Abstracts
Although canola, (Brassica napus L.), is considered a self-pollinating crop, researchers have indicated that crop productivity increases as a result of honey bee Apis mellifera L. pollination. Given this crop's growing importance in Rio Grande do Sul State, Brazil, this work evaluated the increase in pod and seed productivity with respect to interactions with anthophilous insects and manual pollination tests. The visiting frequency of A. mellifera was correlated with the crop's blooming progression, and productivity comparisons were made between plants visited by insects, manually pollinated plants (geitonogamy and xenogamy) and plants without pollination induction. Pod set and seed production per plant were determined for each treatment. Among the 8,624 recorded flower-visiting insects, Hymenoptera representatives were the most prevalent (92.3%), among which 99.8% were A. mellifera. The correlation between these bees and blooming progression was positive (r = 0.87; p = 0.002). Pollination induction increased seed productivity from 28.4% (autogamy) to 50.4% with insect visitations, as well as to 48.7 (geitonogamy) and to 55.1% (xenogamy) through manual pollination.
flower-visiting insects; blooming; canola productivity
A canola (Brassica napus L.) é considerada autocompatível, embora pesquisadores indiquem aumento na produtividade da cultura resultante da polinização efetuada por Apis mellifera L.. Considerando-se a crescente importância dessa cultura no Rio Grande do Sul, avaliou-se o incremento da produtividade de síliquas e sementes a partir de interações com insetos antófilos e com testes de polinização manual. A freqüência de visitas de A. mellifera foi relacionada com o desenvolvimento da floração da cultura e a produtividade foi comparada entre plantas visitadas por insetos, polinizadas manualmente (geitonogamia e xenogamia) e com ausência de indução de polinização. Em cada tratamento avaliou-se a produtividade de síliquas e de sementes formadas por planta. Dentre os 8.624 insetos visitantes florais registrados destacaram-se representantes de Hymenoptera (92,3%), dos quais 99,8% eram A. mellifera. A relação entre essas abelhas e o curso da floração foi positiva (r = 0,87; p = 0,002). A indução de polinização elevou a produtividade de sementes de 28,3% (autogamia) para 50,4% com livre visita de insetos e com polinização manual, respectivamente, para 48,7 (geitonogamia) e 55,1% (xenogamia).
insetos visitantes florais; floração; produtividade agrícola
NOTE
Honey bee contribution to canola pollination in Southern Brazil
Abelhas melíferas na polinização de canola no Sul do Brasil
Annelise de Souza RosaI; Betina BlochteinII,* * Corresponding author < betinabl@pucrs.br> ; Diego Kweco LimaII
IPontifícia Universidade Católica do Rio Grande do Sul/Programa de Pós-Graduação em Zoologia, Av. Ipiranga 6681 90619-900 Porto Alegre, RS Brasil
IIPontifícia Universidade Católica do Rio Grande do Sul/Faculdade de Biociências, Av. Ipiranga 6681 90619-900 Porto Alegre, RS Brasil
ABSTRACT
Although canola, (Brassica napus L.), is considered a self-pollinating crop, researchers have indicated that crop productivity increases as a result of honey bee Apis mellifera L. pollination. Given this crop's growing importance in Rio Grande do Sul State, Brazil, this work evaluated the increase in pod and seed productivity with respect to interactions with anthophilous insects and manual pollination tests. The visiting frequency of A. mellifera was correlated with the crop's blooming progression, and productivity comparisons were made between plants visited by insects, manually pollinated plants (geitonogamy and xenogamy) and plants without pollination induction. Pod set and seed production per plant were determined for each treatment. Among the 8,624 recorded flower-visiting insects, Hymenoptera representatives were the most prevalent (92.3%), among which 99.8% were A. mellifera. The correlation between these bees and blooming progression was positive (r = 0.87; p = 0.002). Pollination induction increased seed productivity from 28.4% (autogamy) to 50.4% with insect visitations, as well as to 48.7 (geitonogamy) and to 55.1% (xenogamy) through manual pollination.
