Abstract
Ammonia (NH3) emissions from broiler production can affect human and animal health and may cause acidification and eutrophication of the surrounding environment. This study aimed to estimate ammonia emissions from broiler litter in two systems of forced ventilation, the tunnel ventilation (TV) and the dark house (DH). The experiment was carried out on eight commercial broiler houses, and the age of the birds (day, d), pH and litter temperature were recorded. Broilers were reared on built-up wood shaving litter using an average flock density of 14 bird m–2. Temperature and relative humidity inside the broiler houses were recorded in the morning during the grow-out period. A factorial experimental design was adopted, with two types of houses, four replicates and two flocks with two replicates each. A deterministic model was used to predict ammonia emissions using the litter pH and temperature, and the day of grow-out. The highest litter temperature and pH were found at 42 d of growth in both housing systems. Mean ambient air temperature and relative humidity did not differ in either system. Mean model predicted ammonia emission was higher in the DH rearing system (5200 mg NH3 m−2h−1 at 42 d) than in the TV system (2700 mg NH3m−2 h−1 at 42 d). TV presented the lowest mean litter temperature and pH at 42 d of growth. In the last week of the broilers’ grow-out cycle, estimated ammonia emissions inside DH reached 5700 mg m−2h−1 in one of the flocks. Ammonia emissions were higher inside DH, and they did not differ between flocks. Assuming a broiler market weight in Brazil of close to 2 kg, ammonia emissions were equivalent to 12 g NH3 bird-marketed−1. Model-predicted ammonia emissions provided comprehensible estimations and might be used in abatement strategies for NH3 emission.
noxious gases; environmental impact; dark house; poultry; tunnel ventilation
Introduction
Agriculture contributes significantly to the anthropogenic emissions of ammonia and greenhouse gases (UNFCCC, 1997UNFCCC. 1997. Kyoto Protocol to the United Nations Framework. Convention on Climate Change. Available at: http://unfccc.int/2860.php [Accessed Mar 30, 2014].
http://unfccc.int/2860.php...
; Beusen et al., 2008Beusen, A.H.W.; Bouwman, A.F.; Heuberger, P.S.C.; Drecht, G. van; Hoek, K.W. van der. 2008. Bottom-up uncertainty estimates of global ammonia emissions from global agricultural production systems. Atmospheric Environment 42: 6067-6077.). As the world's demand for food from livestock production increases, so do the effects of ammonia on rearing surroundings and the atmosphere (Bouwman et al., 1997Bouwman, A.F.; Lee, D.S.; Asman, W.A.H.; Dentener, F.J.; Hoek, K.W. van der; Oliver, G.J. 1997. A global high-resolution emission inventory for ammonia. Global Biogeochemical Cycles 11: 561-587.). Ammonia (NH3) emission is the process of NH3emission from litter to air, and it may cause eutrophication in the environment (UNFCCC, 1997)UNFCCC. 1997. Kyoto Protocol to the United Nations Framework. Convention on Climate Change. Available at: http://unfccc.int/2860.php [Accessed Mar 30, 2014].
http://unfccc.int/2860.php...
. Ammonia emissions from poultry houses are a function of the NH3 concentration inside the housing, which is related to rearing flock density, quality of litter, birds’ diet and ventilation system (Gates et al., 2005Gates, R.S.; Xin, H.; Casey, K.D.; Liang, Y.; Wheeler, E.F. 2005. Method for measuring ammonia emissions from poultry houses. Journal of Applied Poultry Research 14: 622-634.;Ndegwa et al., 2008Ndegwa, P.M.; Hristov, A.N.; Arogo, J.; Sheffield, R.E. 2008. A review of ammonia emission mitigation techniques for concentrated animal feeding operations. Biosystems Engineering 100: 453-469.; Harper et al., 2010Harper, L.A.; Flesch, T.K.; Wilson, J.D. 2010. Ammonia emissions from broiler production in the San Joaquin Valley. Poultry Science 89: 1802-814.; Menegali et al., 2012)Menegali, I.; Tinôco, I.F.F.; Zolnier, S.; Carvalho, C.C.S.; Guimarães, M.C.C. 2012. Influence of different systems of minimum ventilation on air quality in broiler houses. Engenharia Agrícola 32: 1024-1033..
Ammonia emission estimates are needed to assess the broiler industry's impact on local and regional air quality, particularly in countries that are cosigners of the Kyoto protocol. There are limited scientific estimates of NH3 emissions from Brazilian broiler facilities. Broiler houses are similar throughout the country. However, the differences in building dimensions and ventilation systems may affect the environmental control of changing the NH3 emission rate (Wheeler et al., 2006Wheeler, E.F.; Casey, K.D.; Gates, R.; Xin, H.; Zajaczkowski, J.L.; Topper, P.A.; Liang, Y.; Pescatore, A.J. 2006. Ammonia emissions from twelve U.S. broiler chicken houses. Transactions of the ASABE 49: 1495-1512.; Gates et al., 2008Gates, R.S.; Casey, K.D.; Wheeler, E.F.; Xin, H.; Pescatore, A.J. 2008. US broiler housing ammonia emissions inventory. Atmospheric Environment 42: 3342-3350.; Ngwabie et al., 2010)Ngwabie, N.M.; Jeppsson, K.H.; Gustafsson, G.; Nimmermark, S. 2010. Influence of environmental factors and the addition of wood shavings on ammonia and odour emissions from fresh livestock manure. Agricultural Engineering International: CIGR Journal 12: 68-80.. The availability of credible NH3 emission measurements is very limited due to the high number of variables that influence emissions from broiler housings (Gates et al., 2008Gates, R.S.; Casey, K.D.; Wheeler, E.F.; Xin, H.; Pescatore, A.J. 2008. US broiler housing ammonia emissions inventory. Atmospheric Environment 42: 3342-3350.; Snoek et al., 2014)Snoek, D.J.W.; Stigter, J.D.; Oginkc, N.W.M.; Groot Koerkamp, P.W.G. 2014. Sensitivity analysis of mechanistic models for estimating ammonia emission from dairy cow urine puddles. Biosystems Engineering 121: 12-24.. Full-scale in situ measurements are costly and time-consuming, and their accuracy might be compromised by variations in the final broiler market weight (Gates et al., 2008)Gates, R.S.; Casey, K.D.; Wheeler, E.F.; Xin, H.; Pescatore, A.J. 2008. US broiler housing ammonia emissions inventory. Atmospheric Environment 42: 3342-3350..
