Abstracts
Salinity is a major limiting factor to crop productivity in the world especially in semiarid regions. The aim of this study was to evaluate the photosynthetic resistance of Jatropha curcas (L.) young plants subjected to salt stress. The experiment was carried out in a completely randomized design with treatments in a 2 x 3 factorial: two NaCl levels (0 and 100 mmol L-1) and three harvest times: 7 and 14 days of salt exposure and three days of recovery. Leaf Na+ and Cl-concentrations and the K+/Na+ ratios, after seven days of salt exposure, did not reach ionic toxic levels, suggesting that the NaCl-induced osmotic effects prevailed over the ionic ones. Under this condition, the salt stress caused reduction in leaf gas exchange parameters, such as CO2 fixation, stomatal conductance and transpiration. In contrast, salt stress did not change the photochemical efficiency of photossystem II. Conversely, after 14 days of treatment, Na+ and Clions reached very high concentrations, up to toxic levels in leaves. Under such conditions, both leaf gas exchange and photochemistry suffered strong impairment probably caused by ionic toxicity. The recovery treatment for 3d did not significantly decrease the leaf salt concentrations and no improvement was observed in the photosynthetic performance. Physic nut young plants are sensitive to high NaCl-salinity conditions, with high leaf Na+ and Cl- concentrations, low K+/Na+ ratio and great photosynthetic damages due to stomatal and biochemical limitations.
Jatropha curcas; chlorophyll fluorescence; gas exchange; ionic toxicity; salinity
A salinidade é um dos principais fatores que limitam a produtividade das culturas no mundo principalmente em regiões semiáridas. Avaliou-se a resistência da fotossíntese de plantas jovens de pinhãomanso (Jatropha curcas L.) submetidas ao estresse salino. O experimento foi realizado em delineamento inteiramente casualizado com tratamentos em fatorial 2 x 3: duas concentrações de NaCl (0 e 100 mmol L-1) e três tempos de avaliação (7 e 14 dias de exposição e três dias de recuperação). As concentrações de Na+ e Cl- e a relação K+/Na+ nas folhas, após sete dias de exposição ao sal, não indicaram níveis tóxicos, sugerindo os efeitos osmóticos induzidos pelo NaCl prevaleceram sobre as causas iônicas. Sob essas condições, o estresse salino causou redução nos parâmetros de trocas gasosas, como fixação de CO2, condutância estomática e transpiração, mas ao contrário, não alterou a eficiência fotoquímica do fotossistema II. Após 14 dias de tratamento, os íons salinos atingiram concentrações muito elevadas nas folhas, provavelmente atingindo níveis tóxicos. Em tais condições, as trocas gasosas e a atividade fotoquímica sofreram forte redução causada pelo estresse iônico. O tratamento de recuperação não induziu queda intensa nas concentrações dos íons salinos nas folhas e nenhuma melhoria foi observada no desempenho fotossintético. Plantas jovens de pinhão manso são sensíveis a condições de salinidade elevada por NaCl, mostrando altas concentrações de Na+ e Cl-, baixa razão K+/Na+ e danos fotossintéticos intensos causados tanto por limitações estomáticas como por limitações bioquímicas.
