Abstracts
The physiological quality of cotton cultivar seeds (Gossypium hirsutum var. latifolium L.) was evaluated in laboratory by the simulation of water potentials with polyethyleneglycol-6000 (0.0; -0.2; -0.4; -0.6; -0.8 and -1.0 MPa), at 25ºC using germitest paper as substrate. A completely randomized design in a 4 × 6 factorial scheme with four replications of 50 seeds each was used. The studied variables were: germination percentage, first count of germination, germination velocity index, accelerated aging in water, electrical conductivity, humidity, vigor classification, radicle length and radicle/shoot length ratio. The effect of water stress on seed viability and on plantlet vigor was severe at potentials below -0.4 MPa. The 'CNPA 187 8H' cultivar was the least sensitive to the tested osmotic potentials, both in terms of germination and of vigor. The 'BRS-201' cultivar was mostly affected by the viability and vigor tests under water deficit conditions. Differential viability and vigor between cultivars were observed under the water stress levels.
Gossypium hirsutum var. latifolium L.; PEG-6000; vigor; water potential
A qualidade fisiológica de sementes de cultivares de algodoeiro (Gossypium hirsutum var. latifolium L.) foi avaliada em laboratório pela simulação de potenciais hídricos com polietilenoglicol-6000 (0,0; -0,2; -0,4; -0,6; -0,8 e -1,0 MPa), na temperatura de 25ºC, em substrato papel germitest. O delineamento utilizado foi o inteiramente casualizado, em esquema fatorial 4 × 6, com quatro repetições de 50 sementes. As variáveis estudadas foram: porcentagem de germinação, primeira contagem da germinação e índice de velocidade de germinação, envelhecimento acelerado em água, condutividade elétrica, umidade, classificação de vigor (plântulas normais, fortes ou fracas), comprimento de radícula e relação radícula/parte aérea. O efeito do estresse hídrico na viabilidade das sementes e no vigor das plântulas foi severo a partir de -0.4 MPa. O cultivar CNPA 187 8H foi o menos sensível aos níveis de potenciais osmóticos testados tanto em termos de germinação, como de vigor. O cultivar BRS-201 foi o mais afetada nos testes de viabilidade e vigor em condições de déficit hídrico. Observou-se comportamento diferenciado em relação à viabilidade e vigor em diferentes níveis de estresse hídrico simulado por polietilenoglicol-6000.
Gossypium hirsutum var. latifolium L.; PEG-6000; vigor; potencial hídrico
CROP SCIENCE
Germination of cotton cultivar seeds under water stress induced by polyethyleneglycol-6000
Germinação de sementes de cultivares de algodoeiro sob estresse hídrico induzido por polietilenoglicol-6000
Carlos Henrique Salvino Gadelha MenesesI,IV; Riselane de Lucena Alcântara BrunoII; Pedro Dantas FernandesII,* * Corresponding author < pdantas@pq.cnpq.br> ; Walter Esfrain PereiraII; Leonardo Henrique Guedes de Morais LimaIII; Marleide Magalhães de Andrade LimaIII; Márcia Soares VidalIV
IUFRJ/CCS Programa de Pós-Graduação em BiotecnologiaVegetal, Ilha do Fundão, Cidade Universitária, Bloco K Rio de Janeiro, RJ Brasil
IIUFPB/CCA Depto. de Fitotecnia, C.P. 66 Rod. PB/079, km 12 58397-000 Areia, PB Brasil
IIIEmbrapa Algodão Lab. de Biotecnologia Vegetal, C.P. 174, R. Oswaldo Cruz, 1143, Centenário 58428-095 Campina Grande, PB Brasil
IVEmbrapa Agrobiologia Lab. de Genética e Bioquímica, Rod. BR 465, km 7 23890-000 Seropédica, RJ Brasil
ABSTRACT
The physiological quality of cotton cultivar seeds (Gossypium hirsutum var. latifolium L.) was evaluated in laboratory by the simulation of water potentials with polyethyleneglycol-6000 (0.0; -0.2; -0.4; -0.6; -0.8 and -1.0 MPa), at 25ºC using germitest paper as substrate. A completely randomized design in a 4 × 6 factorial scheme with four replications of 50 seeds each was used. The studied variables were: germination percentage, first count of germination, germination velocity index, accelerated aging in water, electrical conductivity, humidity, vigor classification, radicle length and radicle/shoot length ratio. The effect of water stress on seed viability and on plantlet vigor was severe at potentials below -0.4 MPa. The 'CNPA 187 8H' cultivar was the least sensitive to the tested osmotic potentials, both in terms of germination and of vigor. The 'BRS-201' cultivar was mostly affected by the viability and vigor tests under water deficit conditions. Differential viability and vigor between cultivars were observed under the water stress levels.