Field estimations of poultry NH3 emissions have already been calculated in some countries (Groot Koerkamp et al., 1998Groot Koerkamp, P.W.G.; Metz, J.H.M.; Uenk, G.H.; Phillips, V.R.; Holden, M.R.; Sneath, R.W.; Short, J.L.; White, R.P.P.; Hartung, J.; Seedorf, J.; Schröder, M.; Linkert, K.H.; Pedersen, S.; Takai, H.; Johnsen, J.O.; Wathes, C.M. 1998. Concentrations and emissions of ammonia in livestock buildings in Northern Europe Journal of Agricultural Engineering Research 70: 79-95.;Demmers et al., 1999Demmers, T.G.M.; Burgess, L.R.; Short, J.L.; Phillips, V.R.; Clark, J.A.; Wathes, C.M. 1999. Ammonia emissions from two mechanically ventilated UK livestock buildings. Atmospheric Environment 33: 217-227.; Guiziou and Béline, 2005Guiziou, F.; Béline, F. 2005. In situ measurement of ammonia and greenhouse gas emissions from broiler houses in France. Bioresource Technology 96: 203-207.; Pescatore et al., 2005Pescatore, A.J.; Casey, K.D.; Gates, R.S. 2005. Ammonia emissions from broiler houses. Journal of Applied Poultry Research 14: 635-637.; Wheeler et al., 2006Wheeler, E.F.; Casey, K.D.; Gates, R.; Xin, H.; Zajaczkowski, J.L.; Topper, P.A.; Liang, Y.; Pescatore, A.J. 2006. Ammonia emissions from twelve U.S. broiler chicken houses. Transactions of the ASABE 49: 1495-1512.; Gates et al., 2008Gates, R.S.; Casey, K.D.; Wheeler, E.F.; Xin, H.; Pescatore, A.J. 2008. US broiler housing ammonia emissions inventory. Atmospheric Environment 42: 3342-3350.;Harper et al., 2009Harper, L.A.; Flesch, T.K.; Powell, J.M.; Coblentz, W.K.; Jokela, W.E.; Martin, N.P. 2009. Ammonia emissions from dairy production in Wisconsin. Journal of Dairy Science 92: 2326-2337.; Calvet et al., 2011Calvet, S.; Cambra-López, M.; Estellés, F.; Torres, A.G. 2011. Characterization of gas emissions from a Mediterranean broiler farm. Poultry Science 90: 534-542.). Emissions per bird are suggested to be correlated with bird age and litter condition (Groot Koerkamp et al., 1998Groot Koerkamp, P.W.G.; Metz, J.H.M.; Uenk, G.H.; Phillips, V.R.; Holden, M.R.; Sneath, R.W.; Short, J.L.; White, R.P.P.; Hartung, J.; Seedorf, J.; Schröder, M.; Linkert, K.H.; Pedersen, S.; Takai, H.; Johnsen, J.O.; Wathes, C.M. 1998. Concentrations and emissions of ammonia in livestock buildings in Northern Europe Journal of Agricultural Engineering Research 70: 79-95.; Wheeler et al., 2006Wheeler, E.F.; Casey, K.D.; Gates, R.; Xin, H.; Zajaczkowski, J.L.; Topper, P.A.; Liang, Y.; Pescatore, A.J. 2006. Ammonia emissions from twelve U.S. broiler chicken houses. Transactions of the ASABE 49: 1495-1512.; Gates et al., 2008Gates, R.S.; Casey, K.D.; Wheeler, E.F.; Xin, H.; Pescatore, A.J. 2008. US broiler housing ammonia emissions inventory. Atmospheric Environment 42: 3342-3350.), but the results are highly variable (Wheeler et al., 2006Wheeler, E.F.; Casey, K.D.; Gates, R.; Xin, H.; Zajaczkowski, J.L.; Topper, P.A.; Liang, Y.; Pescatore, A.J. 2006. Ammonia emissions from twelve U.S. broiler chicken houses. Transactions of the ASABE 49: 1495-1512.; Casey et al., 2010Casey, K.D.; Gates, R.S.; Shores, R.C.; Thomas, D.; Harris, D.B. 2010. Ammonia emissions from a U.S. broiler house-comparison of concurrent measurements using three different technologies. Journal of the Air & Waste Management Association 60: 939-48.). Mechanistic and deterministic algorithms have been used to estimate NH3 emissions from animal housing under particular circumstances (Bouwman et al., 1997Bouwman, A.F.; Lee, D.S.; Asman, W.A.H.; Dentener, F.J.; Hoek, K.W. van der; Oliver, G.J. 1997. A global high-resolution emission inventory for ammonia. Global Biogeochemical Cycles 11: 561-587.; Miragliotta et al., 2004Miragliotta, M.Y.; Nääs, I.A.; Murayama, M.C.; Moura, D.J. 2004. Software for estimating ammonia emission in broilers housing. Revista Brasileira de Agroinformática 6: 79-89 (in Portuguese, with abstract in English).; Gates et al., 2005Gates, R.S.; Xin, H.; Casey, K.D.; Liang, Y.; Wheeler, E.F. 2005. Method for measuring ammonia emissions from poultry houses. Journal of Applied Poultry Research 14: 622-634.; Siefert and Scudlark, 2008Siefert, R.L.; Scudlark, J.R. 2008. Determination of ammonia emission rates from a tunnel ventilated chicken house using passive samplers and a Gaussian dispersion model. Journal of Atmospheric Chemistry 59: 99-115.; Casey et al., 2010Casey, K.D.; Gates, R.S.; Shores, R.C.; Thomas, D.; Harris, D.B. 2010. Ammonia emissions from a U.S. broiler house-comparison of concurrent measurements using three different technologies. Journal of the Air & Waste Management Association 60: 939-48.). The estimation of ammonia shows high variability influencing the predictive emission models (Snoek et al., 2014Snoek, D.J.W.; Stigter, J.D.; Oginkc, N.W.M.; Groot Koerkamp, P.W.G. 2014. Sensitivity analysis of mechanistic models for estimating ammonia emission from dairy cow urine puddles. Biosystems Engineering 121: 12-24.), and found values have been adopted to define NH3emission inventories in different regions and countries (Groot Koerkamp et al., 1998Groot Koerkamp, P.W.G.; Metz, J.H.M.; Uenk, G.H.; Phillips, V.R.; Holden, M.R.; Sneath, R.W.; Short, J.L.; White, R.P.P.; Hartung, J.; Seedorf, J.; Schröder, M.; Linkert, K.H.; Pedersen, S.; Takai, H.; Johnsen, J.O.; Wathes, C.M. 1998. Concentrations and emissions of ammonia in livestock buildings in Northern Europe Journal of Agricultural Engineering Research 70: 79-95.; Demmers et al., 1999Demmers, T.G.M.; Burgess, L.R.; Short, J.L.; Phillips, V.R.; Clark, J.A.; Wathes, C.M. 1999. Ammonia emissions from two mechanically ventilated UK livestock buildings. Atmospheric Environment 33: 217-227.; Wheeler et al., 2006Wheeler, E.F.; Casey, K.D.; Gates, R.; Xin, H.; Zajaczkowski, J.L.; Topper, P.A.; Liang, Y.; Pescatore, A.J. 2006. Ammonia emissions from twelve U.S. broiler chicken houses. Transactions of the ASABE 49: 1495-1512.; Gates et al., 2008Gates, R.S.; Casey, K.D.; Wheeler, E.F.; Xin, H.; Pescatore, A.J. 2008. US broiler housing ammonia emissions inventory. Atmospheric Environment 42: 3342-3350.; Harper et al., 2009Harper, L.A.; Flesch, T.K.; Powell, J.M.; Coblentz, W.K.; Jokela, W.E.; Martin, N.P. 2009. Ammonia emissions from dairy production in Wisconsin. Journal of Dairy Science 92: 2326-2337.; Harper et al., 2010Harper, L.A.; Flesch, T.K.; Wilson, J.D. 2010. Ammonia emissions from broiler production in the San Joaquin Valley. Poultry Science 89: 1802-814.; Calvet et al., 2011Calvet, S.; Cambra-López, M.; Estellés, F.; Torres, A.G. 2011. Characterization of gas emissions from a Mediterranean broiler farm. Poultry Science 90: 534-542.).
