ABSTRACT:
Arbuscular mycorrhizal fungi (AMF) are very important to plant nutrition, mostly in terms of acquisition of P and micronutrients. While Acacia mangium is closely associated with AMF throughout the whole cycle, Eucalyptus grandis presents this symbiosis primarily at the seedling stage. The aim of this study was to evaluate the dynamics of AMF in these two tree species in both pure and mixed plantations during the first 20 months after planting. We evaluated the abundance, richness and diversity of AMF spores, the rate of AMF mycorrhizal root colonization, enzymatic activity and soil and litter C, N and P. There was an increase in AMF root colonization of E. grandis when intercropped with A. mangium as well as an increase in the activity of acid and alkaline phosphatase in the presence of leguminous trees. AMF colonization and phosphatase activities were both involved in improvements in P cycling and P nutrition in soil. In addition, P cycling was favored in the intercropped plantation, which showed negative correlation with litter C/N and C/P ratios and positive correlation with soil acid phosphatase activity and soil N and P concentrations. Intercropping A. mangium and E. grandis maximized AMF root colonization of E. grandis and phosphatase activity in the soil, both of which accelerate P cycling and forest performance.
Keywords:
AMF spores; mycorrhiza; mixed species; soil enzymes; phosphorus
Introduction
Many studies have been conducted in intercropped plantations of Eucalyptus and leguminous trees to verify and validate the sustainability of these systems. Positive results of Eucalyptus intercropped with leguminous trees (e.g. Acacia mangium and A. mearnsii) have been reported to stimulate the biogeochemical cycles in soil, plant bio-mass productivity, soil fertility, and more (Bachega et al., 2016Bachega, L.R.; Bouillet, J.P.; Piccolo, M.C.; Saint-André, L.; Bouvet, J.M.; Nouvellon, Y.; Gonçalves, J.L.M.; Robin, A.; Laclau, J.P. 2016. Decomposition of Eucalyptus grandis and Acacia mangium leaves and fine roots in tropical conditions did not meet the Home Field Advantage hypothesis. Forest Ecology and Management 359: 33-43.; Binkley et al., 1992Binkley, D.; Dunkin, K.A.; Debell, D.; Ryan, M.G. 1992. Production and nutrient cycling in mixed plantations of Eucalyptus and Albizia in Hawaii. Forest Science 38: 393-408.; Bini et al., 2013aBini, D.; Santos, C.A.; Bouillet, J.; Gonçalves, J.L.D.M.; Cardoso, E.J.B.N. 2013a. Eucalyptus grandis and Acacia mangium in monoculture and intercropped plantations: Evolution of soil and litter microbial and chemical attributes during early stages of plant development. Applied Soil Ecology 63: 57-66.; Bouillet et al., 2013Bouillet, J.P.; Laclau, J.P.; Gonçalves, J.L.M.; Voigtlaender, M.; Gava, J.L.; Leite, F.P.; Hakamada, R.; Mareschal, L.; Mabiala, A.; Tardy, F.; Levillain, J.; Deleporte, P.; Epron, D.; Nouvellon, Y. 2013. Eucalyptus and Acacia tree growth over entire rotation in single- and mixed-species plantations across five sites in Brazil and Congo. Forest Ecology and Management 301: 89-101.; Forrester et al., 2005Forrester, D.I.; Bauhus, J.; Cowie, A.L. 2005. On the success and failure of mixed-species tree plantations: lessons learned from a model system of Eucalyptus globulus and Acacia mearnsii. Forest Ecology and Management 209: 147-155.; Khanna, 1997Khanna, P.K. 1997. Comparison of growth and nutrition of young monocultures and mixed stand of Eucalyptus globulus and Acacia mearnsii. Forest Ecology and Management 94: 105-113.; Paula et al., 2015Paula, R.R.; Bouillet, J.P.; Trivelin, P.C.O.; Zeller, B.; Gonçalves, J.L.M.; Nouvellon, Y.; Bouvet, J.M.; Plassard, C.; Laclau, J.P. 2015. Evidence of short-term belowground transfer of nitrogen from Acacia mangium to Eucalyptus grandis trees in a tropical planted forest. Soil Biology and Biochemistry 91: 99-108.; Voigtlaender et al., 2012Voigtlaender, M.; Laclau, J.P.; Gonçalves, J.L.M.; Piccolo, M.D.C.; Moreira, M.Z.; Nouvellon, Y.; Ranger, J.; Bouillet, J.P. 2012. Introducing Acacia mangium trees in Eucalyptus grandis plantations: consequences for soil organic matter stocks and nitrogen mineralization. Plant and Soil 352: 99-111.). It is known that legumes require more P than non-leguminous plants because maintaining the biological N2 fixation process in root nodules is highly dependent on P (Binkley, 1992Binkley, D. 1992. Mixtures of N2-fixing and non-N2-fixing tree species. p. 99-123. In: Cannell, M.G.R.; Malcom, D.C.; Robertson, P.A., eds. The ecology of mixed-species stands of trees. Blackwell Scientific, London, UK.). In this case, associations with arbuscular mycorrhizal fungi (AMF) may positively influence plant P nutrition and, indirectly, the biological nitrogen fixation process (Cardoso et al., 2013Cardoso, E.J.B.N.; Vasconcellos, R.L.F.; Bini, D.; Miyauchi, M.Y.H.; Santos, C.A.; Alves, P.R.L.; Paula, A.M.; Nakatani, A.S.; Pereira, J.M.; Nogueira, M.A. 2013. Soil health: looking for suitable indicators. What should be considered to assess the effects of use and management on soil health? Scientia Agricola 70: 280-295.).
