Abstract
Water shortage is a major abiotic stress for crop production worldwide, limiting the productivity of crop species, especially in dry-land agricultural areas. This investigation aimed to classify the water-deficit tolerance in mutant rice (Oryza sativa L. spp. indica) genotypes during the reproductive stage. Proline content in the flag leaf of mutant lines increased when plants were subjected to water deficit. Relative water content (RWC) in the flag leaf of different mutant lines dropped in relation to water deficit stress. A decrease RWC was positively related to chlorophyll a degradation. Chlorophyll a , chlorophyll b , total chlorophyll , total carotenoids , maximum quantum yield of PSII , stomatal conductance , transpiration rate and water use efficiency in mutant lines grown under water deficit conditions declined in comparison to the well-watered, leading to a reduction in net-photosynthetic rate. In addition, when exposed to water deficit, panicle traits, including panicle length and fertile grains were dropped. The biochemical and physiological data were subjected to classify the water deficit tolerance. NSG19 (positive control) and DD14 were identified as water deficit tolerant, and AA11, AA12, AA16, BB13, BB16, CC12, CC15, EE12, FF15, FF17, G11 and IR20 (negative control) as water deficit sensitive, using Ward's method.
ward cluster analysis; drought tolerance; fertile grain; photosynthetic pigments; water relation
PLANT PHYSIOLOGY AND BIOCHEMISTRY
Water-deficit tolerant classification in mutant lines of indica rice
Suriyan Cha-umI, * * Corresponding author: < suriyanc@biotec.or.th> ; Suravoot YooyongwechII; Kanyaratt SupaibulwatanaIII
INational Center for Genetic Engineering and Biotechnology (BIOTEC), National Science and Technology Development Agency (NSTDA), 113 Thailand Science Park, Paholyothin Road, Klong 1, Klong Luang - 12120 - Pathumthani - Thailand
IIMahidol University/Faculty of Science and Technology - Dept. of Agricultural Science, Kanchanaburi Campus - 71150 - Kanchanaburi - Thailand
IIIMahidol University/Faculty of Science - Dept. of Biotechnology - 10400 - Payathai, Bangkok - Thailand
ABSTRACT
Water shortage is a major abiotic stress for crop production worldwide, limiting the productivity of crop species, especially in dry-land agricultural areas. This investigation aimed to classify the water-deficit tolerance in mutant rice (Oryza sativa L. spp. indica) genotypes during the reproductive stage. Proline content in the flag leaf of mutant lines increased when plants were subjected to water deficit. Relative water content (RWC) in the flag leaf of different mutant lines dropped in relation to water deficit stress. A decrease RWC was positively related to chlorophyll a degradation. Chlorophyll a , chlorophyll b , total chlorophyll , total carotenoids , maximum quantum yield of PSII , stomatal conductance , transpiration rate and water use efficiency in mutant lines grown under water deficit conditions declined in comparison to the well-watered, leading to a reduction in net-photosynthetic rate. In addition, when exposed to water deficit, panicle traits, including panicle length and fertile grains were dropped. The biochemical and physiological data were subjected to classify the water deficit tolerance. NSG19 (positive control) and DD14 were identified as water deficit tolerant, and AA11, AA12, AA16, BB13, BB16, CC12, CC15, EE12, FF15, FF17, G11 and IR20 (negative control) as water deficit sensitive, using Ward's method.
Keywords: ward cluster analysis, drought tolerance, fertile grain, photosynthetic pigments, water relation
Introduction
Water shortage is a major abiotic stress for crop production worldwide, limiting the productivity of crop species, especially in dry-land agricultural areas (> 1.2 billion hectares) (Chaves and Oliveira, 2004; Kijne, 2006; Passioura, 2007). Rice crop (Oryza sativa L.) improvement including conventional breeding program (Jongdee et al., 2006; Venuprasad et al., 2007; Kumar et al., 2008; Ndijiondjop et al., 2010; Guan et al., 2010) and mutant lines (Koh et al., 2007; Jiang et al., 2007; Cairns et al., 2009; Thang et al., 2010) is one of the strategies for water-deficit tolerant trait. Chemical and irradiation techniques are conventionally approached to induce mutation in a large population of mutant lines and pyramiding drought tolerant trait (Cairns et al., 2009; Thang et al., 2010). Plant biochemical changes, such as enhanced accumulation of stress metabolites and increased antioxidant enzymes are evidently expressed in plant responses to water deficit stress. Moreover, physiological characters include reduced relative water content, pigment degradation, decreased stomatal conductance, reduced internal CO2 concentration, net photosynthetic rate (Pn) reduction and growth inhibition prior to plant death have been well published (Chaves and Oliveira, 2004; Reddy et al., 2004; Cattivelli et al., 2008).