Jatropha curcas; fluorescência da clorofila; trocas gasosas; toxicidade iônica; salinidade
PLANT PHYSIOLOGY AND BIOCHEMISTRY
Salt stress induced damages on the photosynthesis of physic nut young plants
Danos causados por estresse salino sobre a fotossíntese de plantas jovens de pinhão-manso
Evandro Nascimento da SilvaI; Rafael Vasconcelos RibeiroII; Sérgio Luiz Ferreira-SilvaI; Ricardo Almeida ViégasIII; Joaquim Albenisio Gomes SilveiraI,* * Corresponding author < silveira@ufc.br>
IUFC - Depto. de Bioquímica e Biologia Molecular, C.P. 6020 - 60451-970 - Fortaleza - CE, Brasil
IIIAC/Centro de Pesquisa e Desenvolvimento em Ecofisiologia e Biofísica - Setor de Fisiologia Vegetal, C.P. 28 - 13012-970 - Campinas - SP, Brasil
IIIUFPB - Depto. de Engenharia Florestal, C.P. 64 - 58700-970 - Patos - PB, Brasil
ABSTRACT
Salinity is a major limiting factor to crop productivity in the world especially in semiarid regions. The aim of this study was to evaluate the photosynthetic resistance of Jatropha curcas (L.) young plants subjected to salt stress. The experiment was carried out in a completely randomized design with treatments in a 2 × 3 factorial: two NaCl levels (0 and 100 mmol L-1) and three harvest times: 7 and 14 days of salt exposure and three days of recovery. Leaf Na+ and Cl-concentrations and the K+/Na+ ratios, after seven days of salt exposure, did not reach ionic toxic levels, suggesting that the NaCl-induced osmotic effects prevailed over the ionic ones. Under this condition, the salt stress caused reduction in leaf gas exchange parameters, such as CO2 fixation, stomatal conductance and transpiration. In contrast, salt stress did not change the photochemical efficiency of photossystem II. Conversely, after 14 days of treatment, Na+ and Clions reached very high concentrations, up to toxic levels in leaves. Under such conditions, both leaf gas exchange and photochemistry suffered strong impairment probably caused by ionic toxicity. The recovery treatment for 3d did not significantly decrease the leaf salt concentrations and no improvement was observed in the photosynthetic performance. Physic nut young plants are sensitive to high NaCl-salinity conditions, with high leaf Na+ and Cl- concentrations, low K+/Na+ ratio and great photosynthetic damages due to stomatal and biochemical limitations.
Key words:Jatropha curcas, chlorophyll fluorescence, gas exchange, ionic toxicity, salinity
RESUMO
A salinidade é um dos principais fatores que limitam a produtividade das culturas no mundo principalmente em regiões semiáridas. Avaliou-se a resistência da fotossíntese de plantas jovens de pinhãomanso (Jatropha curcas L.) submetidas ao estresse salino. O experimento foi realizado em delineamento inteiramente casualizado com tratamentos em fatorial 2 × 3: duas concentrações de NaCl (0 e 100 mmol L-1) e três tempos de avaliação (7 e 14 dias de exposição e três dias de recuperação). As concentrações de Na+ e Cl- e a relação K+/Na+ nas folhas, após sete dias de exposição ao sal, não indicaram níveis tóxicos, sugerindo os efeitos osmóticos induzidos pelo NaCl prevaleceram sobre as causas iônicas. Sob essas condições, o estresse salino causou redução nos parâmetros de trocas gasosas, como fixação de CO2, condutância estomática e transpiração, mas ao contrário, não alterou a eficiência fotoquímica do fotossistema II. Após 14 dias de tratamento, os íons salinos atingiram concentrações muito elevadas nas folhas, provavelmente atingindo níveis tóxicos. Em tais condições, as trocas gasosas e a atividade fotoquímica sofreram forte redução causada pelo estresse iônico. O tratamento de recuperação não induziu queda intensa nas concentrações dos íons salinos nas folhas e nenhuma melhoria foi observada no desempenho fotossintético. Plantas jovens de pinhão manso são sensíveis a condições de salinidade elevada por NaCl, mostrando altas concentrações de Na+ e Cl-, baixa razão K+/Na+ e danos fotossintéticos intensos causados tanto por limitações estomáticas como por limitações bioquímicas.
Palavras-chave:Jatropha curcas, fluorescência da clorofila, trocas gasosas, toxicidade iônica, salinidade
Introduction
Salinity adversely affects plant growth and development, with nearly 20% of the world cultivated area and about half of the world irrigated lands being affected by salt stress (Sairam and Tyagi, 2004). This problem is more relevant in semiarid regions with low rainfall and high evaporative demand, which strongly contribute to increase soil salinization (Viégas et al., 2001). In these regions, the problem of soil secondary salinization is exacerbated by the use of low quality water associated with inadequate techniques of soil management (Ferreira-Silva et al., 2009).
The accumulation of salt ions in plants can cause osmotic stress, ionic toxicity and induce nutritional deficiencies (Munns, 2002). When Na+ and Cl- ions reach high concentrations in leaves, they cause impairment in both biochemical and photochemical processes of photosynthesis (Munns and Tester, 2008). In addition to these physiological disturbances, abiotic factors such as excess of the salts (Cavalcanti et al., 2007) and heavy metals can trigger secondary stresses as the oxidative stress due to accumulation of reactive oxygen species (Gratão et al., 2005).