Though both species, E. grandis and A. mangium, can associate either with AMF or with ectomycorrhizal fungi (Mendes-Filho et al., 2009Mendes-Filho, P.F.; Vasconcellos, R.L.F.; Paula, A.M.; Cardoso, E.J.B.N. 2009. Evaluating the potential of forest species under “microbial management” for the restoration of degraded mining areas. Water, Air and Soil Pollution 208: 79-89.; Pagano et al., 2008Pagano, M.C.; Scotti, M.R. 2008. Arbuscular and ectomycorrhizal colonization of two Eucalyptus species in semiarid Brazil. Mycoscience 49: 379-384.), our studies were restricted to the first group, since our observations refer to very young plants, beginning at the seedling stage, a period in which there is a predominance of AMF colonization in the roots of both plants (Adams et al., 2006Adams, F.; Reddell, P.; Webb, M.; Shipton, W. 2006. Arbuscular mycorrhizas and ectomycorrhizas on Eucalyptus grandis (Myrtaceae) trees and seedlings in native forests of tropical north-eastern Australia. Australian Journal of Botany 54: 271-281.; Pagano and Scotti, 2008Pagano, M.C.; Scotti, M.R. 2008. Arbuscular and ectomycorrhizal colonization of two Eucalyptus species in semiarid Brazil. Mycoscience 49: 379-384.). However, very little is known about the association between AMF and Eucalyptus, especially when intercropped with A. mangium, which is characterized by forming symbioses with N-fixing bacteria and AMF (Mendes-Filho et al., 2009Mendes-Filho, P.F.; Vasconcellos, R.L.F.; Paula, A.M.; Cardoso, E.J.B.N. 2009. Evaluating the potential of forest species under “microbial management” for the restoration of degraded mining areas. Water, Air and Soil Pollution 208: 79-89.). Intercropped systems generally show greater diversity and abundance of AMF than pure stands (Pagano et al., 2009Pagano, M.C.; Scotti, M.R.; Cabello, M.N. 2009. Effect of the inoculation and distribution of mycorrhizae in Plathymenia reticulata Benth under monoculture and mixed plantation in Brazil. New Forest 38: 197-214.). High diversity and high inoculum potential of AMF in soils, even under low P availability, generally lead to a more efficient use of P and other nutrients (Cardoso et al., 2013Cardoso, E.J.B.N.; Vasconcellos, R.L.F.; Bini, D.; Miyauchi, M.Y.H.; Santos, C.A.; Alves, P.R.L.; Paula, A.M.; Nakatani, A.S.; Pereira, J.M.; Nogueira, M.A. 2013. Soil health: looking for suitable indicators. What should be considered to assess the effects of use and management on soil health? Scientia Agricola 70: 280-295.; Ma and Rengel, 2008Ma, Q.; Rengel, Z. 2008. Phosphorus acquisition and wheat growth are influenced by shoot phosphorus status and soil phosphorus distribution in a split-root system. Journal of Plant Nutrition and Soil Science 171: 266-271.), owing to an increase in the root absorptive surface and to the external fungal hyphae mining the surrounding soil (Hinsinger, 2001Hinsinger, P. 2001. Bioavailability of soil inorganic P in the rhizosphere as affected by root-induced chemical changes: a review. Plant and Soil 237: 173-195.). A decrease in available P in soil cultivated with intercropped trees was attributed to higher P uptake by mycorrhizal plants (Koutika et al., 2014Koutika, L.S.; Epron, D.; Bouillet, J.P.; Mareschal, L. 2014. Changes in N and C concentrations, soil acidity and P availability in tropical mixed Acacia and eucalypt plantations on a nutrient-poor sandy soil. Plant and Soil 379: 205-216.). In addition, decreases in the litter C/P and C/N ratios in mixed plantations is a factor that favors litter decomposition by soil microorganisms and stimulates nutrient cycling (Bini et al., 2013aBini, D.; Santos, C.A.; Bouillet, J.; Gonçalves, J.L.D.M.; Cardoso, E.J.B.N. 2013a. Eucalyptus grandis and Acacia mangium in monoculture and intercropped plantations: Evolution of soil and litter microbial and chemical attributes during early stages of plant development. Applied Soil Ecology 63: 57-66.), mainly because of enzyme activities. Soil enzymes such as cellulase and phosphatases are good indicators of soil quality and mineralization rates of organic matter (Cardoso et al., 2013Cardoso, E.J.B.N.; Vasconcellos, R.L.F.; Bini, D.; Miyauchi, M.Y.H.; Santos, C.A.; Alves, P.R.L.; Paula, A.M.; Nakatani, A.S.; Pereira, J.M.; Nogueira, M.A. 2013. Soil health: looking for suitable indicators. What should be considered to assess the effects of use and management on soil health? Scientia Agricola 70: 280-295.). Phosphatases are enzymes produced by plants and/or microorganisms, which are able to hydrolyze organic P into mineral phosphates and, thus, represent the mineralizing of organic P (Krämer and Green, 2000Kramer, S.; Green, D.M. 2000. Acid and alkaline phosphatase dynamics and their relationship to soil microclimate in a semiarid woodland. Soil Biology and Biochemistry 32: 179-188.). The general aim of this manuscript was to evaluate the dynamics of AMF and soil enzyme activity in A. mangium and E. grandis plantations, and in mixed plantations during the first 20 months after planting.
Materials and Methods
Experimental area
The experiment was conducted in the municipality of Itatinga, SP (23º02′01″ S and 48º37′30″ W), with an average altitude of 830 m. The soil of the area is a Rhodic Ferralsol (FAO classification), with approximately the following constituents: 23 g kg–1 of organic matter, 11 mmolc kg–1 of Al3+ and 52 mmolc kg–1 of cation exchange capacity (CEC), 3.8 of pH (CaCl2) and 5.3 μg g–1 of available P (Bini et al., 2013aBini, D.; Santos, C.A.; Bouillet, J.; Gonçalves, J.L.D.M.; Cardoso, E.J.B.N. 2013a. Eucalyptus grandis and Acacia mangium in monoculture and intercropped plantations: Evolution of soil and litter microbial and chemical attributes during early stages of plant development. Applied Soil Ecology 63: 57-66.).
Prior to the experiment's installation, the experimental area had been occupied by a very old plantation of E. saligna (1940 to 1998) that had never been fertilized. After clear cutting, a plantation of E. grandis with low fertilization (300 kg ha–1 10:20:10 Nitrogen: Phosphorus: Potassium - NPK) was established, and the trees were harvested in Dec 2002. Later, in 2003, a first rotation of pure or intercropped E. grandis and A. mangium were planted and maintained until May 2009.
Experimental design and treatments
The experiment was installed in Nov 2009 by means of the implantation of a second rotation or a second cropping cycle of pure and intercropped E. grandis and A. mangium plantations, consisting of an array of three randomized blocks with tree treatments: (E) E. grandis monoculture (100E:0A); (A) A. mangium mono-culture (0E:100A); and intercropped plantation (50A:50E) of E. grandis (EMS) and A. mangium (AMS), all trees installed within lines, with the same experimental design and minimum tillage as had been used in the first rotation. Each plot measured 30 m × 30 m, in which only an inner plot of 18 m × 18 m (36 trees) was considered for sampling, leaving two buffer rows. Trees were spaced 3 m × 3 m in every treatment.
Soil, roots and litter sampling
Soil and roots (0-10 cm depth) as well as litter (0.25 m2 on the surface) were sampled at three intervals: 7 months after planting (a.p.) (winter – June 2010), 14 months a.p. (summer – Jan 2011) and 20 months a.p. (winter – July 2011). In Brazil's tropical climate we can differentiate only between winter (colder temperature and less rainfall, an average of 16 °C and 45 mm, respectively, from June to Aug) and summer (higher temperature and rainfall, average of 24 °C and 360 mm, respectively, from Dec to Feb). Spring and autumn are intermediary in their characteristics. The first AMF spore counting was performed 2 months after transplanting (Jan 2010), followed by the other three as previously described.
For sampling, nine representative trees were randomly selected in each of the three blocks with mono-cultures for each species. In intercropped areas, 18 trees were chosen, nine A. mangium and nine E. grandis seedlings, as explained above. Three soil, root and litter samples were taken from around each tree (at a distance of 1.5 m from the stem, between lines) and pooled to get three composite samples per block in monocultures and six composite samples in the intercropped plantation, three around A. mangium and three around E. grandis trees. Although considering that there might have been a certain degree of intermingled root growth from both tree species between lines, we made this separation during sampling just to learn about the most expressive influences of each particular root system on the surrounding AMF community.
The soil samples were sieved (2 mm) and stored at 4 °C up to 5 days for microbiological analysis. Air-dried soil was used for chemical analysis. Litter was sampled and consisted of twigs, branches and leaves, that were dried to constant weight at 45 °C.
The available soil P was extracted using ion-exchange resin (Van Raij et al., 2001) and soil pH was determined in a suspension with 0.01 mol L-1 CaCl2 in a 1:2.5 soil: water ratio. The total P in litter was quantified following Murphy and Riley (1962), whereas C and N in litter and in soil were measured in an elemental analyzer. Further information on the chemical analyses (C, N and P in soil and litter) can be found in Bini et al. (2013a)Bini, D.; Santos, C.A.; Bouillet, J.; Gonçalves, J.L.D.M.; Cardoso, E.J.B.N. 2013a. Eucalyptus grandis and Acacia mangium in monoculture and intercropped plantations: Evolution of soil and litter microbial and chemical attributes during early stages of plant development. Applied Soil Ecology 63: 57-66..
