Abstract
In this study, various water-soluble and oil-soluble demulsifiers were selected for separation of water from crude oil emulsions and their productivity measured using the Bottle-test method at 70 °C and 10 ppm concentration. The best ones among 23 demulsifiers examined through the screening process were fatty alcohol ethoxylate, triethanol amine and urea from the water-soluble group and Basororol E2032, Basorol PDB 9935 and TOMAC from the oil-soluble category. Furthermore, the present study investigated the factors effective for demulsification such as temperature, concentration, pH, salinity and modifiers. It was found that the separation improves with increasing demulsifier concentration, increasing salt content, increasing temperature up to 80 °C, keeping the pH values between 5-9. Adding solvent modifiers proved unnecessary. Two formulations were prepared based on suggested optimal concentrations of demulsifier content by experimental design using Qualitec 4 and these proved to be highly effective in treating real and synthetic emulsions.
Crude oil; Demulsifier; Water-in-oil emulsion; Formulation
INTRODUCTION
Water-in-oil emulsions are formed during production of crude oil, which is often accompanied by water. Under the production conditions, a proportion of this water is usually intimately dispersed throughout the crude oil as small droplets. In order to minimize the production problems related to crude oil emulsions and environmental concerns, petroleum operators need to prevent the formation of or break down these emulsions. The emulsification of water in oil is normally difficult due to the immiscibility between these two liquid phases. However, shear mixing imposed on the fluids during production and the existence of natural surfactants in the petroleum's composition contribute to formation of such emulsions (Sjoblom et al., 2005Sjoblom, J., Hemmingsen, P. V., Kallevik, H., The Role of Asphaltenes in Stabilizing Water-in-Crude Oil Emulsions. In: O. C., Mullins, E. Y., Sheu, A., Hammami, A. G., Marshall, (Eds.), Asphaltenes, Heavy Oils, and Petroleomics, Springer, New York, p. 549 (2007).; Ramalho et al., 2010Ramalho, J. B. V. S., Lechuga, F. C., Lucas, E. F., Effect of the structure of commercial poly(ethylene oxide-b-propylene oxide) demulsifier bases on the demulsification of water-in-crude oil emulsions: Elucidation of the demulsification mechanism. Quimica Nova, 33(8) 1664-1670 (2010).). Naturally occurring emulsifiers are concentrated in the higher-boiling polar fraction of the crude oil (Jones et al., 1978Jones, T. J., Neustadter, E. L., Wittingham, K. P., Water-in-crude oil emulsion stability and emulsion destabilization by chemical demulsifiers. J. Cdn. Pet. Tech. 100, April-June (1978).; Strassner, 1968Strassner, J. E., Effect of pH on interfacial films and stability of crude oil-water emulsions. JPT 303, March (1968).; Kimbler et al., 1966Kimbler, O. K., Reed, R. L., Silberberg, I. H., Physical characteristics of natural films formed at the crude oil-water interfaces. JPT 153, June (1966).; Borba, 1990Bobra, M., A Study of the Formation of Water-in-Oil Emulsions, Proc., 1990 Arctic and Marine Oil Spill Program Technical Seminar, Edmonton, Canada (1990).; Kokal and Aljuraid, 1999Kokal, S. L., Al-Juraid, J. I., Quantification of various factors affecting emulsion stability: Water cut, temperature, shear, asphaltene content, demulsifier dosage and mixing different crudes. Paper SPE 56641, SPE Annual Technical Conference and Exhibition, Houston 3-6 October (1999).; Svetgoff, 1989Svetgoff, J. A., Demulsification key to production efficiency. Petroleum Engineer. Intl., 61(8), 28 (1989).; Eley et al., 1998Eley, D. D., Hey, M. J., Symonds, J. D., Emulsions of water in asphaltene containing oils. Colloids & Surfaces, 32, 87(1998).). These include asphaltenes, resins, and oil-soluble organic acids (e.g. naphthenic, carboxylic) and bases, which are the main constituents of the interfacial films surrounding the water droplets and that provide emulsion stability.
Asphaltenes are dark brown to black friable solids with no definite melting point. They consist of condensed aromatic sheets with alkyl and cyclic side chains and heteroatoms (nitrogen, oxygen, sulfur) and trace metals like vanadium and nickel scattered throughout (Daaou et al., 2009Daaou, M., Bendedouch, D., Bouhadda, Y., Vernex-Loset, L., Modaressi, A., Rogalski, M., Explaining the flocculation of Hassi Messaoud asphaltenes in terms of structural characteristics of monomers and aggregates. Energy & Fuels, 23, 5556-5563 (2009).). These molecules can have 30 or more carbons, and they are usually considered to be the heaviest and the most polar components of the petroleum, but recent work shows that the average molecular weight of asphaltenes extracted with n-heptane is about 750 g/mol (Groezin and Mullins, 2007Groezin, H., Mullins, O. C., Asphaltene Molecular Size and Weight by Timeresolved Fluorescence Depolarizarion. In: Mullins, O. C., Sheu, E. Y., Hammami, A., Marshall, A. G., (Eds.), Asphaltenes, Heavy Oils, and Petroleomics, Springer, New York, 2007, p. 17.) and surprisingly, lower values (around 400 g/mol) have also been found (Honse et al., 2012Honse, S. O., Ferreira, S. R., Mansur, C. R. E., Lucas, E. F., Gonzalez, G., Separation and characterization of asphaltenic subfractions. Quimica Nova, 35(10), 1991-1994 (2012).). They are characterized by a relatively constant hydrogen/carbon ratio of 1.15 with a specific gravity near one; however, the nature of asphaltenes in crude oil is still a subject of debate. Asphaltenes are believed to exist in the oil as a colloidal suspension and to be stabilized by resins adsorbed on their surface (Leontaritis and Mansoori, 1998Leontaritis, K. J., Mansoori, G. A., Asphaltene deposition: A comprehensive description of problem manifestations and modeling approaches. Paper SPE 18892, SPE Production and Operations Symposium, Oklahoma City, Oklahoma, 13-14 March (1998).). In this regard, the resins act as peptizing agents for asphaltenes and together form clusters called micelles. These micelles or colloids contain most of the polar material found in the crude oil and possess surface-active properties (interfacial active material). They in turn result from sulfur, nitrogen, oxygen, and metal-containing entities in asphaltene molecules that form polar groups such as aldehydes, carbonyls, carboxylic acids, amines, and amides, and play a key role in stabilizing emulsions. Resins are complex high-molecular-weight compounds that are not soluble in ethylacetate but soluble in n-heptane. It appears that the asphaltene-resin ratio in crude oil is responsible for the type of film formed (solid or mobile) and, therefore, is directly linked to the stability of the emulsion (Strassener, 1968Strassner, J. E., Effect of pH on interfacial films and stability of crude oil-water emulsions. JPT 303, March (1968).; Borba, 1990Bobra, M., A Study of the Formation of Water-in-Oil Emulsions, Proc., 1990 Arctic and Marine Oil Spill Program Technical Seminar, Edmonton, Canada (1990).).