The physic nut (Jatropha curcas) grows in marginal areas where important crop species are not able to survive (Francis et al., 2005). Besides, it presents high economic potential due to its seed oil quality, which can be converted into biodiesel by the industry (Silva et al., 2009a). Although this species had shown satisfactory yield under constraining conditions of semiarid regions such as drought and high temperature, the plant physiological responses to salinity are not yet well known, especially in terms of key process like the photosynthesis.
This study was designed to evaluate the photosynthesis tolerance of physic nut young plants to salt stress. Changes in leaf gas exchange, photochemistry activity, and salt stress indicators (leaf Na+ and Cl- concentrations and K+/Na+ ratios) were measured after 7 and 14 days of salt stress and 3-d recovery.
Material and Methods
The experiment was carried out under greenhouse conditions, localized in the Universidade Federal do Ceará, Fortaleza, Ceará, Brazil (3º44' S, 38º33' W, at sea level), where the environmental conditions were: minimum and maximum mean air temperature of 24 and 36ºC, respectively; mean air relative humidity of 65%; maximum photosynthetic photon flux density (PPFD) of approximately 700 µmol m-2 s-1. Jatropha curcas L. seeds, genotype FT-2, were previously selected taking into account the seed size and weight. Eight days after germination in sand, seedlings were transferred to plastic pots (2 L), containing Hoagland and Arnon (1950) nutrient solution (pH 6.0) with one-forth strength in the first week and full strength afterwards.
The nutrient solution was supplied with 100 mmol L-1 NaCl and then plants were subjected to this stressful treatment during 14 days. The NaCl was added gradually (50 mmol NaCl L-1 d-1) into solution in order to avoid osmotic shock. The treatment with nutrient solution in absence of NaCl was taken as control. At the end of two weeks of treatment, the salt-stressed plants were returned to control conditions for three days. In a previous experiment, a three-day period was sufficient for the full recovery of physic nut young plants subjected to 50 mmol L-1 NaCl.
Leaf gas exchange was measured with an infrared gas analyzer (LCi, ADC, Hoddesdonm, UK), operating in open system and with air flow of 200 mL min-1. Measurements of leaf CO2 assimilation rate (A), transpiration (E), stomatal conductance (gs) and intercellular CO2 concentration (Ci) were taken. The instantaneous carboxilation efficiency (A/Ci) was calculated (Zhang et al., 2001).
The chlorophyll fluorescence was evaluated with a modulated fluorometer (FMS2, Hansatech, King's Lynn, UK). Minimum (Fo), maximum (Fm) and maximum variable (Fv=Fm-Fo) fluorescence intensities were sampled under steady-state conditions in dark-adapted (30 min) leaves. In addition, measurements were also taken under light-adapted conditions, being referred as Fo' (minimum) and Fm' (maximum). The Fo' signal was measured after PSI excitation by far-red light. The fluorescence signal under light-adapted conditions before the saturation pulse is referred as FS' and the variable fluorescence signal under light conditions is ΔF=Fm'-Fs'.
The following photochemical variables were calculated: maximum (Fv/Fm) and actual (ΔF/Fm') quantum yield of primary photochemistry. Apparent electron transport rate (ETR= ΔF/Fm' × PPFD × 0.5 × 0.84), photochemical [qP= (Fm'-Fs)/(Fm'-Fo')] and non-photochemical [NPQ= (Fm-Fm')/Fm'] quenching (Rohácek, 2002). For ETR calculation, 0.5 was used as the fraction of excitation energy distributed to PSII and 0.84 as the fraction of the incoming light absorpted by leaves (Schreiber et al., 1998). Fo' is the basal fluorescence yield measured after PSI excitation by far-red light. The ratio ETR/A was calculated to estimate the use of electrons in other processes not related to the photosynthetic CO2 assimilation rate (Ribeiro et al., 2009). Therefore, an increase in ETR/A indicates that more electrons are driven to other sinks (e.g., photorespiration, nitrogen metabolism or Mehler reaction), suggesting a stressful condition.
Leaf gas exchange and chlorophyll fluorescence were measured simultaneously, in fully expanded and mature leaves of plants exposed to 25ºC and PPFD of 590 µmol m-2s-1 supplied by an external light source. Those measurements were taken after 7-d and 14-d of treatment (0 and 100 mmol L-1 NaCl), and repeated again three days after returning plants to the nutrient solution without the presence of NaCl.