This study aimed to estimate NH3 emissions in two broiler houses during the grow-out cycle using a predictive model.
Materials and Methods
Birds, housing and husbandry
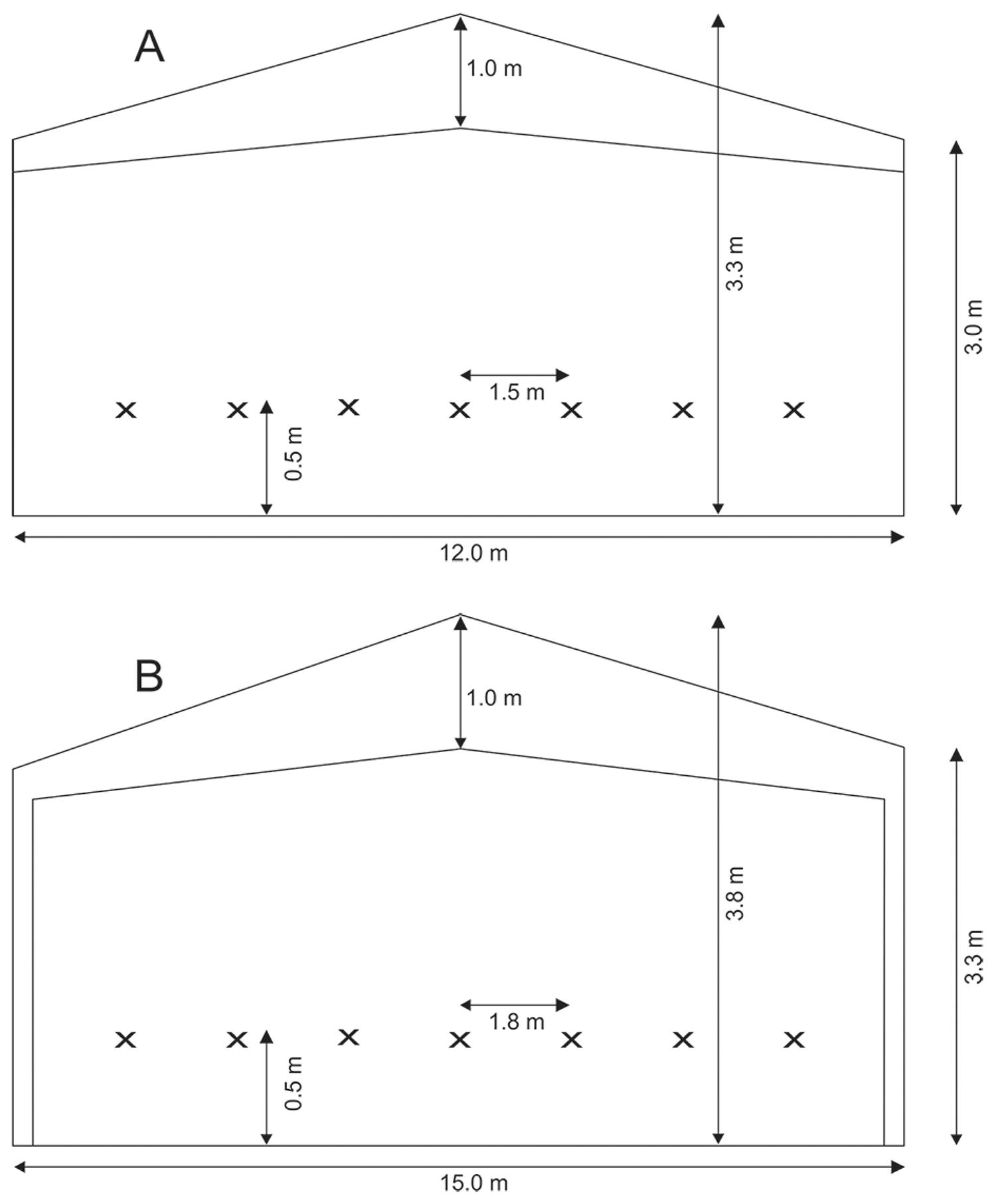
The study was carried out in eight commercial broiler houses located at longitude 54°11’6” W and latitude 23°28’26” S from March to May, 2013. Birds from the Hubbard® genetic strain were reared in mixed flocks (male and female) for 42 d (market age). In the tunnel system (TV, Figure 1A), 25200 broilers were reared, and 31500 broilers were raised in the dark house system (DH, Figure 1B). The adopted average flock density was 14 bird m−2 in TV and DH. The bedding in both houses was 10 cm of build-up wood shavings reused by the third time. The houses were east-west solar oriented.
− Schematic dimensions of the tunnel ventilated commercial broiler houses (TV, A and DH, B). Each “x” marks a location where dry bulb ambient temperature and air relative humidity were recorded. Samples of litter temperature and pH were collected on the place projecting this same location on the floor totaling 28 sampling points (7 in each quadrant).