Key words: flower-visiting insects, blooming, canola productivity
RESUMO
A canola (Brassica napus L.) é considerada autocompatível, embora pesquisadores indiquem aumento na produtividade da cultura resultante da polinização efetuada por Apis mellifera L.. Considerando-se a crescente importância dessa cultura no Rio Grande do Sul, avaliou-se o incremento da produtividade de síliquas e sementes a partir de interações com insetos antófilos e com testes de polinização manual. A freqüência de visitas de A. mellifera foi relacionada com o desenvolvimento da floração da cultura e a produtividade foi comparada entre plantas visitadas por insetos, polinizadas manualmente (geitonogamia e xenogamia) e com ausência de indução de polinização. Em cada tratamento avaliou-se a produtividade de síliquas e de sementes formadas por planta. Dentre os 8.624 insetos visitantes florais registrados destacaram-se representantes de Hymenoptera (92,3%), dos quais 99,8% eram A. mellifera. A relação entre essas abelhas e o curso da floração foi positiva (r = 0,87; p = 0,002). A indução de polinização elevou a produtividade de sementes de 28,3% (autogamia) para 50,4% com livre visita de insetos e com polinização manual, respectivamente, para 48,7 (geitonogamia) e 55,1% (xenogamia).
Palavras-chave: insetos visitantes florais, floração, produtividade agrícola
Introduction
Canola, Brassica napus L., is a genetically modified selection of colza. The levels of erucic acid and glucosinolates present in native rapeseed limit suitability for human and animal consumption. Thus, genetic modifications were made with the aim of reducing the levels of these substances, and thereby allowing the consumption of oil extracted from seeds, which is known as canola oil. Canola belongs to the family of Brassicaceae and is cultivated throughout Brazil's southern grain production systems (Barni et al., 1985; Tomm, 2007), standing out as an excellent economic alternative due to its use in the production of human consumable oils and biodiesel (Cunha, 2007; Tomm, 2007; Marjanovic-Jeromela et al., 2008).
Klein et al. (2007) highlighted the importance of insect pollination in the production of fruits and seeds. Besides contributing to the preservation of natural ecosystems, bee pollination is one of the main alternatives for the improvement of crop productivity (Westcott and Nelson, 2001; D'Ávila and Marchini, 2005). Thus, honey bees, Apis mellifera L., are considered the most important flower visitors (Delaplane and Mayer, 2000) and most efficient B. napus pollinators (Free, 1993; Sabbahi et al., 2005).
Although canola is considered a self-pollinating crop, the presence of insect pollinators, especially A. mellifera, increases its seed productivity (Eisikowitch, 1981; Kevan and Eisikowitch, 1990; Delaplane and Mayer, 2000; Westcott and Nelson, 2001). An adequate pollination process, resulting from consecutive bee visits, ensures the reproductive cycle of the Brassicaceae, increasing their productivity indices (Abrol, 2007). Mussury and Fernandes (2000) demonstrated an increase of 31.9% of seeds per plant under natural pollination conditions, when compared to autogamy conditions. Concerning average seed weight, Williams et al. (1986) and Adegas and Nogueira-Couto (1992) found increases of 34.5% and 19.2%, respectively, when comparing plants under bee influence to plants without any insect influence.
Considering the current worldwide need for canola seed production and the growing importance of this crop in the Rio Grande do Sul State, Brazil, the production of pods and seeds resulting from self-pollination, manual pollination and interactions between honey bees and flowers was evaluated in this study.
Material and Methods
This study evaluated B. napus cultivar Hyola 432 development from pre-blooming to pod harvesting, in a field of 13 ha , in the Três de Maio, Rio Grande do Sul State, Brazil (27º46' S; 54º14' W). There were two apiaries in the region, each with 20 and 18 colonies, respectively, at about 0.2 km and 1 km from the crop's boundaries; potential wild bee areas were very small, since they were reduced due to the presence of native fragments throughout the region. Data gathering was performed between July and October of 2007; temperature, precipitation and relative humidity were respectively 14.6ºC, 2.8 mm and 74.6%. The data were provided by the meteorological station of the existing headquarters in the fields of canola.