There are numerous parameters that contribute to the stability of the interfacial film and as a result to emulsion stability, such as water pH and the additive content (Poteau et al., 2005Poteau, S., Argillier, J. F., Langevin, D., Pincet, F., Perez, E., Influence of pH on stability and dynamic properties of asphaltenes and other amphiphilic molecules at the oil-water interface. Energy & Fuels, 19, 1337-1341 (2005).; Fortuny et al., 2007Fortuny, M., Oliveira, C. B. Z., Melo, R. L. F. V., Nele, M., Coutinho, R. C. C., Santos, A. F., Effect of salinity, temperature, water content, and pH on the microwave demulsification of crude oil emulsions. Energy Fuels, 21, 1358-1364 (2007).; Daaou et al., 2011), but these effects show different behavior for various oil origins (Strassner, 1968Strassner, J. E., Effect of pH on interfacial films and stability of crude oil-water emulsions. JPT 303, March (1968).; Pathak and Kumar, 1995Pathak, A. K., Kumar, T., Study of indigenous crude oil emulsions and their stability. In: Proceedings of PETROTECH-95, the First International Petroleum Conference and Exhibition, New Delhi, January 9-12 (1995).; McLean and Kilpatrick, 1997aMcLean, J. D., Kilpatrick, P. K., Effects of asphaltene aggregation in model heptane-toluene mixtures on stability of water-in-oil emulsions. Journal of Colloid and Interface Science, 196, 23-34 (1997a).; Goldszal et al., 2002Goldszal, A., Bourrel, M., Hurtevent, C., Volle, J.-L., The Third International Conference on Petroleum Phase Behavior and Fouling. New Orleans, USA (2002).). For example, Daou et al. (2011) studied the effect of pH on Algerian crude oil emulsions and suggested that a neutral medium is more efficient than an acidic or basic environment for stabilizing the emulsions. Fortuny et al. (2007)Fortuny, M., Oliveira, C. B. Z., Melo, R. L. F. V., Nele, M., Coutinho, R. C. C., Santos, A. F., Effect of salinity, temperature, water content, and pH on the microwave demulsification of crude oil emulsions. Energy Fuels, 21, 1358-1364 (2007). studied the effects of salinity, temperature, water content and pH on the stability of crude oil emulsions based on microwave treatment and showed that, in emulsions containing high water contents, the rate of demulsification is high, except when high pH and salt content were simultaneously involved. Additionally, Moradi et al. (2011)Moradi, M., Alvarado, V., Huzurbazar, S., Effect of salinity on water-in-crude oil emulsion: Evaluation through drop-size distribution proxy. Energy & Fuels, 25, 260-268 (2011). studied the impact of salinity on crude oil/water emulsions by measuring the droplet-size distribution visualized by an optical microscopy method, and found that emulsions are more stable at lower ionic strength of the aqueous phase.
In the present study, the focus is on the investigation of the effect of pH values, temperature, salinity and alcohol modifiers on the stability of an Iranian crude oil emulsion. In addition, the influence of a wide range of chemical demulsifiers on destabilization of the emulsion is studied in order to better understand the water-in-oil emulsion behavior. Qualitec 4, an experimental design software, was used to achieve an optimized formulation for water separation. These experiments were done by bottle test method, which is the most common method for evaluating the amount of water separated from water-in-oil emulsion (Mat, 2006Mat, H. B., Study on demulsifier formulation for treating Malaysian crude oil emulsion. PhD Thesis, Department of Chemical Engineering, Faculty of Chemical and Natural Resources Engineering, Universiti Teknologi Malaysia (2006).).
EXPERIMENTAL SECTION
Crude Oil Characterization
Crude oils used for these sets of experiments are from two fields, Omidie and Ahwaz heavy crude oil. Their physical characteristics are shown in Table 1. Chemical properties play important roles in the stability of crude oil emulsion. Table 1 shows that the oil from the Omidie field is more stable than the latter as a result of combining four interfacial active agents in the crude oil.
Materials and Equipment
The chemicals and methods used in this study are chosen based on suitability and economic feasibility. The demulsifiers used in this study are shown in Table 2. The majority of the chemicals used in this study were supplied from Merck, BASF and Kavosh Kimia Kerman companies. Table 3 presents the list of solvents used to increase the pour point and solubility of solid and viscous demulsifiers. The homogenizer used in this study is Silent Crusher M manufactured by Heidolph and the incubator is Gerhardt, Model THO 500.
Experimental Methods
This study was carried out using two types of water-in-oil emulsions from real and synthetic oil. The bottle-test method was used in both single and composite demulsifier screening in order to find out the most effective demulsifiers.
Emulsion Preparation
Water-in-oil emulsions were prepared by mixing crude oil and water (4:1 v/v) to obtain 20% (v/v) water content. The emulsification was carried out using a homogenizer at a rate of 10000 rpm for 5 minutes to get a stable emulsion with droplets ~10 μm in diameter. About 10 mL of emulsion sample was prepared and preheated to 70 °C. The synthetic emulsion was prepared by mixing n-heptane (analytical grade) and Toluene (7:3 V/V); 2 grams of asphaltenes are added to a liter of the synthetic crude oil; the resultant solution is then mixed with water in a 4:1 (volumetric) ratio, resulting in a very stable W/O emulsion, as was expected (Bhattacharyya, 1992Bhattacharyya, B. R., Water Soluble Polymer as Water-in-Oil Demulsifiers. US Patent 5,100,582 (1992).). The phases were next mixed using a homogenizer at a speed of 15000 rpm for 6 minutes to get a stable emulsion. The experiments were repeated three times and the reported results are the mean value of each result obtained. Relative errors in estimating the volume of the separated water phase are around 3% for all procedures and steps.
Extraction of Asphaltene
Crude oil was dispersed in n-pentane or n-heptane at a concentration of 1:5 (v/v). The mixture was then sonicated in an ultrasonic bath for 45 minutes and left to settle overnight. After settling, the mixture was sonicated again for 20 minutes. The precipitated asphaltenes were filtered on medium porosity (Hunktell grade 392) filter paper. The collected asphaltenes were mixed again with a 1:2 volume ratio of original crude to n-pentane or n-heptane, sonicated for 45 minutes and left overnight. The asphaltenes were then filtered again and dried at 45 °C under vacuum until the solvent was completely evaporated. The asphaltene-solid fractions were considered to be dry when the change in mass is less than 0.1% over a 24 hour period (Gafonva, 2000Gafonova, O. V., Role of asphaltenes and resins in the stabilization of waterin-hydrocarbon emulsions. The University of Calgary, MSc. Thesis (2000).).