Key words:Gossypium hirsutum var. latifolium L., PEG-6000, vigor, water potential.
RESUMO
A qualidade fisiológica de sementes de cultivares de algodoeiro (Gossypium hirsutum var. latifolium L.) foi avaliada em laboratório pela simulação de potenciais hídricos com polietilenoglicol-6000 (0,0; -0,2; -0,4; -0,6; -0,8 e -1,0 MPa), na temperatura de 25ºC, em substrato papel germitest. O delineamento utilizado foi o inteiramente casualizado, em esquema fatorial 4 × 6, com quatro repetições de 50 sementes. As variáveis estudadas foram: porcentagem de germinação, primeira contagem da germinação e índice de velocidade de germinação, envelhecimento acelerado em água, condutividade elétrica, umidade, classificação de vigor (plântulas normais, fortes ou fracas), comprimento de radícula e relação radícula/parte aérea. O efeito do estresse hídrico na viabilidade das sementes e no vigor das plântulas foi severo a partir de -0.4 MPa. O cultivar CNPA 187 8H foi o menos sensível aos níveis de potenciais osmóticos testados tanto em termos de germinação, como de vigor. O cultivar BRS-201 foi o mais afetada nos testes de viabilidade e vigor em condições de déficit hídrico. Observou-se comportamento diferenciado em relação à viabilidade e vigor em diferentes níveis de estresse hídrico simulado por polietilenoglicol-6000.
Palavras-chave:Gossypium hirsutum var. latifolium L., PEG-6000, vigor, potencial hídrico.
Introduction
Cotton (Gossypium hirsutum var. latifolium L.) is a plant-tolerant crop to water stress. However it may suffer significant growth and production reductions when exposed to such adverse conditions. Nevertheless, the response to water stress varies among cultivars and with the developmental phase of the plant. During the germination phase, the water absorbed is required for several enzymatic reactions, for solubilization and transport of metabolites and as a reagent in the hydraulic digestion of proteins, carbohydrates and lipids from the tissue reserve of the seed towards the embryo (Woodstock, 1998; Carvalho and Nakagawa, 2000; Khajeh-Hosseini et al., 2003).
Low moisture levels in the germination environment induce protection mechanisms against drying or even prevent a stop in the developmental process (Bewley and Oliver, 1992). Severe water stress, however, results in a metabolic imbalance (Blackman et al. , 1992) and a reduction of metabolic activities (Vertucci, 1989).
One of the most commonly methods used to determine the tolerance of plants to abiotic stresses is the evaluation of the germination capacity of seeds under such conditions (Larcher, 2000). Aiming to simulate water stress conditions in the laboratory, germination studies have been carried out with aqueous solutions of polyethyleneglycol-6000 (PEG-6000) and mannitol (Murillo-Amador et al., 2002; Costa et al., 2004; Fanti and Perez, 2004). Laboratory assays simulating water stress circumstances have aided to the identification of cultivars with an elevated level of resistance to such adverse conditions in other crops, such as maize (Tonin et al., 2000) and rice (Pirdashti et al., 2003). Knowing the economic importance of cotton all over the world and of the factors which interfere in its seed germination, the present study aimed to evaluate the effect of drought stress on the viability and vigor of four cotton cultivar seeds in the germination phase, and to select a water stress-tolerant cultivar.
Material and Methods
This study was carried out in Campina Grande, Paraíba, Brazil (07º16' S; 35º53' W and altitude of 552 m), with seeds of four cotton cultivars (G. hirsutum var. latifolium L.): 'BRS-RUBI', 'BRS-SAFIRA', 'BRS-201' and 'CNPA 187 8H', the most indicated cultivars in the northeast region of Brazil due to their water stress tolerance. Seeds had been harvested in the 2005/2006 cropping season, having a relative humidity of 11%. To delint the seeds, portions of 30 g were uninterruptedly stirred in 10 mL of concentrated sulfuric acid (Merck, Darmstadt, Germany) for 3 min and then washed to remove the sulfuric acid (Lopes et al., 2006; Medeiros et al., 2006). Afterwards, seeds were treated with the fungicide n-[(trichloromethyl)thio]-4-cyclohexene-1,2-dicarboxymide.