Activity of soil enzymes in soil samples
Acid and alkaline phosphatases were assessed by spectrophotometry (420 nm). One gram of fresh soil was incubated at 37 °C for 1 h, with 0.2 mL of toluene, 4 mL buffer (pH 6 for acid phosphatase and pH 11 for alkaline phosphatase) and 1 mL of p-nitrophenyl phosphate (0.05 mol L–1). After incubation, the filtered extract was treated with 1 mL of CaCl2 (0.5 mol L–1) and 5 mL of NaOH (0.5 mol L–1) and used for determining enzymatic activities, based on the hydrolysis of p-nitrophenyl and the determination of the p-nitrophenol generated (Tabatabai and Bremner, 1969Tabatabai, M.A.; Bremner, J.M. 1969. Use of p-nitrofenol phosphate for assay of soil phosphatase activity. Soil Biology and Biochemistry 1: 301-307.). Cellulase activity was evaluated as described by Schinner and von Mersi (1990)Schinner, F.; Von Mersi, W. 1990. Xylanase-, CM-cellulase and invertase activity in soil: an improved method. Soil Biology and Biochemistry 22: 511-515., using 10 g of fresh soil incubated at 50 °C for 24 h in phosphate buffer (pH 5.5). Carboxy-methylcellulose was used as substrate and the reducing sugars formed were quantified with a spectrophotometer (690 nm) according to the Prussian blue method.
Mycorrhizal root colonization
To obtain the percentage of AMF root colonization, roots were first cleared with 10 % KOH overnight followed by 10 min in 10 % H2O2. The use of H2O2 was necessary because these tree roots presented a pigmentation that was quite dark and needed bleaching for greater transparency of the roots and their visualization under microscope.
Subsequently, roots were stained with blue ink and submerged in glycerol (Vierheilig et al., 1998Vierheilig, H.; Goughlan, A.P.; Wyss, U.; Piché, Y. 1998. Ink and Vinegar, a simple staining technique for arbuscular mycorrhizal fungi. Applied and Environmental Microbiology 64: 5004-5007.). Stained roots were analyzed under a dissecting microscope at 40x magnification. Mycorrhizal colonization was estimated accounting for the presence or absence of fungal structures such as hyphae, arbuscules and vesicles inside the roots, using the grid-line intersection method (Giovannetti and Mosse, 1980Giovannetti, M.; Mosse, B. 1980. An evaluation of techniques for measuring vesicular-arbuscular mycorrhizal infection in roots. New Phytologist 84: 489-500.).
Quantification and identification of AMF spores
Spores were extracted from 50 g soil samples according to Gerdemann and Nicolson (1963)Gerdemann, J.W.; Nicolson, T.H. 1963. Spores of mycorrhizal Endogone species extracted from soil by wet sieving and decanting. Transactions of the British Mycological Society 46: 235-244.. The soil samples were wet-sieved through mesh sequences (from 0.71 to 0.045 mm). Subsequently, sequential centrifugations were performed with up to 70 % sucrose solutions to separate the spores from the soil particles. The extracted spores were counted, separated into groups of similar morphology, transferred and mounted on semipermanent slides with glycerol and polyvinyl alcohol resin and Melzer's reagent. Spores were identified at species level under a light microscope, using Schenck and Pérez's Manual (1990)Schenck, N.C.; Pérez, Y. 1990. Manual for the identification of VA mycorrhizal fungi. Synergistic, Gainesville. and descriptions provided by the International Culture Collection of (Vesicular) Arbuscular Mycorrhizal Fungi (INVAM http://invam.wvu.edu). After identifying the AMF species, we determined the number of spores in each species together with the total number of species (richness), Simpson's dominance index (Is), and Shannon's diversity index (H).
Statistical analyses
Data were subjected to analysis of variance (ANOVA) and Duncan's test for comparison of means (p ≤ 0.05), and Pearson's correlation analysis using SAS 9.2 software. The environmental and biological variables were tested by multivariate principal components analysis (PCA) to identify correlations between biological data (AMF, acid phosphatase, alkaline phosphatase and cellulase) and environmental data (N, P and C/N and C/P ratios of the soil and litter), for each sampling. The PCA was conducted using the Canoco 4.5 software program.
Results
There was a great fluctuation in spore numbers from one period to the next, sometimes increasing in one treatment and decreasing in another. Generalizing from all treatments, it would be possible to say that there was a quite low spore number at 2 months, with a tendency to decrease even more at 7 and 14 months after planting in a number of cases, followed by a more pronounced tendency to increase at 20 months or, at least, to recuperate equivalent values to those presented at 2 months (Table 1). The total number of AMF species found was 17, five species belonging to the genus Acaulospora, one to Ambispora, three to Gigaspora, three to Glomus, two to Racocetra, two to Scutellospora, and one unidentified specimen. Gigaspora decipiens and Racocetra intraornata stood out for being present in all samplings and all treatments. Acaulospora mellea showed higher numbers of spores at 2 and 20 months after planting. Shannon's index was higher at 2 months for pure Eucalyptus and at 20 months for three treatments (AMS, EMS and A). Simpson's index was always lowest at 20 months, and highest for three treatments at 7 months and, for pure Acacia, highest at 14 months (Table 1).
Number of spores (in 50 g soil) and ecological indices of AMF (Arbuscular mycorrhizal fungi) at 2, 7, 14 and 20 months after planting. A (pure Acacia); E (pure Eucalyptus); AMS (Acacia mixed stand); EMS (Eucalyptus mixed stand). Values followed by different lowercase letters differ within each evaluation period. Values followed by different uppercase letters show differences between the evaluation periods within each treatment (Duncan p < 0.05).
Overall, there was a high degree of AMF root colonization in all treatments and samplings, with roots showing up to 40 % colonization or even more, especially at 14 months (Figure 1). Pure Acacia presented the highest mycorrhizal colonization in all samplings. In contrast, there was overall, lower root colonization in the pure Eucalyptus. However, the E. grandis root colonization in the intercropped plantation was similar to that of pure Acacia and Acacia in mixed stands (AMS), at 14 and 20 months (Figure 1).
Arbuscular mycorrhizal fungi (AMF) root colonization at 7, 14 and 20 months (mths) after planting; values (with standard deviation) followed by different lowercase letters differ within each evaluation period (7, 14 or 20 months); values followed by different uppercase letters show differences between the evaluation periods within each treatment (Duncan p < 0.05).
Mixed plantations stimulated the activity of acid and/or alkaline phosphatase (Figures 2A, B, C and 3A, B and C). Acid phosphatase was more active in pure Acacia at 14 and 20 months (Figure 2A), although the inter-cropped plantation showed similar values at 7 months. On the other hand, alkaline phosphatase had higher activity in the intercropped plantation at all samplings, in the following order: AMS and EMS > pure Eucalyptus and pure Acacia (Figure 2B). In general, cellulase showed the highest activity in treatment E, mainly at 14 and 20 months, since it had little variation in the other treatments over the 20 month period (Figure 2C).
Acid phosphatase (A), alkaline phosphatase (B) and cellulase (C) activities under pure and intercropped plantation of E. grandis and A. mangium at 7, 14 and 20 months (mths) after planting; RS = reducing sugars; PNF = p-nitrophenol phosphate. Values (with standard deviation) followed by different lowercase letters differ within each treatment (Duncan p < 0.05).