Demulsification Tests
The demulsifiers were screened using the bottletest method. Varied amounts of potential demulsifiers were added to a series of tubes or bottles each containing the same amount of an emulsion to be broken (Mat, 2006Mat, H. B., Study on demulsifier formulation for treating Malaysian crude oil emulsion. PhD Thesis, Department of Chemical Engineering, Faculty of Chemical and Natural Resources Engineering, Universiti Teknologi Malaysia (2006).). The screening process is conducted at constant temperature, pH and demulsifier concentration at optimal amounts, which were calculated previously; therefore, the most effective single demulsifier was found based on water separation from the emulsion system. The assays of the effect of pH were conducted by adding HCl or NaOH solutions to adjust the pH of emulsion water.
Bottle-Test
The test was carried out using 10 mL of crude oil in each case in an incubator at a constant temperature (70 °C). An appropriate amount of demulsifiers was injected into 10 mL of the emulsion system to obtain a 10-5 volume fraction of each demulsifier in the emulsion. Then, the bottles were shaken for 1 min to thoroughly mix the demulsifier and the emulsion. The bottles were then returned to the incubator. Separation of phases was monitored by the position of the water/emulsion interface and recorded as a function of time to verify the volume of water separated every 15 min. A bottle containing an emulsion without any additive was employed as a reference (blank). The volume of water separation from the emulsion system was observed. Water separation versus time was plotted, which is defined as:
where V1 is the volume of separated water and V2 is the original volume of water contained.
Optimization of the Demulsifier Formulation
The results from experimental runs were used as a factor in optimizing formulations in order to produce the best results in breaking the water-in-oil emulsion system. The type of material and the concentration were used as the variables in the experimental design. Thus, the concentrations of watersoluble demulsifiers (fatty alcohol ethoxylate, triethanolamine and urea) change in all runs while keeping the pH and temperature constant at 5.5 and 70 °C, respectively. The L-8 array method of experimental design that was applied in this work considers two levels of variables (high and low); thus, eight runs of the experimental design were used to optimize the concentration for both oil-soluble and water-soluble demulsifiers. The optimal concentration for both factors was then combined as a new formulation for treating crude oil emulsion systems.
RESULTS AND DISCUSSIONS
Effect of pH Values
According to the literature (Tambe and Sharma, 1993Tambe, D. E. and Sharma, M. M., Factors controlling the stability of colloid-stabilized emulsions. J. Colloid Interface Sci., 157, 244 (1993).; Binks, 1993Binks, B. P., Surfactant monolayers at the oil-water interface. Chemistry and Industry, 14, 537-541 (July 1993).; Johansen et al., 1988Johansen, E. J., Skjarvo, M., Lund, T., Water-incrude oil emulsions from the Norwegian continental shelf, Part I. Formation, charachterization and stability correlations. Colloids and Surfaces, 34, 353-370 (1988/89).; Porter, 1994Porter, M. R., Use of Surfactant Theory. Handbook of Surfactants, Blackie Academic & Professional, United Kingdom, 26-93 (1994).; Poteau et al., 2005Poteau, S., Argillier, J. F., Langevin, D., Pincet, F., Perez, E., Influence of pH on stability and dynamic properties of asphaltenes and other amphiphilic molecules at the oil-water interface. Energy & Fuels, 19, 1337-1341 (2005).; Fortuny et al., 2007Fortuny, M., Oliveira, C. B. Z., Melo, R. L. F. V., Nele, M., Coutinho, R. C. C., Santos, A. F., Effect of salinity, temperature, water content, and pH on the microwave demulsification of crude oil emulsions. Energy Fuels, 21, 1358-1364 (2007).; Daaou et al., 2011), adjusting the pH of emulsions seems indeed to be effective in resolution of waterin-oil emulsions; thus, a series of tests were prepared as a function of pH. The effects of pH on emulsion properties were evaluated by adding HCl or NaOH solutions to the aqueous phase prior to emulsion preparation. Figure 1 summarizes the results of pH adjustments on the ability of the demulsifier to separate water from water-in-oil emulsions under various pH values. Obviously, changing pH values influence emulsion stability. For acidic medium, the separated water volume varied from 12% (of the total added water) at pH 3.0 to 49% V/V at pH 5.5. The same pattern was observed when using Basorol E 2032 as a demusifier from the oil soluble group, which showed separation of water varying from 20% V/V to 57% V/V. Again the optimum (57% separation of the total water) was reached at pH 5.5. In the alkaline medium (pH 9.3-13), separated water varied from 5% V/V to 30% V/V and from 15% V/V to 45% V/V by injecting triethanolamine and Basorol E 2032, respectively. At pH 13, the emulsion shows the greatest stability since little water separates. Therefore, we can conclude that, at very high and very low pH values, the emulsions seem to be stable, while intermediate pH values cause instability. The range and degree of emulsion stability are very dependent on the crude oil from which the emulsion was prepared. Consequently, the optimal pH for treating crude oil emulsions seem vary from 5 to 9. According to Johansen et al. (1989)Johansen, E. J., Skjarvo, M., Lund, T., Water-incrude oil emulsions from the Norwegian continental shelf, Part I. Formation, charachterization and stability correlations. Colloids and Surfaces, 34, 353-370 (1988/89)., a main reason for this is that a low permittivity medium causes the cancellation of electrostatic repulsion and of any importance of multivalent counter-ions as destabilizers. In addition, Poteau et al. (2005)Poteau, S., Argillier, J. F., Langevin, D., Pincet, F., Perez, E., Influence of pH on stability and dynamic properties of asphaltenes and other amphiphilic molecules at the oil-water interface. Energy & Fuels, 19, 1337-1341 (2005). showed that pH has a strong influence on the interfacial properties of asphaltenes at a Venezuelan crude oil/water interface at high or low pH. This is because, at these pH values, asphaltene functional groups become charged, leading to enhancing surface activity.
Effects of water separation using various pH values. Experimental conditions: T = 70 °C, Demulsifier concentration = 10-5 volume fraction.
