ABSTRACT:
Heavy metal pollution of the soil environment has become a major source of concern and continues to pose serious health problems to both humans and ecological systems worldwide. Phytoremediation is a biological treatment whereby plants are used to remove pollutant from the environment. An experiment was conducted to evaluate the potential of Melastoma malabathricum as a phytoremediator to absorb heavy metals from soil contaminated with sewage sludge. Melastoma malabathricum seedlings were planted on six different growth media: T0 - Control (100 % soil), T1 (80 % soil + 20 % sewage sludge), T2 (60 % soil + 40 % sewage sludge), T3 (40 % soil + 60 % sewage sludge), T4 (20 % soil + 80 % sewage sludge) and T5 (100 % sludge). There were differences found in both growth parameters and plant biomass. The highest growth performance such as plant height and number of leaves was found in T3. Iron was highly accumulated in the roots, Cu in the stems in T3, while Pb was accumulated in leaves in T5. The results showed the lowest Translocation Factor (TF) and highest Bioconcentration Factor (BCF) values in relation to the following elements: Cu, Fe, Mn, Pb and Zn. Melastoma malabathricum roots are able to uptake and translocate the elements into the plant's shoots. Therefore, it can be considered a good accumulator plant due to its capability of concentrating contaminants in aerial tissue. Melastoma malabathricum were thus found to be suitable for absorbing heavy metals in contaminated soils, and this species can also be considered an effective phtyoremediator of contaminated soil and mitigator of soil pollution.
Keywords:
soil pollution; phytoremediation; translocation factor; bioconcentration factor
Introduction
The ever expanding urbanization and population boom has caused a huge increase in the volume of domestic and industrial waste such as sewage sludge which now poses a major threat to the environment and human beings. Malaysia produces about 5 million cubic meters of sewage sludge per year, and by the year 2022, the amount has been estimated to reach 7 million cubic meters per year (Indah Water Konsortium, 1997Indah Water Konsortium. 1997. A Potty History of Sewage and its Treatment. Kuala Lumpur, Malaysia.). According to Majid et al. (2012)Majid, N.M.; Islam, M.M.; Mathew, L. 2012. Heavy metals uptake and translocation by mangium (Acacia mangium) from sewage contaminated soil. Australian Journal of Crop Science 6: 1228-1235., sewage sludge in Malaysia can cause serious environmental problems, especially soil pollution, with particular concern for its disposal. The most common way to reduce sewage sludge is to dispose of it in the land or in the sea (Odegaard et al., 2002Odegaard, H.; Paulsrud, B.; Karlsson, I. 2002. Wastewater sludge as a resource: sludge disposal strategies and corresponding treatment technologies aimed at sustainable handling of wastewater sludge. Water Science and Technology 46: 295-303.). It is suitable as fertilizer material and suitable for being used as an amendment to the soil because it contains high amounts of nutrient (Korentajer, 1991Korentajer, L. 1991. A review of agricultural use of sewage sludge: benefits and potential hazards. Water, Air, & Soil Pollution 17: 189-196.; Abdu et al., 2011Abdu, A.; Aderis, N.; Abdul-hamid, H.; Majid, N.M. 2011. Using Orthosiphon stamineus B. for phytoremediation of heavy metals in soils amended with sewage sludge. American Journal of Applied Sciences 8: 323-331.). However, sewage sludge such as anthropogenic sources of heavy metals contains numerous contaminants which can be hazardous to living organisms (Murtaza et al., 2010Murtaza, G.; Ghafoor, A.; Qadir, M.; Owens, G.; Aziz, M.; Zia, M. 2010. Disposal and use of sewage on agricultural lands in Pakistan: a review. Pedosphere 20: 23-34.).
Several methods have been developed in removing the heavy metal content in sewage sludge, such as detoxification and physicochemical remediation (Ghosh and Singh, 2005Ghosh, M.; Singh, S.P. 2005. A review on phytoremediation of heavy metals and utilization of its byproducts. Applied Ecology and Environmental Research 3: 1-18.). However, these methods are not suitable for detoxifying large amounts of sewage sludge, since they are expensive, time consuming, and increase the mobilization of contaminants, and destroy the biotics and structure of the soil (Danh et al., 2009Danh, L.T.; Truong, P.; Mammucari, R.; Tran, T.; Foster, N. 2009. Vetiver grass, Vetiveria zizanioides: a choice plant for phytoremediation of heavy metals and organic wastes. International Journal of Phytoremediation 11: 664-691.). Phytoremediation has been perceived as a more environmentally-friendly, “green” and comparatively low-cost where living plants are used to remediate the polluted areas (Jadia and Fulekar, 2009Jadia, C.D.; Fulekar, M.H. 2009. Phytoremediation of heavy metals: recent techniques. African Journal of Biotechnology 8: 921-928.; Shuhe et al., 2005Shuhe, W.; Qixing, Z.; Xin, W.; Kaisong, Z.; Guanlin, G.; Qiying, M.A.L. 2005. A newly discovered Cd-hyperaccumulato Solanum nigrum L. Chinese Science Bulletin 50: 33-38; Garbisu and Alkorta, 2001Garbisu, C.; Alkorta, I. 2001. Phytoextraction: a cost-effective plant-based technology for the removal of metals from the environment. Bioresource Technology 77: 229-236.). The information about the potential of herbal plant species to remediate the contaminated soil with sewage sludge is still lacking. Therefore, Melastoma malabathricum was selected to determine its efficacy in cleaning up heavy metals in soil treated with sewage sludge. Melastoma malabathricum grows in the humid tropics of Asia, is able to grow in polluted soil and is tolerant to acidic conditions (Osaki et al., 1995Osaki, M.; Sittibush, C.; Nuyim, T. 1995. Nutritional characteristics of wild plants grown in peat and acid sulfate soils distributed in Thailand and Malaysia. p. 63-76. In: Vijarsorn, P.; Suzuki, K.; Kyuma, K.; Wada, E.; Nagano, T.; Takai, Y., eds. A tropical swamp forest ecosystem and its greenhouse gas emission. Nodai Research Institute, Tokyo University of Agriculture, Tokyo, Japan., 1997Osaki, M.; Watanabe, T.; Tadano, T. 1997. Beneficial effect of aluminum on growth of plants adapted to low pH soils. Soil Science and Plant Nutrition 43: 827-837.; Watanabe et al., 1997Watanabe, T.; Osaki, M.; Tadano, T. 1997. Aluminum induced growth stimulation in relation to calcium, magnesium, and silicate nutrition in Melastoma malabathricum L. Soil Science and Plant Nutrition 43: 827-837.). The specific aims of this study were as follows: to determine the effectiveness of Melastoma malabathricum as a remover of heavy metals in soil contaminated with sewage sludge; to evaluate heavy metal uptake in plant parts; and to quantify heavy metal concentrations in growth media before planting and after harvesting.