At the end of experiment, leaves were sampled, frozen and stored at -80ºC. After lyophilization, samples were placed into hermetically closed tubes containing deionized water and heated under water-bath at 100ºC for 1 h. The extracts were then filtered and used for determination of Na+ and K+ contents by flame photometry. The Cl- content was determinate through titration with AgNO3 as described by Silveira et al. (2009).
The experiment was arranged in a completely randomized design, with two factors: NaCl (0 and 100 mmol L-1); time (seven and 14 days of salt stress and three days of recovery). Data were subjected to analysis of variance and mean from four replicates were compared by the Tukey test at the 0.05 level of confidence.
Results and Discussion
Leaves of physic nut young plants treated with NaCl (100 mmol L-1) were affected by the Na+, Cl- and K+ concentrations compared to control plants (Table 1). After seven days of treatment, the Na+ and K+ concentrations reached values around 603 and 581 mmol (kg DM)-1 respectively, while the K+/Na+ ratio was reduced from 5.40 to 0.96 in stressed plants. In spite of the Na+ accumulation associated with a reduction in K+ concentration, and consequent decrease in K+/Na+ ratio, in the treated plants, the saline condition was not sufficient to induce an ionic stress and a great disturbance in the leaf ion homeostasis, taken as basis the Na+ and K+ concentrations. In fact, Silva et al. (2009b) observed that toxicity symptoms in physic nut leaves appeared only when the leaf K+/Na+ ratio is lower than 0.50.
After seven days of treatment with 100 mmol L-1 NaCl, the leaf Cl- content was just 326 mmol (kg DM)-1 (Table 1). This concentration represents only 54% of that noticed for the Na+ concentration, evidencing that J. curcas is a species more excluder of Cl- than Na+. The stress imposed by NaCl was predominantly osmotic over the ionic effects. This evidence is consistent with the results obtained by Silva et al. (2009a) working with several NaCl levels under experimental conditions similar to those employed in this current study. In addition, this species actually has a high affinity for Na+ and/or greater ability to exclude Cl- ions from roots and leaves (Silva et al., 2009b).
After 14-d of exposure to salt stress, the Na+ and Cl- concentrations reached very high levels, possibly achieving toxic values. The Na+ and Cl- concentrations were about 1,721 and 1,498 mmol (kg DM)-1 respectively. These concentrations of Na+ and Cl- ions are able to trigger salt toxicity symptoms in physic nut leaves such as leaf chlorosis followed by necrotic areas (Silva et al., 2009b), as shown in the Figure 1. Corroborating these observations, the leaf K+ concentration decreased strongly in salt-stressed plants, varying from 1,071 (control) to 423 mmol (kg DM)-1. As a consequence, the K+/ Na+ ratio was reduced from 5.0 to 0.24, evidencing clearly that after 14-d of exposure the salinity induced great disturbances in the K+-Na+ ionic homeostasis (Munns and Tester, 2008).
The salt-induced osmotic effects after 7-d and the toxic ionic effects after 14-d of salt exposure were evidenced by the appearance of visual symptoms in physic nut leaves (Figure 1). Indeed, after 7-d of treatment the leaves exhibited minor senescence symptoms while the leaves showed drastic toxicity visual symptoms after 14d as indicated by intense necrotic areas. These symptoms persisted even after the NaCl removal from the nutrient medium (recovery). After 3-d recovery, the Na+ and Cl- concentrations in the pre-stressed plants decreased by 15% and 17% only, respectively while that the K+/Na+ ratio was not changed, in comparison to the 14-d stressed plants (Table 1). Thus, most J. curcas plants suffered an osmotic stress after 7-d. However, a strong ionic stress was established in physic nut leaves in the following seven days, i.e., after 14-d of exposure to NaCl. Overall, after the first days of salt exposure glycophyte plants display an osmotic response and that after some weeks the ionic component of the salinity is predominant (Munns and Tester, 2008).