These results are consistent with Tambe and Sharma (1993)Tambe, D. E. and Sharma, M. M., Factors controlling the stability of colloid-stabilized emulsions. J. Colloid Interface Sci., 157, 244 (1993).. According to their experiments, the stability of oil-in-water emulsions increased as the pH increased from 4 to 6; however, a further increase in pH, from 6 to 8 and finally to 10, resulted in formation of relatively less stable oil-in-water emulsions and more stable water-in-oil emulsions. This is consistent with data obtained by Strassner (1968)Strassner, J. E., Effect of pH on interfacial films and stability of crude oil-water emulsions. JPT 303, March (1968)., who studied crude oil emulsions at different pH values and found that Venezuelan crude oil emulsions at pH < 6 are highly stable, while those at pH> 10 exhibit low stability or are highly unstable, although at pH =13 the emulsion was very stable.
Effect of Temperature
Figure 2 presents the separation process at five different temperatures as a function of time. In this case, 5 samples were prepared and 10 ppm of demulsifier (fatty alcohol ethoxylate) was injected into each of them. The bottle-test was then carried out at different temperatures (10 °C, 20 °C, 50 °C, 60 °C, 70 °C and 80 °C) and constant pH of 5.5 for 72 hours. According to Figure 2, increasing the temperature increased the water separation dramatically from 15% to 50% for 10 °C and 50 °C, respectively. This trend continued with a lower slope up to 70 °C, which represented 57% water separation. However, raising the temperature from 70 to 80 °C resulted in only 2% further separation, which is within the experimental error limits.
Effects of water separation using various temperatures. Experimental conditions: Demulsifier concentration = 10-5 volume fraction, pH=5.5. Demulsifier: Fatty alcohol ethoxylate.
Consequently, we can conclude that higher temperatures promote destabilization effects caused by increased Brownian motion and mass transfer across the interface, which is mainly due to the fact that the interfacial viscosity of the internal phase decreases as the temperature increases; as a result, the momentum between two water droplets increases, coalescence occurs, and the two phases of immiscible liquids separate due to their different densities and polarities.
Effect of Salinity
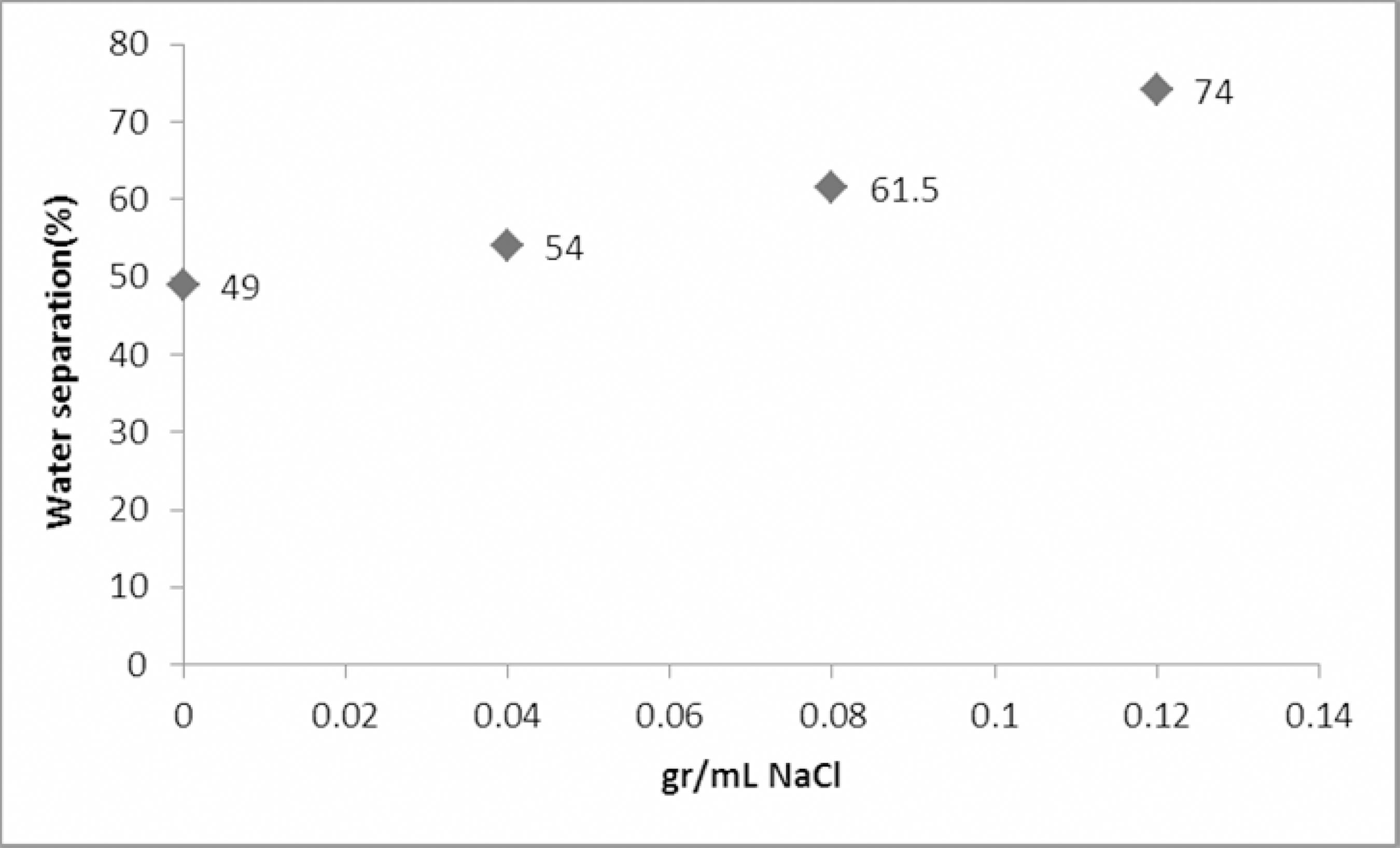
According to Binks (1993)Binks, B. P., Surfactant monolayers at the oil-water interface. Chemistry and Industry, 14, 537-541 (July 1993)., O/W droplets increase in size upon increasing salt concentration, while W/O droplets decrease in size; hence, the presence of salt seems to have an adverse effect on emulsion stability. In order to test this, four samples of emulsion of different salt contents (zero, 0.04, 0.08, 0.12 gr/mL) were prepared, each sample containing an equal amount (10 ppm) of triethanolamine as a demulsifier. We chose this chemical because of its moderate rate and productivity of separation, in order to observe the water-in-oil emulsion's behavior more precisely. Figure 3 shows the result of adding inorganic salt (sodium chloride) to the crude oil emulsion. As expected, the presence of inorganic cations in the system had an adverse effect on emulsion stability; thus, the best separation of water was achieved for the sample containing the highest concentration of 0.12 g/mL NaCl (74% separation of total water) compared to the blank with no NaCl (49% separation of total water). This phenomenon could be explained by the pronounced change in interfacial film behavior. The salt ions lead to an increase in relaxation of the formed film (Binks, 1993Binks, B. P., Surfactant monolayers at the oil-water interface. Chemistry and Industry, 14, 537-541 (July 1993).). These results are in agreement with those obtained by Fortuny et al. (2007)Fortuny, M., Oliveira, C. B. Z., Melo, R. L. F. V., Nele, M., Coutinho, R. C. C., Santos, A. F., Effect of salinity, temperature, water content, and pH on the microwave demulsification of crude oil emulsions. Energy Fuels, 21, 1358-1364 (2007)., who studied the effects of salinity, temperature, water content and pH on the stability of crude oil emulsions upon microwave treatment and found that the demulsification process was achieved with high efficiencies for emulsions containing high water contents, except when high pH and salt contents were simultaneously involved. Additionally, Moradi et al. (2011)Moradi, M., Alvarado, V., Huzurbazar, S., Effect of salinity on water-in-crude oil emulsion: Evaluation through drop-size distribution proxy. Energy & Fuels, 25, 260-268 (2011). indicated that emulsions are more stable at lower ionic strength of the aqueous phase.