Apis mellifera, as well as other pollinators, were counted throughout the blooming period in order to determine their abundance. This procedure was performed by three 30-minute periods (as of 12h00, 14h00 and 16h00), twice a week, at temperatures over 12ºC, totaling 27 h of records. Records were made over a transect of 300 m in length and 1 m wide, at an average speed of about 0.6 km h-1. Insect identification was only made at the order level, while A. mellifera was identified at the species level. The blooming progression was monitored using 120 plants, which were labeled with colored ribbons and distributed in the four ends of the field and at eight points in the inner portion of the crop field. The number of flowers on each plant's main inflorescence was evaluated during the entire blooming period.
Concerning the pollination efficiency test, seed productivity was compared between four experiments: (i) autogamy, covering the inflorescences during the entire anthesis period; (ii) control, allowing spontaneous insect access; (iii) geitonogamy - manual pollination between flowers of the same plant; (iv) xenogamy - manual pollination between flowers from different plants. Four replicates were performed for each treatment, where each replicate included seven plants. To evaluate the autogamy, geitonogamy and xenogamy treatments, the plants were protected with wooden frames (1 × 1 × 1.8 m), covered with a thin mesh (Voala tissue), allowing air circulation, to and excluding insects. The control treatment was marked with wooden stakes and kept unprotected to allow insect visits. As this treatment is unprotected, it also includes abiotic factors. For manual pollination purposes, pollen grains were transferred between vigorous flowers using a thin brush, in four-day intervals, because the anthesis period has approximately the same time interval. Considering that a few flowers outlived their vigorous stage over untreated time periods, the index of manually treated flowers was calculated based on the total number of flowers produced, amounting to 51.9% and 42.6% concerning the geitonogamy and xenogamy treatments, respectively. At the end of crop cycle, the plants were harvested and the following parameters were evaluated: total number of pods / plant (n = 28 plants / treatment); number of seeds / pod (n = 160 pods / treatment); and seed weight (n = 160 pods / treatment).
The percent of seed / plant was estimated for each treatment, using the formula: average number of seeds per pods multiplied by 100 and divided by the average number of ovules obtained from 100 flowers with mature ovaries. The total seed weight per plant was calculated by multiplying the average individual seed weight by the number of seeds per plant.
To determine flower attractiveness to the bees, the relation between the foraging behavior of A. mellifera and the blooming progression of B. napus was calculated by Pearson's correlation test. Since the parameters total number of pods / plant and seed weight showed parametric data, the averages of the pollination treatments were compared using variance analysis (ANOVA), followed by the Games-Howell test (95% confidence intervals) for non-homogeneous samples (number of pods / plant, pod weight and seed weight). The number of seeds / pod parameter was characterized by non-parametric data. In this case, the Kruskal-Wallis test and the Student - Newman - Keuls test (95% confidence intervals) were used to compare the averages. SPSS version 11.5 statistics package for MS Windows was used in all analyses, except the Student Newman Keuls test, for which the Bioestat version 5.0 program was used.
Results and Discussion
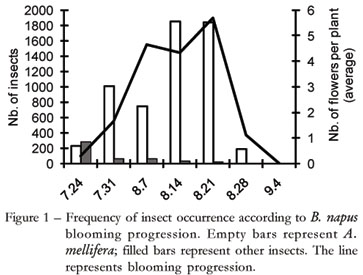
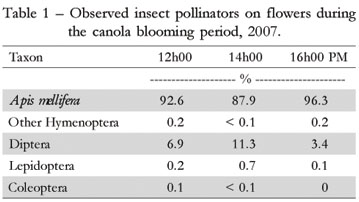
During the blooming process of B. napus plants, 8.624 insects were recorded on their flowers. They included species of Hymenoptera, especially A. mellifera (99.83%), Diptera, Lepidoptera and Coleoptera (Table 1). The attractiveness of B. napus flowers to A. mellifera is associated with the availability of food resources, whether nectar or pollen, according to previous records by Free (1993), Delaplane and Mayer (2000) and Mussury et al. (2003). Another explanation for the intense visitation of bees to flowers is that canola is a crop that blooms in winter. During this period, food resources from other sources are scare because the plants usually are not in bloom during the winter.