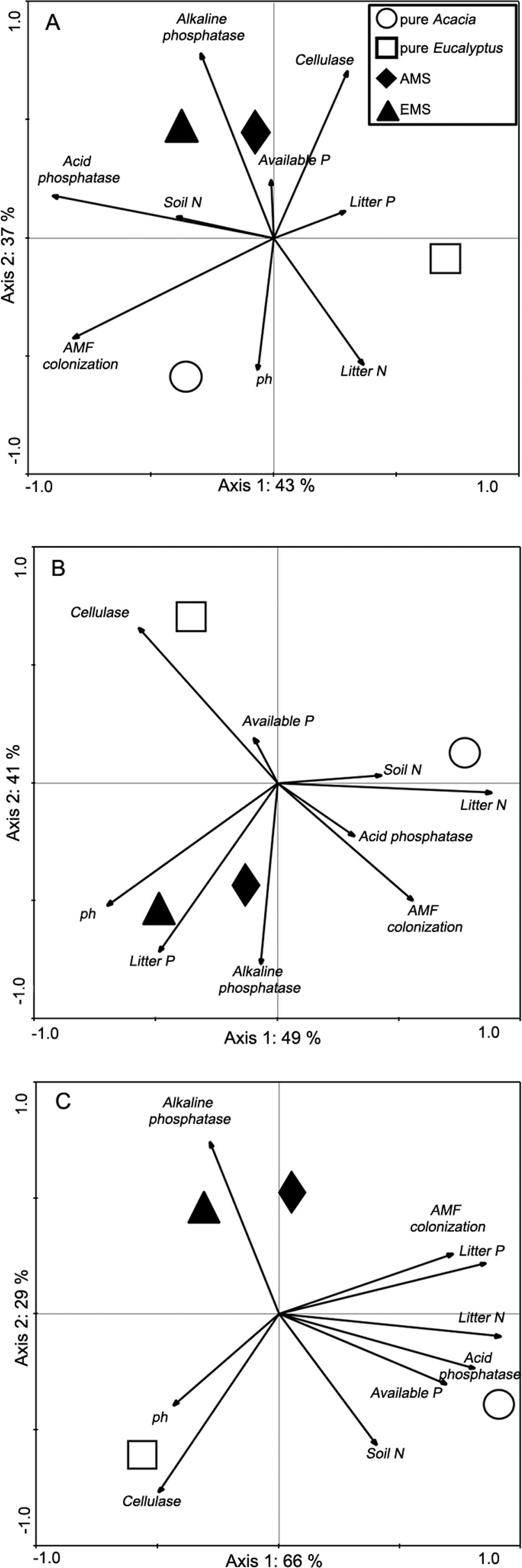
Principal Components Analysis (PCA) of the environmental and biological variables at 7 months (A), 14 months (B), 20 months (C). Geometrical forms (triangles, squares, diamonds and circles) represent the centroid of each treatment, from nine subsamples. AMS = Acacia mixed stand; EMS = Eucalyptus mixed stand; AMF = Arbuscular mycorrhizal fungi.
At 7 months, mycorrhizal root colonization correlated positively with soil total C and acid phosphatase activity. For the same 7 months, there was negative correlation between root colonization and cellulase activity, litter C content and litter C/N ratio, the latter persisting at all samplings (Table 2). There was positive correlation between AMF root colonization and litter N at 14 and 20 months, negative correlation between AMF root colonization and litter C/P ratio, and positive correlation between root colonization and litter P concentration and acid phosphatase activity was also recorded at 20 months (Table 2).
Pearson's correlation between litter or soil chemical data and AMF (Arbuscular mycorrhizal fungi) root colonization at 7, 14 and 20 months after planting.
For the PCA (Figure 3A, B and C) there was great similarity between samples originated from around Acacia or Eucalyptus trees in the mixed plantation, and this pattern was repeated at all samplings. In addition, pure Eucalyptus always appeared opposite pure Acacia in the figure. Furthermore, during the whole study period, we found a very close association between alkaline phosphatase and the intercropped plantation, in both the Eucalyptus or Acacia root systems (Figure 3A, B and C). Acid phosphatase activity was closer to pure Acacia at 20 months, while AMF colonization was equidistant from pure Acacia and the mixed plantation at 14 and 20 months. However, pure Eucalyptus was associated only with cellulase and pH, while pure Acacia was associated with AMF colonization, litter P, litter N, acid phosphatase, available P, and soil N. At 20 months (Figure 3C) there was closer association between alkaline phosphatase and the mixed plantation. At this sampling there was greater separation between the three treatments: axis 1 separated pure Acacia from pure Eucalyptus, while axis 2 separated the mixed plantation (represented by both plant species) from both monocultures.
Discussion
AMF sporulation dynamics at the initial stages after planting
High root colonization rates starting at 7 months after planting, when it was possible to get the first root samples, are probably due to an inoculum potential left by previous plantations of Eucalyptus grown for more than 50 years, and also by intercropped stands from the first rotation. However, this inoculum potential may have been due mostly to other AMF propagules, different from spores, perhaps as fungal external hyphae or colonized root pieces. At early successional stages in pioneer or initial secondary species, tree development shows greater dependence on AMF than at the more advanced stages (climax). In fact, this evidence can be found in both, in initial secondary forests (Zangaro et al., 2003Zangaro, W.; Nisizaki, S.M.A.; Domingos, J.C.B.; Nakano, E.M. 2003. Mycorrhizal response and successional status in 80 woody species from south Brazil. Journal of Tropical Ecology 19: 315-324.; Zangaro et al., 2007Zangaro, W.; Nishidate, F.; Vandresen, J.; Andrade, A.; Nogueira, M.A. 2007. Root mycorrhizal colonization and plant responsiveness are related to root plasticity, soil fertility and successional status of native woody species in southern Brazil. Journal of Tropical Ecology 23: 53-62.) and renewed Eucalyptus plantations (Adams et al., 2006Adams, F.; Reddell, P.; Webb, M.; Shipton, W. 2006. Arbuscular mycorrhizas and ectomycorrhizas on Eucalyptus grandis (Myrtaceae) trees and seedlings in native forests of tropical north-eastern Australia. Australian Journal of Botany 54: 271-281.). Initial plant development depends mostly on soil nutrients, and early AMF root colonization can be an advantage for the plant. At advanced growth stages, the plants become more efficient in recycling nutrients (biochemical cycling) which can decrease their demand for soil nutrients and fungal associations (Dighton and Mason, 1985Dighton, J.; Mason, P.A. 1985. Mycorrhizal dynamics during forest tree development. p. 117-139. In: Moore, D.; Casselton, L.A.; Wood, D.A.; Frankland, J.C., eds. Developmental Biology of Higher Fungi. Cambridge University Press, Cambrigde.). This has been verified generally in forest environments. Sometimes for certain plants this can also be said for ectomycorrhizae (Adams et al., 2006Adams, F.; Reddell, P.; Webb, M.; Shipton, W. 2006. Arbuscular mycorrhizas and ectomycorrhizas on Eucalyptus grandis (Myrtaceae) trees and seedlings in native forests of tropical north-eastern Australia. Australian Journal of Botany 54: 271-281.; Cheng et al., 2000Chen, Y.L.; Brundrett, M.C.; Dell, B. 2000. Effects of ectomycorrhizas and vesicular-arbuscular mycorrhizas, alone or in competition, on root colonization and growth of Eucalyptus globulus and E. urophylla. New Phytologist 146: 545-556.). No ectomycorrhizal structures were found on the roots during our study. It is known that ectomycorrhizae may predominate in Eucalyptus at later stages, after two to three years of growth, and are also frequent in certain leguminous trees (Pagano and Scotti, 2008Pagano, M.C.; Scotti, M.R. 2008. Arbuscular and ectomycorrhizal colonization of two Eucalyptus species in semiarid Brazil. Mycoscience 49: 379-384.).