Effects of water separation using various concentrations of NaCl. Experimental conditions: T = 70 °C, Demulsifier concentration = 10-5 volume fraction, pH=5.5, Demulsifier:Triethanolamine.
Effect of the Type of Demulsifiers
The emulsion destabilization, an essential step for the efficient separation of water from petroleum, can be carried out by the addition of chemical substances, named demulsifiers (Ramalho et al., 2010Ramalho, J. B. V. S., Lechuga, F. C., Lucas, E. F., Effect of the structure of commercial poly(ethylene oxide-b-propylene oxide) demulsifier bases on the demulsification of water-in-crude oil emulsions: Elucidation of the demulsification mechanism. Quimica Nova, 33(8) 1664-1670 (2010).; Kelland et al., 2009Kelland, M. A., Production Chemical for the Oil and Gas Industry. CRC Press, New York, (2009).). According to Lucas et al. (2009)Lucas, E. F., Mansur, C. R. E., Spinelli, L., Queirs, Y. G. C., Polymer science applied to petroleum production. Pure and Applied Chemistry, 81(3) 473-494 (2009). and Pacheco et al. (2011)Pacheco, V. F., Spinelli, L., Lucas, E. F., Mansur, C. R. E., Destabilization of petroleum emulsions: Evaluation of the influence of the solvent on additives. Energy & Fuels, 5, 1659-1666 (2011). these substances include block copolymers based on ethylene oxide and propylene oxide (PEO-b-PPO). In the present study, the focus was on the selection of chemicals which possess this appropriate functionality. For instance, glycols (mono, di, tri and poly) change the density and polarity of the water phase, but the majority of applied demulsifiers accelerate the rate of flocculation and coagulation, which leads to faster separation of the water phase from the oil phase.
The screening process was carried out at optimal pH values and temperatures to facilitate the breaking of the emulsion. The results using water-soluble demulsifiers are shown in Figures 4 and 5. It was found that the three most effective demulsifiers for water separation are fatty alcohol ethoxylate (52% V/V), triethanolamine (49% V/V) and urea (45% V/V). The main reason for selecting urea, in spite of better productivity of polyethylene glycol (47%), is economic concerns, as urea is more cost effective. In addition, it showed a higher rate of separation compared to polyethylene glycol. Nearly half of the chemicals in this group did not perform well in breaking W/O emulsion, because water separation ranged from 0.0% V/V to 30.0% V/V. The water-soluble demulsifiers cause film drainage and help in aggregation and coalescence of the water phase.
Water separation using water-soluble demulsifiers. Experimental conditions: T = 70 °C, Demulsifier concentration = 10 ppm, pH=5.5.
Water separation using water-soluble demulsifiers. Experimental conditions: T = 70 °C, Demulsifier concentration = 10-5 volume fraction, pH=5.5.
However, according to the literature, an oil-soluble type of demulsifier is very effective in W/O emulsion resolution (Bhattacharyya, 1992Bhattacharyya, B. R., Water Soluble Polymer as Water-in-Oil Demulsifiers. US Patent 5,100,582 (1992).). This is because oil is the continuous phase, while water is the dispersed phase. Thus, the surfactants dissolve in the continuous phase with less mass transfer resistance at optimum temperature, and the dispersion of the demulsifiers injected in the system becomes easier. The results for water separation using oil-soluble demulsifiers are shown in Figure 6. Basorol E 2032 gives the best results compared to the others. Although this is an expensive chemical, its role in resolving emulsion problems is very effective with 57% V/V and higher separations, followed by Basorol PDB 9935 and TOMAC with 55% V/V and 50% V/V separation, respectively. Except for naphthalene, which shows the lowest separation ability, the other oil-soluble demulsifiers show separation above 40% V/V, which supports the claim that oil-soluble demulsifiers are more effective than water-soluble demulsifiers.
Water separation using water-soluble demulsifiers. Experimental conditions: T = 70 °C, Demulsifier concentration = 10-5 volume fraction, pH=5.5.
Effects of Various Concentrations
Selection of a representative demulsifier is based on ability of the demulsifier to separate water from an emulsion system. As a result, fatty alcohol ethoxylate and Basorol E 2032 were chosen for this test, as shown in Figures 7 and 8, respectively. The concentrations used in this test were 10-5, 2×10-5, 5×10-5 and 10-4 volume fraction of additives and experimental conditions were kept constant at optimal values. This test aims to obtain the relationship between concentration and emulsion resolution efficiency. As Figure 7 shows, water separations were lower than 65.0% V/V for all concentrations. The lowest concentration gave 52% V/V separation, and 64% V/V was achieved by using the highest concentration of fatty alcohol ethoxylate.
Effects of water separation using various concentrations of fatty alcohol ethoxylate. Experimental conditions: T = 70 °C, pH=5.5.
Effects of water separation using various concentrations of Basorol E 2032. Experimental conditions: T = 70 °C, pH=5.5.
Therefore, in the industrial application of these demulsifiers, the user can choose the proper balance between the increased cost of using a higher concentration to save time and equipment capacity and a lower concentration and cost of demulsifier, which will result in longer separation time and probably a higher investment in the capacity of equipment. Figure 8 shows that injecting 100 ppm of Basorol E 2032 results in 80% V/V of water separation followed by 65% V/V, 60% V/V and 50% V/V using 5×10-5, 2×10-5 and 10-5 volume fractions, respectively. Therefore, the concentration plays a significant role in the demulsification process because higher concentrations increase the rate of coalescence of droplets because of interfacial film thinning.
