Abstract
Recently public health problems resulting from scorpion stings have shown an alarming increase in various tropical and subtropical countries such as Brazil, Mexico, Tunisia and Morocco. In some regions of Brazil, particularly in the States of Minas Gerais, São Paulo, Bahia, Goiás and the Federal District, more than 6000 scorpion stings with over 100 deaths were reported during a three-year period. In this paper, we attempt to demostrate the effects of human activities on the environment and their consequences on the distribution of dangerous species of scorpions. This topic is discussed in relation to the evolutionary ecology, life history strategies and asexual reproduction of scorpions.
Scorpionism; scorpion; life history strategy; parthenogenesis; Brazil
Review article
SCORPIONS, SCORPIONISM, LIFE HISTORY STRATEGIES AND PARTHENOGENESIS
W.R. LOURENÇO
 CORRESPONDENCE TO:
W.R. LOURENÇO, Société de Biogéographie, 57 rue Cuvier 75005 Paris, France.
, O. CUELLAR 2.
CORRESPONDENCE TO:
W.R. LOURENÇO, Société de Biogéographie, 57 rue Cuvier 75005 Paris, France.
, O. CUELLAR 2.
1 Société de Biogéographie, Paris, France, 2 Department of Biology, The University of Utah, 201 Biology Building, Salt Lake City, Utah, U.S.A.
ABSTRACT. Recently public health problems resulting from scorpion stings have shown an alarming increase in various tropical and subtropical countries such as Brazil, Mexico, Tunisia and Morocco. In some regions of Brazil, particularly in the States of Minas Gerais, São Paulo, Bahia, Goiás and the Federal District, more than 6000 scorpion stings with over 100 deaths were reported during a three-year period. In this paper, we attempt to demostrate the effects of human activities on the environment and their consequences on the distribution of dangerous species of scorpions. This topic is discussed in relation to the evolutionary ecology, life history strategies and asexual reproduction of scorpions.
KEY WORDS: Scorpionism, scorpion, life history strategy, parthenogenesis, Brazil.INTRODUCTION
In recent years, public health reports of severe poisoning from scorpion stings have shown an alarming increase in various tropical and subtropical countries such as Brazil, Mexico, Tunisia and Morocco. In Brazil alone, more than six thousand scorpion stings with over one hundred deaths were reported during a brief three-year interval(29). Additional deaths by scorpions not previously suspected of being dangereous to humans have recently been documented from Peruvian and Brazilian Amazonia. Scorpions are chelicerate arthropods with paired poison glands at the distal extremity of the abdomen, which ends in a sharp sting (Figure 1). All scorpions possess venom glands, but less than 25 of the 1400 known species are considered dangerous and capable of inflicting human death. The evolution of venom in scorpions is associated with prey capture and digestion, and is only secondarily related to defensive behavior. Nevertheless, physical contact between humans and scorpions usually results in a sting. If the poison exerts a severe or lethal outcome, the incident is defined as "scorpionism". Why scorpionism is more common in some regions of the world than in others has not been clearly ascertained. In this paper, we suggest that scorpionism is related to the association of the offending species with man-made environments, and at least in one species, Tityus serrulatus Lutz & Mello, to its all-female mode of reproduction(13).
SCORPIONS AND LIFE HISTORY STRATEGIES
Although some scorpions tend to be associated with man-made or disturbed habitats, the majority are adapted to very specific and predictable natural environments(9,10,22,23,24). For instance, all species of the genus Rhopalurus occur exclusively in savanna formations. Two South American species Opisthacanthus cayaporum Vellard and Tityus fasciolatus Pessôa, occur exclusively in termite mounds. Several species in the family Bothriuridae occupy burrows and most Chactidae (Chactinae) live exclusively in rainforests. The specificity of micro habitats required by some species is thought to be associated with the long-term stability of their specific ecosystems, which has persisted for thousands or millions of years. Even in some extremely unfavorable ecosystems, such as deserts, underground burrowing provides remarkable environmental stability by protecting scorpions from aridity and temperature extremes(25,26).