Materials and Methods
The study was conducted at the greenhouse at the Faculty of Forestry, Universiti Putra Malaysia, Serdang, Malaysia (2°59' N latitude and 101°42' E longitude) for 6 months (Feb 2014 to July 2014). The soil was collected from Puchong (3°00' N latitude and 101°38' E longitude) and the soil series was Munchong developed over fined grained sedimentary rocks (shale) and metamorphic rocks. The soils are very fine, kaolinitic, isohyperthermic, red-yellow Tipik Tempalemoks. Based on USDA soil taxonomy, the soil is classified as Oxisols. The temperature recorded in this greenhouse ranged between 27 °C in the morning to 35 °C in the afternoon with an average relative humidity of 65 %.
The seedlings of Melastoma malabathricum were prepared from cuttings of the mature stem and planted in polybags (16.0 × 16.0 cm). The growing medium for Melastoma malabathricum was soil:organic matter:river sand in a 3:2:1 ratio. The seedlings were transplanted into suitable pots (392 mm diameter × 324 mm height) that were with the mixture of soil and sewage sludge.
There were six different treatments with four replicates; T0 - Control (100 % soil), T1 (80 % soil + 20 % sewage sludge), T2 (60 % soil + 40 % sewage sludge), T3 (40 % soil + 60 % sewage sludge), T4 (20 % soil+ 80 % sewage sludge) and T5 (100 % sewage sludge). The pots were arranged in a Randomized Complete Block Design (RCBD) in a greenhouse. Soil samples were collected from each pot before planting and after harvesting. All soils and sewage sludge were air dried and sieved to < 2 mm. Soil pH was determined in a suspension in soil:water ratio of 1:2.5 by using a glass electrode pH meter (Ahmadpour et al., 2010Ahmadpour, P.; Nawi, A.M.; Abdu, A.; Abdul-Hamid, H.; Singh, D.K.; Hassan, A.; Majid, N.M.; Jusop, S. 2010. Uptake of heavy metals by Jatropha curcas L. planted in soils containing sewage sludge. American Journal of Applied Sciences 7: 1291-1299.). Total carbon (C) was determined by the conventional method, with a loss on ignition, using 5 g of air dried soil, which was kept in an oven for 8 h at 550 °C (Heiri et al., 2001Heiri, O.; Lotter, A.F.; Lemcke, G. 2001. Loss on ignition as a method for estimating organic and carbonate content in sediments: reproducibility and comparability of results. Journal of Paleolimnology 25: 101-110.). Total nitrogen (N) contents were measured using a CHNS analyzer (Ye et al., 2002Ye, Z.H.; Shu, W.S.; Zhang, Z.Q.; Lan, C.Y.; Wong, M.H. 2002. Evaluation of major constraints to revegetation of lead/zinc mine tailings using bioassay techniques. Chemosphere 47: 1103-1111.).
The soil and plant digests were analyzed for the heavy metals selected such as zinc (Zn), lead (Pb), copper (Cu), iron (Fe) and manganese (Mn) by using an Atomic Absorption Spectrophotometer (AAS) (Justin et al., 2011Justin, V.; Majid, N.M.; Islam, M.M.; Abdu, A. 2011. Assessment of heavy metal uptake and translocation in Acacia mangium for phytoremediation of cadmium-contaminated soil. Journal of Food, Agriculture and Environment 9: 588-592.). The soil was digested according to the aqua regia method while plants were digested using the wet digestion method (Justin et al., 2011Justin, V.; Majid, N.M.; Islam, M.M.; Abdu, A. 2011. Assessment of heavy metal uptake and translocation in Acacia mangium for phytoremediation of cadmium-contaminated soil. Journal of Food, Agriculture and Environment 9: 588-592.). The plant height and number of leaves were measured every month. Plant biomass was measured separately for leaves, stems and roots.
In order to indicate the potential of plant species, two indicators are used. Firstly, Bioconcentration Factors (BCF) which indicate the ability of a plant species to accumulate metals in its tissue from the soils or water. Secondly, Translocation Factors (TF) which indicate the competence of the plant species to translocate metals from roots to shoots. The plant's ability to accumulate metals from soils and translocate metals were estimated using the TF in equation 1 and the BCF in equation 2 according to Yoon et al., 2006Yoon, J.; Cao, X.D.; Zhou, Q.X.; Ma, L.Q. 2006. Accumulation of Pb, Cu and Zn in native plants growing on a contaminated Florida site. Science of the Total Environment 368: 456-464.; Ho et al., 2008Ho, W.M.; Ang, L.H.; Lee, D.K. 2008. Assessment of Pb uptake, translocation and immobilization in kenaf (Hibiscus cannabinus L.) for phytoremediation of sand tailings. Journal of Environmental Sciences 20: 1341-1347.:
The leachates obtained from watering with tap water were collected after harvesting (July 2014). About 200 mL of leachate from each pot was collected and stored for chemical analysis. The leachate drainage systems consisted of about 2 kg of stone which was placed under the soil layer at the bottom of containers. PVC tubes were inserted into the soil layer in the container for collection of leachate. Analysis of variance (ANOVA) was used for growth parameters, heavy metals in the soil, sludge and plant parts and mean separation tests were carried out using Tukey's test (p ≤ 0.05). A comparison using Student's t-test at a 5 % level was made to detect any significant differences between samples taken before planting and after harvesting. The computation and preparation of graphs were done by the use of the SPSS 16.00 and Microsoft EXCEL 2003 software programs.