To obtain information on seed quality, germination, accelerated aging and conductivity tests were performed. The germination test was made with 200 seeds (four subsamples of 50 seeds) in BOD-Biological Oxygen Demand (Biosystems, Mangelsdorf, Curitiba, Brazil), as described in Gonela et al. (2004), at 25ºC and without photoperiod control. The first count was performed on the 4th day, and the final count 12 days after the start of the germination experiment (Brasil, 1992). For the accelerated aging test, four subsamples of 150 seeds were distributed as thin, uniform layers on an aluminum screen fixed onto a plastic box containing 40 mL distilled water. The boxes were maintained at 42ºC for 72 h and then the percentage of germination was determined, taking into account only the morphologically perfect and well-developed plantlets.
For the electrical conductivity test, four replications with 50 seeds each (not treated with the fungicide) were used per treatment. The seeds were weighed, immersed in 75 mL distilled water at 25ºC for 24 h. Then the electric conductivity was measured with a conductivity-meter. The moisture level of the seeds was determined by the oven method (at 105 ± 3ºC), for a 24 h, according to standard procedures (Rules for Seed Analysis RSA; Brasil, 1992), before and after the accelerated aging test.
To evaluate the effect of water potential on the physiological quality of seeds, germitest paper was used, satu rated with PEG-6000 solutions adjusted to osmotic potentials (0.0; -0.2; -0.4; -0.6; -0.8; -1.0 MPa). The concentrations of PEG-6000 required to obtain these values were determined by using the equation of Michel and Kaufmann (1973): Ys = - (1.18 × 102) C - (1.18 × 104) C2 + (2.67 × 104) CT + (8.39 × 107) C2T, where Ys = osmotic potential (bar); C = concentration (g L-1 PEG-6000 in water); T = temperature (ºC). As a control, a solution with osmotic potential Ys = 0.0 MPa was used. The water used in this experiment was demineralized, containing 0.02% of fungicide.
For the germination test under conditions of water restriction, 200 seeds were used (four subsamples of 50 seeds) for each treatment. Experiments were carried out in a germination chamber at 25ºC, evaluating germination four and 12 days after seeding using standard procedures RSA (Brasil, 1992). Along with the germination test, the following vigor tests were conducted as described above: first germination counting, germination velocity index (GVI) and vigor classification (classification into normal, strong and weak seedlings, according to Vieira and Carvalho, 1994). The GVI was determined by daily counting and the index was calculated by using the equation described by Nakagawa (1994):
 , where GVI= Germination Velocity Index; G1, G2, Gn = number of normal plantlets from the first, second and nth counting; N1, N2, Nn = number of days.
, where GVI= Germination Velocity Index; G1, G2, Gn = number of normal plantlets from the first, second and nth counting; N1, N2, Nn = number of days.
Radicle length and the radicle/shoot length ratio were determined using the methodology of Vieira and Carvalho (1994) with 80 plantlets (four subsamples of 20 plantlets) for each treatment. The duration of the tests varied with the concentrations of PEG-6000, since water deficit circumstances delay the germination process. The evaluation occurred when 80% of germination was attained for each treatment and the maximum duration was 20 days. Germination was scored as positive when seeds formed plantlets with well-defined radicles and shoots, with a length of at least 0.5 cm. Taking into account these criteria, the control treatment (0.0 MPa) was evaluated on the 4th day, -0.2 and -0.4 MPa on the 12th day and below -0.6 MPa on the 20th day.
For the data-analysis, a completely randomized design was used, and the variables were submitted to deviance analysis (likelihood tests for the sources of variability controlled in the model), and the averages of the variables were compared using the χ2-test at 5%. The same approach was applied for water restriction tests, using a 4 × 6 factorial scheme (cultivars × water potentials). The variables of the water restriction experiment were submitted to both deviance analysis and logistic regression analysis (Andersson et al., 2001; Verhoeven et al., 2004) the data in percentage were estimated by using the formula: p=[exp(nij)/1+exp(nij)] (Paula, 2001).
Moisture data, moisture of aged seeds, electrical conductivity, radicle length and the radicl/shoot length ratio were submitted to variance analysis and the averages of these variables were compared by Tukey test (p < 0.05).
Results
For all cultivars, the average germination values were higher than the standard values (± 75%) for commercially available cotton seeds in Brazil (Table 1), indicating that the seeds used in this study were of good physiological quality. The cultivar "CNPA 187 8H' presented a higher germination percentage than the cultivars "BRS 201', "BRS RUBI' and "BRS SAFIRA', although it was different only from "BRS RUBI' and "BRS SAFIRA' (p < 0.05). The mean values obtained in the first count varied from 80 to 87% and permitted differentiating the cultivars. The "CNPA 187 8H' cultivar presented a larger number of normal plantlets, when compared to "BRS RUBI'. Differences were observed regarding the percentage of normal plantlets, permitting the separation of the cultivars into two groups; "BRS SAFIRA',"BRS 201' and "CNPA 187 8H' had a higher physiological potential than the BRS RUBI cultivar.