Scorpions have been divided into two broad ecological categories," equilibrium" and "oportunistic", depending on their type of environment and life history strategies(24). Equilibrium species occupy stable, natural environments, produce single egg clutches, do not store sperm, have long life-spans, low population density, low rmax, weak mobility, and are highly endemic, often known from a single locality(9,10,12,24). In contrast, opportunistic species, such as some members of the genera Centruroides, Tityus and Isometrus, readily invade disturbed environments, produce multiple clutches from a single insemination, have elaborate sperm storage capabilities(6), have short embryonic development, short life spans, high population density, rapid mobility and are widely distributed (Figure 2 and Figure 3).
INVASION OF OPPORTUNISTIC SCORPIONS INTO HUMAN SETTLEMENTS
The recent expansion of human communities in western Brazil was followed by the invasion of a highly toxic scorpion species Tityus serrulatus, which previously had been restricted to a small region of natural savannas in the State of Minas Gerais(13). This species now poses an enormous health problem in Brazil due to its great toxicity, its invasion into human dwellings and its rapid prolificity resulting from parthenogenesis. The first comprehensive study of scorpionism in Brazil was presented by Maurano(17), but dealt with another native species, Tityus bahiensis (Perty, 1834), the second most toxic in South America. Although the main preoccupation with T.serrulatus since its description had been its great toxicity and medical importance, the absence of males from all known populations of this species had remained enigmatic. This puzzle was eventually answered by Matthiesen(16), who first demonstrated that T.serrulatus was an all-female species. Parthenogenesis has since been reported in several other species of scorpions(13). Tityus serrulatus is probably an obligate parthenospecies, since bisexual populations have never been found. The absence of related bisexuals within its present geographic range suggests that the generating parental was either eliminated after producing T.serrulatus, or that T.serrulatus evolved elsewhere and has since invaded an extensive region devoid of its bisexual progenitors(13).
SELECTION OF A PARTHENOSPECIES BY HUMAN ENVIRONMENTS
Several authors have noted that parthenogenetic animals tend to occur in habitats that are different from those of their related bisexuals(2,5,30,31), a pattern which Vandel(31) coined" geographic parthenogenesis". According to White(35,36), parthenogenetic animals are distinguished from their bisexual counterparts by two unique features, a stronger dispersal ability and a much higher reproductive potential. Additionally, Cuellar(2) has suggested that parthenogenetic animals can only evolve in areas devoid of the generating bisexuals because fertilization would disrupt an all-female lineage, and competition would impede its successful establishement in nature.
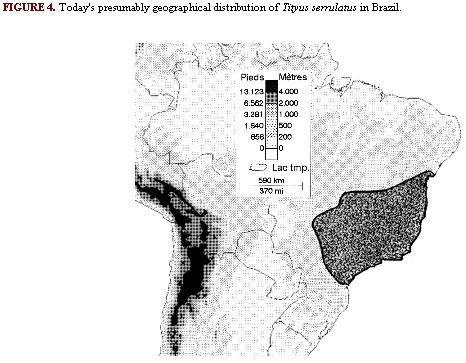
The highly toxic Brazilian scorpion Tityus serrulatus appears to conform with the above predictions. Although this species previously occupied a restricted area in the State of Minas Gerais in Brazil, today it is widely distributed throughout the southeast region of the country (Figure 4). The geographical expansion of this species was undoubtelly related to human colonization, which started about 300 years ago from the Atlantic coastal region westward. Typically, newly erected towns are invaded within a few years after their foundation, although the surrounding natural areas are virtually devoid of this scorpion. The rapid expansion of T.serrulatus into human dwellings was recently demonstrated by its invasion into Brasilia, the capital of Brazil(15). Construction of Brasilia was initiated in 1956 and completed during the 1970s. From 1971 to 1975, a precise inventory of the local scorpions and their densities was conducted in this region, yielding three species: Tityus fasciolatus, Bothriurus araguayae and Ananteris balzani(8). Among them, T. fasciolatus, represents 93% of the total population. This species is similar to T.serrulatus in several traits. Both average essentially the same adult size (65 versus 67 mm), same brood size (16), same embryonic development (2.5-3 months) and same age to maturity (2.0 to 2.5 years). However, T.fasciolatus is bisexual, with a sex ratio of 1 male to 3 females. They also differ in the type of population regulation, that of T. fasciolatus being density dependent and that of T.serrulatus density independent. Consequentely, the populations of T.fasciolatus have been stable for many years(7), whereas those of T.serrulatus have fluctated widely(15). Since the introduction of T.serrulatus into Brasilia and the adjoining Federal District during the late 1980s and early 1990s, the populations of T.fasciolatus have been rapidly declining. A new inventory conducted recently revealed that T.serrulatus now contitutes 70% of the total in this urbanized region(15), and is undoubtedly displacing the bisexual. Other cities in Brazil have been similarly invaded(28).