Results
General properties of the soils and sewage sludge
The particle texture of the soil was sandy clay (not tabulated). The pH values of soil and sewage sludge were 5.51 and 6.61, respectively (Table 1). The soil pH values were significantly different (p ≤ 0.05) before planting and after harvesting (Table 2). The minimum (5.51) and maximum (6.61) soil pH values were found before planting for control (T0) and after harvesting for 100 % sewage treatment (T5), respectively (Table 2). The highest change in soil pH was in the T3 treatment which was from 5.71 to 6.33. After harvesting, a trend of increasing pH in the soil emerged. Meanwhile, the value of total carbon was 9 % in the control and 64 % in sewage sludge, respectively (Table 1). The highest change in total carbon level was found in the T4 treatment (1 %) followed by the T3 (1 %). Total carbon decreased in all treatments after harvesting (Table 2). The values of Cu (3.90 mg kg–1), Fe (1549.13 mg kg–1), Mn (1.10 mg kg–1), Pb (2.36) and Zn (8.85 mg kg–1) in the soil were lower, whereas the values of Cu (4.93 mg kg–1), Fe (1602.13 mg kg–1), Mn (9.73 mg kg–1), Pb (10.35) and Zn (68.38 mg kg–1) in sewage sludge were always higher compared with the control (Table 1).
Growth performance and plant biomass of Melastoma malabathricum
There were significant differences in plant height at different levels of treatments (Table 3). The Melastoma malabathricum planted in the T1 medium had the highest plant height (128 cm) compared to the rest of the treatments (Table 3). The lowest plant height was found in the T5 treatment which is 9.30 cm. There were no differences between the treatments in terms of number of leaves. The T3 treatment showed the highest number of leaves (1045) followed by T2 (1034). The lowest number of leaves was recorded by T0 (control), which produced 431 leaves. Furthermore, the plant leaves, stems and roots showed differences between treatments after 6 months (Table 3). The results indicated that the majority of plant biomass increased from T0 to T3 but appeared to slightly decrease in T5.
Heavy metal concentrations of Melastoma malabathricum in growth media before planting and after harvesting
The concentrations of heavy metal in growth media before planting and after harvesting are shown in Figures 1A, B, C, D and E. The highest level of Fe was observed in T5 (1602 mg kg–1) which decreased after harvesting (1555 mg kg–1). Maximum reduction in Zn levels in the Melastoma malabathricum growth media was observed in T5 where the initial level of Zn was 68 mg kg–1 while the level of Zn after harvesting was 48 mg kg–1. Maximum reduction in Cu levels in the Melastoma malabathricum growth media was observed in T5, where the initial level of Cu was 4.93 mg kg–1 while the level of Cu after harvesting was 0.49 mg kg–1. Maximum Pb reduction was found in T5 (3.99 mg kg–1), while maximum reduction of Mn was also found in T5 (7.00 mg kg–1). Over the experimental period of 6 month, the concentration of heavy metals in the growth medium before planting decreased substantially.
Concentrations of Fe (A), Zn (B), Cu (C), Pb (D) and Mn (E) in growth medium before planting and after harvesting. Different letters indicate significant differences between means for each treatment before planting and after harvesting; T0 = Control; T1 = 80 % soil + 20 % sewage sludge; T2 = 60 % soil + 40 % sewage sludge; T3 = 40 % soil + 60 % sewage sludge; T4 = 20 % soil + 80 % sewage sludge; T5 = 100 % sludge.
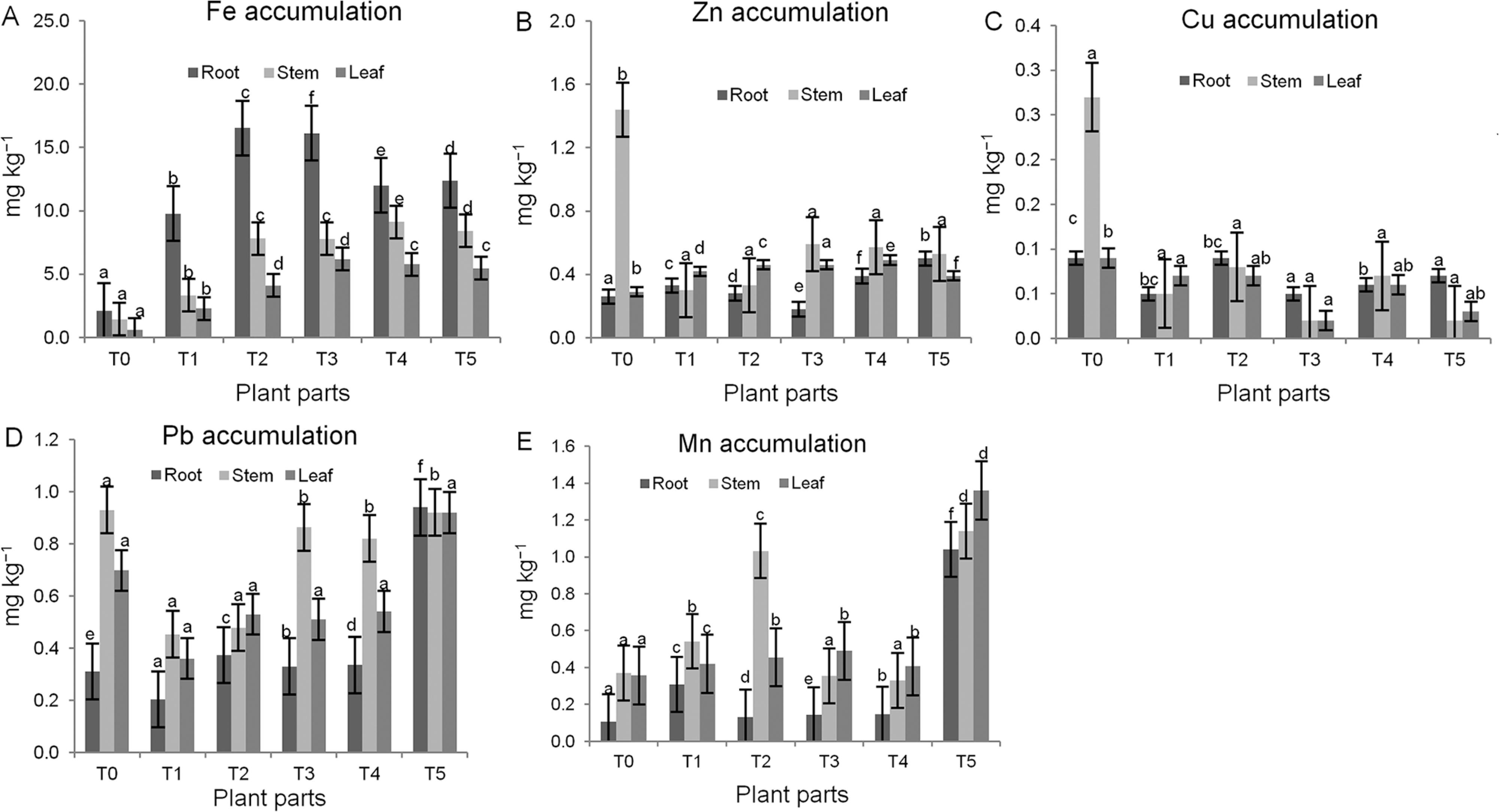
