Abstract
In this work the modelling of a process of textile dyeing of a single cotton thread is presented. This thread moves at a constant velocity within a homogeneous dye solution under steady state conditions. The method of volume averaging is applied to obtain the mass transfer equations related to the diffusion and adsorption process inside the cotton thread on a small scale. The one-equation model is developed for the fiber and dye solution system, assuming the principle of local mass equilibrium to be valid. On a large scale, the governing equations for the cotton thread, including the expression for effective diffusivity tensor, are obtained. Solution of these equations permits the dye concentration profile for inside the cotton thread and in the dyeing batch to be obtained and the best conditions for the dyeing process to be chosen.
textile; dyeing; modeling
TRANSPORT PHENOMENA UNIT OPERATION
The modelling of a textile dyeing process utilizing the method of volume averaging
A.A.Ulson de SouzaI; S.WhitakerII
IDepartamento de Engenharia Química e Engenharia de Alimentos, Universidade Federal de Santa Catarina, Phone: (+55) (48) 331-9448, Fax: (+55) (48) 331-9687, Cx. P. 476, 88040-900, Florianópolis - SC, Brazil E-mail: augusto@enq.ufsc.br
IIDepartment of Chemical Engineering and Material Science, Phone (+1) (916) 752-8775, Fax (+1) (916) 752-1031 University of California at Davis, CA 95616, U.S.A. E-mail: swhitaker@ucdavis.edu
ABSTRACT
In this work the modelling of a process of textile dyeing of a single cotton thread is presented. This thread moves at a constant velocity within a homogeneous dye solution under steady state conditions. The method of volume averaging is applied to obtain the mass transfer equations related to the diffusion and adsorption process inside the cotton thread on a small scale. The one-equation model is developed for the fiber and dye solution system, assuming the principle of local mass equilibrium to be valid. On a large scale, the governing equations for the cotton thread, including the expression for effective diffusivity tensor, are obtained. Solution of these equations permits the dye concentration profile for inside the cotton thread and in the dyeing batch to be obtained and the best conditions for the dyeing process to be chosen.
Keywords: textile, dyeing, modeling.
INTRODUCTION
The problem under study is illustrated in Figure 1, which shows a uniform cotton thread (w-region), moving at a constant velocity, uo, within a homogeneous dye solution. The w-region is composed of fibers (s-region) and the dye solution inside the thread (b-phase). The concentration of dye in the thread at x = 0 is CAwo, and the concentration in the h-region at y ~ ¥ is a constant value, CA¥.
A small scale can be identified inside the s-region as shown in Figure 1. On this small scale, two phases can be characterized: liquid in the microfibers, g-phase, and solid, k-phase (Plumb and Whitaker, 1988a, b, 1990). The k-phase refers to the cotton microfibers (Trotman, 1975; Holme, 1986), where the adsorption process occurs.
g - k SYSTEM AVERAGING
The governing differential equations and boundary conditions for the mass transfer process in both the g-phase and the k-phase, illustrated in Figure 1, are given by
It is assumed that in the interface the diffusive flux from the g-phase to the k-phase is equal to the adsorption rate.
The k-phase is assumed to be a rigid phase and the adsorption isotherm is a linear function expressed as
Here CAg represents the molar concentration of chemical species under study (mol/m3), CAs represents the surface concentration (mol/m2), and Dg is the g-phase molecular diffusivity of species A (Whitaker, 1992). The entrances and exits of the g-phase at the boundary of the s-region are represented by variable  . Variable
. Variable  is used to represent the entire interfacial area within that region. The g-phase and the k-phase and the s-b system move at the same velocity in relation to the coordinate system; these two scales are confined to within the cotton fibers.
is used to represent the entire interfacial area within that region. The g-phase and the k-phase and the s-b system move at the same velocity in relation to the coordinate system; these two scales are confined to within the cotton fibers.
The intrinsic average concentration is defined by
The spatial averaging theorem (Howes and Whitaker, 1985) for volume  can be expressed as
can be expressed as
in which Agk represents the interfacial area g-k contained within averaging volume  .
.