Four houses had the TV system and four the DH system. TV houses (12 m wide, 150 m long and 3.2 m high, and a floor area of 1800 m2) had forced ventilation, exhaust fans (11 with air flow of 450 m3s−1), a high-pressure fogging system and the lateral areas were closed off with yellow polypropylene curtains. DH houses (15 m wide, 150 m long and 3.8 m high, and a floor area of 31500 m2) had forced ventilation; exhaust fans (12 with air flow of 580 m3 s−1), a high-pressure fogging system, and internal built walls painted with a black coating. In both houses the chick heating system used during day 1 to 14 of the grow-out period consisted of a stove (burned wood) with the heat distributed through metallic tubes with sensors controlling the inlet air temperature. Light intensity inside the houses was set at 25 lx for the first weeks of grow-out (7-21 d), and at 5 lx for the remainder of the grow-out cycle (42 d) using automatic light control based sensors placed in the houses. The two systems had similar ventilation systems with the air inlet at one end, and the outlet at the opposite side. They differed in the reflectivity inside, as the inside walls in the DH houses were coated with black paint.
Ambient air temperature was controlled automatically using the association of the evaporative cooling system and negative pressure ventilation. The air flow was programmed to decrease air temperature from 32 °C on the first day to 27 °C after two weeks, and finally to 24 °C during the end of the grow-out phase.
Data recording
Air temperature (AT, ºC) and relative humidity (RH, %) were measured in the morning (10h00) in the houses using sensors equally distributed over 28 points during the grow-out cycle (Figure 2). Litter pH and temperature (LT, ºC) were recorded at the same time using a portable meter. Data recording in areas nearby or below drinkers and feeders were avoided. The age of birds was computed in days (d).
− Schematic plan view of the tunnel ventilated broiler houses TV (A) and DH (B) used in the study.
Ammonia emissions were assessed in the area near the air outlet of the houses using the capsule method suggested by Jeppsson (1999)Jeppsson, K.H. 1999. Volatilization of ammonia in deep litter systems with different bedding materials for young cattle. Journal of Agricultural Engineering Research 7: 49-57. at 21, 35 and 40 d of the grow-out cycle. The methodology allows for estimating the amount of ammonia emissions in a particular area of the litter. The closed cubic capsule had an air inlet using a PVC tube (Φ 0.075 m) and inside it a small fan (12V, 60mA) was placed to promote homogeneity in the capsule’s inside air. The air outlet had a small exhaust fan (maximum flow of 90 m3 h−1) adjusted with a dimmer. The samples of ammonia concentration were taken at a height of 1 m at a point in the PVC tube, after allowing the capsule to be under continuous flow for 20 min in the same place. Ammonia concentrations were measured using a gas pump. Ammonia emissions (ε, mg m−2 h−1) were estimated using Equation 1 (Jeppsson, 1999Jeppsson, K.H. 1999. Volatilization of ammonia in deep litter systems with different bedding materials for young cattle. Journal of Agricultural Engineering Research 7: 49-57.).
in which Ccaps = ammonia concentration inside the capsule (ppm), C2 = ammonia concentration at 2 m of height (ppm), σ = the corrected density of ammonia at the capsule outlet (0.765 kg m−2), and ϕ = air flow (m3 m−2 h−1).
Model-prediction of ammonia emission
Mean values of recorded variables were calculated, and they were used as input to the software developed using the deterministic algorithm described in Equation 2 (Miragliotta et al., 2004Miragliotta, M.Y.; Nääs, I.A.; Murayama, M.C.; Moura, D.J. 2004. Software for estimating ammonia emission in broilers housing. Revista Brasileira de Agroinformática 6: 79-89 (in Portuguese, with abstract in English).).
in which E = model-predicted ammonia emission (mg m−2 h−1), d = day of grow out, LT = mean litter temperature (°C), and pH = litter pH.
Statistical analysis
The experimental design was completely randomized in a 2 × 4 factorial design (TV and DH; ages 7, 21, 35 and 42 d of growth) with four replicates per treatment, totaling 32 experimental units. The Kolmogorov-Smirnov test was used to verify the hypothesis of normality for all variables, using a 95 % confidence limit. Analysis of variance was used to compare means using the Tukey and the F-test at a 95 % confidence level. For the model-predicted ammonia emission, a 2 × 2 factorial design was adopted, considering the type of the broiler house (TV and DH) and two replications for the number of flocks. All statistical analysis was carried out according to Wheater and Cook (2000)Wheater, C.P.; Cook, P.A. 2000. Using Statistics to Understand the Environment. Routledge, New York, NY, USA. using ASSISTAT® software (Assistat-Statistical Attendance Software, version 7.6).
Results and Discussion
The Kolmogorov-Smirnov test showed a normal distribution of the data. An effect of the type of house (factor, F1) or age of birds (F2) was found (p> 0.05) in the ambient air temperature and relative humidity only at 35 d (p = 0.1; Table 1). Environmental data inside the houses did not differ, indicating that the chosen variables should not be the cause of possible variation in the estimation of NH3 emissions. Ambient air temperature is considered a primary factor in regulating the chemical processes involved in ammonia emissions (Wheeler et al., 2006Wheeler, E.F.; Casey, K.D.; Gates, R.; Xin, H.; Zajaczkowski, J.L.; Topper, P.A.; Liang, Y.; Pescatore, A.J. 2006. Ammonia emissions from twelve U.S. broiler chicken houses. Transactions of the ASABE 49: 1495-1512.; Sommer et al., 2006Sommer, S.G.; Zhang, G.Q.; Bannink, A.; Chadwick, D.; Misselbrook, T.; Harison, R.; Hutchings, N.J.; Menzi, H.; Monteny, G.J.; Ni, J.Q.; Oenema, O.; Webb, J. 2006. Algorithms determining ammonia emission from buildings housing cattle and pigs and from manure stores. Advances in Agronomy 89: 261-335.; Casey et al., 2010)Casey, K.D.; Gates, R.S.; Shores, R.C.; Thomas, D.; Harris, D.B. 2010. Ammonia emissions from a U.S. broiler house-comparison of concurrent measurements using three different technologies. Journal of the Air & Waste Management Association 60: 939-48.. Previous studies showed that the distribution uniformity of environmental features may affect broiler weight gain (Miragliotta et al., 2006Miragliotta, M.Y.; Nääs, I.A.; Manzione, R. L.; Nascimento, F.F. 2006. Spatial analysis of stress conditions inside broiler house under tunnel ventilation. Scientia Agricola 63: 426-432.; Lima et al., 2011)Lima, K.A.O.; Moura, D.J.; Carvalho, T.M.R.; Bueno, L.G.F.; Vercellino, R.A. 2011. Ammonia emissions in tunnel-ventilated broiler houses. Brazilian Journal of Poultry Science 13: 265-270..