Apis mellifera frequency was directly related to the progression of B. napus blooming, and it differed from that of other insect groups (Figure 1). The correlation between the number of B. napus flowers in the crop and the number of recorded honey bees during the blooming period was positive (n = 9 days; r = 0.87; p < 0.05). The abundance of food resources offered by B. napus increased flower attractiveness and, consequently, bee pollination, resulting in higher seed productivity (Manning and Boland, 2004). The introduction of A. mellifera colonies in the crop is an alternative for productivity improvements and should be performed when approximately 10% of the blooming has occurred (Abrol, 2007). If the beehives are established before this period, the bees may be forced to feed on other plants which may be in blooming, preventing pollination increases in the crop itself (Abrol, 2007). Concerning the cultivar studied, Hyola 432, this 10% index occurred between the 2nd and 3rd weeks of blooming, a period during which the beehives should be introduced.
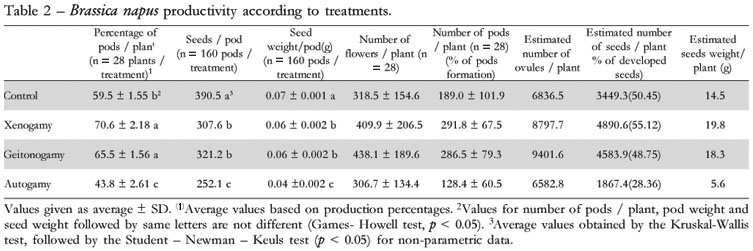
Brassica napus Hyola 432 productivity in all pollination induction situations, whether manually (geitonogamy and xenogamy) or by insects and abiotic conditions (control), exceeded autogamic productivity (Table 2). The productivity results for manually pollinated plants, through geitonogamy and xenogamy, were statistically similar regarding the number of pods / plant and seeds / pod, as well as pod weight and seed weight / pod (Table 2). The number of pods in these plants was higher than in the other treatments. However, in plants subjected to normal insect visitation (control), the num ber of seeds / pod, pod weight and seed weight / pod were higher than with manual pollination treatments, and even higher when compared to autogamic pollination. This lower productivity in terms of number of seeds / pod, as well as pod and seed weights obtained with manual pollination treatments can be attributed to low stigma receptivity at the moment of pollination (Muthugapatti et al., 1993). Stigmas are receptive for only two days (Free, 1993), while the intervals between manual pollination procedures were four days. Given that manual treatments were only applied to vigorous flowers, it is plausible to infer that certain flowers were not yet receptive. On the other hand, once pollen grains are deposited on yet unreceptive stigmas, their viability decreases with time, according to Bots and Mariani (2005). Therefore, any pollen deposited prior to stigma receptivity becomes less viable, affecting seed production.
High rates of A. mellifera visitations were recorded during the anthesis period. Due to this intense pollination, the flowers were most likely visited more than once during this period. Hence, it is also likely that pollen grains came in contact with the stigma during its receptive period. On the other hand, flowers under manual pollination treatments were only supplied once with pollen. Production losses in manual pollination processes can be attributed to insufficient pollen placement on flower stigmas (Kwak and Jennersten, 1991). The pollinating efficiency of A. mellifera is due to morphological characteristics that favor the transportation and deposition of large quantities of pollen on flowers (Abrol, 2007). The manual pollination procedures chosen were most likely not as efficient as honey bee pollination concerning the placement of pollen grains on flower stigmas.
All parameters concerning the production of pods and seeds in manually pollinated plants, especially in xenogamy treatments, were higher than those of the other treatments, except for the parameter "estimation of the number of seeds per plant" in geitonogamy, which was 1.7% below the estimate of the control (Table 2). Mussury and Fernandes (2000) found different results while working with the cultivar CTC-4 at Dourados, Mato Grosso do Sul State, Brazil, where spontaneous insect pollination (control) produced greater numbers of seeds per plant (51.5% and 77.9%) than manual treatments through geitonogamy and xenogamy, respectively. This variation may be related to the differences in pod development between the cultivars in question, the study methods applied, and the climatic conditions in the study regions.