The results of the accelerated aging test indicated low germination efficiency and consequently a gradual reduction of seed vigor. This test has been considered efficient to separate lots of cotton seeds according to vigor level and consequently, to show loss of physiological quality (Mendonça et al., 2008). In our study, the "BRS 201' cultivar was less vigorous than the "BRS RUBI' cultivar was also shown that in a period of 72 h at 41ºC. The "BRS SAFIRA' and "CNPA 187 8H' cultivars constituted an intermediate level, and there was not difference between them (Table 1). The moisture content of seeds of the cultivars did not vary among the phases before (HB) and after (HA) the accelerated aging test. The same was observed for the electric conductivity (Table 1).
Germination efficiency diminished with the osmotic potential and germination was completely inhibited at -1.0 MPa (Figure 1). The "CNPA 187 8H' cultivar could be classified as the least sensitive to water stress, because its sensitivity was different from the others at potentials -0.4; -0.6 and -0.8 MPa."BRS RUBI', "BRS SAFIRA' and "BRS 201' cultivars are in the group of susceptibility to water restriction conditions because these genotypes presented the lowest germination indexes. An exception occurred at potential -0.6 MPa, where cultivar "BRS 201' was more sensitive than the others from the group, and therefore the vigor of this cultivar was considered inferior. The "BRS RUBI', "BRS SAFIRA' and "BRS 201' cultivars lost viability more rapidly when the osmotic potential diminished. In contrast, the "CNPA 187 8H' cultivar showed a higher level of tolerance to water restriction conditions.
For the first germination count experiment, more than 80% of the seeds of all the cultivars germinated four days after the start of the test at osmotic potentials of 0.0 MPa. For all cultivars, 8 to 12% germination at the first count was only observed at osmotic potentials equal to, or less than -0.2 MPa, without any differentiation among cultivars at any potential (data not shown). Comparing the results of vigor at the 1st count, with the results of the germination, the vigor of the cotton seeds was more affected than the germination percentage. These results may justify the delay in seed germination, observed at more negative osmotic potentials.
GVI values for all cultivars were reduced when the osmotic potential of the substrate diminished (Figure 2). These data show the sensitivity to elevated levels of water restriction, completely inhibiting the germination process at the most negative value (-1.0 MPa), except for the cultivar "CNPA 187 8H'. Cultivar "BRS 201' was the most sensitive to high drought stress levels, although its sensibility was not different from cultivar "CNPA 187 8H' at -0.2 MPa. Down to -0.4 MPa, all cultivars presented GVI values superior to 35.0 and reductions occurred only at osmotic potentials below -0.4 MPa.
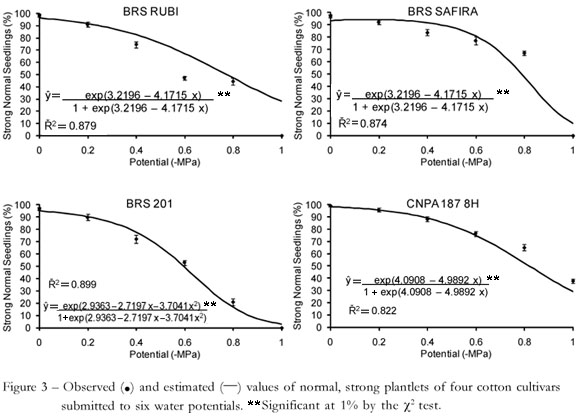
The vigor diminished as the water potential dropped (Figure 3), and the number of normal, strong plants fell. There were differences between treatments at all osmotic potentials, and "CNPA 187 8H' was the least affected by treatments with PEG-6000. "CNPA 187 8H' had the highest percentage of normal and strong plants at potentials -0.2 and - 1.0 MPa. At the other tested potentials (-0.4, -0.6 and -0.8 MPa), both cultivars "CNPA 187 8H' and "BRS SAFIRA' showed the best results. The "BRS 201' cultivar had the lowest percentage of normal strong plantlets and the highest percentage of normal weak plantlets at -0.8 MPa.