Plant biochemical and physiological changes in crop species in response to water deficit stress have been implemented as criteria for screening water-deficit tolerance in plant breeding programs (Ashraf and Foolad, 2007; Ashraf, 2010). Yield traits are the most important criteria for water-deficit tolerance screening (Fukai et al., 1999; Yang et al., 2001; Pantuwan et al., 2002; Kumar et al., 2008). Some previous studies suggested that multivariate cluster analysis is practically required for water-deficit classification in rice breeding programs (Cabuslay et al., 2002; Cha-um et al., 2010). In Asian countries, rice plays a key role as major carbohydrate crop. It is a basal food, feeding more than 3 billion people and providing 50-80 % of their daily calorie intake (Khush, 2005). In rainfed paddy fields, water shortage has been well known as being a serious issue, especially in the reproductive stage, during which plants are particularly sensitive, leading to low crop yield (Fukai et al., 1999; Pantuwan et al., 2002; Bouman et al., 2006). In the genetic resources, NSG19 is a positive check of water-deficit tolerance, which is utilized in drought tolerance screening, whereas IR20 is a negative check (Mitchell et al., 1998; Wade et al., 1999; Pantuwan et al., 2002; Uyprasert et al., 2004; Kumar et al., 2006). This investigation aimed to classify the water-deficit tolerance in mutant rice genotypes during the reproductive stage.
Materials and Methods
M4-seeds of twelve mutant rice cultivars, MT 4-01 (code AA11), MT 4-02 (code AA12), MT 4-03 (code AA16), MT 4-04 (code BB13), MT 4-05 (code BB16), MT 4-06 (code CC12), MT 4-07 (code CC15), MT 4-08 (code DD14), MT 4-09 (code EE12), MT 4-10 (code FF15), MT 4-11 (code FF17) and MT 4-12 (code G11), derived from Γ-irradiation and ethyl methane sulfonate (EMS) mutagens of jasmine rice (Oryza sativa L. ssp. indica cv. KDML 105) were germinated and transplanted to pots containing clay soil (EC = 2.687 dS m-1; pH = 5.5; organic matter = 10.36 %; total nitrogen = 0.17 %; total phosphorus = 0.07 %; total potassium = 1.19 %) in 50 % shading (acclimatization) light intensity and grown on for 2 weeks. The pots were arranged on plastic trays (30 × 45 cm). Water irrigation was supplied using a moisture spray. Acclimatized plants were transferred directly to water-flooded pots (15 cm in diameter × 30 cm in height) containing clay soil. The experiment site was located at Klong Luang, Pathumthani, Thailand (Latitude 14°01'12" N and Longitude 100°31'12" E) and conducted between Aug. and Nov. 2010. In the booting stage [85 days after sowing (DAS)], soil water content (SWC) was adjusted to 56 % (WW; full irrigation or well-watered) and 7 % (WD; 14 days withholding irrigation or water-deficit). The SWC was calculated using the weight fraction: SWC (%) = [(FW-DW)/DW] × 100, where FW was the fresh weight of a soil portion of the internal area of each pot and DW was the dry weight of the soil portion after drying in a hot air oven at 85 °C for 4 days (Coombs et al., 1987). Relative water content (RWC), proline content in the leaf blade, photosynthetic pigments, chlorophyll fluorescence, net-photosynthetic rate (Pn), transpiration rate (E), stomatal conductance (gs) in flag leaf and panicle traits in rice plants were measured.
Relative water content (RWC) was calculated according to Bonnet et al. (2000). This parameter was calculated from fresh weight (FW) dry weight (DW) and turgid weight (TW) following the equation; RWC (%) = [(FW-DW)/(TW-DW)] × 100.
