck virus by reverse transcriptase polymerase chain reaction and analysis of isolate relationships. Journal of Phytopathology 149: 713-718.
- Boari, A.J.; Freitas-Astúa, J.; Ferreira, P.T.O.; Neder, D.G.; Nogueira, N.L.; Rossi, M.L.; Kitajima, E.W. 2004. Purification and serology of the coffee ringspot virus. Summa Phytopathologica 30: 453-458.
- Chagas, C.M. 1973. Association of the mite Brevipalpus phoenicis (Geijskes) with the coffee ringspot. O Biológico 39: 229-232 (in Portuguese).
- Chagas, C.M.; July, J.R.; Alba, A.P.C. 1961. Mechanical transmission and structural features of coffee ringspot virus. Phytopathologische Zeitschrift 102: 100-106.
- Childers, C.C.; French, J.V.; Rodrigues, J.C.V. 2003a. Brevipalpus californicus, B. obovatus, B. phoenicis, and B. lewisi (Acari: Tenuipalpidae): a review of their biology, feeding injury and economic importance. Experimental and Applied Acarology 30:5-28.
- Childers, C.C.; Rodrigues, J.C.V.; Welbourn, W.C. 2003b. Host plants of Brevipalpus californicus, B. obovatus and B. phoenicis (Acari: Tenuipalpidae) and their potential involvement in the spread of viral diseases vectored by these mites. Experimental and Applied Acarology 30: 29-105.
- Childers, C.C.; Rodrigues, J.C.V.; Derrick, K.S.; Achor. D.S.; French, J.V.; Welbourn, W.C.; Ochoa, R.; Kitajima, E.W. 2003c. Citrus leprosis and its status in Florida and Texas: past and present. Experimental and Applied Acarology 30: 181-202.
- Colariccio, A.; Lovisolo, O.; Chagas, C.M.; Galletti, S.R.; Rossetti, V.; Kitajima, E.W. 1995. Mechanical transmission and ultrastructural aspects of citrus leprosis disease. Fitopatologia Brasileira 20: 208-213.
- Dal Bo, E.; Peña, E.; Fernandez, R.; Kubo, K.; Freitas-Astúa, J.; Bedendo, I.P.; Kitajima, E.W. 2007. Preliminary survey of virus diseases in ornamentals at La Plata, Argentina. Fitopatologia Brasileira 32 (supplement): S135, 2007 (in Spanish).
- Doi, Y.; Chang, M.U.; Yora, K. 1977. Orchid fleck virus. Description of Plant Viruses, No. 183. Comonwealth Mycological Institute/Association of Applied Biology, Wellesbourne, UK, 4 p.
- Fawcett, H.S. 1911. Scaly bark or nail-head rust of citrus. Bulletin No. 106, Florida Agriculture Experimental Station. Gainesville, FL, US, 41p.
- Ferreira, P.T.O.; Kubo, K.S.; Kitajima, E.W. 2004a. Ringspots in Anthurium sp. and Cordyline terminalis associated with cytoplasmic type of Brevipalpus-borne viruses. Virus Review and Research 9 (supplement): 249, 2004a.
- Ferreira, P.T.O.; Buso Jr.; A.A.; Freitas-Astúa, J.; Kitajima, E.W. 2004b. Kenaf (Hibiscus cannabis): experimental host for the Hibiscus green spot and Hibiscus chlorotic spot viruses. Summa Phytopathologica 30: 68-69 (In Portuguese).
- Ferreira, P.T.O.; Locali-Fabris, E.C.; Freitas-Astúa, J.; Antonioli-Luizon, R.; Gomes, R.T.; Machado, M.A.; Kitajima, E.W. 2007. Characterization of a bacilliform virus isolated from Solanum violaefolium transmitted by the tenuipalpid mites Brevipalpus phoenicis and B. obovatus Summa Phytopathologica 33: 264-269 (in Portuguese, with abstract in English).
- Freitas-Astúa, J.; Rezende, J.A.M.; Kitajima, E.W. 1999. Incidence of orchid viruses in the state of São Paulo, Brazil. Fitopatologia Brasileira 24: 125-130.
- Freitas-Astúa, J.; Moreira, L.; Rivera, C.; Rodriguez C.M.; Kitajima, E.W. 2002. First report of orchid fleck vírus in Costa Rica. Plant Disease 86: 1402.
- Freitas-Astúa, J.; Kitajima, E.W.; Astúa-Monge, G.; Locali, E.C.; Machado, M.A. 2004. Leadwort (Plumbago auriculata): a new host for a bacilliform, rhabdoviruslike virus. Summa Phytopathologica 30: 80 (in Portuguese).