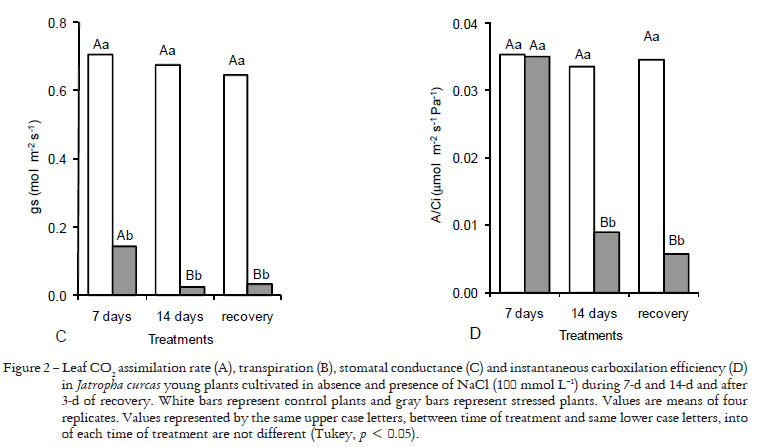
The leaf CO2 assimilation rate was strongly reduced in plants exposed to salt stress compared to control ones. Plants submitted to NaCl had 23% and 85% reductions after 7 and 14-d of treatment, respectively (Figure 2A). The transpiration rate was reduced in 39% and 84% (Figure 2B), the stomatal conductance by 80% and 97% (Figure 2C) and the intercellular CO2 concentration by 23% and 47% (data not shown) after 7 and 14-d of treatment, respectively. In contrast, the instantaneous carboxylation efficiency, estimated by the A/Ci ratio (Figure 2D), remained unchanged after 7-d of treatment but it was strongly reduced (73%) after 14-d of exposure to NaCl. Interestingly, after 3-d of recovery the A/Ci ratio did not change as compared to stressed plants (Figure 2D).
The reduction of photosynthesis after seven days of salt treatment was caused only by a stomatal limitation, i.e. reduced CO2 availability to carboxylation, while both the stomatal and biochemical limitations were observed after 14 days of treatment. As expected, the negative effects caused by salt stress on the leaf gas exchange variables were progressively increased as the time of salt exposure increased. In addition, no improvement in the photosynthesis was observed after the salt removal (recovery treatment), suggesting that after 14-d of treatment irreversible damage on the photosynthesis occurred provably due to ionic toxicity caused in the chloroplasts (López-Climent et al., 2008).
The decrease of photosynthesis associated with low stomatal conductance in plants subjected to saline conditions (Figure 2) is in accordance to Meloni et al. (2003), who observed impairment in the photosynthetic efficiency of cotton cultivars exposed to 50 and 100 mmol L-1 NaCl. On the other hand, decreases in intercellular CO2 concentration and A/Ci ratio also indicate that salt stress affected the photosynthesis by metabolic limitation. Reductions of A/Ci ratio are probably associated with a decrease of Rubisco carboxylase activity, which occurred in parallel with Na+ and Cl- intense accumulation in leaf tissues (Table 1). Thus, the reduction of photosynthesis may, at least in part, be a direct effect of Na+ and Cl- ions, as observed in sorghum (Netondo et al., 2004) and orange (López-Climent et al., 2008) plants.
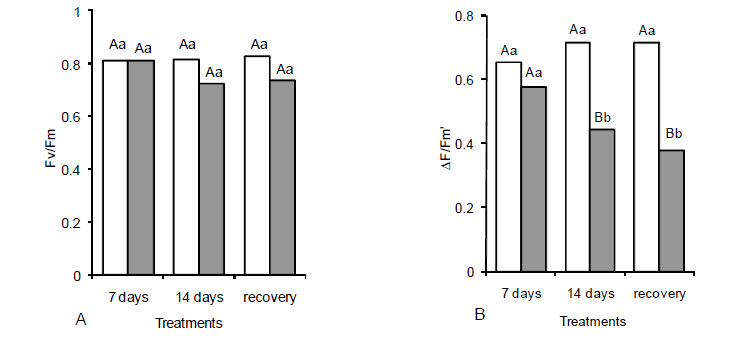
Regarding to photochemistry effects, maximum quantum yield (Fv/Fm) was not affected by salinity, whereas the actual quantum yield of primary photochemistry (ΔF/Fm') decreased (39%) after 14-d of exposure to NaCl (Figure 3A,B). The photochemical quenching (qP) was not affected by salt stress, while the non photochemical quenching (NPQ) increased in plants subjected to salt stress (Figure 3C,D). The increase of NPQ was already significant after seven days of treatment and progressively increased after 14-d of salt stress (Figure 3D).