Even under stress conditions, a more pronounced radicle development was observed for the "BRS RUBI', "BRS SAFIRA' and "CNPA 187 8H' cultivars at -0.4 and - 0.8 MPa (Figure 4). At potential -0.2 MPa, the "BRS SAFIRA', "BRS 201' and "CNPA 187 8H' cultivars had higher values than "BRS RUBI'. At -0.4 and -0.6 MPa, cultivar "BRS 201' had the lowest level of adaptability to the treatments with PEG-6000, because it had the smallest average radicle length during the study. The averages of all cultivars fell drastically, with values close to zero, at osmotic potentials -1.0 MPa.
For the shoot data, the cultivars were only different at potential -1.0 MPa, where cultivar "CNPA 187 8H' presented the best shoot growth (data not shown). For the radicle/ shoot length ratio, the greatest differences among cultivars were observed at -0.4 MPa, where this ratio showed the highest value as a natural reaction of the plantlets to water stress (Figure 5). At -0.4 MPa, the radicle/shoot length ratio had higher values for cultivars "BRS RUBI', "BRS 201' and "CNPA 187 8H', presenting a higher level of tolerance to water stress as compared to the "BRS SAFIRA' cultivar, which showed less adaptability. For the other osmotic potentials (0.0; -0.2; -0.6; -0.8 and -1.0 MPa) there were no differences among cultivars.
For the majority of the cultivars, the embryonic axis dry mass tended to increase with the concentration of PEG-6000 (data not shown). At almost all levels, there were no differences among cultivars, with the exception to the potential - 0.6 MPa, in which the cultivar CNPA 187 8H was more tolerant to water restriction, showing better development.
Discussion
The results observed in Figure 1 were as expected, since the concentration of salts in the germination medium (PEG- 6000 in this case) had controlled the water absorption by the seed tissues, thus interfering in, or impeding, the germination process. Higher concentrations of PEG-6000 in the solution reduced water absorption by the seeds of the different cultivars, consequently diminishing the percentage of germination.
Germination was completely inhibited at water potential -1.0 MPa. Similar data were obtained by Pereira et al. (1998) who found significant effects on germination percentages for ten genotypes, PEG-6000 simulated osmotic potentials, and for the interaction between these two factors. Similarly to the observations in the present study, they also observed complete inhibition of average germination percentage at -1.0 MPa. Ghajari and Zeinali (2003) studied the effect of drought and salinity on the germination and growth of plantlets of two cotton cultivars ("Sahel' and "Bakhtegan'). They applied water potentials varying from -0.1 to -1.0 MPa induced by PEG-6000 and NaCl, and found that the germination percentage and the percentage of normal plants increased, both under salinity and osmosis. Ghajari and Zeinali (2003) also observed an increase in radicle length until -0.2 MPa when using PEG-6000. The "Bakhtegan' cultivar was more tolerant to salinity than the "Sahel' cultivar, but under the conditions simulated with PEG-6000, tolerance and germination of "Bakhtegan' were inferior to "Sahel', thus classifying the two cultivars according to sensitivity to this type of stress, as seen in this work.
Pirdashti et al. (2003) observed similar effects in studies with 15 rice (Oryza sativa) genotypes and five osmotic potentials. The germination potential diminished at higher osmotic potentials in PEG-6000 solutions, and was completely inhibited at -1.0 MPa. In contrast, Leucaena leucocephala germination occurred at all levels of osmotic stress of 0.0; - 0.3; -0.6; -0.9 and -1.2 MPa (Souza Filho, 2006). Okçu et al. (2005) evaluated three pea (Pisum sativum L.) genotypes subjected to five water potentials and to both water and saline stresses. The authors observed that saline stress had no impact on germination percentage, although PEG-6000 simulated water stress and reduced germination drastically. Fanti and Perez (2003) verified a decrease in the germination of "paineira' seeds (Chorisia speciosa St. Hil. - Bombacaceae), as a consequence of low osmotic potentials, and this was attributed to diminished water diffusion through the seed coat.
Germination was retarded when stress levels increased (Figures 2 and 3), probably because of a diminished movement and accessibility of water for seed imbibitions (Rauf, 2007). In general, the decreasing osmotic potential levels progressively reduced the number of normal strong plantlets of all cultivars. Therefore, with the lower osmotic potential of the substrate and water absorption by the seeds, germination was affected, resulting in plants with less vigor.
Moraes et al. (2005) studied the effects of PEG-6000 on bean (Phaseolus vulgaris L.) seeds and observed that the seeds also presented a progressive decrease of vigor as osmotic potentials became more negative. Braccini et al. (1998) found similar results, with an accentuated reduction of vigor of bean seeds caused by more negative values of osmotic potential of PEG-6000 solutions.