TV houses had lower mean values of LT and pH (30.0 ºC and 8.4, respectively) than DH houses (33.3 °C and 8.6, respectively), Table 2. The factor age of broilers presented the highest mean amount at the end of the grow-out cycle (42 d). The distribution uniformity of environmental features may affect broiler weight gain (Miragliotta et al., 2006Miragliotta, M.Y.; Nääs, I.A.; Manzione, R. L.; Nascimento, F.F. 2006. Spatial analysis of stress conditions inside broiler house under tunnel ventilation. Scientia Agricola 63: 426-432.; Lima et al., 2011Lima, K.A.O.; Moura, D.J.; Carvalho, T.M.R.; Bueno, L.G.F.; Vercellino, R.A. 2011. Ammonia emissions in tunnel-ventilated broiler houses. Brazilian Journal of Poultry Science 13: 265-270.). The mean pH values of the two houses (TV and DH) showed a slight difference from the 7 d (8.2) to 42 d of growth. In the present study litter pH was 8.9 in the DH houses and 8.8 in the TV houses at 42 d of grow-out.
An important aspect of ammonia emission is the litter characteristics (Groot Koerkamp et al., 1998Groot Koerkamp, P.W.G.; Metz, J.H.M.; Uenk, G.H.; Phillips, V.R.; Holden, M.R.; Sneath, R.W.; Short, J.L.; White, R.P.P.; Hartung, J.; Seedorf, J.; Schröder, M.; Linkert, K.H.; Pedersen, S.; Takai, H.; Johnsen, J.O.; Wathes, C.M. 1998. Concentrations and emissions of ammonia in livestock buildings in Northern Europe Journal of Agricultural Engineering Research 70: 79-95.; Gates et al., 2008Gates, R.S.; Casey, K.D.; Wheeler, E.F.; Xin, H.; Pescatore, A.J. 2008. US broiler housing ammonia emissions inventory. Atmospheric Environment 42: 3342-3350.). Excreta accumulation on litter increases the litter pH and leads to a rise in NH3 concentrations inside broiler houses (Wheeler et al., 2006Wheeler, E.F.; Casey, K.D.; Gates, R.; Xin, H.; Zajaczkowski, J.L.; Topper, P.A.; Liang, Y.; Pescatore, A.J. 2006. Ammonia emissions from twelve U.S. broiler chicken houses. Transactions of the ASABE 49: 1495-1512.). The higher the litter pH, the lower the conversion of NH3 (volatile) into NH4+ (nonvolatile). Consequently, high ammonia volatilization in built-up litter without proper management is usual (Kristensen et al., 2000Kristensen, H.H.; Burgess, L.R.; Demmers, T.G.M.; Wathes, C.M. 2000. The preferences of laying hens for different concentrations of atmospheric ammonia. Applied Animal Behaviour Science 68: 307–318.). The dark house presented higher litter temperature than the tunnel ventilation system and the ammonia emission in the DH should be greater than in the TV system (Table 2). Broiler housing presents a broad range of NH3 emission rates depending on the flock harvest day and the ambient temperature during the grow-out period (Guiziou and Béline, 2005Guiziou, F.; Béline, F. 2005. In situ measurement of ammonia and greenhouse gas emissions from broiler houses in France. Bioresource Technology 96: 203-207.; Wheeler et al., 2006Wheeler, E.F.; Casey, K.D.; Gates, R.; Xin, H.; Zajaczkowski, J.L.; Topper, P.A.; Liang, Y.; Pescatore, A.J. 2006. Ammonia emissions from twelve U.S. broiler chicken houses. Transactions of the ASABE 49: 1495-1512.).
NH3 emissions increased with bird age (Figure 3), which corroborates previous studies (Wheeler et al., 2006Wheeler, E.F.; Casey, K.D.; Gates, R.; Xin, H.; Zajaczkowski, J.L.; Topper, P.A.; Liang, Y.; Pescatore, A.J. 2006. Ammonia emissions from twelve U.S. broiler chicken houses. Transactions of the ASABE 49: 1495-1512.; Calvet et al., 2011Calvet, S.; Cambra-López, M.; Estellés, F.; Torres, A.G. 2011. Characterization of gas emissions from a Mediterranean broiler farm. Poultry Science 90: 534-542.; Lin et al., 2012Lin, X.J.; Cortus, E.L.; Zhang, R.; Jiang, S.; Heber, A.J. 2012. Air emissions from broiler houses in California. Transactions of the ASABE 55: 1895-1908.). Ammonia emissions increased with broiler growth due to the consequent excreta build-up (Ngwabie et al., 2010Ngwabie, N.M.; Jeppsson, K.H.; Gustafsson, G.; Nimmermark, S. 2010. Influence of environmental factors and the addition of wood shavings on ammonia and odour emissions from fresh livestock manure. Agricultural Engineering International: CIGR Journal 12: 68-80., Calvet et al., 2011Calvet, S.; Cambra-López, M.; Estellés, F.; Torres, A.G. 2011. Characterization of gas emissions from a Mediterranean broiler farm. Poultry Science 90: 534-542.). There is a strong association between emissions, bird age, and litter condition (Gates et al., 2008Gates, R.S.; Casey, K.D.; Wheeler, E.F.; Xin, H.; Pescatore, A.J. 2008. US broiler housing ammonia emissions inventory. Atmospheric Environment 42: 3342-3350.; Casey et al., 2010Casey, K.D.; Gates, R.S.; Shores, R.C.; Thomas, D.; Harris, D.B. 2010. Ammonia emissions from a U.S. broiler house-comparison of concurrent measurements using three different technologies. Journal of the Air & Waste Management Association 60: 939-48.). Broilers were reared on built-up litter, which is known to increase NH3 emissions. Gates et al. (2008)Gates, R.S.; Casey, K.D.; Wheeler, E.F.; Xin, H.; Pescatore, A.J. 2008. US broiler housing ammonia emissions inventory. Atmospheric Environment 42: 3342-3350. and Lima et al. (2011)Lima, K.A.O.; Moura, D.J.; Carvalho, T.M.R.; Bueno, L.G.F.; Vercellino, R.A. 2011. Ammonia emissions in tunnel-ventilated broiler houses. Brazilian Journal of Poultry Science 13: 265-270.assessed the ammonia emissions in tunnel ventilation houses and found lower rates of NH3 emission in new bedding than in those with built-up litter. However, the renewal of bedding in each flock is not used by farmers as it may lead to an increase in production costs.
− Model-predicted ammonia emissions from two broiler houses (Tunnel ventilation, TV and dark house, DH) at four grow-out phases, 7 days (A), 21 days (B), 35 days (C) and 42 days (D).