Interestingly, this photochemical parameter increased in the recovery treatment. The reduction of ΔF/ Fm' accompanied by a decrease in the apparent electron transport rate (data not shown) in the stressed plants differs from results reported by Lu et al. (2002), who did not observe effects of salt stress in photochemical reactions of S. salsa leaves. Impairments in photochemistry of plants exposed to salinity may be related to possible damages in primary electron acceptors, such as plastoquinone pool (Foyer and Noctor, 2000). In addition, the continuous increase of NPQ during stress and recovery treatments suggest the activation of a protective mechanism for dissipation of excessive energy not used in photochemical reactions (Ribeiro et al., 2009; Rohácek, 2002). However, this protective defense was not sufficient to avoid oxidative and irreversible damage after 14-d of treatment, as suggested by the toxicity symptoms in leaves (Figure 1). Our data reveal a reasonable photochemical activity even under ionic stress, which was not compatible with the photosynthetic rates (Figures 2 and 3). In fact, full inhibition of PSII photochemistry occurs only under severe oxidative damage (Chagas et al., 2008).
After 14-d under salt stress, plants showed ETR/A ratio 4-fold higher than the control. This difference was maintained even after the recovery period (Figure 4). The increase in ETR/A ratio represents an imbalance between the electron flow and the CO2 assimilation during photosynthesis, which is frequently associated with increases in oxygenase activity of Rubisco and might represent an electron flow to other physiological processes rather than to CO2 assimilation reactions (Baker et al., 2007; Ribeiro et al., 2009). Thus, the occurrence of increases in ETR/A and decreases in A/Ci might indicate a loss of photosynthetic efficiency in physic nut plants under salinity, especially in the ionic phase of salt stress.
The accumulation of toxic ions (Na+ and Cl-) accompanied by decreases in K+ concentration and severe photosynthetic damages indicate that physic nut young plants are sensitive to high salinity caused by NaCl. The absence of any recovery in photosynthesis, after three days of salt removing, also indicates that high Na+ and Cl- leaf concentrations might cause acute damages on the photochemical and gas exchange of the photosynthesis.
Conclusion
Physic nut young plants are sensitive to high salinity, showing high leaf Na+ and Cl- concentrations and very low K+/Na+ ratios, after 14 days of exposure to 100 mmol L-1 NaCl. Under this condition, the salt-induced ionic toxicity effects are capable to induce acute photosynthetic damages (photochemistry and gas exchange) due to stomatal and biochemical limitations.
Acknowledgements
To CNPq, CAPES and "Fundação Cearense de Apoio ao Desenvolvimento Científico e Tecnológico" (FUNCAP) for financial support. J.A.G.S. and R.V.R. are CNPq researchers and E.N.S. and S.L.F.S. hold CNPq fellowships. The authors gratefully acknowledge the Tamanduá Farm Institute, Santa Terezinha-PB (Brazil), for supplying of physic nut seeds.
Received September 23, 2009
Accepted April 27, 2010
- Baker, N.R.; Harbinson, J.; Kramer, D.M. 2007. Determining the limitations and regulation of photosynthetic energy transduction in leaves. Plant, Cell and Environment 30: 1107-1125.
- Cavalcanti, F.R.; Oliveira, J.T.A.; Martins-Miranda, A.S.; Viégas, R.A.; Silveira, J.A.G. 2007. Superoxide dismutase, catalase and peroxide activities do not confer protection against oxidative damage in salt-stressed cowpea leaves. Journal of Plant Physiology 163: 563-571.
- Chagas, R.M.; Silveira, J.A.G.; Ribeiro, R.V.; Vitorrelo, V.A.; Carrer, H. 2008. Photochemical damage and comparative performance of superoxide dismutase and ascorbate peroxidase in sugarcane leaves exposure to paraquat-induced oxidative stress. Pesticide Biochemistry and Physiology 80: 181-188.
- Ferreira-Silva, S.L.; Voigt, E.L.; Viégas, R.A.; Paiva, J.R.; Silveira, J.A.G. 2009. Influence of rootstocks on the resistance of cashew plantlets to salt stress. Pesquisa Agropecuária Brasileira 44: 361-367. (in Portuguese, with abstract in English).
- Foyer, C.H.; Noctor, R. 2000. Oxygen processing in photosynthesis: regulation and signaling. New Phytologist 146: 359-388.