Effects of Modifier Addition
Addition of an alcohol as a modifier helps the demulsification process. Alcohols seem to destabilize the film through the diffusion or partioning mechanism. In order to determine the effect of the addition of modifier in this formulation, four alcohols were used: methanol, butanol, pentanol and cyclohexanol. Figure 9 shows the percentage of water separation using these alcohols. It shows that methanol has a better ability in promoting water separation from the emulsion system compared to butanol, pentanol and cyclohexanol. This is because short chain alcohols are very soluble in water and long chain alcohols are very soluble in oil. As expected, cyclohexanol showed the lowest separation (0.5% V/V) because of its low polarity. Methanol itself results in 6% V/V water separation followed by butanol (3% V/V) and pentanol (1% V/V). Thus, methanol was selected as a modifier in this test based on the result of the previous test; ioctylamine was chosen as an oil-soluble demulsifier (because of its moderate productivity, which allowed us to observe the water-in-oil emulsiom more precisely, and of the type of emulsion that promotes the efficiency of oilsoluble demulsifiers) . For comparison, a blank was prepared where the emulsion system was injected with demulsifier without modifier. The second system of emulsion was injected with addition of modifier. Figure 10 shows the results of water separation from this test. Accordingly, it can be concluded that the difference in water separation is only 4% V/V, i.e., 53% V/V with modifier (methanol) addition and 49% V/V without it. Hence, the presence of methanol in this demulsifiers had a negligible effect on the demulsification process. Thus, it seems that modifier addition is unnecessary.
Percentage of water separation using alcohols. Experimental conditions: T = 70 °C, alcohol concentration = 5×10-5 volume fraction, pH=5.5
Comparison of water separation with and without modifier. Experimental conditions: T = 70 °C, Demulsifier concentration = 10-5 volume fraction, modifier concentration = 5×10-5 volume fraction, pH=5.5
Optimization by the Software Qualitec 4
Table 4 shows the range and levels of the independent variables for the water-soluble and oil-soluble demulsifiers investigated in this study. The concentration values for both water-soluble and oil-soluble demulsifiers were obtained from trial and error tests using the bottle-test method. By this method, eight different runs were performed for water-soluble and oil-soluble demulsifiers, as shown in Tables 5 and 6, respectively. According to the results, it is clear that the percentage of water separation using water-soluble demulsifiers is different from those achieved by applying oil-soluble demulsifiers in different runs. This difference in separation is noticeably dependant on dosage and fraction of each demulsifier. According to Table 5, the best separation clearly occurred in runs 4 and 6 (80% V/V), while run 5, with separation of around 20% V/V, came at the bottom of the list. The average amount of separation in these runs was almost 58% V/V. Turning to Table 6, the average separation was roughly 63% V/V and run 8 had the best separation (75% V/V) compared to run 1, which had the lowest separation of ~3.7% V/V. The significance of the factors and interactions is shown in a variance analysis (ANOVA) (Tables 7 and 8).
Table 7 shows that fatty alcohol ethoxylate is the most effective factor in separating water (42.08% V/V); while urea had the lowest effect, separating around 0% V/V. According to Table 8, Basorol E 2032 with a separation of ~44% V/V and TOMAC with a productivity of ~14% V/V are the most significant and the least crucial factors among oil-soluble demulsifiers, respectively. Information about the optimum and productivity of each factor is provided in Table 9. In this table, the optimal quantity relevant to each parameter and its level are shown.
According to these results, the optimal formulation for treating water-in-oil emulsions is clearly achieved by mixing 60 ppm urea, 50 ppm triethanolamine and 40 ppm fatty alcohol ethoxylate, and the best demulsifier was fatty alcohol ethoxylate in proportion to the other demulsifiers in the water-soluble group. Similarly, for oil-soluble demulsifiers, the optimum formulation is Basorol E 2032, with the optimum amount around 15 ppm in addition to 30 ppm TOMAC and 20 ppm Basorol PDB 9935. In this case, Qualitec 4 predicts a separation of around 86% V/V of water from emulsion for both groups. It is noteworthy that, due to the fact that urea is not effective in the final formulation (0% V/V effectiveness), as shown in Table 7, we did not employ it in creating formula A and the composition of this formulation were achieved practically; hence, formula A was produced according to the suggested composition: 0% urea, 16% triethanolamine, 20% fatty alcohol ethoxylate, 6% Basorol E2032, 8% Basorol PDB 9935, 12% TOMAC and 39% aromatic solvent. However, another formula (B) containing urea was produced according to the Qualitec 4 suggestion: 1.53% urea, 11.84% triethanolamine, 14.65% fatty alcohol ethoxylate, 17.82% Basorol E2032, 12.75% Basorol PDB 9935, 10.89% TOMAC and 30.49% aromatic solvent.
Comparison of the Optimum Formulae with Commercial Demulsifier Formulations
Two commercial demulsifier formulations were used in this experiment to compare the yield of emulsion resolution. They include demulsifiers VZB1413 and VZB1414 supplied by Kavosh Kimia Kerman Co. Emulsions were prepared from Omidie field oil. Figure 11 presents the result of water separation by applying a 10-5 volume fraction of formulations A, B, VZB 1413 and VZB 1414. As Figure 11 shows, formulation A effects the best separation, which is almost 90% V/V, followed by formulation B at 84%, VZB1414 at 72% and VZB1413 at 65%. As expected, the mixture of oil-soluble and water-soluble demulsifiers results in the best separation.
Water separation of real emulsion using A, B and commercial demulsifier formulations. Experimental conditions: T = 70 °C; formulation concentration = 10-5 volume fraction, pH=5.5
Among these, formulation A, suggested by the results of Experimental Design in Qualitec 4, is even more effective than the commercial formulations.