Heavy metal concentrations in plant parts
The concentrations of heavy metal in plant parts before planting and after harvesting are shown in Figures 2A, B, C, D and E. The highest Fe accumulation was observed in the root of Melastoma malabathricum in T3 (16.13 mg kg–1), while the lowest Fe was found in T0 (0.62 mg kg–1) in the leaf. Figure 2B showed the highest Zn concentration was observed in the stems in T0 (1.44 mg kg–1), and the lowest Zn concentration was observed in the roots in T3 (0.18 mg kg–1). The highest value of Cu was observed in the stem parts of the plant in T0 (0.27 mg kg–1). The minimum value of Cu was found in both the leaves and the stems both in T3 and T5 (0.02 mg kg–1). The highest Pb uptake (0.94 mg kg–1) in T5 was found in the roots followed by T0 (0.93 mg kg–1) in the stem. The highest concentration of Mn (1.36 mg kg–1) was accumulated in the leaf of the T5 treatment, while the lowest concentration of Mn was noted in the roots of the control (0.11 mg kg–1).
Concentrations of Fe (A), Zn (B), Cu (C), Pb (D) and Mn (E) in plant parts after harvesting; Different letters indicate significant differences between means for each treatment before planting and after harvesting; T0 = Control; T1 = 80 % soil + 20 % sewage sludge; T2 = 60 % soil + 40 % sewage sludge; T3 = 40 % soil + 60 % sewage sludge; T4 = 20 % soil + 80 % sewage sludge; T5 = 100 % sludge.
Bioconcentration Factor (BCF) and Translocation Factor (TF) of heavy metals
The BCF and TF of heavy metals are shown in Table 4. Cu showed the highest BCF value (1.925) for T1 followed by Pb (1.356) and Zn (0.230) in T0 (Control). In the cases of Fe and Mn, the highest BCF values were in T4 and T1 (0.078 and 0.075, respectively). The lowest BCF value (0.001) was found in Fe for T0 out of all the other metals. In the case of Mn, T5 showed the lowest BCF and the value was 0.011, while in Zn, T3 showed the lowest BCF with a value of 0.040. Meanwhile for Cu and Pb, the lowest values for BCF were recorded in T4 (0.100 and 0.066, respectively). The BCFs of Melastoma malabathricum ranged from 0.001 to 1.925 and only two samples showed BCFs more than 1, probably due to the high amount of heavy metal elements in the aerial parts (shoots) of these plants.
The highest TF for Pb was recorded in T3 (12.65) compared to all other treatments. The maximum value for Cu was recorded in T2 (11.240), while for Mn and Zn maximum value was recorded in T0 (6.518 and 4.210, respectively). The highest TF value for Fe occurred in T4 with values of 1.244. Out of all the treatments the lowest values for TF were recorded in Fe (0.574). The minimum value of TF for Cu, Pb and Zn was observed in T5 with values of 2.405, 1.957 and 1.860, respectively, whereas in the case of Zn, the minimum values were recorded in T3 (0.620). Overall, the TF for Melastoma malabathricum was more than 1 (TF > 1).
Leachates of heavy metals
Figures 3A, B, C, D and E show the concentrations of Fe, Zn, Cu, Pb and Mn in leachates. The concentrations of Cu in T3 (0.06 mg L–1) were lower compared to other treatments. The highest concentration of heavy metals in leachates were found in Fe in T2 (26.83 mg L–1).
Concentrations of Fe (A), Zn (B), Cu (C), Pb (D) and Mn (E) in leacheate after harvesting; Different letters indicate significant differences between means for each treatment before planting and after harvesting; T0 = Control; T1 = 80 % soil + 20 % sewage sludge; T2 = 60 % soil + 40 % sewage sludge; T3 = 40 % soil + 60 % sewage sludge; T4 = 20 % soil + 80 % sewage sludge; T5 = 100 % sludge.