Proline in the root and leaf tissues was extracted and analyzed according to the method of Bates et al. (1973). Fifty milligrams of fresh material was ground with liquid nitrogen in a mortar. The homogenate powder was mixed with 1 mL aqueous sulfosalicylic acid (3 % w/v) and filtered through filter paper (Whatman #1, England). The extracted solution was reacted with an equal volume of glacial acetic acid and ninhydrin reagent (1.25 mg ninhydrin in 30 mL glacial acetic acid and 20 mL 6 M H3PO4) and incubated at 95 °C for 1 h. The reaction was terminated by placing the container in an ice bath. The reaction mixture was mixed vigorously with 2 mL toluene. After cooling to 25 °C, the chromophore was measured by spectrophotometer (HACH DR/4000; Model 48000, HACH Company, Loveland, Colorado, USA) at 520 nm using L-proline as a standard.
Chlorophyll a (Chla), chlorophyll b (Chlb) and total chlorophyll (TC) content, were analyzed following the methods of Shabala et al. (1998) and total carotenoid (Cx+c) concentrations were assayed according to Lichtenthaler (1987). One hundred milligrams of leaf material was collected and placed in a 25 mL glass vial along with 10 mL 95.5 % acetone, and blended using a homogenizer. The glass vials were sealed with parafilm to prevent evaporation, and then stored at 4 °C for 48 h. Chla and Chlb concentrations were measured using a UV-visible spectrophotometer at 662 nm and 644 nm wavelengths. The Cx+c concentration was also measured by spectrophotometer (HACH DR/4000; Model 48000, HACH Company, Loveland, Colorado, USA) at 470 nm. A solution of 95.5 % acetone was used as a blank.
Chlorophyll fluorescence emission from the adaxial surface on the leaf was measured using a fluorescence monitoring system (FMS 2; Hansatech Instruments Ltd., Norfolk, UK) in the pulse amplitude modulation mode, as previously described by Loggini et al. (1999). A leaf, adapted to dark conditions for 30 min using leaf-clips, was initially exposed to the modulated measuring beam of far-red light (LED source with typical peak at wavelength 735 nm). Original (F0) and maximum (Fm) fluorescence yields were measured under weak modulated red light (< 0.5µmol m-2 s-1) with 1.6 s pulses of saturating light (> 6.8µmol m-2 s-1 PAR) and calculated using FMS software for WindowsÒ. The variable fluorescence yield (Fv) was calculated by the equation of Fm-F0. The ratio of variable to maximum fluorescence (Fv/Fm) was calculated as maximum quantum yield of PSII photochemistry.
Net photosynthetic rate (Pn; µmol m-2 s-1), transpiration rate (E; mmol m-2 s-1), stomatal conductance (gs; mmol H2O m-2 s-1) and water use efficiency (WUE; %) were measured using a Portable Photosynthesis System (Model LI 6400, LI-CORÒ Inc, Lincoln, Nebraska, USA) with an Infra-red Gas Analyser following Cha-um et al. (2007). WUE was calculated according to equation: WUE (%) = [Pn/E] × 100. Panicle length, fertile grains, sterile grains, total grains and one-hundred seed weight per panicle in the well-watered (WW) or water deficit (WD) were measured. Yield loss, RWC and WUE abilities, proline accumulation, Pn reduction, Fv/Fm diminution, and pigment degradation were calculated following Cha-um et al. (2009).
The experiment was arranged as 12 × 2 factorials in Completely Randomized Block Design (CRBD) with eight replicates (n = 8). The mean values obtained were compared using Duncan's New Multiple Range Test (DMRT) and analyzed with SPSS software. Yield loss, RWC and WUE abilities, proline accumulation, Pn reduction, Fv/Fm diminution, and pigment degradation in the flag leaves of rice grown under severe water deficit stress were assessed in order to classify cultivars as either tolerant or sensitive using Ward's method of Hierarchical cluster analysis in SPSS software.
Results and Discussion
Proline content in flag leaf of water-deficit stressed plant was increased (p < 0.01) and higher than that in controlled plant for 1.3-3.5 folds. For example, proline in AA11 and CC15 grown under water deficit stress was accumulated for 3.5 and 3.4 folds, respectively when compared with those in well-water condition (Figure 1). Relative water content (RWC) in the flag leaf of mutant rice lines dropped in plants exposed to water deficit stress (Table 1). The reduction of RWC in AA16 and CC15 lines of mutant rice was 15.4 % and 14.6 % when subjected to water deficit stress. A positive correlation between RWC and chlorophyll content was demonstrated (Figure 2).