Another interesting form of behavior of early plantings refers to the dynamics of mycorrhizal fungi in terms of spore density, which was similar for all treatments. Although the ecological indices showed high variation, we found low diversity, abundance and richness of spores at 7 and 14 months, which may be related to the spore germination process in response to the presence of tree roots and the release of chemical precursors that stimulate germination and root infection (Smith and Read, 1997Smith, S.E.; Read, D.J. 1997. Mycorrhizal symbiosis. Academic Press, New York, NY, USA.). Mycelial growth and root colonization can be prioritized at a number of developmental stages of AMF instead of spore multiplication (Moreira-Souza et al., 2003Moreira-Souza, M.; Trufem, S.F.B.; Gomes-da-Costa, S.M.; Cardoso, E.J.B.N. 2003. Arbuscular mycorrhizal fungi associated with Araucaria angustifolia (Bert.) O. Ktze. Mycorrhiza 13: 211-215.; Stürmer and Siqueira, 2011Stürmer, S.L.; Siqueira, J.O. 2011. Species richness and spore abundance of arbuscular mycorrhizal fungi across distinct land uses in Western Brazilian Amazon. Mycorrhiza 21: 255-267.), as probably occurred at 7 and 14 months, corroborated by high rates of root colonization. After 20 months of planting, there might have been a change in strategy, leading to emphasis on reproduction, triggered by several factors, such as seasonal weather changes or even the developmental phase of the host plant (Moreira-Souza et al., 2003Moreira-Souza, M.; Trufem, S.F.B.; Gomes-da-Costa, S.M.; Cardoso, E.J.B.N. 2003. Arbuscular mycorrhizal fungi associated with Araucaria angustifolia (Bert.) O. Ktze. Mycorrhiza 13: 211-215.; Pereira et al., 2014Pereira, C.M.R.; Silva, D.K.A.; Ferreira, A.C.D.A.; Goto, B.T.; Maia, L.C. 2014. Diversity of arbuscular mycorrhizal fungi in Atlantic forest areas under different land uses. Agriculture, Ecosystems and Environment 185: 245-252.). According to Pereira et al. (2014), instable areas, different host species, soil preparation, and crop rotation among others, may induce AMF sporulation to ensure their survival. The increase in diversity, abundance and richness of AMF spores in all areas at 20 months may be due to the reproduction of AMF associated with the host roots. Pagano et al. (2009) studied AMF in plantations of E. camaldulensis and Tabebuia heptaphylla and found greater spore diversity in inter-cropped plantations. In our experiment, this probably also occured, since by two months after planting the intercropped plantation had already presented greater diversity and richness of AMF spores (Table 1).
AMF colonization
In addition to the successional stage of forests, other factors can affect the degree of mycorrhizal root colonization, such as host specificity, seasonality, soil pH and texture, and other physical or chemical attributes, as well as available P (Zangaro et al., 2007Zangaro, W.; Nishidate, F.; Vandresen, J.; Andrade, A.; Nogueira, M.A. 2007. Root mycorrhizal colonization and plant responsiveness are related to root plasticity, soil fertility and successional status of native woody species in southern Brazil. Journal of Tropical Ecology 23: 53-62.). Higher root colonization observed during summer, at 14 months, regardless of the treatment, contrasted with lower root colonization in winter, displaying a seasonal effect. Regardless of the treatment, the low P availability (5.4 μg g–1) (see Bini et al., 2013aBini, D.; Santos, C.A.; Bouillet, J.; Gonçalves, J.L.D.M.; Cardoso, E.J.B.N. 2013a. Eucalyptus grandis and Acacia mangium in monoculture and intercropped plantations: Evolution of soil and litter microbial and chemical attributes during early stages of plant development. Applied Soil Ecology 63: 57-66.) in the soil may have stimulated the colonization rate even more than other factors (Smith and Read, 1997Smith, S.E.; Read, D.J. 1997. Mycorrhizal symbiosis. Academic Press, New York, NY, USA.).
Ignoring seasonal effects, AMF colonization was always higher in A. mangium monoculture and inter-cropped plantations (AMS and EMS), since this is a highly mycotrophic species (Mendes-Filho et al., 2009Mendes-Filho, P.F.; Vasconcellos, R.L.F.; Paula, A.M.; Cardoso, E.J.B.N. 2009. Evaluating the potential of forest species under “microbial management” for the restoration of degraded mining areas. Water, Air and Soil Pollution 208: 79-89.). According to Ghosh and Verma (2006)Ghosh, S.; Verma, N.K. 2006. Growth and mycorrhizal dependency of Acacia mangium Willd. inoculated with three vesicular arbuscular mycorrhizal fungi in Lateritic Soil. New Forest 31: 75-81., Acacia seedlings inoculated with AMF (Glomus mosseae, G. occultum and G. aggregatum) show an increase in shoot dry matter, root diameter, leaf area, chlorophyll content and bio-mass of A. mangium, when compared with the non-inoculated control plants. The greater mycorrhizal dependency of A. mangium and, consequently, maintenance of a higher inoculum potential in the surrounding soil, may have been the main reason for the higher rate of mycorrhizal colonization in the intercropped E. grandis at 20 months of transplanting, as reported by Pagano et al. (2009). Our results are corroborated by Adams et al. (2006), for whom the presence of understory may increase AMF colonization in monocultures of E. grandis. Thus, the presence of AMF colonized Acacia roots seems to stimulate AMF colonization in E. grandis. Many authors have reported an increase in AMF diversity and density in soils under mixed vegetation, in contrast to monocultures (Bainard et al., 2011Bainard, L.D.; Klironomos, J.N.; Gordon, A.M. 2011. Arbuscular mycorrhizal fungi in tree-based intercropping systems: A review of their abundance and diversity. Pedobiologia 54: 57-61.; Dobo et al., 2016Dobo, B.; Asefa, F.; Asfaw, Z. 2016. Diversity of arbuscular mycorrhizal fungi of different plant species grown in three land use types in Wensho and Shebidino Districts of Sidama in Southern Ethiopia. Advances in Bioscience and Bioengineering 4: 25-34.).
It is generally assumed that Eucalyptus trees form arbuscular mycorrhiza only at the seedling or plantlet stages (Adams et al., 2006Adams, F.; Reddell, P.; Webb, M.; Shipton, W. 2006. Arbuscular mycorrhizas and ectomycorrhizas on Eucalyptus grandis (Myrtaceae) trees and seedlings in native forests of tropical north-eastern Australia. Australian Journal of Botany 54: 271-281.; Chen et al., 2000Chen, Y.L.; Brundrett, M.C.; Dell, B. 2000. Effects of ectomycorrhizas and vesicular-arbuscular mycorrhizas, alone or in competition, on root colonization and growth of Eucalyptus globulus and E. urophylla. New Phytologist 146: 545-556.), while there is increased association with ectomycorrhizal fungi in older plants (Adams et al., 2006Adams, F.; Reddell, P.; Webb, M.; Shipton, W. 2006. Arbuscular mycorrhizas and ectomycorrhizas on Eucalyptus grandis (Myrtaceae) trees and seedlings in native forests of tropical north-eastern Australia. Australian Journal of Botany 54: 271-281.; Santos et al., 2001Santos, V.L.; Muchovej, R.M.; Borges, A.C.; Neves, J.C.L.; Kasuya, M.C.M. 2001. Vesicular-arbuscular-ectomycorrhiza succession in seedlings of Eucalyptus spp. Brazilian Journal of Microbiology 32: 81-86.), although it is not yet clear if this phenomenon always prevails. We showed an average of 37 % and 43 % AMF colonization at 20 months after planting in E and EMS, respectively, which is almost quadruple the colonization reported by Adams et al. (2005). Knowledge of the association between AMF and Eucalyptus and the dynamics of the mycorrhizal symbiosis in different management systems and soil types is precarious. According to Bainard et al. (2016), soil management can play an important role in maintaining the association between E. grandis and AMF. Therefore, a management system that enables the non-disruption of the network of underground hyphae, and a greater abundance of AMF spores in predominantly low-P soils, would have two highly desirable features that may result in higher AMF colonization.