Discussion
Our general observation of reduced biomass production of Melastoma malabathricum with the addition of high amounts of heavy metals is similar to the findings by other authors for sunflower, ricinus, alfalfa and mustard as phytoextraction (Niu et al., 2007Niu, Z.X.; Sun, L.N.; Sun, T.H.; Li, Y.S.; Wong, H. 2007. Evaluation of phytoextracting cadmium and lead by sunflower, ricinus, alfalfa and mustard in hydroponic culture. Journal of Environmental Sciences 19: 961-967.). According to Justin et al. (2011)Justin, V.; Majid, N.M.; Islam, M.M.; Abdu, A. 2011. Assessment of heavy metal uptake and translocation in Acacia mangium for phytoremediation of cadmium-contaminated soil. Journal of Food, Agriculture and Environment 9: 588-592. plant biomass is an important factor in growth analysis and the success of phytoremediation depends on it. Thus, plants must have a high potential for absorbing elements from a large biomass. High amounts of heavy metal depressed the growth of Melastoma malabathricum by decreasing the numbers of leaf/plant and plant height, and possible contamination of heavy metals in the soil by sewage sludge in the T5 treatment. Thus, this plant is not suitable for cultivation in 100 % sewage sludge media. Plant growth depends on nutrient supply, rate of absorption and the degree of mobility within the plant. The ability of Melastoma malabathricum to accumulate different heavy metals is highly dependent on soil pH, nature and the concentration of other contaminants. The results showed that, the soil pH increased after harvesting from 5.51 to 6.61. For better plant growth, the soil should be kept in the range of 6.4 to 7.0 (Abdu et al., 2011Abdu, A.; Aderis, N.; Abdul-hamid, H.; Majid, N.M. 2011. Using Orthosiphon stamineus B. for phytoremediation of heavy metals in soils amended with sewage sludge. American Journal of Applied Sciences 8: 323-331.). However, the pH is a very important factor that can control heavy metal uptake by plants. According to Lin et al. (2003)Lin, J.; Jiang, W.; Liu, D. 2003. Accumulation of copper by roots, hypocotyls, cotyledons and leaves of sunflower (Helianthus annuus L.). Bioresource Technology 86: 151-155., the solubility and bioavailability of heavy metals decrease with increased soil pH. Melastoma malabathricum have the potential to remove toxic elements, especially heavy metals, and this may have increased the soil pH after harvesting. In the low pH of soil, plants can uptake and remove toxic elements such as Fe, Mn, Cu, Zn and Pb. The percentage of total carbon decreased after harvesting and total carbon content increased with increasing sludge in the growth media. Total carbon in the soil decreased and may have been lost through soil decomposition, leaching of dissolved organic carbon (DOC), and conversion of carbon into soil organic matter and carbon dioxide. Temperature, sunlight, type of plants, and soil clay content were the factors that caused loss of carbon in the soil. It was observed that sludge had high organic matter levels compared to control. Sewage sludge contains organic matter that can improve the bulk density, CEC, soil fertility, soil water holding capacity, and help plant turgidity in a short period of drought (Keeren et al., 2013Keeren, S.R.; Arifin, A.; Daljit, D.K.; Hazandy, A.H.; Shamshuddin, J.; Wong, W.Z. 2013. Heavy metal uptake and translocation by Dipterocarpus verrucosus from sewage sludge contaminated soil. American Journal of Environmental Sciences 9: 259-268.). Moreover, the experiment indicated that heavy metals in sewage sludge were higher compared to control. It has been proved that sewage sludge contains essential organic and toxic materials from industrial and commercial waste (Alvarez et al., 2000Alvarez, E.A.; Mochon, M.C.; Sanchez, J.C.J.; Rodrigues, M.T. 2000. Determination of heavy metals in sewage sludge by microwave acid digestion and inductively coupled plasma atomic emission spectrometry. Toxicological & Environmental Chemistry 75: 207-214.).
From our observations the distribution of heavy metal elements were not the same among the plant parts (roots, stems and leaves). Heavy metal concentration in plant parts varies according to the different plant species and certain heavy metals are expected to accumulate in the roots because of direct contact from the soil. Other factors that can affect the mechanisms of plant uptake are the root zones which help to absorb contaminant from the soil and metabolize it inside the plant tissue (Tangahu et al., 2011Tangahu, B.V.; Sheikh-Abdullah, S.R.; Basri, H.; Idris, M.; Anuar, N.; Mukhlisin, M. 2011. A review in heavy metals (As, Pb and Hg) uptake by plants through phytoremediation. International Journal of Chemical Engineering 2011: 1-31.). Other studies have observed that the movement of elements can be limited, because of the high cation-exchange capacity of the cell wall (Raskin et al., 1997Raskin, I.; Smith, R.D.; Salt, D.E. 1997. Phytoremediation of metals: using plants to remove pollutants from the environment. Current Opinion in Biotechnology 8: 221-226.). Tangahu et al. (2011)Tangahu, B.V.; Sheikh-Abdullah, S.R.; Basri, H.; Idris, M.; Anuar, N.; Mukhlisin, M. 2011. A review in heavy metals (As, Pb and Hg) uptake by plants through phytoremediation. International Journal of Chemical Engineering 2011: 1-31. stated that the concentration of heavy metals in roots was higher compared to above ground tissue with high concentrations in fine roots. The highest Fe accumulation was found in roots and it shows that the roots are efficient in storing Fe. According to Yoon et al. (2006)Yoon, J.; Cao, X.D.; Zhou, Q.X.; Ma, L.Q. 2006. Accumulation of Pb, Cu and Zn in native plants growing on a contaminated Florida site. Science of the Total Environment 368: 456-464., plants take up heavy metals from soils through the plasma membrane of the root epidermal cells or through mass flow of water into the roots. Furthermore, plant roots have special biochemical pathways for absorbing essential elements (Madrid et al., 2003Madrid, F.; Liphadzi, M.S.; Kirkham, M.B. 2003. Heavy metal displacement in chelate-irrigated soil during phytoremediation. Journal of Hydrology 272: 107-119.). Pulford et al. (2002)Pulford, I.D.; Riddell-Black, D.; Stewart, C. 2002. Heavy metal uptake by willow clones from sewage sludge-treated soil: the potential for phytoremediation. International Journal of Phytoremediation 4: 59-72. stated that the change in metal availability of heavy metals to plants also depends on root respiration, root exudates and the decomposition of dead root tissue. Further detailed experiments are required to clarify root distribution, rhizophere characteristics, and the density and morphology of Melastoma malabathricum grown in pot experiments and at the field site.
One of the important roles and successes of phytoremediation is the ability of plants to absorb heavy metals from soil and water. There were differences found in heavy metal concentrations between treatments in the growth media before planting and after harvesting (p < 0.05). It was found that, the concentration of heavy metals was higher in sewage sludge treated soil. However, the concentration of heavy metals such as Fe, Zn, Cu, Pb and Mn decreased after harvesting. Any changes in soil heavy metal concentration in the growth media are likely due to plant uptake and probably little loss from leaching. In this experiment, the highest Fe was found in the control because of acidic conditions of clay soil in the Munchong soil series (Darus, 1979Darus, A. 1979. Mineralogy and genesis of soils in Universiti Pertanian Malaysia, Serdang, Selangor. Pertanika 2: 141-148.). The highest values for Zn, Mn, and Pb were noted in T5 and it was shown that these concentrations of elements increase with an ascending percentage of sludge and high heavy metal concentrations in soil restricted germination as well as reduced root and shoot growth, and finally biomass production in Melastoma malabathricum. The findings presented here are in accord with similar investigations on other species (Shanker et al., 2005). Soil analysis from growth media provided good information about the relative changes in soil and it could be used to refine the estimates for phytoremediation.