- Frezzi, M.S. 1940. The citrus leprosis. Researches conducted at the laboratory of pathology of Bella Vista (Corrientes). Boletín Frutas y Hortalizas. 5. Ministerio de la Agricultura de la Nación, Buenos Aires, Argentina. 16p (in Spanish).
- Gibbs, A.; Mackenzie, A.; Blanchfield, A.; Cross, P.; Wilson, C.; Kitajima, E.W., Nightingale, M.; Clemens, M. 2000. Viruses of orchids in Australia: their identification, biology and control. Orchid Review 65 (3); 10-21.
- Gowanlock, D.H.; Dietzegen, R.G. 1995. Small rhabdovirus-like particles in violet (Viola spp.). Australasian Plant Pathology 24: 215-216.
- Groot, TVM. 2006. The effect of symbiont induced haploid thelytoky on the evolution of Brevipalpus mites. PhD dissertation, Institute of Biodiversity and Ecosystem Dynamics. University of Amsterdam, Amsterdam, the Netherlands, 154p.
- Guidotti, A.R.; Rodrigues, V.; Machado, M.A.; Freitas-Astúa, J. 2006. Transmission of the Cestrum ringspot virus by different active phases of the mite Brevipalpus obovatus. Summa Phytopathologica 32 (supplement): S 75 (in Portuguese).
- Hume, H.H. 1901. Some fungus diseases of citrus and others fruits. Proceedings Florida State Horticulture Society 14: 6470.
- Kitajima, E.W.; Chagas, C.M. 2009. Viral diseases in coffee. p. 550-556. In Wintgens, J.N. ed. Coffee: growing, processing, sustainable production. 2nd ed, Wiley-VCH, Weinheim, Germany. 976p.
- Kitajima, E.W.; Costa, A.S. 1972. Bacilliform particles associated with the coffee ringspot. Ciência e Cultura 24: 542-545 (in Portuguese, with abstract in English).
- Kitajima,E.W.; Kondo, H.; Mackenzie, A.; Rezende, J.A.M.; Gioria, R.; Gibbs, A.; Tamada, T. 2001. Comparative cytopathology and immunocytochemistry of Japanese, Australian and Brazilian isolates of Orchid fleck virus. Journal of General Plant Pathology 67: 231-237.
- Kitajima, E.W.; Chagas, C.M.; Rodrigues, J.C.V. 2003a. Brevipalpus-transmitted plant vírus and virus-like diseases: cytopathology and some recent cases. Experimental and Applied Acarology 30: 135-160.
- Kitajima, E.W.; Rezende, J.A.M.; Rodrigues, J.C.V. 2003b. Passion fruit green spot virus vectored by Brevipalpus phoenicis (Acari: Tenuipalpidae). Experimental and Applied Acarology 30: 225-231.
- Kitajima, E.W.; Kubo, K.S.; Gomes, R.T.; Ferreira, P.T.O. 2004. Chlorotic spots on Solanum violaefolium, transmissible by Brevipalpus mite, associated with a nuclear type of virus. Fitopatologia Brasileira 29 (supplement): S65 (in Portuguese).
- Kitajima, E.W.; Boari, A.J.; Salaroli, R.B. 2006. Green spots on Beaumontia leaves associated with a cytoplasmic type of Brevipalpus transmitted virus. Summa Phytopathologica 32 (supplement): S 11 (in Portuguese).
- Kitajima, E.W.; Groot, T.V.M.; Freitas-Astúa, J.; Alberti, G.; Morais, G.J. 2007. In situ detecion of the Cardinium symbionts of Brevipalpus (Acari: Tenuipalpidae) by electron microscopy. Experimental and Applied Acarology 43: 263-271.
- Kitajima, E.W.; Kubo, K.S.; Ferreira, P.T.O.; Alcântara, B.K.; Boari, A.J.; Gomes, R.T.; Freitas-Astúa, J.; Rezende, J.A.M.; Morais, G.J.; Salaroli R.B. 2008. Chlorotic spots on Clerodendrum, a disease caused by a nuclear type of Brevipalpus (Acari: Tenuipalpidae) transmitted virus. Scientia Agricola 65: 36-49.
- Kitajima, E.W.; Calegario, R.F.; Freitas-Astúa, J.; Alberti, G. 2009. Electron microscopic evidences that Citrus leprosis virus, cytoplamisc type (CiLV-C) only circulate in the vector Brevipalpus phoenicis (Acari: Tenuipalpid), while other nuclear type Brevipalpus transmitted viruses do replicate. Abstracts from Milbenkundliches Klloquium in Posen. 7. Uniwersytet im Adama Mickiewicza. Posnan, Polland, p. 28-29.