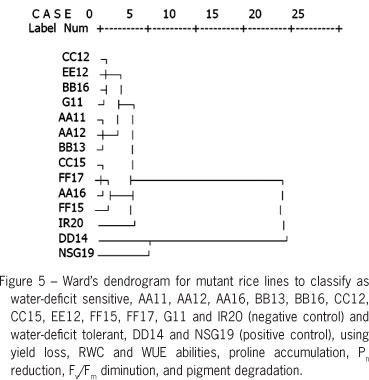
Photosynthetic pigments, chlorophyll a (Chla), chlorophyll b (Chlb), total chlorophyll a (TC) and total carotenoids (Cx+c) in rice crop were decreased when subjected to water deficit stress (Table 1). In DD14 mutant lines, Chla content in water-deficit stressed plants was maintained (Table 1). The degradation percentage of Chla (5.1 %) and TC (27.4 %) in DD14 plants subjected to water-deficit was lower than in other rice lines (65.1-71.7 %). Degradation of Chla pigments was positively correlated to maximum quantum yield of PSII (Fv/Fm) (Figure 3), leading to reduced net photosynthetic rate (Pn) (Figure 4). Photosynthetic abilities including Fv/Fm, Pn, stomatal conductance (gs) and transpiration rate (E) drastically decreased when exposed to water deficit conditions (Table 2). In addition, water use efficiency (WUE) was similarly trended, except in AA12, BB13, BB16, CC12 and CC15 was alleviated (16.9, 24.4, 8.3, 27.6 and 11.1 % reduction). Panicle length in rice lines grown under water deficit stress showed no differences, except in CC12 (25.4 % reduction) and FF15 (35.0 % reduction). Number of fertile grains decreased (p < 0.01) when plants subjected to water-deficit conditions whereas number of sterile grains was increased (Table 3). A thousand-grain weight was unchanged when subjected to water deficit except in lines, BB13 and FF15. In addition, yield failure, RWC and WUE abilities, proline accumulation, Pn reduction, Fv/Fm diminution, and pigment degradation data were subjected to classify clusters of the group as water deficit tolerant, NSG19, DD14, and water deficit sensitive, AA11, AA12, AA16, BB13, BB16, CC12, CC15, EE12, FF15, FF17, G11 and IR20 using Ward's method (Figure 5).
Proline content in the flag leaf blade increased depending on the reduction of RWC in the leaf tissues. Proline accumulation in the rice crop has been well established as an effective indicator of plant responses to water deficit stress. For example, proline content in PT1 and IR20 (water deficit sensitive) peaked in plants subjected to severe water deficit (7 % SWC) and was higher than in the tolerant cultivars (KDML105 and NSG19) (Cha-um et al., 2010). Similarly, proline in the leaf tissues of drought sensitive, CR203 (3.12 times) is accumulated higher than in drought tolerant cultivars, DR2 (1.41 times) and Cuom (2.06 times), when plants are subjected to water deficit stress (Hien et al., 2003). In contrast, P5CR and P5CS genes in proline biosynthesis in drought tolerant (Ca/H680) are up-regulated resulting in enriched proline contents greater than those in drought sensitive (Ca/H148) when exposed to drought stress (Parida et al., 2008). So, proline accumulation in rice crop responses to drought stress is still unclear and unsuitable to play a critical role as good indicator. Relative water content (RWC) in mutant lines of rice had a similar decreasing trend when subjected to water deficit stress. For some rice lines (AA16 and CC15), RWC in the flag leaf tissues was maintained for 84.6 % and 85.4 %, respectively when exposed to water deficit stress. In Gangyon 527, Yixiangyou 9 and Gangyou 188 cultivars of rice, RWC in the leaves decreased depending on a degree of water deficit stress (Wang et al., 2010). In addition, the RWC in drought tolerant Ca/H 680 was maintained at higher levels than that in drought sensitive Ca/H 148 when subjected to drought conditions (Parida et al., 2008). However, only RWC in the leaf tissues is inappropriate to make a decision for the drought tolerance in rice cultivar (Pantuwan et al., 2002; Jongdee et al., 2006).