ADVANTAGES OF SEXUAL AND PARTHENOGENETIC REPRODUCTION
Approximately 95% of all living species reproduce sexually. Yet the origin of sexual reproduction is not clear and it has probably evolved independently several times. Since sexual reproduction allows genetic recombination, it should also allow the rapid incorporation of favorable mutations. Muller(21) was the first to propose that sex should accelerate evolution because two favorable mutations (A and B) are more likely to arise in different individuals of the same population than in a single individual. In asexual species, AB can only arise when two similar mutations occur simultaneously in the same individual(19,34).
A similar theory attempting to explain the advantages of sexual reproduction was formulated by Van Valen(33). According to this author, the environment of any given species is comprised of two major factors, the abiotic and biotic. The abiotic constitutes physical factors such as climate, and the biotic constitutes other species in the environment, particularly closely related ones competing with each other for limited resources such as food. Among such species, any evolutionary modifications adapting one to the environment may be detrimental to the other, but their evolution does not influence the abiotic factors. Therefore, each of the competing species must evolve constantly and rapidly in response to the modifications of the other. Otherwise, the least alterable will ultimately be eliminated by the selective forces of competition. This is what Van Valen(33) called "the Red Queen hypothesis", in analogy to Lewis Carol's book of Alice in Wonderland, in which the Red Queen said to Alice "Here you see, it takes all the running you can do to keep in the same place". Therefore, in environments such as rain forests, where competition is extremely intense, sexual reproduction is not only advantageous, but a necessity.
The Red Queen hypothesis, however, does not seem to accord with the geographic distribution of parthenogenetic animals, the majority of which occur in remote habitats isolated from their bisexual congeners(4). According to Cuellar(4), the major reason for the insular distribution of parthenogenetic species is the ability of single individuals to found a new colony without a member of the opposite sex. Assuming that parthenospecies are truly superior colonizers and have evolved in isolation from their bisexual progenitors(2,4), then competition does not appear to have played an important role in their evolution. Aside from the potentially disruptive influence of competition on the establishment of unisexual clones, fertilization of the virgin females would also eliminate the unisexual lineages by disrupting all-femalesness and the meiotic process, the latter of which regulates constancy of ploidy and the integrity of the species(2,3). Therefore, at least initially, parthenospecies must escape their bisexual counterparts in order to found new colonies. As long as they remain isolated from the bisexuals, they can circumvent extinction.
Although sexuality is the predominant mode of reproduction among all organisms, it is not entirely devoid of costs, the most common of which are meiosis and the production of males(19,34). As stated by Mayr(20), "in parthenogenetic animals, all zygotes are egg-producing females that do not waste half of their eggs on males." Still, sexual reproduction has the long-term advantage and is undoubtely the reason why it has appeared several times during evolutionary history and predominates as the principal reproductive mechanism in most organisms(1,18,34). On the other hand, parthenogenesis is only advantageous under special environments(4), may not be very old evolutionarily(1) and is considered an evolutionary blind alley(35,36).
PARTHENOGENETIC SCORPIONS AND SCORPIONISM
Since the initial demonstration of parthenogenesis in the scorpion Tityus serrulatus by Matthiesen(16), four additional parthenogenetic species have been recognized(13) and a few others have been suggested(27). Parthenogenesis in this group may be more common than presently suspected, since males in many species tend to be rare or even unknown. In fact, the recent demonstration of parthenogenesis in the above four species was based on rarity of males, and 1:1 sex ratios tend to be rare among scorpions in general(24). Except for T. serrulatus, which is probably an obligate parthenospecies, all of the other unisexuals are associated with bisexual populations (Figure 5). Within the range of these unisexual bisexual complexes, the bisexuals tend to be dominant and the unisexuals seem to occupy the peripheral sites(11,14), although as indicated by Lourenço & Cuellar(13) very little is still known about their specific ecologies. In Colombia, the parthenogenetic populations of Tityus columbianus (Thorell) occur on the western edge of the savannas within very dry canyons (Figure 6 and Figure 7). This region is known as the Mondonedo formation, which according to Van der Hammen(32) has been dry for the last 10,000 years. The bisexual populations of this species occupy the central and northern part of the range, consisting of a moist valley with extensive underground water. In contrast, the obligate parthenogen Tityus serrulatus has colonized all of the environments modified by human activity, and is presently expanding at an alarming pace toward the States of Mato Grosso, Tocantins and Rondonia, keeping pace with the ever expanding human population. What measures should be taken to prevent this highly toxic species from further expansion are not clear, but ultimately they must stem from the combined efforts of both field and laboratory investigations. Field biologists should strive to understand ecological and life history patterns, whereas laboratory studies should focus on behavioral and reproductive traits.