The high concentration of Fe in the growth medium did not restrict plant growth. The results showed that, Melastoma malabathricum are most efficient in up-taking and storing Fe in the roots. According to Keller et al. (2003)Keller, C.; Hammer, D.; Kayser, A.; Richner, W.; Brodbeck, M. 2003. Root development and heavy metal phytoextraction efficiency: comparison of different plant species in the field. Plant and Soil 249: 67-81., plants with extensive root systems are able to absorb more amounts of heavy metals due to soil penetration. Iron is highly insoluble, the presence of organic ligands can convert Fe to a mobile form; thus, the high uptakes of Fe by the plant roots (Perk, 2013Perk, M.V.D. 2013. Soil and Water Contamination: From Molecular to Catchment Scale. 2ed. Taylor and Francis, New York, NY, USA.). The nutrient availability in the T0 (Control) was lower compared to other treatments and it may limit absorption to the growing plants which caused low biomass production. The T5 Treatment also faced the same problem where biomass was lower due to the high concentration of sewage sludge in growth media. This high concentration might be the cause of heavy metal toxicity in growing plants. Zinc was concentrated in almost all plant parts and it showed that Zn was well distributed in Melastoma malabathricum. Plant growth was disrupted in T5 because of the high content of heavy metals in growth media. There were no symptoms of toxicity found due to Pb and the inhibition of growth of the plants studied throughout the experiments. It proved that Melastoma malabathricum was tolerant towards Pb. The results showed that, there were imbalances of distribution of heavy metals in plant parts and soil due to the restriction in the Pb extraction method in the soil that only extracts the bioavailability of heavy metals (Selamat et al., 2014Selamat, S.N.; Abdullah, S.R.S.; Idris, M. 2014. Phytoremediation of lead (Pb) and Arsenic (As) by Melastoma malabathricum L. from contaminated soil in separate exposure. International Journal of Phytoremediation 16: 694-703.).
TF and BCF values are important to indicate the potential of a plant for phytoremediation. Generally, the accumulation of heavy metals is higher in plants compared to soil and water. Plants should store heavy metals in stems to be classified as phytoremediator plants. Yoon et al. (2006)Yoon, J.; Cao, X.D.; Zhou, Q.X.; Ma, L.Q. 2006. Accumulation of Pb, Cu and Zn in native plants growing on a contaminated Florida site. Science of the Total Environment 368: 456-464. found that plants which can accumulate heavy metals with high BCF and low TF can be classified as phytostabilization plants. However, phytostabilization plants can release metals into the environment again through decomposing roots. These plants are not suitable for extended usage, especially for cultivating food crops. Based on Table 3, Cu showed the highest BCF which may be due to the effect of the physiological and accumulative abilities of Melastoma malabahricum when coming up against this element. According to Niu et al. (2007)Niu, Z.X.; Sun, L.N.; Sun, T.H.; Li, Y.S.; Wong, H. 2007. Evaluation of phytoextracting cadmium and lead by sunflower, ricinus, alfalfa and mustard in hydroponic culture. Journal of Environmental Sciences 19: 961-967., factors that can affect the BCF values depend on type and concentration of heavy metals. The highest TF was found in Pb in all the heavy metals, followed by Cu. It showed that, Melastoma malabathricum have the ability to absorb more Pb compared to other heavy metals and to transfer to aerial parts at the same time. According to Yoon et al. (2006)Yoon, J.; Cao, X.D.; Zhou, Q.X.; Ma, L.Q. 2006. Accumulation of Pb, Cu and Zn in native plants growing on a contaminated Florida site. Science of the Total Environment 368: 456-464., Cu is an important nutrient which supports plant systems and higher TF from the roots to the shoots is acceptable. The BCF and TF for Fe was the lowest out of all the other elements. Leachate analysis for Fe proved that the concentration of Fe in leachate was high and t indicates that Melastoma malabathricum absorbs less Fe in plant tissue. TF were more than 1 and BCF values are very low in all treatments. A translocation factor value greater than 1 indicates the translocation of metal from the root to the aerial parts (Jamil et al., 2009Jamil, S.; Abhilash, P.C.; Singh, N.; Sharma, P.N. 2009. Jatropha curcas: a potential crop for phytoremediation of a coal fly ash. Journal of Hazardous Materials 172: 269-275.). According to Yoon et al. (2006)Yoon, J.; Cao, X.D.; Zhou, Q.X.; Ma, L.Q. 2006. Accumulation of Pb, Cu and Zn in native plants growing on a contaminated Florida site. Science of the Total Environment 368: 456-464., only plant species with TF greater than 1 with low BCF values have the potential to be used for phytoextraction. In this experiment, more than 1 TF value indicated that, heavy metals were transported easily in Melastoma malabathricum. Hence, Melastoma malabathricum can be a phytoremediator plant for Fe, Zn, Mn, Cu and Pb.