The integration of Eqs. (1) through (4) in volume  , using the spatial averaging theorem as presented by Ochoa-Tapia et al. (1993) and Whitaker (1999), results in the volume-averaged form of Eq. (1), given by
, using the spatial averaging theorem as presented by Ochoa-Tapia et al. (1993) and Whitaker (1999), results in the volume-averaged form of Eq. (1), given by
A key aspect of the process of spatial smoothing isthat the boundary condition given by Eq. (2) is combined with the governing equation. The area average concentration can be replaced by the intrinsic average concentration,  when the following length-scale constraints, lg << rs and
when the following length-scale constraints, lg << rs and  << l, are satisfied (Ochoa-Tapia et al., 1993; Whitaker, 1999).
<< l, are satisfied (Ochoa-Tapia et al., 1993; Whitaker, 1999).
Here avgkrepresents the surface area per unit volume, given by
and the spatial deviation concentration can be expressed as
The Closure Problem
At this point a representation for the spatial deviation concentration needs to be developed.
Subtracting Eq. (8) divided by eg from Eq. (1), one can obtain
The interfacial boundary condition for the deviation concentration can be expressed as
Since the source  only influences the
only influences the  field over a distance on the order of lg, we can generally replace the boundary condition imposed at
field over a distance on the order of lg, we can generally replace the boundary condition imposed at  with a spatially periodic condition for
with a spatially periodic condition for  (Whitaker, 1999). So, when the spatially periodic model is used and
(Whitaker, 1999). So, when the spatially periodic model is used and  is much greater than one, the boundary value problem can be rewritten as
is much greater than one, the boundary value problem can be rewritten as
Closure Variables
The boundary value problem for deviation concentration is solved by the method of superposition, where a proposed solution is given by
Whitaker (1999) proves that y = constant is the only solution. Since this additive constant will not pass through the filter, the value of y plays no role in the closed form of the volume averaged diffusion equation.
Here b and the scalar s are the closure variables and y is an arbitrary function (Whitaker, 1999). The two closure variables can be determined according to the following two boundary value problems:
Problem I
Problem II
Closed Form
The closed form of the governing equation for the intrinsic average concentration, <CAg >g, can be obtained by substitution of Eq. (18) into Eq. (8). The resulting equation can be expressed as
where the effective diffusivity tensor is defined by
and vector u is defined by
Here the diffusive tensor, Deff, depends only on the geometry of the porous medium (Whitaker, 1999).
One can use Eq. (23) and Eq. (27) for estimating the order of s and u. Using these results in Eq. (25), Whitaker (1999) demonstrated that the advective term can be neglected for the case of diffusion in porous solids. The final form of the local average diffusion and transport equation is given by
s-b SYSTEM AVERAGING
In this section we will develop the spatially smoothed equations associated with volume  , shown in Figure 1. The length scales related to this averaging volume are identified in Figure 1. The boundary value problem associated with the local volume averaging procedure is given by
, shown in Figure 1. The length scales related to this averaging volume are identified in Figure 1. The boundary value problem associated with the local volume averaging procedure is given by
in the s-region
The s-Region
Integration of Eq. (29) over  , illustrated in Figure 1, results in
, illustrated in Figure 1, results in
in which the nomenclature for the s-region has been simplified by using the relationship  , where
, where
By using the averaging theorem and following the same procedure as that adopted previously and assuming that the restriction ls << rw is satisfied, Eq. (34) can be expressed as
Here js represents the volume fraction of the s-region contained in the volume  .
.
The Closure Problem
Analogously to the previous procedure, here a representation for the spatial deviation concentration is required. The use of the spatial deviation concentration defined by Gray (1975) and applied to the s-region results in
The spatial deviation concentration equation can be obtained by subtracting Eq. (36) divided by js from Eq. (29), and the resulting equation can be simplified when the following restrictions are satisfied:  . Under these circumstances Eq. (36) can be rewritten as
. Under these circumstances Eq. (36) can be rewritten as
The b-Phase
The volume averaging form of Eq. (33) in volume  , using the averaging theorem, is given by
, using the averaging theorem, is given by
The deviation concentration for the b-phase is given by  .
.
The Closure Problem
One can see that the subtraction of Eq. (41) divided by jb from Eq. (33) results in the governing equation for the deviation concentration, which is given by
By analysis of the order of the terms in Eq. (42), and assuming the length-scale constraints given by lb << rw and  <<1, one can conclude that the nonlocal term can be considered negligible compared to the diffusion term and the closure process can be considered quasi-steady. Under these circumstances, Eq. (42) can be rewritten as
<<1, one can conclude that the nonlocal term can be considered negligible compared to the diffusion term and the closure process can be considered quasi-steady. Under these circumstances, Eq. (42) can be rewritten as
One-Equation Model
Making the assumption that the principle of local mass equilibrium (Quintard and Whitaker, 1993; Whitaker, 1986 a, b) is valid, we can write
Here <CA>* is the spatial average concentration defined as
The following definitions
can be used with Eqs. (36) and (41) to give
Here we have defined the overall effective diffusivity as
Closure Variables
Considering that the local closure problem has a unique nonhomogeneous term proportional to the gradient of the spatial average concentration evaluated on the centroid, one can write
where bb and bs are the closure variables.