GVI decreased with more negative osmotic potential values (Figure 2), leading to complete inhibition at -0.8 MPa, suggesting that the resistance limit of these cultivars to water stress is between -0.6 and -0.8 MPa. The higher the GVI number obtained, the higher the velocity of germination and the vigor, since the index estimates the average number of normal plants per day (Nakagawa, 1994). Similar to what was found in the present study, Fanti and and Perez (2003) observed a decrease in the GVI of "paineira' seeds with more negative osmotic potentials.
The GVI of Bowdichia virgilioides seeds was also reduced at higher osmotic potentials and totally inhibited at - 0.9 MPa, suggesting that the resistance limit for B. virgilioides to water stress is between -0.7 and -0.9 MPa (Silva et al., 2001). Similarly, the GVI of L. leucocephala seeds responds negatively to elevated levels of osmotic stress; the germination percentage presented a reduction in the order of 60%, while the GVI was reduced by 80%. Lower germination velocities were observed for the control as the osmotic potential increased (Figure 2). This can be explained by the fact that the seeds needed more time to germinate, resulting in a lower germination velocity. GVI is a more sensitive measure for the effects of osmotic stress than the germination potential measure (Dias and Marcos Filho, 1996).
Differences were observed in the percentage of strong plantlets for each osmotic potential and the treatments could generally be differentiated from the control (0.0 MPa). Comparing the percentages of strong plantlets (Figure 3) with those of weak plantlets (data not shown), a relationship could be observed in the results obtained for each treatment, since the total of strong and weak plantlets constitutes the final germination percentage. The classification of the vigor of plantlets was more robust than the germination test to compare different cultivars. This is in accordance with Marcos Filho (1999), who suggested that this type of test is important for identifying differences in the physiological potential of seeds that constitute lots with a high and homogeneous level of germination potential.
Radicle length diminished more drastically at stress levels below -0.4 MPa (Figure 4). Radicle length at potential - 0.2 MPa was favored by the use of PEG-6000, since the radicles obtained under these conditions were longer in all cultivars, mainly in "CNPA 187 8H'. Pace et al. (1999) evaluates the effect of water stress on two cotton cultivars under water stress and found that radicle lengths increased under drought conditions. This is a common response of cotton, for which the exploration of the soil and the intimate contact between the surfaces of the root and the soil particles are essential for the effective absorption of water by the roots. The contact surface and the consequent water absorption capacity are maximized by the production of root hairs. For this reason, the development of the root system of cotton is also involved in the adaptation of this plant to water stress. However, drought has also been shown to reduce growth, development, and root distribution in some drought-sensitive cotton genotypes (Malik, 1979).
Pereira et al. (2008) observed increments in primary root length of carrots (Daucus carota L.) as a response to drought. Similar results were also obtained for mustard (Brassica juncea L.) plantlets by Srinivasan et al. (1999). During water stress conditions, levels of soluble proteins and specific enzymes rise, causing higher concentrations of solutes, resulting in a more rapid growth, and as a consequence, leading to higher biomass accumulation (Smith and Cobb, 1991).
In general, more negative values of osmotic potentials progressively diminished the vigor of all cultivars (Figure 4). Therefore, germination is affected and smaller and less vigorous plants are obtained after reduction of the water potential of the substrate and because of the deficiency in water absorption by the seeds,. These effects negatively influence the development and the establishment of the plants under field conditions, as a consequence of reduced velocities of physiological and biochemical processes. Likewise, this was observed in this study for the vigor at the first count and for the germination at the final count.
For all cultivars, the average values of radicle length were always superior to the average shoot lengths (Figure 5). This indicates that under different stress conditions during the germination phase of cotton, roots tend to elongate more than shoots. This leads to a higher root/shoot ratio, which is a common response of plants to water restriction conditions.
Conclusions
Cotton is relatively tolerant to water stress induced by PEG-6000. The water deficit was most effective in reducing seed viability and vigor at osmotic potentials equal to or more negative than -0.4 MPa. Based on the viability tests, cultivar CNPA 187 8H was the least sensitive to all osmotic potentials, while cultivar BRS 201 was the most sensitive. Regarding the vigor tests, cultivar CNPA 187 8H was more tolerant to low osmotic potentials, while cultivar BRS 201 was the most sensitive.
Acknowledgements
To the colleagues of the experimental station of Embrapa Cotton and to members of Laboratory for Seed Analysis of the Center of Agricultural Sciences of the Federal University of Paraíba for their technical assistance during the analyses. The first author acknowledges a fellowship granted by CAPES, Brazil.