The self-fertility index for B. napus Hyola 432, obtained through the autogamy test, indicated that 28.4% of ovules developed into seeds. However, the results for the other treatment, whether through spontaneous insect visits or manual pollination, indicated that pollination induction increased productivity, yielding up to 55.1% in ovule fertilization in plants of the xenogamy treatment. Seed productivity may be increased through the introduction of bees in the crop during the blooming period, given that seed numbers and weights were higher for plants treated through xenogamy or receiving spontaneous insect visits. However, honey bee foraging activities and, consequently, their pollinating potential, were limited by low temperatures in the study area , since the effective recording of bee visits always began at 12h00, when the temperature reached 12ºC (Woyke et al., 2003).
Many authors indicate ideal numbers of bee colonies needed to improve canola productivity, where recommendations vary, among other factors, according to locality, cultivar, plant density, number of flowers / inflorescence on each plant, blooming duration, internal colony conditions and number of flowers / hectare (Abrol, 2007). Abrol (2007) recommends the introduction of three to four colonies per hectare, while Sabbahi et al. (2005) states that it is important to insert at least three colonies per hectare in order to reach satisfactory productivity levels. Colonies must be placed according to their foraging radius, and pollinating activities decrease when this distance exceeds 0.5 km (Abrol, 2007). Manning and Wallis (2005) have found losses in productivity in areas even 0.2 km from established the apiaries. Manning and Boland (2004) indicated that the number of pods per B. napus plant (cv. Karoo) decreased as the distance between apiaries increased, observing that the pod set decreased by 16% at a distance of 1 km from the apiary. To establish the number of hives and the distance that crop must be from them, it is important that studies be carried out prior to planting, since there can be wide agrometeorological variations from one region to another. Agrometeorological factors possibly interfere in some way with the number of hives for a given crop, for a satisfactory yield.
There were two apiaries in the surrounding area of the canola crop studied, one with 20 colonies of A. mellifera (0.2 km) and another with 18 colonies, located at a distance of 1 km. Sabbahi et al. (2005) demonstrated that at least three colonies per hectare are necessary in order to obtain satisfactory productivity levels. Based on this study, 39 colonies were necessary in order to yield such results for the canola crop studied, since its area is 13 ha. Considering that there were 20 colonies already within a satisfactory foraging radius, it would be necessary to double this number to achieve increases in the production of B. napus pods and seeds.
As a conclusion, manual pollination increases B. napus productivity. Apis mellifera was the most frequent pollinator of B. napus flowers. Colonies of this insect should be introduced in order to increase the seed production of canola.
Acknowledgements
To Dr. Nídio Barni and Dr. Gilberto Tomm for their suggestions concerning the crop and the area of study, as well as Prof. Marcos Garrafa, Valdir Benedetti and Dalziro Valdameri for allowing and supporting field work at the Sociedade Educacional Três de Maio (SETREM, Três de Maio Educational Society). We are also grateful to Ione Maria de Souza Rosa, Ney Telles Ferreira Júnior, Andressa Paladini and Nadílson Roberto Ferreira for their support in field work, Prof. João Feliz de Moraes for his help with statistical analyses and Daniela Loose Ferreira for her suggestions during manuscript review. We thank CNPq (National Council for Scientific and Technological Development) for granting the master's scholarship to the first author and PUCRS for the infrastructure that allowed this research to be carried out.
Received August 18, 2009
Accepted July 13, 2010
- Abrol, D.P. 2007. Honeybees rapeseed pollinator plant interaction. Advances in Botanical Research 45: 337-369.
- Adegas, J.E.B.; Nogueira-Couto, R.H. 1992. Entomophilous pollination in rape (Brassica napus L. var oleifera) in Brazil. Apidologie 23: 203-209.
- Barni, N.D.; Hilgert, E.R.; Zanotelli, V.; Vargas, J.N.R.; Tedesco, A.; Bohn, D.; Gomes, J.E.S.; Gonçalves, J.C. 1985. Introduction and evaluation of cultivars of rapessed (Brassica napus L. var. oleifera Metzg.) in Rio Grande do Sul State. Agronomia Sulriograndense 21: 21-54. (in Portuguese).
- Bots, M.; Mariani, C. Pollen viability in the field. 2005. COGEM. Available at: http://www.cogem.net/ContentFiles/Pollen_viability.pdf [Accessed May 8, 2007]
- Cunha, G.R. 2007. Agroenergy: The Future that Arrived. O Nacional, Passo Fundo, RS ,. Brazil. (in Portuguese).