Cu concentration was lower compared to other treatments. Studies have shown that Cu is relatively unavailable for leaching due to organic matter in the sludge which has bonded to Cu (Madrid et al., 2003Madrid, F.; Liphadzi, M.S.; Kirkham, M.B. 2003. Heavy metal displacement in chelate-irrigated soil during phytoremediation. Journal of Hydrology 272: 107-119.). Fe leached more amounts compared to other elements. According to Madrid et al. (2003)Madrid, F.; Liphadzi, M.S.; Kirkham, M.B. 2003. Heavy metal displacement in chelate-irrigated soil during phytoremediation. Journal of Hydrology 272: 107-119., the concentrations of Fe in water are 500 times higher compared to the amount allowed in drinking water. The Pb concentration in leachate was also high in this experiment. The leached Pb exceeded the limits of drinking water standards (0.05 mg L–1) and human consumption of more than 0.05 mg L–1 Pb, poses risks to human health. The previous study revealed that the presence of leachate on agricultural soil results in heavy metal accumulation in soils and bioaccumulation in plants (Onwuka et al., 2014Onwuka, S.U.; Solomon, B.E.; Kekpeazu, F.O. 2014. Leachate pollution of cocoyam and pawpaw crops grown around Anaekie Obiakor illegal dumpsite, Awka, Anambra State, Nigeria. International Journal of Engineering Research and General Science 2: 2091-2730.). These metals do not decompose easily in the environment and can become concentrated in the food chain; furthermore, they can create many problems for both the environment and humans. Phytoremediation is a biological method that can solve this problem whereby plants have been used to stabilize resident pollutants in, or even remove from the environment (Parvaresh et al., 2001Parvaresh, A.A.R.; Movahedian, H.; Zozli, M.A. 2001. Survey of heavy metals concentration in municipal solid wastes leachate of Isfahan city and their reduction method. Journal of Research in Medical Sciences 6: 4.; Umar et al., 2010Umar, M.; Aziz, H.A.; Yusoff, M.S. 2010. Variability of parameter involved in leachate pollution index and determination of LPI from four landfills in Malaysia. International Journal of Chemical Engineering 2010: 1-6.). The plants used in phytoremediation must be the hyper-accumulative. This kind of vegetation is chosen mainly because of its physiological potential to tolerate and assimilate toxic substances, its growth rates, the depth of its roots and the ability to degrade and/or bio-accumulate contaminants in its roots, branches or leaves (Salt et al., 1998Salt, D.E.; Smith, R.D.; Raskin, J. 1998. Phytoremediation. Annual Review of Plant Physiology and Plant Molecular Biology 49: 643-668.; Llugany et al., 2007Llugany, M.; Tolra, R.; Poschnrieder, C.; Barcelo, J. 2007. Metal hyperaccumulation: advantage for both the plant and the human being? Ecosistemas 16: 4-9 (in Spanish, with abstract in English).). In our current study, we found heavy metal accumulation in the root, stem and leaf of Melastoma malabathricum. Thus, Melastoma malabathricum species can be used as a highly effective heavy metal accumulator and can remove the toxic effect of leachates.
Conclusions
Phytoremediation is an emerging technology that employs the use of higher plants for the cleanup of contaminated environments. The main advantage of phytoremediation is environmental friendliness. Traditional methods which are used for cleaning up heavy metal contaminated soil disrupt the soil structure and reduce soil productivity, whereas phytoremediation can clean up the soil without causing any kind of harm to the quality of the soil. In our current study, Melastoma malabathricum was found to effectively absorb heavy metals from soil contaminated with sewage sludge and the TF values were above 1 while the BCF was lower. These showed that plants are able to accumulate high levels of heavy metals. Therefore, Melastoma malabathricum could be a highly effective phytoextractor of Mn, Pb, Cu, Fe and Zn. Even the phytoremediation technique seems to be one of the best alternatives, but it also has certain limitations. Prolonged research needs to be conducted to minimize this limitation in order to apply this technique effectively. With greater diffusion of this technique, field trials and experiments can be carried out to examine the real potential for its use, and promising results for the remediation of heavy metals in contaminated areas may be attained. Therefore this experiment needs to be conducted in the field in order to verify if this species can be used as an effective accumulator of heavy metals in large areas.
Acknowledgements
We are thankful to the Ministry of Higher Education Malaysia and University Putra Malaysia for financial support.
References
- Abdu, A.; Aderis, N.; Abdul-hamid, H.; Majid, N.M. 2011. Using Orthosiphon stamineus B. for phytoremediation of heavy metals in soils amended with sewage sludge. American Journal of Applied Sciences 8: 323-331.
- Ahmadpour, P.; Nawi, A.M.; Abdu, A.; Abdul-Hamid, H.; Singh, D.K.; Hassan, A.; Majid, N.M.; Jusop, S. 2010. Uptake of heavy metals by Jatropha curcas L. planted in soils containing sewage sludge. American Journal of Applied Sciences 7: 1291-1299.
- Alvarez, E.A.; Mochon, M.C.; Sanchez, J.C.J.; Rodrigues, M.T. 2000. Determination of heavy metals in sewage sludge by microwave acid digestion and inductively coupled plasma atomic emission spectrometry. Toxicological & Environmental Chemistry 75: 207-214.
- Darus, A. 1979. Mineralogy and genesis of soils in Universiti Pertanian Malaysia, Serdang, Selangor. Pertanika 2: 141-148.
- Danh, L.T.; Truong, P.; Mammucari, R.; Tran, T.; Foster, N. 2009. Vetiver grass, Vetiveria zizanioides: a choice plant for phytoremediation of heavy metals and organic wastes. International Journal of Phytoremediation 11: 664-691.
- Garbisu, C.; Alkorta, I. 2001. Phytoextraction: a cost-effective plant-based technology for the removal of metals from the environment. Bioresource Technology 77: 229-236.
- Ghosh, M.; Singh, S.P. 2005. A review on phytoremediation of heavy metals and utilization of its byproducts. Applied Ecology and Environmental Research 3: 1-18.
- Heiri, O.; Lotter, A.F.; Lemcke, G. 2001. Loss on ignition as a method for estimating organic and carbonate content in sediments: reproducibility and comparability of results. Journal of Paleolimnology 25: 101-110.
- Ho, W.M.; Ang, L.H.; Lee, D.K. 2008. Assessment of Pb uptake, translocation and immobilization in kenaf (Hibiscus cannabinus L.) for phytoremediation of sand tailings. Journal of Environmental Sciences 20: 1341-1347.
- Indah Water Konsortium. 1997. A Potty History of Sewage and its Treatment. Kuala Lumpur, Malaysia.
- Jadia, C.D.; Fulekar, M.H. 2009. Phytoremediation of heavy metals: recent techniques. African Journal of Biotechnology 8: 921-928.
- Jamil, S.; Abhilash, P.C.; Singh, N.; Sharma, P.N. 2009. Jatropha curcas: a potential crop for phytoremediation of a coal fly ash. Journal of Hazardous Materials 172: 269-275.
- Justin, V.; Majid, N.M.; Islam, M.M.; Abdu, A. 2011. Assessment of heavy metal uptake and translocation in Acacia mangium for phytoremediation of cadmium-contaminated soil. Journal of Food, Agriculture and Environment 9: 588-592.