A reduction of RWC in the flag leaf was positively related to pigment degradation. In the present study, Chla and TC degradation percentage in "DD14" mutant lines in water deficit condition was lower than other lines. Similar results are presented in NSG19 and KDML105 grown under water deficit stress (Cha-um et al., 2010). Also, the photosynthetic abilities i.e. chlorophyll fluorescence and net photosynthetic rate (Pn) in drought tolerant genotype (NSG19) are also better than in drought sensitive genotype (IR20). For example, the Pn in the flag leaf of NSG19 was reduced in 19.5 % whereas Pn of IR20 was reduced in 64.3 % (Cha-um et al., 2010). In this investigation, two rice genotypes were chosen as positive (drought tolerance) and negative control (drought sensitive). Moreover, relative transpiration in NSG19 (0.35) and IR20 (0.48) quite differ when plants are subjected to water deficit (PEG-induced -0.5 MPa) for 6 days (Cabuslay et al., 2002).
The normalized transpiration rate (NTR) in three modern rice, BRS Primavera, BRSMG Curinga and BRS Soberana and one traditional rice, Douradão has been investigated as indicator in plant responses to the fraction of transpirable soil water (FTSW), which is identified the p factor (at 0.95 NTR) as the adaptability of rice genotypes to drought prone environments (Heinemann et al., 2011). In previous publications, the plant morphological characters i.e. leaf rolling, leaf chlorosis and green leaf area have been well established (Cabuslay et al., 2002; Bernier et al., 2008). Moreover, yield traits, such as grain yield, productivity and grain sterility are the most popular parameters used to identify water deficit tolerance in rice breeding programs (Fukai et al., 1999; Yang et al., 2001; Pantuwan et al., 2002; Kumar et al., 2008; Venuprasad et al., 2008; Wang et al., 2010). Multivariate indices including yield failure, RWC and WUE abilities, proline accumulation, Pn reduction, Fv/Fm diminution, and pigment degradation data have been employed to classify members of water deficit tolerance, NSG19 and DD14. In addition, water-deficit tolerance classification in rice crop using multivariate parameters has been well established (Cubusley et al., 2002; Jongdee et al., 2006; Cha-um et al., 2010).
In conclusion, chlorophyll a, total chlorophyll, total carotenoids and fertile grain traits of mutant rice genotypes "DD14" in response to water deficit stress were superior to those in other cultivars and can be played as multivariate criteria for water-deficit tolerance classification. Mutant rice line "DD14" was identified as water deficit tolerance using Ward's cluster analysis. The water-deficit tolerant line of rice from this investigation should be further verified in the rainfed paddy field.
Acknowledgments
To Mahidol University, as a funding source (Grant code A04/1/2553), and to Jonathan Shore, for English grammatical proofing.
Received February 10, 2011
Accepted October 26, 2011
Edited by: Antonio Costa de Oliveira
- Ashraf, M. 2010. Inducing drought tolerance in plants: recent advances. Biotechnology Advance 28: 169-183.
- Ashraf, M.; Foolad, M.R. 2007. Role of glycine betaine and proline in improving plant abiotic stress resistance. Environmental and Experimental Botany 59: 206-216.
- Bates, L.S.; Waldren, R.P.; Teare, I.D. 1973. Rapid determination of free proline for water-stress studies. Plant and Soil 39: 205-207.
- Bernier, J.; Atlin, G.N.; Serraj, R.; Kumar, A.; Spaner, D. 2008. Breeding upland rice for drought resistance. Journal of the Science of Food and Agriculture 88: 927-939.
- Bonnet, M.; Camares, O.; Veisseire, P. 2000. Effect of zinc and influence of Acremonium lolii on growth parameters, chlorophyll a fluorescence and antioxidant enzyme activities of ryegrass (Lolium perenne L. cv Apollo). Journal of Experimental Botany 51: 945-953.
- Bouman, B.A.M.; Humphreys, E.; Tuong, T.P.; Barker, R. 2006. Rice and water. Advances in Agronomy 92: 187-237.
- Cabuslay, G.S.; Ito, O.; Alejar, A.A. 2002. Physiological evaluation of responses of rice (Oryza sativa L.) to water deficit. Plant Science 163: 815-827.
- Cairns, J.E.; Acuna, T.L.B.; Simborio, F.A.; Dimayuga, G.; Praba, M.L.; Leung, H.; Torres, R.; Lafitte, H.R. 2009. Identification of deletion mutants with improved performance under water-limited environments in rice (Oryza sativa L.). Field Crops Research 114: 159-168.
- Cattivelli, L.; Rizza, F.; Badeck, F.W.; Mazzucotelli, E.; Mastrangelo, A.N.; Francia, E.; Marè, C.; Tondelli, A.; Stanca, A.M. 2008. Drought tolerance improvement in crop plants: An integrated view from breeding to genomics. Field Crops Research 105: 1-14.