- Francis, G. Edinger, R. Becker, K. 2005. A concept for simultaneous wasteland reclamation, fuel production, and socio-economic development in degraded areas in India: Need, potential and perspectives of Jatropha plantations. Nature Resources Forum 29: 12-24.
- Gratão, P.L.; Polle, A.; Lea, P.J.; Azevedo, R.A. 2005. Making the life of heavy metal-stressed plants a little easier. Functional Plant Biology 32: 481-494.
- Hoagland, D.R.; Arnon, D.I. 1950. The water culture method for growing plants without soil. University of California, Berkeley, CA, USA.
- López-Climent, M.F.; Arbona, V.; Pérez-Clemente, R.M.; Gómez-Cadenas, A. 2008. Relationship between salt tolerance and photosynthetic machinery performance in citrus. Environmental and Experimental Botany 62: 176-184.
- Lu, C.; Qiu, N.; Lu, Q. 2002. Does salt stress lead to increased susceptibility of photosystem II to photoinhibition and changes in photosynthetic pigment composition in halophyte Suaeda salsa grown outdoors? Plant Science 63: 1063-1068.
- Meloni, D.A.; Oliva, M.A.; Martinez, C.A.; Cambraia, J. 2003. Photosynthesis and activity of superoxide dismutase, peroxidase and glutathione reductase in cotton under salt stress. Environmental and Experimental Botany 49: 69-76.
- Munns, R. 2002. Comparative physiology of salt and water stress. Plant, Cell and Environment 25: 239-250.
- Munns, R.; Tester, M. 2008. Mechanisms of salinity tolerance. Annual Review of Plant Biology 59: 651-681.
- Netondo, G.W.; Onyango, J.C.; Beck, E. 2004. Sorghum and salinity. II. Gas exchange and chlorophyll fluorescence of sorghum under salt stress. Crop Science 44: 806-811.
- Ribeiro, R.V.; Machado, E.C.; Santos, M.G.; Oliveira, R.F. 2009. Photosynthesis and water relations of well-watered orange plants as affected by winter and summer conditions. Photosynthetica 47: 215-222.
- Rohácek, K. 2002. Chlorophyll fluorescence parameters: the definitions, photosynthetic meaning, and mutual relationships. Photosynthetica 40: 13-29.
- Sairam, R.K.; Tyagi, A. 2004. Physiology and molecular biology of salinity stress tolerance in plants. Current Science 86: 407-421.
- Schreiber, U.; Bilger, W.; Hormann, H.; Neubauer, C. 1998. Chlorophyll fluorescence as a diagnostic tool: basics and some aspects of practical relevance. In: Raghavendra, A.S., ed. Photosynthesis: a comprehensive treatise. Cambridge University Press, Cambridge, MA, USA.
- Silva, E.N.; Silveira, J.A.G.; Fernandes, C.R.R.; Lima, C.S.; Viégas, R.A. 2009a. Contribution of organic and inorganic solutes to osmotic adjustment of physic nut under salinity. Pesquisa Agropecuária Brasileira 44: 437- 445.
- Silva, E.N.; Silveira, J.A.G.; Fernandes, C.R.R.; Dutra, A.T.B.; Aragão, R.M. 2009b. Ion uptake and growth of physic nut under different salinity levels. Revista Ciência Agronômica 40: 240-246.
- Silveira, J.A.G.; Sandro, S.A.M.; Lima, J.P.M.S.; Viégas, R.A. 2009. Roots and leaves display contrasting osmotic adjustment mechanisms in response to NaCl-salinity in Atriplex nummularia Environmental and Experimental Botany 66: 1-8.
- Viégas, R.A.; Silveira, J.A.G.; Lima Júnior, A.R.; Queiroz, J.E.; Fausto, M.J.M. 2001. Effects of NaCl-salinity on growth and inorganic solute accumulation in young cashew plants. Revista Brasileira de Engenharia Agrícola e Ambiental 5: 216-222.
- Zhang, S.; Li, Q.; Ma, K.; Chen, L. 2001. Temperature-dependent gas exchange and stomatal/nonstomatal limitation to CO2 assimilation of Quercus liaotungensis under midday higher irradiance. Photosynthetica 39: 383-388.
Publication Dates
-
Publication in this collection
13 Jan 2011 -
Date of issue
Feb 2011
History
-
Received
23 Sept 2009 -
Accepted
27 Apr 2010