Intercropping, AMF and nutrient cycling
The presence of leguminous species and root colonization increases the diversity and quality of litter in intercropped plantations and, therefore, decreases cellulase activity. Cellulase decomposes the recalcitrant and hard to break down cellulose, which is dominant in plant species such as Eucalyptus and presents a high C/N ratio. In all samplings cellulase activity was positively correlated with C concentrations of litter and high C/N and C/P ratios, and negatively with litter N concentrations and AMF colonization (Table 1; Figure 3A, B and C). Higher activity of cellulase was also found in soil that received plant residues with high C/N from Pinus, in comparison to residues from Araucaria from either native or secondary forest (Bini et al., 2013bBini, D.; Alcantara, C.; Banhos, K.; Kishino, N.; Andrade, G.; Zangaro, W.; Nogueira, M.A. 2013b. Effects of land use on soil organic carbon and microbial processes associated with soil health in southern Brazil. European Journal of Soil Biology 55: 117-123.). On the other hand, the intercropped system produces higher quality litter (Bini et al., 2013aBini, D.; Santos, C.A.; Bouillet, J.; Gonçalves, J.L.D.M.; Cardoso, E.J.B.N. 2013a. Eucalyptus grandis and Acacia mangium in monoculture and intercropped plantations: Evolution of soil and litter microbial and chemical attributes during early stages of plant development. Applied Soil Ecology 63: 57-66.), resulting in changes in soil enzymatic activity. The presence of A. mangium in the intercropped plantation with E. grandis offers a less recalcitrant mixture of plant litter with higher N concentration, thereby suppressing cellulase activity, especially in the rhizosphere. There are many reports on Eucalyptus intercropped with leguminous trees favoring N and P cycling, mostly due to a higher biological N fixation of the legume and higher mycorrhizal colonization of both plant species (Forrester et al., 2005Forrester, D.I.; Bauhus, J.; Cowie, A.L. 2005. On the success and failure of mixed-species tree plantations: lessons learned from a model system of Eucalyptus globulus and Acacia mearnsii. Forest Ecology and Management 209: 147-155.; Khanna, 1997Khanna, P.K. 1997. Comparison of growth and nutrition of young monocultures and mixed stand of Eucalyptus globulus and Acacia mearnsii. Forest Ecology and Management 94: 105-113.). According to Treseder (2013)Treseder, K.K. 2013. The extent of mycorrhizal colonization of roots and its influence on plant growth and phosphorus content. Plant and Soil 371: 1-13., high AMF colonization levels correlate with increased P concentration and plant growth. In our study, the increase in plant P concentration and P cycling in soil seem to be consequences of mycorrhizal colonization and phosphatase activities. This also explains the negative correlations between root colonization and litter C/N (in all samplings) as well as litter C/P (at 20 months) (Table 1), which indicate that systems with higher root colonization may generate litter of better quality, with narrower C/N and C/P ratios. We also found negative correlation between AMF and litter C/N ratios at all samplings, suggesting that AMF may be involved in the transference of soil N to plants, mainly for EMS, or that AMF are responsible for greater P acquisition which is fundamental to biological N fixation in legumes (Binkley, 1992Binkley, D. 1992. Mixtures of N2-fixing and non-N2-fixing tree species. p. 99-123. In: Cannell, M.G.R.; Malcom, D.C.; Robertson, P.A., eds. The ecology of mixed-species stands of trees. Blackwell Scientific, London, UK.; Hodge and Fitter, 2010Hodge, A.; Fitter, A.H. 2010. Substantial nitrogen acquisition by arbuscular mycorrhizal fungi from organic material has implications for N cycling. Proceedings of the National Academy of Sciences of the United States of America 107: 13754-13759.). However, we observed only negative correlation between AMF colonization and litter C/P at 20 months, since the cycling of P is slower than that of N (Forrester et al., 2005Forrester, D.I.; Bauhus, J.; Cowie, A.L. 2005. On the success and failure of mixed-species tree plantations: lessons learned from a model system of Eucalyptus globulus and Acacia mearnsii. Forest Ecology and Management 209: 147-155.). Furthermore, P cycling and phosphatase activity were maximized by N cycling in our study and AMF are possibly involved, especially with regard to EMS. According to Koutika et al. (2014), high translocation of P from soil to tree may explain the lower soil P availability in intercropped plantations of A. mangium and a first generation of the cross between E. urophylla × E. grandis. In fact, Tang et al. (2013)Tang, G.L.I.K.; Zhang, C.; Gao, C.; Li, B. 2013. Accelerated nutrient cycling via leaf litter, and not root interaction, increases growth of Eucalyptus in mixed-species plantations with Leucaena. Forest Ecology and Management 310: 45-53. have shown the growth stimulation of E. camaldulensis intercropped with legumes caused by the return of nutrients to the soil via litter. Therefore, mycorrhizal transfer of nutrients such as P to plants promotes the enrichment of the deposited litter, decreasing the C/P ratio. Furthermore, certain mycorrhizal plants acquire nutrients through an extensive hyphal network connecting one plant to another of the same or different species (Simard et al., 2003Simard, W.S.; Jones, M.D.; Durall, D.M. 2003. Carbon and nutrient fluxes within and between mycorrhizal plants. p. 34-74. In: Van der Heijden, M.G.A.; Sanders, I.R., eds. Mycorrhizal ecology. Springer, Berlin.). Thus plantations of E. grandis and A. mangium would help in the N nutrition of Eucalyptus via N transported from Acacia by AMF.
The increased AMF colonization may have additional positive effects, such as promoting the activity of acid phosphatase and alkaline phosphatase in inter-cropped plantations and pure plantations of A. mangium. In our study, conducted in soils with low available P, the dependency of plants on phosphatase activity (acid and alkaline) for mineralization of organic P may have been crucial for improving soil health and plant growth (Cardoso et al., 2013Cardoso, E.J.B.N.; Vasconcellos, R.L.F.; Bini, D.; Miyauchi, M.Y.H.; Santos, C.A.; Alves, P.R.L.; Paula, A.M.; Nakatani, A.S.; Pereira, J.M.; Nogueira, M.A. 2013. Soil health: looking for suitable indicators. What should be considered to assess the effects of use and management on soil health? Scientia Agricola 70: 280-295.; Hinsinger, 2001Hinsinger, P. 2001. Bioavailability of soil inorganic P in the rhizosphere as affected by root-induced chemical changes: a review. Plant and Soil 237: 173-195.; Vance et al., 2003Vance, C.P.; Uhde-Stone, C.; Allan, D.L. 2003. Phosphorus acquisition and use: critical adaptations by plants for securing a nonrenewable resource. New Phytologist 157: 423-447.). In intercropped or pure A. mangium we found maximum P cycling, also inferred by positive correlation between AMF colonization and acid phosphatase at 7 and 20 months. In general, the production of acid phosphatase is assigned mainly to plant roots and fungi (Dakora and Phillips, 2002Dakora, F.D.; Phillips, D.A. 2002. Root as mediators of mineral acquisition in low-nutrient environments. Plant and Soil 245: 35-47.) and there might have been additional stimulation for acquisition of organic P through the release of phosphatases by AMF. In addition, alkaline phosphatase activity was stimulated by intercropped plantations. This enzyme is produced mostly by bacterial groups (Dakora and Phillips, 2002Dakora, F.D.; Phillips, D.A. 2002. Root as mediators of mineral acquisition in low-nutrient environments. Plant and Soil 245: 35-47.). In this case, AMF may affect the diversity and structure of bacterial communities in the rhizosphere and hyphosphere (Toljander et al., 2006Toljander, J.F.; Artursson, V.; Paul, L.R.; Jansson, J.K.; Finlay, R.D. 2006. Attachment of different soil bacteria to arbuscular mycorrhizal fungal extraradical hyphae is determined by hyphal vitality and fungal species. FEMS Microbiology Letters 254: 34-40.; Andrade et al., 1997Andrade, G.; Mihara, K.L.; Linderman, R.G.; Bethlenfalvay, G.J. 1997. Bacteria from rhizosphere and hyphosphere soils of different arbuscular-mycorrhizal fungi. Plant and Soil 192: 71-79.). Hyphosphere refers only to the soil zone surrounding individual fungal hyphae (Andrade et al., 1997Andrade, G.; Mihara, K.L.; Linderman, R.G.; Bethlenfalvay, G.J. 1997. Bacteria from rhizosphere and hyphosphere soils of different arbuscular-mycorrhizal fungi. Plant and Soil 192: 71-79.), which may stimulate phosphatase activity (Taktek et al., 2015Taktek, S.; Trepanier, M.; Servin, P.M.; St-Arnaud, M.; Piche, Y.; Fortin, J.A.; Antoun, H. 2015. Trapping of phosphate solubilizing bacteria on hyphae of the arbuscular mycorrhizal fungus Rhizophagus irregularis DAOM 197198. Soil Biology and Biochemistry 90: 1-9.; Zhang et al., 2014Zhang, L.; Fan, J.; Ding, X.; He, X.; Zhang, F.; Feng, G. 2014. Hyphosphere interactions between an arbuscular mycorrhizal fungus and a phosphate solubilizing bacterium promote phytate mineralization in soil. Soil Biology and Biochemistry 74: 177-183.). Thus, we suggest that in either pure or intercropped A. mangium a double strategy for P acquisition occurs, i.e., via mycorrhizal hyphae and stimulation of phosphatase activity. This result is a clear demonstration that, during the first 20 months of growth in the mixed plantation there were no relevant differences between the root regions of the intercropped species, at least for the parameters that were evaluated in our study.