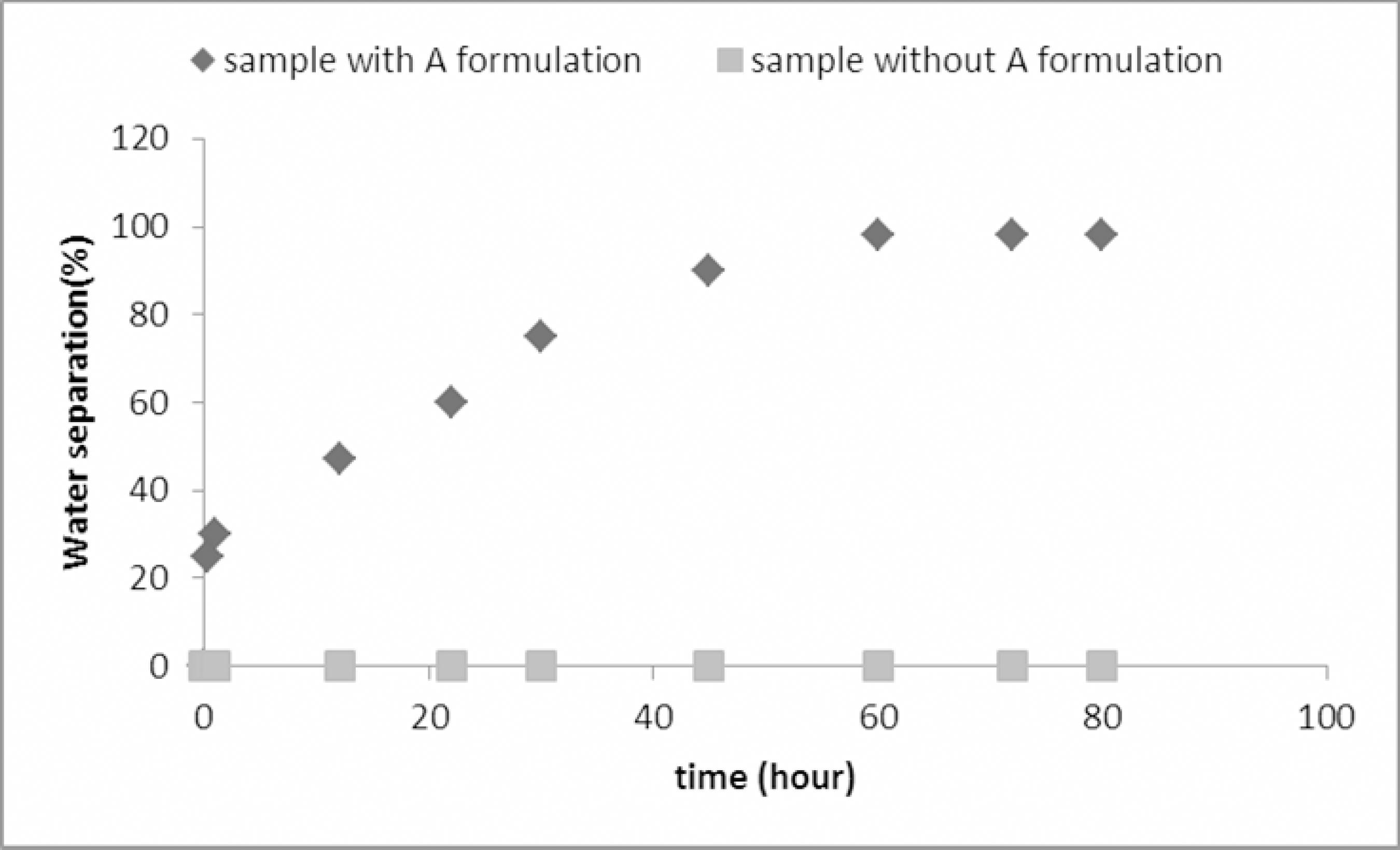
Effectiveness of Formulation A on Synthetic Oil Emulsion
Formulation A was also tested on a synthetic oil containing only one interfacial active agent, i.e., it contained asphaltene and no resins or waxes. According to Gafonova (2000)Gafonova, O. V., Role of asphaltenes and resins in the stabilization of waterin-hydrocarbon emulsions. The University of Calgary, MSc. Thesis (2000)., resins and waxes could not individually form emulsions, only asphaltenes can individually form emulsions. Figure 12 shows the results of applying 10 ppm of Formula A to the synthetic oil. A separation of 98% shows that this is an easier emulsion to break, most likely due to low viscosity (because of the absence of resins and waxes), which decrease the mass transfer limitations for the movement of the demulsifiers through the continuous phase. This phenomenon provides for better film drainage by lowering the interfacial and surface tensions of the aqueous phase and for almost complete separation.
Water separation of real emulsion using A, B and commercial demulsifier formulations. Experimental conditions: T = 70 °C; formulation concentration = 10-5 volume fraction, pH=5.5
CONCLUSIONS
To choose the most effective demulsifiers, a screening process was used considering the price of demulsifiers. This was carried out using the bottletest method. Based on results for single water-soluble demulsifiers, fatty alcohol ethoxylate (52% v/v), triethanolamine (49% v/v) and urea (42% v/v) are relatively effective; among oil-soluble demulsifiers, Basorol E2032 (57%), Basorol PDB 9935 (55%) and TOMAC (50%) showed the best results in treating the Omidie Oil Field emulsion systems. These tests were run by injecting ppm levels of demulsifiers under optimal conditions in terms of temperature (70 °C) and pH (5.5). As the process temperature increased, the concentration of demulsifier and the salt content of the water of the emulsion positively influenced demulsification, while the optimum pH level was 5.5.
The present study suggested two formulations according to the Qualitec 4 Analysis System; the more effective formulation (B), which separated almost 90% of a real crude-water emulsion using 10 ppm volume fraction contained: 16% triethanolamine, 20% fatty alcohol ethoxylate, 6% Basorol E2032, 8% Basorol PDB 9935, 12% TOMAC and 39% aromatic solvent, but no urea. Also, this new formulation was more effective than commercial demulsifier formulations (VZB 1413 and VZB 1414 with 65% and 72% separation, respectively).
REFERENCES
- Bhattacharyya, B. R., Water Soluble Polymer as Water-in-Oil Demulsifiers. US Patent 5,100,582 (1992).
- Bobra, M., A Study of the Formation of Water-in-Oil Emulsions, Proc., 1990 Arctic and Marine Oil Spill Program Technical Seminar, Edmonton, Canada (1990).
- Binks, B. P., Surfactant monolayers at the oil-water interface. Chemistry and Industry, 14, 537-541 (July 1993).
- Daaou, M., Bendedouch, D., Bouhadda, Y., Vernex-Loset, L., Modaressi, A., Rogalski, M., Explaining the flocculation of Hassi Messaoud asphaltenes in terms of structural characteristics of monomers and aggregates. Energy & Fuels, 23, 5556-5563 (2009).
- Daaou, M., Bendedouch, D., Water pH and surfactant addition effects on the stability of an Algerian crude oil emulsion. Journal of Saudi Chemical Society, 16, 333-337,(2012)
- Eley, D. D., Hey, M. J., Symonds, J. D., Emulsions of water in asphaltene containing oils. Colloids & Surfaces, 32, 87(1998).