- Keeren, S.R.; Arifin, A.; Daljit, D.K.; Hazandy, A.H.; Shamshuddin, J.; Wong, W.Z. 2013. Heavy metal uptake and translocation by Dipterocarpus verrucosus from sewage sludge contaminated soil. American Journal of Environmental Sciences 9: 259-268.
- Keller, C.; Hammer, D.; Kayser, A.; Richner, W.; Brodbeck, M. 2003. Root development and heavy metal phytoextraction efficiency: comparison of different plant species in the field. Plant and Soil 249: 67-81.
- Korentajer, L. 1991. A review of agricultural use of sewage sludge: benefits and potential hazards. Water, Air, & Soil Pollution 17: 189-196.
- Lin, J.; Jiang, W.; Liu, D. 2003. Accumulation of copper by roots, hypocotyls, cotyledons and leaves of sunflower (Helianthus annuus L.). Bioresource Technology 86: 151-155.
- Llugany, M.; Tolra, R.; Poschnrieder, C.; Barcelo, J. 2007. Metal hyperaccumulation: advantage for both the plant and the human being? Ecosistemas 16: 4-9 (in Spanish, with abstract in English).
- Madrid, F.; Liphadzi, M.S.; Kirkham, M.B. 2003. Heavy metal displacement in chelate-irrigated soil during phytoremediation. Journal of Hydrology 272: 107-119.
- Majid, N.M.; Islam, M.M.; Mathew, L. 2012. Heavy metals uptake and translocation by mangium (Acacia mangium) from sewage contaminated soil. Australian Journal of Crop Science 6: 1228-1235.
- Murtaza, G.; Ghafoor, A.; Qadir, M.; Owens, G.; Aziz, M.; Zia, M. 2010. Disposal and use of sewage on agricultural lands in Pakistan: a review. Pedosphere 20: 23-34.
- Niu, Z.X.; Sun, L.N.; Sun, T.H.; Li, Y.S.; Wong, H. 2007. Evaluation of phytoextracting cadmium and lead by sunflower, ricinus, alfalfa and mustard in hydroponic culture. Journal of Environmental Sciences 19: 961-967.
- Odegaard, H.; Paulsrud, B.; Karlsson, I. 2002. Wastewater sludge as a resource: sludge disposal strategies and corresponding treatment technologies aimed at sustainable handling of wastewater sludge. Water Science and Technology 46: 295-303.
- Onwuka, S.U.; Solomon, B.E.; Kekpeazu, F.O. 2014. Leachate pollution of cocoyam and pawpaw crops grown around Anaekie Obiakor illegal dumpsite, Awka, Anambra State, Nigeria. International Journal of Engineering Research and General Science 2: 2091-2730.
- Osaki, M.; Sittibush, C.; Nuyim, T. 1995. Nutritional characteristics of wild plants grown in peat and acid sulfate soils distributed in Thailand and Malaysia. p. 63-76. In: Vijarsorn, P.; Suzuki, K.; Kyuma, K.; Wada, E.; Nagano, T.; Takai, Y., eds. A tropical swamp forest ecosystem and its greenhouse gas emission. Nodai Research Institute, Tokyo University of Agriculture, Tokyo, Japan.
- Osaki, M.; Watanabe, T.; Tadano, T. 1997. Beneficial effect of aluminum on growth of plants adapted to low pH soils. Soil Science and Plant Nutrition 43: 827-837.
- Parvaresh, A.A.R.; Movahedian, H.; Zozli, M.A. 2001. Survey of heavy metals concentration in municipal solid wastes leachate of Isfahan city and their reduction method. Journal of Research in Medical Sciences 6: 4.
- Perk, M.V.D. 2013. Soil and Water Contamination: From Molecular to Catchment Scale. 2ed. Taylor and Francis, New York, NY, USA.
- Pulford, I.D.; Riddell-Black, D.; Stewart, C. 2002. Heavy metal uptake by willow clones from sewage sludge-treated soil: the potential for phytoremediation. International Journal of Phytoremediation 4: 59-72.
- Raskin, I.; Smith, R.D.; Salt, D.E. 1997. Phytoremediation of metals: using plants to remove pollutants from the environment. Current Opinion in Biotechnology 8: 221-226.
- Salt, D.E.; Smith, R.D.; Raskin, J. 1998. Phytoremediation. Annual Review of Plant Physiology and Plant Molecular Biology 49: 643-668.
- Shuhe, W.; Qixing, Z.; Xin, W.; Kaisong, Z.; Guanlin, G.; Qiying, M.A.L. 2005. A newly discovered Cd-hyperaccumulato Solanum nigrum L. Chinese Science Bulletin 50: 33-38
- Selamat, S.N.; Abdullah, S.R.S.; Idris, M. 2014. Phytoremediation of lead (Pb) and Arsenic (As) by Melastoma malabathricum L. from contaminated soil in separate exposure. International Journal of Phytoremediation 16: 694-703.
- Tangahu, B.V.; Sheikh-Abdullah, S.R.; Basri, H.; Idris, M.; Anuar, N.; Mukhlisin, M. 2011. A review in heavy metals (As, Pb and Hg) uptake by plants through phytoremediation. International Journal of Chemical Engineering 2011: 1-31.
- Umar, M.; Aziz, H.A.; Yusoff, M.S. 2010. Variability of parameter involved in leachate pollution index and determination of LPI from four landfills in Malaysia. International Journal of Chemical Engineering 2010: 1-6.
- Watanabe, T.; Osaki, M.; Tadano, T. 1997. Aluminum induced growth stimulation in relation to calcium, magnesium, and silicate nutrition in Melastoma malabathricum L. Soil Science and Plant Nutrition 43: 827-837.
- Yoon, J.; Cao, X.D.; Zhou, Q.X.; Ma, L.Q. 2006. Accumulation of Pb, Cu and Zn in native plants growing on a contaminated Florida site. Science of the Total Environment 368: 456-464.
- Ye, Z.H.; Shu, W.S.; Zhang, Z.Q.; Lan, C.Y.; Wong, M.H. 2002. Evaluation of major constraints to revegetation of lead/zinc mine tailings using bioassay techniques. Chemosphere 47: 1103-1111.
Edited by
Publication Dates
-
Publication in this collection
Jan-Feb 2018
History
-
Received
07 Jan 2016 -
Accepted
11 Nov 2016