The following boundary value problem needs to be solved:
One can show that y = x = constant. This constant will not pass through the filter represented by area integrals in Eq. (49), as suggested by Whitaker (1999). So the value of this constant plays no role in the closed-form equation.
The Closed Form
Substituting the expressions given by Eq. (50) and Eq. (51) for the spatial deviation concentrations in Eq. (49), taking into consideration solution of the boundary value problem, one can obtain
where
THE w-h SYSTEM
The w-Region
At this point we need to consider the w-region motion related to the h-phase and for this circumstance the time derivative of average concentration of species A in the w-region can be expressed as
By simplification Eq. (59), we can write
The subscript on the left side of Eq. (60) does not indicate what is being held constant, but instead indicates the velocity of the observer who is measuring the concentration. On the basis of Eq. (60) the governing equation for the w-region can be written as
The h-Phase
The governing equation for the h-phase is given by
One can assume that the boundary layer solution for the hydrodynamic problem in the h-phase is acceptable, and in this circumstance the velocity profiles obtained by Sakiadis (1961a, b, c) can be used in Eq. (62).
CONCLUSIONS
The model of a single cylinder cotton thread, developed using the method of volume averaging for the adsorption dyeing process, represents a fundamental approach in this area. Two scales were considered in order to formulate this problem. The k-phase, inside the s-region, is composed of microfibers where the adsorption process occurs. The w-region, containing the s-b system, moves at a constant velocity. The method of volume averaging is applied to obtain the mass transfer equations related to the adsorption process on the small and the large scale. The one-equation model is developed for the b - s system, assuming the local mass equilibrium. The simulation results and validation of this model as well as the effective mass diffusivity obtained by solution of closure problems will be presented in a subsequent paper.
ACKNOWLEGEMENTS
This work was done when Antônio Augusto Ulson de Souza was a postdoctoral student at University of California, Davis, with financial support from the Fundação Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, CAPES, Brazil.
NOMENCLATURE
gk
Interfacial area of the g-k system, m2
ge
Area of entrances and exits for the g-phase, m2
sb
Interfacial area of the s-b system, m2
se
Area of entrances and exits for the s-region, m2
be
Area of entrances and exits for the b-phase, m2
Agk
The g-k interfacial area contained within the averaging volume,
s, m2Age
Area of entrances and exits for the g-phase contained within the averaging volume,
s, m2Asb
The s-b interfacial area contained within the averaging volume,
w, m2avgk
The g-k interfacial area per unit volume, m-1
CA¥
Concentration in the h-phase outside the boundary layer, kgmol/ m3
CAg
Point concentration in the g-phase, kgmol/ m3
<CAg>g=Cs
Intrinsic averaged concentration in the g-phase, kgmol/ m3
Ag
Spatial deviation concentration in the g-phase, kgmol/ m3
CAb
Point concentration in the b-phase, kgmol/ m3
CAh
Point concentration in the h-phase, kgmol/ m3
<CAb>b
Intrinsic regional averaged concentration for the b-phase, kgmol/ m3
<Cs>
Superficial regional averaged concentration for the s-region, kgmol/ m3
<Cs>s
Intrinsic regional averaged concentration for the s-region, kgmol/ m3
s
Spatial deviation concentration in the s-region, kgmol/ m3
Ab
Spatial deviation concentration in the b-phase, kgmol/ m3
Intrinsic spatial averaged concentration for the s-b system, kgmol/ m3
Dg
The g-phase molecular diffusivity, m2/s
Deff
The g-phase effective diffusivity tensor, m2/s
Db
The b-phase molecular diffusivity, m2/s
Dh
The h-region molecular diffusivity, m2/s
Effective diffusivity tensor for the s-b system , m2/s
I
Unit tensor
<K>
The averaged adsorption equilibrium constant, m
Keq
Adsorption equilibrium constant, m
w
Characteristic length of the w-region, m
s
Characteristic length of the s-region, m
b
Characteristic length of the b-phase, m
g
Characteristic length of the g-phase, m
l i
Lattice vectors describing a spatially periodic porous medium, m
Long length for volume averaged quantities associated with the w-h system, m
ngk
Outwardly directed unit normal vector pointing from the g-phase toward the k-phase
nsb
Outwardly directed unit normal vector pointing from the s-region toward the b-phase
Radius of the g-k system averaging volume,
s, mRadius of the s-b system averaging volume,
w, mTime, s
Characteristic time, s
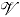 s
s
Small-scale averaging volume, m3
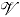 w
w
Large-scale averaging volume, m3
The w-region velocity vector, m/s
The h-phase velocity vector, m/s
Volume of the g-phase contained within
s, m3Volume of the s-region contained within
w, m3Mass boundary layer
Hydrodynamic boundary layer
eg
The g-phase volume fraction in the g-k system
The s-region volume fraction in the s-b system
The b-phase volume fraction in the s-b system
The averaged porosity
Received: October 25, 2001
Accepted: July 3, 2003
- Gray, W.G., A Deviation of the Equations for Multiphase Transport, Chemical Engineering Science, 30, 229-233 (1975).