- Cha-um, S.; Supaibulwatana, K.; Kirdmanee, C. 2007. Glycinebetaine accumulation, physiological characterizations and growth efficiency in salt tolerant and salt sensitive lines of indica rice (Oryza sativa L. spp. indica) response to salt stress. Journal of Agronomy and Crop Science 193: 157-166.
- Cha-um, S.; Boriboonkaset, T.; Pichakum, A.; Kirdmanee, C. 2009. Multivariate physiological indices for salt tolerant classification in indica rice (Oryza sativa L. spp. indica). General and Applied Plant Physiology 35: 75-87.
- Cha-um, S.; Yooyongwech, S.; Supaibulwatana, K. 2010. Water deficit stress in the reproductive stage of four indica rice (Oryza sativa L.) genotypes. Pakistan Journal of Botany 42: 3387-3398.
- Chaves, M.M.; Oliveira, M.M. 2004. Mechanisms underlying plant resilience to water deficits: prospects for water-saving agriculture. Journal of Experimental Botany 55: 2365-2384.
- Coombs, J.; Hall, D.O.; Long, S.P.; Scurlock, J.M.O. 1987. Techniques in Bioproductivity and Photosynthesis. Pergamon, Oxford, UK.
- Fukai, S.; Pantuwan, G.; Jongdee, B.; Cooper, M. 1999. Screening for drought resistance in rainfed lowland rice. Field Crops Research 64: 61-74.
- Guan, Y.S.; Serraj, R.; Liu, S.H.; Xu, J.L.; Ali, J.; Wang, W.S.; Venus, E.; Zhu, L.H.; Li, Z.K. 2010. Simultaneously improving yield under drought stress and non-stress conditions: a case study of rice (Oryza sativa L.). Journal of Experimental Botany 61: 4145-4156.
- Heinemann, A.B.; Stone, L.F.; Fageria, N.K. 2011. Transpiration rate response to water deficit during vegetative and reproductive phases of upland rice cultivars. Scientia Agricola 68: 24-30.
- Hien, D.T.; Jacobs, M.; Angenon, G.; Hermans, C.; Thu, T.T.; Son, L.V.; Roosens, N.H. 2003. Proline accumulation and ∆1-pyrroline-5-carboxylate synthetase gene properties in three rice cultivars differing in salinity and drought tolerance. Plant Science 165: 1059-1068.
- Jiang, S.Y.; Bachmann, D.; La, H.; Ma, Z.; Venkatesh, P.N.; Ramamoorthy, R.; Ramachandran, S. 2007. Ds insertion mutagenesis as an efficient tool to produce diverse variation for rice breeding. Plant Molecular Biology 65: 385-402.
- Jongdee, B.; Pantuwan, G.; Fukai, S.; Fischer, K. 2006. Improving drought tolerance in rainfed lowland rice: an example from Thailand. Agricultural Water Management 80: 223-240.
- Khush, G.S. 2005. What it will take to feed 5.0 billion rice consumers in 2030. Plant Molecular Biology 59: 1-6.
- Kijne, J.W. 2006. Abiotic stress and water scarcity: identifying and resolving conflicts from plant level to global level. Field Crops Research 97: 3-18.
- Koh, S.; Lee, S.C.; Kim, M.K.; Koh, J.H.; Lee, S.; An, G.; Choe, S.; Kim, S.R. 2007. T-DNA tagged knockout mutation of rice OsGSK1, an orthologue of Arabidopsis BIN2, with enhanced tolerance to various abiotic stresses. Plant Molecular Biology 65: 453-466.
- Kumar, A.; Bernier, J.; Verulkar, S.; Lafitte, H.R.; Atlin, G.N. 2008. Breeding for drought tolerance: direct selection for yield, response to selection and use of drought-tolerant donors in upland and lowland-adapted populations. Field Crops Research 107: 221-231.
- Kumar, R.; Sarawgi, A.K.; Ramos, C.; Amarante, S.T.; Ismail, A.M.; Wade, L.J. 2006. Partitioning of dry matter during drought stress in rainfed lowland rice. Field Crops Research 98: 1-11.
- Lichtenthaler, H.K. 1987. Chlorophylls and carotenoids: pigments of photosynthetic biomembranes. Methods in Enzymology 148: 350-380.