REFERENCES
02 CUELLAR O. Animal parthenogenesis.Science, 1977, 197, 837-43.
03 CUELLAR O. The evolution of parthenogenesis. A historical perspective, In: MOENS, PB. Ed. Meiosis. New York: Academic Press, 1987: 43-104.
04 CUELLAR O. Biogeography of parthenogenetic animals. Biogeographica,1994,70, 1-13.
05 GLESENER RR., TILMAN D. Sexuality and the components of environmental uncertainty: clues from geographic parthenogenesis in terrestrial animals. Am. Nat., 1978,112, 659-73.
06 KOVOOR J., LOURENÇO WR., MUÑOZ-CUEVAS A. Conservation des spermatozoïdes dans les voies génitales des femelles et biologie de la reproduction des Scorpions (Chélicérates). C.R. Acad. Sci. Paris, 1987, 304, 259-64.
07 LOURENÇO WR. La biologie sexuelle et le dévelopment post-embryonnaire du Scorpion Buthidae: Tityus trivittatus fasciolatus Pessôa, 1935. Rev. Nordest. Biol., 1979, 2, 49-96.
08 LOURENÇO WR. Sur l'écologie du Scorpion Buthidae: Tityus trivittatus fasciolatus Pessôa, 1935. Vie Milieu, 1981, 31, 71-6.
09 LOURENÇO WR. Biogéographie évolutive, écologie et les stratégies biodémographiques chez les Scorpions néotropicaux. C.R. Soc. Biogéogr., 1991a, 67, 171-90.
10 LOURENÇO WR. La province biogéographique guyanaise: étude de la biodiversité et des centres d'endémisme en vue de la conservation des patrimoines génétiques. C.R. Soc. Biogéogr., 1991b, 67, 113-31.
11 LOURENÇO WR. Parthenogenesis in the scorpion Tityus columbianus Thorell (Scorpiones: Buthidae). Bull. Br.Arachnol. Soc., 1991c, 8, 274-6.
12 LOURENÇO WR. Diversity and endemism in tropical versus temperate scorpion communities. Biogeographica, 1994, 70, 155-60.
13 LOURENÇO WR., CUELLAR O. Notes on the geography of parthenogenetic scorpions. Biogeographica, 1994, 70, 19-23.
14 LOURENÇO WR., CUELLAR O., MÉNDEZ DE LA CRUZ FR. Geographic variation in brood size between parthenogenetic and sexual populations of the Colombian scorpion Tityus columbianus. (submited).
15 LOURENÇO WR., KNOX MB., YOSHIZAWA MAC. L'invasion d'une communauté à le stade initial d'une succession secondaire par une espèce parthénogénétique de Scorpion. Biogeographica, 1994, 70, 77-91.
16 MATTHIESEN FA. Parthenogenesis in scorpions. Evolution, 1962,16, 255-6.
17 MAURANO HR. O escorpionismo. These, Rio de Janeiro: Faculdade Medicina. 1915.
18 MAYNARD SMITH J. The evolution of sex. Cambridge: Cambridge University Press. 1978.
19 MAYNARD SMITH J. Evolutionary Genetics. Oxford: Oxford University Press. 1989.
20 MAYR E. Animal species and evolution. Cambridge: Belknap. 1963.
21 MULLER HJ. Some genetic aspects of sex. Am. Nat., 1932,66: 118-38.
22 PIANKA ER. On r-and K-selection. Am. Nat., 1970, 104, 592-7.
23 PIANKA ER. Evolutionary Ecology. New York: Harper & Row. 1988.
24 POLIS GA. Ecology. In: POLIS, GA. Ed. The biology of Scorpions. Stanford: Stanford University Press, 1990: 247-93.
25 POLIS GA., FARLEY RD. Characteristics and environmental determinants of natality, growth and maturity in a natural population of the desert scorpion Paruroctonus mesaensis (Scorpionida, Vaejovidae). J.Zool., 1979, 187, 517-42.