Mean values of ambient AT did not differ (p > 0.05) and varied according to broiler requirements in each grow-out period (Table 2). The values were 26.4 ºC in TV and 26.3 ºC in DH. Mean values of RH did not differ between houses; however, an interaction between the “house” and “age of broilers” factors was identified at 35 and 42 d of growth. The change in pH depends on the age of the birds, and it may be attributed to the effect of litter temperature, ambient temperature and density of birds (Pescatore et al., 2005Pescatore, A.J.; Casey, K.D.; Gates, R.S. 2005. Ammonia emissions from broiler houses. Journal of Applied Poultry Research 14: 635-637.; Menegali et al., 2012)Menegali, I.; Tinôco, I.F.F.; Zolnier, S.; Carvalho, C.C.S.; Guimarães, M.C.C. 2012. Influence of different systems of minimum ventilation on air quality in broiler houses. Engenharia Agrícola 32: 1024-1033.. The pH of litter also influences the release of ammonia, which is maximized at levels of pH above 7.0. The difference between the temperature and litter pH at different ages provided a growing impact on the degradation reaction of nitrogen compounds in poultry litter and tended to be alkaline. The increase in temperature and pH of the litter provided greater microbial activity and the formation of noxious gases (Calvet et al., 2011)Calvet, S.; Cambra-López, M.; Estellés, F.; Torres, A.G. 2011. Characterization of gas emissions from a Mediterranean broiler farm. Poultry Science 90: 534-542.. Thus, the quantity and quality of litter affect the ability of NH3 emissions from broiler housing (Toghyani et al., 2010)Toghyani, M.; Gheisari, A.; Modaresi, M.; Tabeidian, S.A.; Toghyani, M. 2010. Effect of different litter material on performance and behavior of broiler chickens. Applied Animal Behaviour Science 122: 48-52..
The provision of minimum ventilation in the most uniform way is essential to ensuring good air quality control inside broiler housing. The small air exchange, high humidity, temperature and pH, and type of litter management may induce greenhouse gas emissions (Menegali et al., 2012Menegali, I.; Tinôco, I.F.F.; Zolnier, S.; Carvalho, C.C.S.; Guimarães, M.C.C. 2012. Influence of different systems of minimum ventilation on air quality in broiler houses. Engenharia Agrícola 32: 1024-1033.).
Model-predicted ammonia emissions
Mean ammonia emission amounts measured inside the TV using the capsule were similar for both houses studied at the first stage of the grow-out cycle (Figure 4). However, results varied during the broiler grow-out phase, 725 mg NH3 m−2 h−1at 21 d, 2200 mg NH3 m−2 h−1 at 35 d, and 2700 mg NH3 m−2 h−1 at 42 d of grow-out.
− Box plot of model-predicted ammonia emissions from two broiler houses (tunnel ventilation, TV, dark house, DH) and the measured values using the capsule method (TVM) at 21, 35 days and 42 days of the grow-out phase.
No effect was found for the studied factors at 7 d of growth (Table 3) from the model-predicted value. The house (F1) affected ammonia emission at 21 d. At 35 d, there was an interaction (p ≤ 0.05) between the factors (F1 and F2), meaning that the type of broiler house and the number of flocks were interacting. The average model-predicted value of ammonia emission in DH in the second flock was 5200 mg m−2 h−1, and also on TV with lower emission (2200 mg m−2 h−1; Table 4).
Siefert and Scudlark (2008)Siefert, R.L.; Scudlark, J.R. 2008. Determination of ammonia emission rates from a tunnel ventilated chicken house using passive samplers and a Gaussian dispersion model. Journal of Atmospheric Chemistry 59: 99-115. studied ammonia emissions from tunnel system houses and found values of 2700 mg NH3bird−1, equivalent to 2.7 g NH3bird−1. Results obtained in the present experiment, at 35 d were lower than that with an average emission (in TV and DH) of ~ 4000 mg NH3m−2 h−1 (12 g NH3 bird−1).Gates et al. (2008)Gates, R.S.; Casey, K.D.; Wheeler, E.F.; Xin, H.; Pescatore, A.J. 2008. US broiler housing ammonia emissions inventory. Atmospheric Environment 42: 3342-3350. found values of ammonia emission near 20 g NH3 bird−1, practically double the results found in the present study. Harper et al. (2010)Harper, L.A.; Flesch, T.K.; Wilson, J.D. 2010. Ammonia emissions from broiler production in the San Joaquin Valley. Poultry Science 89: 1802-814. evaluated the weekly average emission during the growth cycle of the birds, which resulted in a total of 4 kg NH3 per tunnel ventilation house during the summer. The result is similar to that found in the present study, on average 5 kg of NH3 per flock in tunnel ventilation. Redwine et al. (2002)Redwine, J.S.; Lacey, R.E.; Mukhtar, S.; Carey, J.B. 2002. Concentration and emissions of ammonia and particulate matter in tunnel-ventilated broiler houses under summer conditions in Texas. Transactions of the ASAE 45: 1101-1109. found that NH3 emission rates in broiler production varied from 38 to 2105 mg NH3 h–1. Despite the high variations between the results cited, the lowest value in the current literature is similar to that obtained in the present study, when considering the house with a total area of 1800 m2. Assuming a market bird weight of 2 kg the highest ammonia emission value found was 12 g NH3 bird-marketed−1.
The highest model-predicted ammonia emission occurred on the 21st d of growth in DH (1000 mg NH3 m−2 h−1). On the 35th d, there was an interaction between the “house” and “number of flocks” factors, and the largest model-predicted value of ammonia emission was for DH (4200 mg m−2 h−1) during flock 2, different from flock 1 (2600 mg m−2 h−1). On the last day of growth (market age, 42 d) considering the type of house, the highest model-predicted mean value of ammonia emission in DH (5700 mg m−2 h−1) was found. Model-predicted NH3 emissions increased along the grow-out cycle, arising from an increase in excreta deposition on litter (Figure 3). The ammonia emission found using the model-predicted ammonia, and those measured inside the houses were similar on the grow-out days 21, 35 and 42 in TV. Nevertheless, the values differed from those found in the DH on days 35 and 42 (Figure 4).
The emission rate of ammonia tends to be relatively stable in the first weeks of growth, rising by the third week of life. This increase coincides with the growth period of the birds, and the feed intake is higher than in other phases of the cycle. These emission factors such as concentration of excreta in litter temperature, pH and rate of ventilation may be associated (Harper et al., 2010Harper, L.A.; Flesch, T.K.; Wilson, J.D. 2010. Ammonia emissions from broiler production in the San Joaquin Valley. Poultry Science 89: 1802-814.).