Received November 23, 2009
Accepted October 05, 2010
- Andersson, L.; Milberg, P.; Schutz, W.; Steinmetz, O. 2001. Germination characteristics and emergence time of annual Bromus species of differing weediness in Sweeden. Weed Research 42: 135-147.
- Bewley, J.D.; Oliver, M.J. 1992. Desiccation tolerance in vegetative plant tissues and seeds: protein synthesis in relations to desiccations and a potential role for protection and repair mechanisms. p. 141- 160. In: Osmond, C.D.; Somero, G.; Bolis, C.L., eds. Water and life: a comparative analysis of water relationships at the organism, cellular and molecular levels. Spring, Berlin, German.
- Blackman, S.A.; Obendorf, R.L.; Leopold, A.C. 1992. Maturation proteins and sugars in desiccation tolerance of developing soybean seeds. Plant Physiology 100: 225-230.
- Braccini, A.L.; Reis, M.S.; Sediyama, C.S.; Sediyama, T.; Rocha, V.S. 1998. Effects of water potential induced by polyethyleneglycol on soybean seed physiological quality. Pesquisa Agropecuária Brasileira 33: 1451-1459. (in Portuguese with abstract in English).
- Brasil. Ministério da Agricultura..1992. Rules for seed analysis. MA/SNAD/LANARV. Brasília, DF, Brazil. (in Portuguese).
- Carvalho, N.M.; Nakagawa, J. 2000. Seeds: science, technology and production. Funep, Jaboticabal, SP, Brazil. (in Portuguese).
- Costa, P.R.; Custódio, C.C.; Machado Neto, N.B.; Marubayashi, O.M. 2004. Water deficit induced by mannitol on soybean seed classified in different sizes. Revista Brasileira de Sementes 26: 105-113. (in Portuguese with abstract in English).
- Dias, D.C.F.S; Marcos Filho, J. 1996. Electrical conductivity tests to evaluate the vigor of soybean (Glycine max (L.) Merrill) seeds. Scientia Agricola 53: 31-42. (in Portuguese with abstract in English).
- Fanti, S.C.; Perez, S.C.J.G.A. 2003. Water stress and accelerated aging effects on the viability of osmoconditioned Chorisia speciosa seeds. Pesquisa Agropecuária Brasileira 38: 537-543. (in Portuguese with abstract in English).
- Fanti, S.C.; Perez, S.C.J.G.A. 2004. Seed germination of paineira under water and salt stress. Pesquisa Agropecuária Brasileira 39: 903-909. (in Portuguese with abstract in English).
- Ghajari, A.; Zeinali, E. 2003. Effects of salinity and drought stresses on germination and seedling growth of two cotton cultivars. Seed and Plant 18: 506-509.
- Gonela, A.; Lemos, E.G.M.; Rodrigues, T.J.D.; Paterniani, M.L.S. 2004. Enzymatic reaction to saline stress during stylosanthes germination. Pesquisa Agropecuária Brasileira 39: 93-95.
- Khajeh-Hosseini, M.; Powell, A.A.; Bingham, I.J. 2003. The interaction between salinity stress and seed vigor during germination of soybean seeds. Seed Science and Technology 31: 715-725.
- Larcher, W. 2000. Plant ecophysiology. RIMA, São Carlos, SP, Brazil. 531 p. (in Portuguese).
- Lopes, K.P.; Bruno, R.L.A.; Costa, R.F.; Bruno, G.B.; Rocha, M.S. 2006. Effects of seed processing on physiological and sanitary qualities of seeds of herbaceous cotton. Revista Brasileira de Engenharia Agrícola e Ambiental 10: 426-435. (in Portuguese with abstract in English).
- Malik, R.S.; Dhankar, J.S.; Turner, N.C. 1979. Influence of soil water deficits on root growth and cotton seedlings. Plant Soil 53: 109-115.
- Marcos Filho, J. 1999. Vigor tests: importance and use. In: Krzyzanowski, F.C.; Vieira, R.D.; França Neto, J.B. Seeds Vigor: concepts and tests. ABRATES, Londrina, PR, Brazil. (in Portuguese).
- Medeiros Filho, S.; Silva, S.O.; Dutra, A.S.; Torres, S.B. 2006. Comparison of germination test methodologies of linted and delinted cotton seeds. Revista Caatinga 19: 56-60. (in Portuguese with abstract in English).
- Mendonça, E.A.F.; Azevedo, S.C.; Guimarães, S.C.; Albuquerque, M.C.F. 2008. Vigor tests in upland cotton seeds. Revista Brasileira de Sementes 30: 1-9. (in Portuguese with abstract in English).