- Loggini, B.; Scartazza, A.; Brugnoli, E.; Navari-Izzo, F. 1999. Antioxidant defense system, pigment composition, and photosynthetic efficiency in two wheat cultivars subjected to drought. Plant Physiology 119: 1091-1099.
- Mitchell, J.H.; Siamhan, D.; Wamala, M.H.; Risimeri, J.B.; Chinyamakobvu, E.; Henderson, S.A.; Fukai, S. 1998. The use of seedling leaf date score for evaluation of drought resistance of rice. Field Crops Research 55: 129-139.
- Ndijiondjop, M.N.; Manneh, B.; Cissoko, M.; Drame, N.K.; Kakai, R.G.; Bocco, R.; Baimey, H.; Wopereis, M. 2010. Drought resistance in an interspecific backcross population of rice (Oryza spp.) derived from the cross WAB56-104 (O. sativa) × CG14 (O. glaberrima). Plant Science 179: 364-373.
- Pantuwan, G.; Fukai, S.; Cooper, M.; Rajatasereekul, S.; O'Toole, J.C. 2002. Yield response of rice (Oryza sativa L.) genotypes to drought under rainfed lowlands. 2. Selection of drought resistant genotypes. Field Crops Research 73: 169-180.
- Parida, A.K.; Dagaonkar, V.S.; Phalak, M.S.; Aurangabadkar, L.P. 2008. Differential responses of the enzymes involved in proline biosynthesis and degradation in drought tolerant cotton genotypes during drought stress and recovery. Acta Physiologae Plantarum 30: 619-627.
- Passioura, J. 2007. The drought environment: physical, biological and agricultural perspectives. Journal of Experimental Botany 58: 113-117.
- Reddy, A.R.; Chaitanya, K.V.; Vivekanandan, M. 2004. Drought-induced responses of photosynthesis and antioxidant metabolism in higher plants. Journal of Plant Physiology 161: 1189-1202.
- Shabala, S.N.; Shabala, S.I.; Martynenko, A.I.; Babourina, O.; Newman, I.A. 1998. Salinity effect on bioelectric activity, growth, Na+ accumulation and chlorophyll fluorescence of maize leaves: a comparative survey and prospects for screening. Australian Journal of Plant Physiology 25: 609-616.
- Thang, N.B.; Wu, J.; Zhou, W.; Shi, C. 2010. The screening of mutants and construction of mutant library for Oryza sativa cv. Nipponbare via ethyl methane sulphonate inducing. Biologia 65: 660-669.
- Uyprasert, S.; Toojinda, T.; Udomprasert, N.; Tragoonrung, S.; Vanavichit, A. 2004. Proline accumulation and rooting pattern in rice in response to water deficit under rainfed lowlands. Science Asia 30: 301-311.
- Venuprasad, R.; Lafitte, H.R.; Atlin, G.N. 2007. Response to direct selection for grain yield under drought stress in rice. Crop Science 47: 285-293.
- Venuprasad, R.; Cruz, M.T.S.; Amante, M.; Magbanua, R.; Kumar, A.; Atlin, G.N. 2008. Response to two cycles of divergent selection for grain yield under drought stress in four rice breeding populations. Field Crops Research 107: 232-244.
- Wade, L.J.; McLaren, C.G.; Quintana, L.; Harnpichitvitaya, D.; Rajatasereekul, S.; Sarawgi, A.K.; Kumar, A.; Ahmed, H.U.; Sarwoto; Singh, A.K.; Rodriguez, R.; Siopongco, J.; Sarkarung, S. 1999. Genotype by environment interactions across diverse rainfed lowland rice environments. Field Crops Research 64: 35-50.
- Wang, H.; Zhang, L.; Ma, J.; Li, X.; Li, Y.; Zhang, R.; Wang, R. 2010. Effects of water stress on reactive oxygen species generation and protection system in rice during grain-filling stage. Agricultural Science in China 9: 633-641.
- Yang, J.; Zhang, J.; Wang, Z.; Zhu, Q.; Wang, W. 2001. Remobilization of carbon reserves in response to water deficit during grain filling of rice. Field Crops Research 71: 47-55.
Publication Dates
-
Publication in this collection
29 Feb 2012 -
Date of issue
Apr 2012
History
-
Received
10 Feb 2011 -
Accepted
26 Oct 2011