Groot Koerkamp et al. (1998)Groot Koerkamp, P.W.G.; Metz, J.H.M.; Uenk, G.H.; Phillips, V.R.; Holden, M.R.; Sneath, R.W.; Short, J.L.; White, R.P.P.; Hartung, J.; Seedorf, J.; Schröder, M.; Linkert, K.H.; Pedersen, S.; Takai, H.; Johnsen, J.O.; Wathes, C.M. 1998. Concentrations and emissions of ammonia in livestock buildings in Northern Europe Journal of Agricultural Engineering Research 70: 79-95. found values of ammonia emissions in European countries near 11000 mg m−2h−1. The highest value found in the present study was ~6000 mg m−2 h−1 in the DH house (Table 4), which is nearly half of the values found previously. Studying NH3 emissions from new and built-up litter Gates et al. (2008)Gates, R.S.; Casey, K.D.; Wheeler, E.F.; Xin, H.; Pescatore, A.J. 2008. US broiler housing ammonia emissions inventory. Atmospheric Environment 42: 3342-3350. found that broilers raised on built-up litter emit ~25 g NH3 bird-marketed−1. Our highest findings of NH3 emissions (with market weight in Brazil near 2 kg) in built-up litter was equivalent to 12 g NH3bird-marketed−1, which is lower than the results for broilers reared in new bedding. Wheeler et al. (2006)Wheeler, E.F.; Casey, K.D.; Gates, R.; Xin, H.; Zajaczkowski, J.L.; Topper, P.A.; Liang, Y.; Pescatore, A.J. 2006. Ammonia emissions from twelve U.S. broiler chicken houses. Transactions of the ASABE 49: 1495-1512. found a mean value of 17 g NH3bird-marketed−1, which was similar to the results found in the present study. Values found here were similar to those found by Calvet et al. (2011)Calvet, S.; Cambra-López, M.; Estellés, F.; Torres, A.G. 2011. Characterization of gas emissions from a Mediterranean broiler farm. Poultry Science 90: 534-542. in southern Europe (24 g NH3 bird-marketed−1).
According to Liu et al. (2011)Liu, Z.; Wang-Li, L.; Beasley, D.B.; Shah, S.B. 2011. Validation and uncertainty analysis of an ammonia emission model for broiler litter. Transactions of the ASABE 54: 1051-1057. ventilation rate changes inside the housing may be the cause of different results in ammonia emission estimation. Liu et al. (2011)Liu, Z.; Wang-Li, L.; Beasley, D.B.; Shah, S.B. 2011. Validation and uncertainty analysis of an ammonia emission model for broiler litter. Transactions of the ASABE 54: 1051-1057.showed that when air velocity increased to a predetermined level (≥ 1.6 m s–1), emission flux increases. The loss of nitrogen and water from the litter is no longer negligible, which results in reduced measurements of emission fluxes. The differences in air flow inside the houses might be the reason for significant differences in the ammonia emission results found in current literature (Groot Koerkamp et al., 1998Groot Koerkamp, P.W.G.; Metz, J.H.M.; Uenk, G.H.; Phillips, V.R.; Holden, M.R.; Sneath, R.W.; Short, J.L.; White, R.P.P.; Hartung, J.; Seedorf, J.; Schröder, M.; Linkert, K.H.; Pedersen, S.; Takai, H.; Johnsen, J.O.; Wathes, C.M. 1998. Concentrations and emissions of ammonia in livestock buildings in Northern Europe Journal of Agricultural Engineering Research 70: 79-95.; Wheeler et al., 2006Wheeler, E.F.; Casey, K.D.; Gates, R.; Xin, H.; Zajaczkowski, J.L.; Topper, P.A.; Liang, Y.; Pescatore, A.J. 2006. Ammonia emissions from twelve U.S. broiler chicken houses. Transactions of the ASABE 49: 1495-1512.;Gates et al., 2008Gates, R.S.; Casey, K.D.; Wheeler, E.F.; Xin, H.; Pescatore, A.J. 2008. US broiler housing ammonia emissions inventory. Atmospheric Environment 42: 3342-3350.; Harper et al., 2009Harper, L.A.; Flesch, T.K.; Powell, J.M.; Coblentz, W.K.; Jokela, W.E.; Martin, N.P. 2009. Ammonia emissions from dairy production in Wisconsin. Journal of Dairy Science 92: 2326-2337.; Calvet et al., 2011Calvet, S.; Cambra-López, M.; Estellés, F.; Torres, A.G. 2011. Characterization of gas emissions from a Mediterranean broiler farm. Poultry Science 90: 534-542.).
Predicted levels of NH3 emissions from the model had a sharp increase after the 35th day of grow-out in the DH, when compared to the predictive value of TV, and the measured quantity using the capsule (TVM, Figure 2). The NH3 emissions were similar to those estimated in the TV houses but differed substantially from those in the DH house after the 35th d of the grow-out cycle.
Whereas the methods applied to this study can be used for estimating ammonia emission trends, the information presented should not be used for making regulatory determinations. However, the model provided comprehensible estimation and might be used as NH3 emission abatement strategies. The estimated ammonia emission was higher in the dark house than in the tunnel ventilation system. The amount of ammonia increased gradually throughout the grow-out cycle in both types of houses, and the highest mean value was attributed to the dark house system. The results from the predictive model showed a similar level of ammonia emissions in the tunnel ventilated house, but the level differed from the dark house starting during the broiler grow-out stage.
Acknowledgment
The authors wish to thank the Coordination for the Improvement of Higher Level Personnel (CAPES) for supporting this research.
References
- Beusen, A.H.W.; Bouwman, A.F.; Heuberger, P.S.C.; Drecht, G. van; Hoek, K.W. van der. 2008. Bottom-up uncertainty estimates of global ammonia emissions from global agricultural production systems. Atmospheric Environment 42: 6067-6077.
- Bouwman, A.F.; Lee, D.S.; Asman, W.A.H.; Dentener, F.J.; Hoek, K.W. van der; Oliver, G.J. 1997. A global high-resolution emission inventory for ammonia. Global Biogeochemical Cycles 11: 561-587.
- Calvet, S.; Cambra-López, M.; Estellés, F.; Torres, A.G. 2011. Characterization of gas emissions from a Mediterranean broiler farm. Poultry Science 90: 534-542.
- Casey, K.D.; Gates, R.S.; Shores, R.C.; Thomas, D.; Harris, D.B. 2010. Ammonia emissions from a U.S. broiler house-comparison of concurrent measurements using three different technologies. Journal of the Air & Waste Management Association 60: 939-48.