- Holme, I., The Effects of Chemical and Physical Properties on Dyeing and Finishing, in C. Preston (ed.), The Dyeing of Cellulosic Fibres, Chap. 3, Dyers Company Publications Trust (1986).
- Howes, F.A. and Whitaker, S., The Spatial Averaging Theorem Revisited, Chemical Engineering Science, vol. 40, pp. 1387-1392 (1985).
- Ochoa-Tapia, Chem. Engng. Sci., c.48 (1993).
- Plumb, O.A. and Whitaker, S., Dispersion in Heterogeneous Porous Media I. Local Volume Averaging and Large Scale Averaging, Water Resources Research, 24, 913-926 (1988a).
- Plumb, O.A. and Whitaker, S., Dispersion in Heterogeneous Porous Media II. Predictions for Stratified and Two-dimensional Spatially Periodic Systems, Water Resources Research, 24, 927-938 (1988b).
- Plumb, O.A. and Whitaker, S., Diffusion, Adsorption and Dispersion in Porous Media: Small-scale Averaging and Local Volume Averaging, in J. H. Cushman (ed.), Dynamics of Fluids in Hierarchical Porous Media, Chap. 5, Academic Press, New York (1990).
- Quintard, M. and Whitaker, S., One and two-equation Models for Transient Diffusion Processes in Two-phase Systems, in Advances in Heat Transfer, 23, 369-465, Academic Press, New York (1993).
- Sakiadis, B. C., Boundary-layer Behavior on Continuous Solid Surfaces: I. Boundary-layer Equations for Two-dimensional and Axisymmetric Flow, A.I.Ch.E. Journal, 7, 26-28 (1961a).
- Sakiadis, B.C., Boundary-layer Behavior on Continuous Solid Surfaces: II. The Boundary-layer on a Continuous Flat Surface, A.I.Ch.E. Journal, 7, 221-225 (1961b).
- Sakiadis, B.C., Boundary-layer Behavior on Continuous Solid Surfaces: III. The Boundary-layer on a Continuous Cylindrical Surface, A.I.Ch.E. Journal, 7, 467-472 (1961c).
- Trotman E. R., Dyeing and Chemical Technology of Textile Fibres, Charles Griffin & Co. LTD, London (1975).
- Whitaker, S., Transient Diffusion Adsorption and Reaction in Porous Catalysts: The Reaction Controlled, Quasi-steady Catalytic Surface, Chemical Engineering Science, 41, 3015-3022 (1986a).
- Whitaker, S., Local Thermal Equilibrium: An Application to Packed Bed Catalytic Reactor Design, Chemical Engineering Science, 41, 2029-2039 (1986b).
- Whitaker, S., The Species Mass Jump Condition at a Singular Surface, Chemical Engineering Science, 47, 1677-1685 (1992).
- Whitaker, S., Theory and Application of Transport in Porous Media: The Method of Volume Averaging, London, Kluwer Academic, 219p (1999).
Publication Dates
-
Publication in this collection
04 Dec 2003 -
Date of issue
Oct 2003
History
-
Accepted
03 July 2003 -
Received
25 Oct 2001














