- Fortuny, M., Oliveira, C. B. Z., Melo, R. L. F. V., Nele, M., Coutinho, R. C. C., Santos, A. F., Effect of salinity, temperature, water content, and pH on the microwave demulsification of crude oil emulsions. Energy Fuels, 21, 1358-1364 (2007).
- Gafonova, O. V., Role of asphaltenes and resins in the stabilization of waterin-hydrocarbon emulsions. The University of Calgary, MSc. Thesis (2000).
- Goldszal, A., Bourrel, M., Hurtevent, C., Volle, J.-L., The Third International Conference on Petroleum Phase Behavior and Fouling. New Orleans, USA (2002).
- Groezin, H., Mullins, O. C., Asphaltene Molecular Size and Weight by Timeresolved Fluorescence Depolarizarion. In: Mullins, O. C., Sheu, E. Y., Hammami, A., Marshall, A. G., (Eds.), Asphaltenes, Heavy Oils, and Petroleomics, Springer, New York, 2007, p. 17.
- Honse, S. O., Ferreira, S. R., Mansur, C. R. E., Lucas, E. F., Gonzalez, G., Separation and characterization of asphaltenic subfractions. Quimica Nova, 35(10), 1991-1994 (2012).
- Johansen, E. J., Skjarvo, M., Lund, T., Water-incrude oil emulsions from the Norwegian continental shelf, Part I. Formation, charachterization and stability correlations. Colloids and Surfaces, 34, 353-370 (1988/89).
- Jones, T. J., Neustadter, E. L., Wittingham, K. P., Water-in-crude oil emulsion stability and emulsion destabilization by chemical demulsifiers. J. Cdn. Pet. Tech. 100, April-June (1978).
- Kelland, M. A., Production Chemical for the Oil and Gas Industry. CRC Press, New York, (2009).
- Kimbler, O. K., Reed, R. L., Silberberg, I. H., Physical characteristics of natural films formed at the crude oil-water interfaces. JPT 153, June (1966).
- Kokal, S. L., Al-Juraid, J. I., Quantification of various factors affecting emulsion stability: Water cut, temperature, shear, asphaltene content, demulsifier dosage and mixing different crudes. Paper SPE 56641, SPE Annual Technical Conference and Exhibition, Houston 3-6 October (1999).
- Leontaritis, K. J., Mansoori, G. A., Asphaltene deposition: A comprehensive description of problem manifestations and modeling approaches. Paper SPE 18892, SPE Production and Operations Symposium, Oklahoma City, Oklahoma, 13-14 March (1998).
- Levine, S., Sanford, E., Stabilization of emulsion droplets by fine powders. Can. J. Chem. Eng., 62, 258 (1985).
- Lucas, E. F., Mansur, C. R. E., Spinelli, L., Queirs, Y. G. C., Polymer science applied to petroleum production. Pure and Applied Chemistry, 81(3) 473-494 (2009).
- Mat, H. B., Study on demulsifier formulation for treating Malaysian crude oil emulsion. PhD Thesis, Department of Chemical Engineering, Faculty of Chemical and Natural Resources Engineering, Universiti Teknologi Malaysia (2006).
- McLean, J. D., Kilpatrick, P. K., Effects of asphaltene aggregation in model heptane-toluene mixtures on stability of water-in-oil emulsions. Journal of Colloid and Interface Science, 196, 23-34 (1997a).
- McLean, J. D., Kilpatrick, P. K., Effect of asphaltene solvency on stability of water-in-crude-oil emulsion. Journal of Colloid and Interface Science, 189, 242-253 (1997b).
- Menon, V. B., Nikolov, A. D., Wasan, D. T., Interfacial effects of solids-stabilized emulsions: Measurements of film tension and particle interaction energy. J. Colloid Interface Sci., 124(1), 317 (1988).
- Moradi, M., Alvarado, V., Huzurbazar, S., Effect of salinity on water-in-crude oil emulsion: Evaluation through drop-size distribution proxy. Energy & Fuels, 25, 260-268 (2011).
- Pacheco, V. F., Spinelli, L., Lucas, E. F., Mansur, C. R. E., Destabilization of petroleum emulsions: Evaluation of the influence of the solvent on additives. Energy & Fuels, 5, 1659-1666 (2011).
- Pathak, A. K., Kumar, T., Study of indigenous crude oil emulsions and their stability. In: Proceedings of PETROTECH-95, the First International Petroleum Conference and Exhibition, New Delhi, January 9-12 (1995).
- Poteau, S., Argillier, J. F., Langevin, D., Pincet, F., Perez, E., Influence of pH on stability and dynamic properties of asphaltenes and other amphiphilic molecules at the oil-water interface. Energy & Fuels, 19, 1337-1341 (2005).
- Porter, M. R., Use of Surfactant Theory. Handbook of Surfactants, Blackie Academic & Professional, United Kingdom, 26-93 (1994).
- Ramalho, J. B. V. S., Lechuga, F. C., Lucas, E. F., Effect of the structure of commercial poly(ethylene oxide-b-propylene oxide) demulsifier bases on the demulsification of water-in-crude oil emulsions: Elucidation of the demulsification mechanism. Quimica Nova, 33(8) 1664-1670 (2010).
- Sjoblom, J., Hemmingsen, P. V., Kallevik, H., The Role of Asphaltenes in Stabilizing Water-in-Crude Oil Emulsions. In: O. C., Mullins, E. Y., Sheu, A., Hammami, A. G., Marshall, (Eds.), Asphaltenes, Heavy Oils, and Petroleomics, Springer, New York, p. 549 (2007).
- Staiss, F., Bohm, R. and Kupfer, R., Improved demulsifier chemistry: A novel approach in the dehydration of crude oil. Society of Petroleum Engineers Production Engineering, SPE Production Engineering, 334-338 (1991).
- Strassner, J. E., Effect of pH on interfacial films and stability of crude oil-water emulsions. JPT 303, March (1968).
- Svetgoff, J. A., Demulsification key to production efficiency. Petroleum Engineer. Intl., 61(8), 28 (1989).
- Tambe, D. E. and Sharma, M. M., Factors controlling the stability of colloid-stabilized emulsions. J. Colloid Interface Sci., 157, 244 (1993).
- Wang, X. Y., Alvarado, V. J., Direct current electrorheological stability determination of water-incrude oil emulsions. Journal of Physical Chemistry, B 113, 13811-13816 (2009).
Publication Dates
-
Publication in this collection
Jan-Mar 2015
History
-
Received
09 June 2013 -
Reviewed
26 Oct 2013 -
Accepted
09 Dec 2013