- Demmers, T.G.M.; Burgess, L.R.; Short, J.L.; Phillips, V.R.; Clark, J.A.; Wathes, C.M. 1999. Ammonia emissions from two mechanically ventilated UK livestock buildings. Atmospheric Environment 33: 217-227.
- Gates, R.S.; Casey, K.D.; Wheeler, E.F.; Xin, H.; Pescatore, A.J. 2008. US broiler housing ammonia emissions inventory. Atmospheric Environment 42: 3342-3350.
- Gates, R.S.; Xin, H.; Casey, K.D.; Liang, Y.; Wheeler, E.F. 2005. Method for measuring ammonia emissions from poultry houses. Journal of Applied Poultry Research 14: 622-634.
- Groot Koerkamp, P.W.G.; Metz, J.H.M.; Uenk, G.H.; Phillips, V.R.; Holden, M.R.; Sneath, R.W.; Short, J.L.; White, R.P.P.; Hartung, J.; Seedorf, J.; Schröder, M.; Linkert, K.H.; Pedersen, S.; Takai, H.; Johnsen, J.O.; Wathes, C.M. 1998. Concentrations and emissions of ammonia in livestock buildings in Northern Europe Journal of Agricultural Engineering Research 70: 79-95.
- Guiziou, F.; Béline, F. 2005. In situ measurement of ammonia and greenhouse gas emissions from broiler houses in France. Bioresource Technology 96: 203-207.
- Harper, L.A.; Flesch, T.K.; Wilson, J.D. 2010. Ammonia emissions from broiler production in the San Joaquin Valley. Poultry Science 89: 1802-814.
- Harper, L.A.; Flesch, T.K.; Powell, J.M.; Coblentz, W.K.; Jokela, W.E.; Martin, N.P. 2009. Ammonia emissions from dairy production in Wisconsin. Journal of Dairy Science 92: 2326-2337.
- Jeppsson, K.H. 1999. Volatilization of ammonia in deep litter systems with different bedding materials for young cattle. Journal of Agricultural Engineering Research 7: 49-57.
- Kristensen, H.H.; Burgess, L.R.; Demmers, T.G.M.; Wathes, C.M. 2000. The preferences of laying hens for different concentrations of atmospheric ammonia. Applied Animal Behaviour Science 68: 307–318.
- Lima, K.A.O.; Moura, D.J.; Carvalho, T.M.R.; Bueno, L.G.F.; Vercellino, R.A. 2011. Ammonia emissions in tunnel-ventilated broiler houses. Brazilian Journal of Poultry Science 13: 265-270.
- Lin, X.J.; Cortus, E.L.; Zhang, R.; Jiang, S.; Heber, A.J. 2012. Air emissions from broiler houses in California. Transactions of the ASABE 55: 1895-1908.
- Liu, Z.; Wang-Li, L.; Beasley, D.B.; Shah, S.B. 2011. Validation and uncertainty analysis of an ammonia emission model for broiler litter. Transactions of the ASABE 54: 1051-1057.
- Menegali, I.; Tinôco, I.F.F.; Zolnier, S.; Carvalho, C.C.S.; Guimarães, M.C.C. 2012. Influence of different systems of minimum ventilation on air quality in broiler houses. Engenharia Agrícola 32: 1024-1033.
- Miragliotta, M.Y.; Nääs, I.A.; Manzione, R. L.; Nascimento, F.F. 2006. Spatial analysis of stress conditions inside broiler house under tunnel ventilation. Scientia Agricola 63: 426-432.
- Miragliotta, M.Y.; Nääs, I.A.; Murayama, M.C.; Moura, D.J. 2004. Software for estimating ammonia emission in broilers housing. Revista Brasileira de Agroinformática 6: 79-89 (in Portuguese, with abstract in English).
- Ndegwa, P.M.; Hristov, A.N.; Arogo, J.; Sheffield, R.E. 2008. A review of ammonia emission mitigation techniques for concentrated animal feeding operations. Biosystems Engineering 100: 453-469.
- Ngwabie, N.M.; Jeppsson, K.H.; Gustafsson, G.; Nimmermark, S. 2010. Influence of environmental factors and the addition of wood shavings on ammonia and odour emissions from fresh livestock manure. Agricultural Engineering International: CIGR Journal 12: 68-80.
- Pescatore, A.J.; Casey, K.D.; Gates, R.S. 2005. Ammonia emissions from broiler houses. Journal of Applied Poultry Research 14: 635-637.
- Redwine, J.S.; Lacey, R.E.; Mukhtar, S.; Carey, J.B. 2002. Concentration and emissions of ammonia and particulate matter in tunnel-ventilated broiler houses under summer conditions in Texas. Transactions of the ASAE 45: 1101-1109.
- Siefert, R.L.; Scudlark, J.R. 2008. Determination of ammonia emission rates from a tunnel ventilated chicken house using passive samplers and a Gaussian dispersion model. Journal of Atmospheric Chemistry 59: 99-115.
- Snoek, D.J.W.; Stigter, J.D.; Oginkc, N.W.M.; Groot Koerkamp, P.W.G. 2014. Sensitivity analysis of mechanistic models for estimating ammonia emission from dairy cow urine puddles. Biosystems Engineering 121: 12-24.
- Sommer, S.G.; Zhang, G.Q.; Bannink, A.; Chadwick, D.; Misselbrook, T.; Harison, R.; Hutchings, N.J.; Menzi, H.; Monteny, G.J.; Ni, J.Q.; Oenema, O.; Webb, J. 2006. Algorithms determining ammonia emission from buildings housing cattle and pigs and from manure stores. Advances in Agronomy 89: 261-335.
- Toghyani, M.; Gheisari, A.; Modaresi, M.; Tabeidian, S.A.; Toghyani, M. 2010. Effect of different litter material on performance and behavior of broiler chickens. Applied Animal Behaviour Science 122: 48-52.
- UNFCCC. 1997. Kyoto Protocol to the United Nations Framework. Convention on Climate Change. Available at: http://unfccc.int/2860.php [Accessed Mar 30, 2014].
» http://unfccc.int/2860.php - Wheater, C.P.; Cook, P.A. 2000. Using Statistics to Understand the Environment. Routledge, New York, NY, USA.
- Wheeler, E.F.; Casey, K.D.; Gates, R.; Xin, H.; Zajaczkowski, J.L.; Topper, P.A.; Liang, Y.; Pescatore, A.J. 2006. Ammonia emissions from twelve U.S. broiler chicken houses. Transactions of the ASABE 49: 1495-1512.
Edited by
Publication Dates
-
Publication in this collection
Sep-Oct 2015
History
-
Received
27 June 2014 -
Accepted
02 Apr 2015