Acknowledgements
We would like to extend thanks to the São Paulo Research Foundation - FAPESP (Project n°. 2009/17525-6 and n°. 2010/16623-9), the National Agency for Research (ANR - 2010- STRA-004), the Programmed Thematic Action (ATP Neucapalm)/Centre for International Cooperation in Agricultural Research for Development (CIRAD) and the University of São Paulo (USP)/French Committee for the Evaluation of Academic and Scientific Cooperation with Brazil (COFECUB) for funding this project (n°. 22193PA). Thanks are also due to the staff of the Itatinga Forest Sciences Experimental Station and of the Laboratory of Soil Microbiology at the Luiz de Queiroz College of Agriculture for their assistance in conducting the experiments. D. Bini was supported by a doctoral grant (n°. 140893/2011-3) and E.J.B.N. Cardoso by a productivity grant (n°. 307563/2009-0) of the Brazilian National Council for Scientific and Technological Development (CNPq).
References
- Adams, F.; Reddell, P.; Webb, M.; Shipton, W. 2006. Arbuscular mycorrhizas and ectomycorrhizas on Eucalyptus grandis (Myrtaceae) trees and seedlings in native forests of tropical north-eastern Australia. Australian Journal of Botany 54: 271-281.
- Andrade, G.; Mihara, K.L.; Linderman, R.G.; Bethlenfalvay, G.J. 1997. Bacteria from rhizosphere and hyphosphere soils of different arbuscular-mycorrhizal fungi. Plant and Soil 192: 71-79.
- Bachega, L.R.; Bouillet, J.P.; Piccolo, M.C.; Saint-André, L.; Bouvet, J.M.; Nouvellon, Y.; Gonçalves, J.L.M.; Robin, A.; Laclau, J.P. 2016. Decomposition of Eucalyptus grandis and Acacia mangium leaves and fine roots in tropical conditions did not meet the Home Field Advantage hypothesis. Forest Ecology and Management 359: 33-43.
- Bainard, L.D.; Klironomos, J.N.; Gordon, A.M. 2011. Arbuscular mycorrhizal fungi in tree-based intercropping systems: A review of their abundance and diversity. Pedobiologia 54: 57-61.
- Binkley, D.; Dunkin, K.A.; Debell, D.; Ryan, M.G. 1992. Production and nutrient cycling in mixed plantations of Eucalyptus and Albizia in Hawaii. Forest Science 38: 393-408.
- Binkley, D. 1992. Mixtures of N2-fixing and non-N2-fixing tree species. p. 99-123. In: Cannell, M.G.R.; Malcom, D.C.; Robertson, P.A., eds. The ecology of mixed-species stands of trees. Blackwell Scientific, London, UK.
- Bini, D.; Santos, C.A.; Bouillet, J.; Gonçalves, J.L.D.M.; Cardoso, E.J.B.N. 2013a. Eucalyptus grandis and Acacia mangium in monoculture and intercropped plantations: Evolution of soil and litter microbial and chemical attributes during early stages of plant development. Applied Soil Ecology 63: 57-66.
- Bini, D.; Alcantara, C.; Banhos, K.; Kishino, N.; Andrade, G.; Zangaro, W.; Nogueira, M.A. 2013b. Effects of land use on soil organic carbon and microbial processes associated with soil health in southern Brazil. European Journal of Soil Biology 55: 117-123.
- Bouillet, J.P.; Laclau, J.P.; Gonçalves, J.L.M.; Voigtlaender, M.; Gava, J.L.; Leite, F.P.; Hakamada, R.; Mareschal, L.; Mabiala, A.; Tardy, F.; Levillain, J.; Deleporte, P.; Epron, D.; Nouvellon, Y. 2013. Eucalyptus and Acacia tree growth over entire rotation in single- and mixed-species plantations across five sites in Brazil and Congo. Forest Ecology and Management 301: 89-101.
- Cardoso, E.J.B.N.; Vasconcellos, R.L.F.; Bini, D.; Miyauchi, M.Y.H.; Santos, C.A.; Alves, P.R.L.; Paula, A.M.; Nakatani, A.S.; Pereira, J.M.; Nogueira, M.A. 2013. Soil health: looking for suitable indicators. What should be considered to assess the effects of use and management on soil health? Scientia Agricola 70: 280-295.
- Chen, Y.L.; Brundrett, M.C.; Dell, B. 2000. Effects of ectomycorrhizas and vesicular-arbuscular mycorrhizas, alone or in competition, on root colonization and growth of Eucalyptus globulus and E urophylla New Phytologist 146: 545-556.
- Dakora, F.D.; Phillips, D.A. 2002. Root as mediators of mineral acquisition in low-nutrient environments. Plant and Soil 245: 35-47.
- Dighton, J.; Mason, P.A. 1985. Mycorrhizal dynamics during forest tree development. p. 117-139. In: Moore, D.; Casselton, L.A.; Wood, D.A.; Frankland, J.C., eds. Developmental Biology of Higher Fungi. Cambridge University Press, Cambrigde.
- Dobo, B.; Asefa, F.; Asfaw, Z. 2016. Diversity of arbuscular mycorrhizal fungi of different plant species grown in three land use types in Wensho and Shebidino Districts of Sidama in Southern Ethiopia. Advances in Bioscience and Bioengineering 4: 25-34.
- Forrester, D.I.; Bauhus, J.; Cowie, A.L. 2005. On the success and failure of mixed-species tree plantations: lessons learned from a model system of Eucalyptus globulus and Acacia mearnsii Forest Ecology and Management 209: 147-155.
- Gerdemann, J.W.; Nicolson, T.H. 1963. Spores of mycorrhizal Endogone species extracted from soil by wet sieving and decanting. Transactions of the British Mycological Society 46: 235-244.
- Giovannetti, M.; Mosse, B. 1980. An evaluation of techniques for measuring vesicular-arbuscular mycorrhizal infection in roots. New Phytologist 84: 489-500.
- Ghosh, S.; Verma, N.K. 2006. Growth and mycorrhizal dependency of Acacia mangium Willd. inoculated with three vesicular arbuscular mycorrhizal fungi in Lateritic Soil. New Forest 31: 75-81.
- Hinsinger, P. 2001. Bioavailability of soil inorganic P in the rhizosphere as affected by root-induced chemical changes: a review. Plant and Soil 237: 173-195.