- Knorr, L.C. 1968. On the etiology of leprosis in Citrus. Proceedins from Conference International Organization of Citrus Virologists; 4. Rome, Italy, 1966. University of Florida Press, Gainesville, FL, USA, p.332-341.
- Kondo, H.; Matsumoto, J.; Maeda, T.; Inouye, N. 1995. Host range and some properties of orchid fleck virus isolated from oriental Cymbidium in Japan. Bulletin Research Institute of Bioresource, Okayama University 2: 151-161 (in Japanese, with abstract in English).
- Kondo, H.; Maeda, T.; Tamada, T. 2003. Orchid fleck virus: Brevipalpus californicus mite transmission, biological properties and genome structure. Experimental and Applied Acarology 30: 215-223.
- Kondo, H.; Maeda, T.; Shirako, Y.; Tamada, T. 2006. Orchid fleck virus is a rhabdovirus with an unusual bipartite genome Journal of General Virology 87: 2413-2421.
- Kubo, K.S.; Antonioli-Luizon, R.; Freitas-Astúa, J.; Kitajima, E.W. 2007. Molecular diagnosis of the Clerodendron chlorotic spot virus (ClCSV). Summa Phytopathologica 33 (supplement): S12 (in Portuguese).
- Kubo, K.S.; Freitas-Astúa, J.; Machado, M.A.; Ktajima, E.W. 2009a. Orchid fleck symptoms may be caused naturally by two different Brevipalpus transmitted viruses. Journal of General Plant Pathology 75: 250-256.
- Kubo, K.S.; Stuart, R.M.; Freitas-Astua, J.; Antonioli-Luizon, R.; Locali-Fabris, E.C.; Coletta F.,H.D; Machado, M.A.; Kitajima, E.W. 2009b. Evaluation of the genetic variability of orchid fleck virus by single-strand conformational polymorphism analysis and nucleotide sequencing of a fragment from the nucleocapsid gene. Archives of Virology 154: 1009-1014.
- Locali, E.C.; Freitas-Astúa, J.; Souza, A.A.; Takita, M.A.; Astúa-Monge, G.; Antonioli-Luizon, R.; Kitajima, E.W.; Machado, M.A. 2003. Development of a molecular tool for the diagnosis of leprosis, a major threat to citrus production in the Americas. Plant Disease 87: 1317-1321.
- Locali, E.C.; Freits-Astúa, J.; Antonioli-Lauizon, R.; Boari, A.J.; Machado, M.A. 2005. Diagnosis of the coffee ringspot by RT-PCR. Fitopatolologia Brasileira 30 (supplement): S 185 (in Portuguese).
- Locali-Fabris, E.C.; Freitas-Astúa, J.; Souza, A.A.; Takita, M.A.; Astúa-Monge, G.; Antonioli-Luizon, A.; Rodrigues, V.; Targon, M.L.P.N.; Machado, M.A. 2006. Complete nucleotide sequence, genomic organization and phylogenetic analysis of Citrus leprosis virus cytoplasmic type. Journal of General Virology 87: 2721-2729.
- Lorenzi, H.; Souza, H.M. 2001. Ornamental plants in Brazil. Woody, herbaceous and climbing. 3 ed. Instituto Plantarum, Nova Odessa, SP, Brazil. 1088 p. (in Portuguese).
- Marques, J.P.R.; Freitas-Astúa, J.; Kitajima, E.W.; Appezzato-da-Glória, B. 2007. Leaf and stem lesions of sweet orange plants caused by the Citrus leprosis vírus. Pesquisa Agropecuária Brasileira 42: 1531-1536 (in Portuguese, with English abstract).
- Musumeci, M.R.; Rossetti, V.V. 1963. Transmission of citrus leprosis symptoms by the mite Brevipalpus phoenicis Ciência e Cultura 15: 228 (in Portuguese).
- Mesa, N.C.; Ochoa, R.; Welbourn, W.C.; Evans, G.A.; Moraes, G.J. 2009. A catalog of the tenuipalpid (Acari) of the world with a key to genera. Zootaxa 2008: 1-185.
- Nogueira, N.L.; Rossi, M.L. 2005. Rhabdovirus-like bacilliform particles in the leaves of creeping Jenny (Lysimaquia congestifolia Hemsi, Primulaceae) associated to typical lesions caused by the mite Brevipalpus Fitopatologia Brasileira 30 (supplement): S 189 (in Portuguese).