- D'Ávila, M.; Marchini, L.C. 2005. Pollination provided by bees in economically important crops in Brazil. Boletim da Indústria Animal 62: 79-90. (in Portuguese).
- Delaplane, K.S.; Mayer, D.F. 2000. Crop Pollination by Bees. CABI Publishing, Wallington, UK.
- Eisikowitch, D. 1981. Some aspects of pollination of oil seed rape (Brassica napus L.). Journal of Agricultural Science 96: 321-326.
- Free, J.B. 1993. Insect pollination of crops. Academic Press, London, UK.
- Kevan, G.; Eisikowith, D. 1990. The effects of insect pollination on canola (Brassica napus L. cv. O. A. C. Triton) of seed germination. Euphytica 45: 39-41.
- Klein, A.; Vaissiere, B.E.; Cane, J.H., Steffan-Dewenter, I.; Cunningham, S.A.; Kremen, C.; Tscharntke, T. 2007. Importance of pollinators in changing landscapes for world crops. Proceedings of the Royal Society 274: 303-313.
- Kwak, M.M.; Jennersten, O. 1991. Bumblebee visitation and seed set in Melampyrum pratense and Viscaria vulgaris: heterospecific pollen limitation. Oecologia 86: 99-104.
- Manning, R.; Boland, J. 2004. A preliminary investigation into honey bee (Apis mellifera) pollination of canola (Brassica napus cv. Karoo) in Western Australia. Australian Journal of Experimental Agriculture 40: 439-442.
- Manning, R.; Wallis, R. 2005. Seed yields in canola (Brassica napus cv. Karoo) depend on the distance of plants from honeybee apiaries. 2005. Australian Journal of Experimental Agriculture 45: 1307-1313.
- Marjanovic-Jeromela, A.; Marinkovic, R.; Mijic, A.; Zdunic, Z.; Ivanovska, S.; Jankulovska, M. 2008. Correlation and path analysis of quantitative traits in winter rapeseed (Brassica napus L.). Agriculturae Conspectus Scientificus 73: 13-18.
- Mussury, R.M.; Fernandes, W. 2000. Studies of the floral biolgy and reproductive system of Brassica napus L. (Cruciferae). Brazilian Archives of Biology and Technology 43: 111-117.
- Mussury, R.M.; Fernandes, W.D.; Scalon, S.P.Q. 2003. Activity of some insects in Brassica napus L. flowers at Dourados-MS and the interaction with climatic factors. Ciência e Agrotecnologia 27: 382-388. (in Portuguese, with abstract in English).
- Muthugapatti, K.K.; Thorsness, M.K.; Rundle, S.J.; Goldeberg, M.L.; Nasrallah, J.B.; Nasrallah, M.E. 1993. Ablation of papillar cell function in Brassica flowers results in the loss of stigma receptivity to pollination. The Plant Cell 5: 263-275.
- Sabbahi, R.; Oliveira, D.; Marceau, J. 2005. Influence of Honey Bee (Hymenopetra: Apidae) Density on the Production of Canola (Cruciferae: Brassicaceae). Journal of Economic Entomology 98: 367-372.
- Tomm, G.O. 2007. Technological indicatives for canola productivity in Rio Grande do Sul. Embrapa. Available at: http://www.cnpt.embrapa.br/culturas/canola/p_sp03_2007.pdf [Accessed Jan. 15, 2009]
- Westcott, L.; Nelson, D. 2001. Canola pollination: an update. Bee World 82: 115-129.
- Williams, I.; Martin, A.P.; White, R.P. 1986. The pollination requirementes of oil-seed rape (Brassica napus L.). Journal of Agricultural Science 106: 27-30.
- Woyke, J.; Wilde, J.; Wilde, M. 2003. Flight activity reaction to temperature changes in Apis dorsata, Apis laboriosa and Apis mellifera Journal of Apicultural Science 47: 73-80.
Publication Dates
-
Publication in this collection
31 Mar 2011 -
Date of issue
Apr 2011
History
-
Accepted
13 July 2010 -
Received
18 Aug 2009