- Hodge, A.; Fitter, A.H. 2010. Substantial nitrogen acquisition by arbuscular mycorrhizal fungi from organic material has implications for N cycling. Proceedings of the National Academy of Sciences of the United States of America 107: 13754-13759.
- Khanna, P.K. 1997. Comparison of growth and nutrition of young monocultures and mixed stand of Eucalyptus globulus and Acacia mearnsii Forest Ecology and Management 94: 105-113.
- Koutika, L.S.; Epron, D.; Bouillet, J.P.; Mareschal, L. 2014. Changes in N and C concentrations, soil acidity and P availability in tropical mixed Acacia and eucalypt plantations on a nutrient-poor sandy soil. Plant and Soil 379: 205-216.
- Kramer, S.; Green, D.M. 2000. Acid and alkaline phosphatase dynamics and their relationship to soil microclimate in a semiarid woodland. Soil Biology and Biochemistry 32: 179-188.
- Laclau, J.P.; Bouillet, J.P.; Gonçalves, J.L.M.; Silva, E.V.; Jourdan, C.; Cunha, M.C.S.; Moreira, M.R.; Saint-André, L.; Maquère, V.; Nouvellon, Y.; Ranger, J. 2008. Mixed-species plantations of Acacia mangium and Eucalyptus grandis in Brazil: Biomass allocation and net primary production. Forest Ecology and Management 255: 3905-3917.
- Ma, Q.; Rengel, Z. 2008. Phosphorus acquisition and wheat growth are influenced by shoot phosphorus status and soil phosphorus distribution in a split-root system. Journal of Plant Nutrition and Soil Science 171: 266-271.
- Mendes-Filho, P.F.; Vasconcellos, R.L.F.; Paula, A.M.; Cardoso, E.J.B.N. 2009. Evaluating the potential of forest species under “microbial management” for the restoration of degraded mining areas. Water, Air and Soil Pollution 208: 79-89.
- Moreira-Souza, M.; Trufem, S.F.B.; Gomes-da-Costa, S.M.; Cardoso, E.J.B.N. 2003. Arbuscular mycorrhizal fungi associated with Araucaria angustifolia (Bert.) O. Ktze. Mycorrhiza 13: 211-215.
- Pagano, M.C.; Scotti, M.R. 2008. Arbuscular and ectomycorrhizal colonization of two Eucalyptus species in semiarid Brazil. Mycoscience 49: 379-384.
- Pagano, M.C.; Scotti, M.R.; Cabello, M.N. 2009. Effect of the inoculation and distribution of mycorrhizae in Plathymenia reticulata Benth under monoculture and mixed plantation in Brazil. New Forest 38: 197-214.
- Paula, R.R.; Bouillet, J.P.; Trivelin, P.C.O.; Zeller, B.; Gonçalves, J.L.M.; Nouvellon, Y.; Bouvet, J.M.; Plassard, C.; Laclau, J.P. 2015. Evidence of short-term belowground transfer of nitrogen from Acacia mangium to Eucalyptus grandis trees in a tropical planted forest. Soil Biology and Biochemistry 91: 99-108.
- Pereira, C.M.R.; Silva, D.K.A.; Ferreira, A.C.D.A.; Goto, B.T.; Maia, L.C. 2014. Diversity of arbuscular mycorrhizal fungi in Atlantic forest areas under different land uses. Agriculture, Ecosystems and Environment 185: 245-252.
- Santos, V.L.; Muchovej, R.M.; Borges, A.C.; Neves, J.C.L.; Kasuya, M.C.M. 2001. Vesicular-arbuscular-ectomycorrhiza succession in seedlings of Eucalyptus spp. Brazilian Journal of Microbiology 32: 81-86.
- Schenck, N.C.; Pérez, Y. 1990. Manual for the identification of VA mycorrhizal fungi. Synergistic, Gainesville.
- Schinner, F.; Von Mersi, W. 1990. Xylanase-, CM-cellulase and invertase activity in soil: an improved method. Soil Biology and Biochemistry 22: 511-515.
- Simard, W.S.; Jones, M.D.; Durall, D.M. 2003. Carbon and nutrient fluxes within and between mycorrhizal plants. p. 34-74. In: Van der Heijden, M.G.A.; Sanders, I.R., eds. Mycorrhizal ecology. Springer, Berlin.
- Smith, S.E.; Read, D.J. 1997. Mycorrhizal symbiosis. Academic Press, New York, NY, USA.
- Stürmer, S.L.; Siqueira, J.O. 2011. Species richness and spore abundance of arbuscular mycorrhizal fungi across distinct land uses in Western Brazilian Amazon. Mycorrhiza 21: 255-267.
- Tabatabai, M.A.; Bremner, J.M. 1969. Use of p-nitrofenol phosphate for assay of soil phosphatase activity. Soil Biology and Biochemistry 1: 301-307.
- Taktek, S.; Trepanier, M.; Servin, P.M.; St-Arnaud, M.; Piche, Y.; Fortin, J.A.; Antoun, H. 2015. Trapping of phosphate solubilizing bacteria on hyphae of the arbuscular mycorrhizal fungus Rhizophagus irregularis DAOM 197198. Soil Biology and Biochemistry 90: 1-9.
- Tang, G.L.I.K.; Zhang, C.; Gao, C.; Li, B. 2013. Accelerated nutrient cycling via leaf litter, and not root interaction, increases growth of Eucalyptus in mixed-species plantations with Leucaena Forest Ecology and Management 310: 45-53.
- Toljander, J.F.; Artursson, V.; Paul, L.R.; Jansson, J.K.; Finlay, R.D. 2006. Attachment of different soil bacteria to arbuscular mycorrhizal fungal extraradical hyphae is determined by hyphal vitality and fungal species. FEMS Microbiology Letters 254: 34-40.
- Treseder, K.K. 2013. The extent of mycorrhizal colonization of roots and its influence on plant growth and phosphorus content. Plant and Soil 371: 1-13.
- Vance, C.P.; Uhde-Stone, C.; Allan, D.L. 2003. Phosphorus acquisition and use: critical adaptations by plants for securing a nonrenewable resource. New Phytologist 157: 423-447.
- Vierheilig, H.; Goughlan, A.P.; Wyss, U.; Piché, Y. 1998. Ink and Vinegar, a simple staining technique for arbuscular mycorrhizal fungi. Applied and Environmental Microbiology 64: 5004-5007.
- Voigtlaender, M.; Laclau, J.P.; Gonçalves, J.L.M.; Piccolo, M.D.C.; Moreira, M.Z.; Nouvellon, Y.; Ranger, J.; Bouillet, J.P. 2012. Introducing Acacia mangium trees in Eucalyptus grandis plantations: consequences for soil organic matter stocks and nitrogen mineralization. Plant and Soil 352: 99-111.
- Zangaro, W.; Nisizaki, S.M.A.; Domingos, J.C.B.; Nakano, E.M. 2003. Mycorrhizal response and successional status in 80 woody species from south Brazil. Journal of Tropical Ecology 19: 315-324.
- Zangaro, W.; Nishidate, F.; Vandresen, J.; Andrade, A.; Nogueira, M.A. 2007. Root mycorrhizal colonization and plant responsiveness are related to root plasticity, soil fertility and successional status of native woody species in southern Brazil. Journal of Tropical Ecology 23: 53-62.
- Zhang, L.; Fan, J.; Ding, X.; He, X.; Zhang, F.; Feng, G. 2014. Hyphosphere interactions between an arbuscular mycorrhizal fungus and a phosphate solubilizing bacterium promote phytate mineralization in soil. Soil Biology and Biochemistry 74: 177-183.
Edited by
Publication Dates
-
Publication in this collection
Mar-Apr 2018
History
-
Received
27 Aug 2016 -
Accepted
02 Feb 2017