- Nogueira, N.L.; Rodrigues, J.C.V.; Rossi, M.L. 2003. Rhabdovirus-like particles in three ornamental species showing local lesions and the presence of Brevipalpus mites. Summa Phytopathologica 29: 278-282 (in Portuguese, with abstract in English).
- Nogueira, N.L.; Rodrigues, J.C.V.; Rossi, M.L. 2004. Dracaena marginata Lam and Ligustrum sinensis Loureiro, two new host plants for Brevipalpus borne vírus. Fitopatologia Brasileira 29: S234 (In Portuguese).
- Novelli, V.M.; Kubo, K.S.; Freitas-Astúa, J.; Machado, M.A., Salaroli, R.B.; Kitajima, E.W. 2008. Natural infection of peace lily (Spatiphyllum wallisii) by a nuclear type of Brevipalpus-transmitted virus. Abstracts Simposio Latinamericano y del Caribe "La Biodiversidad acarina: utilización, protección y conservación", 2, Centro Nacional de Sanidad Agropecuaria, Havana, Cuba, 2008, CDRom, Resumen BA-P14.
- Pascon, R.C.; Kitajima, J.P.; Breton, M.C.; Assumpção, L., Greggio, C., Zanca, A.S.; Okura, V.K.; Alegria, M.C.; Camargo, M.E.; Silva, G.G.C.; Cardozo, J.C.; Vallin, M.A.; Franco, S.F.; Silva, V.H., Jordão Jr., H.; Oliveira, F.; Giachetto, P.F.; Ferrri, F.; Aguillar-Vildoso, C.I.; Francischini, F.J.B.; Silva, J.M.F.; Arruda, P.; Ferro, J.A.; Reinach, F.; Silva A.C.R. 2006. The complete nucleotide sequence and genomic organization of Citrus leprosis associated vírus, cytoplasmic type (CiLV-C). Vírus Genes 32: 289-298.
- Pijnacker, L.P.; Ferwerda, M.A.; Helle, W. 1981. Cytological investigations on the female and male reproductive system of the parthenogetetic privet mite, Brevipalpus obovatus (Donnadieu) (Phytoptipalpidae, Acari). Acarologia 22: 157-163.
- Rodrigues, J.C.V.; Kitajima, E.W.; Childers, C.C.; Chagas, C.M. 2003. Citrus leprosies virus vectored by Brevipalpus phoenicis (Acari: Tenuipalpidae) on citrus in Brazil. Experimental and Applied Acarology 30: 161-179.
- Rodrigues, J.C.V.; Gallo-Meagher, M.; Ochoa, R.; Childers, C.C.; Adams, B.J. 2004. Mitochondrial DNA and RAPD polymorphisms in the haploid mite Brevipalpus phoenicis (Acari: Tenuipalpidae). Experimental and Applied Acarology 34: 275290.
- Rodrigues, J.C.V.; Locali, E.C.; Freitas-Astúa, J.; Kitajima, E.W. 2005. Transmissibility of citrus leprosis virus by Brevipalpus phoenicis in Solanum violaefolium Plant Disease 89: 911.
- Rodrigues, J.C.V.; Antony, L.M.K.; Salaroli, R.B.; Kitajima, E.W. 2008. Brevipalpus-associated viruses in the central Amazon basin. Tropical Plant Pathology 33: 12-19.
- Steane, D.A.; De Kok, R.P.J.; Olmstead, R.G. 2004. Phylogenetic relationships between Clerodendrum (Lamiaceae) and other Ajugoid genera inferred from nuclear and chloroplast DNA sequence data. Molecular Phylogenetics and Evolution 32: 39-45.
- Vergani, A.R. 1942. The transmission of the "lepra explosiva" of the tree privet by mites. Revista Agricultura y Agronomia 9: 292-294 (in Spanish).
- Weeks, A.R.; Marec, F.; Breeuwer, J.A.J. 2001. A mite species that consists entirely of haploid females. Science 292: 2479-2483.
- Welbourn, W.C.; Ochoa, R.; Kane, E.C.; Erbe, E.F. 2003. Morphological observations on Brevipalpus phoenicis (Acari: Tenuipalpidae) including comparisons with B. californicus and B. obovatus. Experimental and Applied Acarology 30: 107-133.
Publication Dates
-
Publication in this collection
17 June 2010 -
Date of issue
2010
History
-
Received
13 May 2009 -
Accepted
26 Feb 2010