- Michel, B.E.; Kaufmann, M.R. 1973. The osmotic potential of polyethylene glycol 6000. Plant Physiology 51: 914-916.
- Moraes, G.A.F.; Menezes, N.L.; Pasqualli, L.L. 2005. Bean seed performance under different osmotic potentials. Ciência Rural 35: 776-780. (in Portuguese with abstract in English).
- Murillo-Amador, B.; Lopez-Aguilar, R.; Kaya, C.; Larrinaga- Mayoral, J.; Flores-Hernandez, A. 2002. Comparative effects of NaCl and polyethylene glycol on germination, emergence and seedling growth of cowpea. Journal of Agronomy and Crop Science 188: 235-247.
- Nakagawa, J. 1994. Seed vigour tests based on seedling characteristics. p. 49-85. In: Vieira, R.D.; Carvalho, N.M. Vigor tests on seeds. FUNEP, Jaboticabal, SP, Brazil. (in Portuguese).
- Okçu, G.; Kaya, M.D.; Atak, M. 2005. Effects of salt and drought stresses on germination and seedling growth of pea (Pisum sativum L.). Turkish Journal of Agriculture and Forestry 29: 237-242.
- Pace, P.F.; Cralle, H.T.; El-Halawany, S.H.M.; Cothren, J.T.; Senseman, S.A. 1999. Drought-induced changes in shoot and root growth of young cotton plants. Journal of Cotton Science 3: 183-187.
- Paula, G.A. 2001. Regression Models with Computational Support. EDUSP, São Paulo, SP, Brazil.
- Pereira, J.R.; Fernandes, P.D.; Beltrão, N.E.M. 1998. Deterioration, hardening and abnormalities in seed and seedling of upland cotton (Gossypium hirsutum l. r. latifolium H.) genotypes under water stress. Revista Brasileira de Engenharia Agrícola e Ambiental 2: 186-194. (in Portuguese with abstract in English).
- Pereira, M.D.; Dias, D.C.F.S.; Dias, L.A.S.; Araújo, E.F. 2008. Germination and vigor of carrot seeds primed in moistened paper and aerated solution. Revista Brasileira de Sementes 30: 137-145. (in Portuguese with abstract in English).
- Pirdashti, H.; Sarvestani Tahmasebi, Z.; Nematzadeh, G.H.; Ismail A. 2003. Effect of water stress on seed germination and seedling growth of rice (Oryza sativa L.) genotypes. Pakistan Journal of Agronomy 2: 217-222.
- Silva, L.M.M.; Aguiar, I.B.; Rodrigues, T.J.D. 2001. Seed germination of Bowdichia virgilioides Kunth, under water stress. Revista Brasileira de Engenharia Agrícola e Ambiental 5: 115- 118. (in Portuguese with abstract in English).
- Smith, P.T.; Cobb, B.G. 1991. Accelerated germination of pepper seed by priming with salt solutions and water. HortScience 26: 417-419.
- Souza Filho, A.P.S. 2006. Influence of temperature, light and osmotic and saline stresses on seed germination of Leucaena leucocephala Pasturas Tropicales 22: 47-53. (in Spanish with abstract in English).
- Srinivasan, K.; Saxena, S.; Singh, B.B. 1999. Osmo- and hydropriming of mustard seeds to improve vigour and some biochemical activities. Seed Science and Technology 27: 785-793.
- Tonin, G.A.; Carvalho, N.M.; Kronka, S.N.; Ferraudo, A.S. 2000. Culture systems, velvet bean and mineral fertilization influence on maize seeds physiological quality. Revista Brasileira de Sementes 22: 276-279. (in Portuguese with abstract in English).
- Verhoeven, K.J.F.; Biere, A.; Nevo, E.; Damme, J.M.M. van. 2004. Can a genetic correlation with seed mass constrain adaptive evolution of seedling desiccation tolerance in wild barley? International Journal of Plant Sciences 165: 281-288.
- Vertucci, C.W. 1989. The kinetics of seed imbibition. p. 93-115. In: Crop Science Society Of America. Seed moisture. CSSA, Madison, WI USA. (Special Publication, 14).
- Vieira, R.D.; Carvalho, N.M. 1994. Vigor Tests on Seeds. FUNEP, Jaboticabal, SP, Brazil. (in Portuguese).
- Woodstock, L.W. 1998. Seed imbibition: a critical period for successful germination. Journal of Seed Technology 12: 1-15.
Publication Dates
-
Publication in this collection
31 Mar 2011 -
Date of issue
Apr 2011
History
-
Accepted
05 Oct 2010 -
Received
23 Nov 2009