26 POLIS GA., FARLEY RD. Population biology of a desert scorpion: survivorship, microhabitat, and the evolution of life history strategy. Ecology, 1980,61, 620-9.
27 POLIS GA., SISSOM WD. Life History. In: POLIS, GA. Ed. The Biology of Scorpions. Stanford: Stanford University Press, 1990: 161-223.
28 SPIRANDELLI CRUZ EF., YASSUDA CRW., JIM J., BARRAVIERA B. Programa de controle de surto de escorpião Tityus serrulatus Lutz e Mello, 1922, no Município de Aparecida (SP), (Scorpiones, Buthidae). Rev. Soc. Bras. Med. Trop , 1995, 28, 123-8.
29 STUTZ WH. Ocorrência de escorpionídeos no municipio de Uberlândia-MG-Brasil. Tese, Uberlandia: Universidade Federal de Uberlândia, 1990.
30 UDVARDY MDF. Dynamic zoogeography. With special reference to land animals. New York: R. V. Nostrand, 1969.
31 VANDEL A. La parthénogenèse géographique: Contribution à l'étude biologique et cytologique de la parthénogenèse naturelle. Bull. Biol. France Belg., 1928, 62, 164-281.
32 VAN DER HAMMEN T. The age of the Mondonedo formation and the Mastodon Fauna of Mosquera (Sabana de Bogota). Geologie Mijnbouw, 1965, 44, 384-90.
33 VAN VALEN L. A new evolutionary law. Evol. Theory, 1973, 1, 1-30.
34 WILLIAMS GC. Sex and evolution. Princeton: Princeton University Press, 1975.
35 WHITE MJD. Animal Cytology and Evolution. Cambridge: Cambridge University Press, 1954, 454.
36 WHITE MJD. Animal Cytology and Evolution. New York: Cambridge University Press, 1973, 931.
- 01 BELL G. The Masterpiece of Nature Berkeley: University of California Press. 1982
- 02 CUELLAR O. Animal parthenogenesis.Science, 1977, 197, 837-43.
- 03 CUELLAR O. The evolution of parthenogenesis. A historical perspective, In: MOENS, PB. Ed. Meiosis New York: Academic Press, 1987: 43-104.
- 04 CUELLAR O. Biogeography of parthenogenetic animals. Biogeographica,1994,70, 1-13.
- 05 GLESENER RR., TILMAN D. Sexuality and the components of environmental uncertainty: clues from geographic parthenogenesis in terrestrial animals. Am. Nat., 1978,112, 659-73.
- 06 KOVOOR J., LOURENÇO WR., MUÑOZ-CUEVAS A. Conservation des spermatozoïdes dans les voies génitales des femelles et biologie de la reproduction des Scorpions (Chélicérates). C.R. Acad. Sci. Paris, 1987, 304, 259-64.
- 07 LOURENÇO WR. La biologie sexuelle et le dévelopment post-embryonnaire du Scorpion Buthidae: Tityus trivittatus fasciolatus Pessôa, 1935. Rev. Nordest. Biol., 1979, 2, 49-96.
- 08 LOURENÇO WR. Sur l'écologie du Scorpion Buthidae: Tityus trivittatus fasciolatus Pessôa, 1935. Vie Milieu, 1981, 31, 71-6.
- 09 LOURENÇO WR. Biogéographie évolutive, écologie et les stratégies biodémographiques chez les Scorpions néotropicaux. C.R. Soc. Biogéogr., 1991a, 67, 171-90.
-
1010 LOURENÇO WR. La province biogéographique guyanaise: étude de la biodiversité et des centres d'endémisme en vue de la conservation des patrimoines génétiques. C.R. Soc. Biogéogr., 1991b, 67, 113-31.
- 11 LOURENÇO WR. Parthenogenesis in the scorpion Tityus columbianus Thorell (Scorpiones: Buthidae). Bull. Br.Arachnol. Soc., 1991c, 8, 274-6.
- 12 LOURENÇO WR. Diversity and endemism in tropical versus temperate scorpion communities. Biogeographica, 1994, 70, 155-60.
- 13 LOURENÇO WR., CUELLAR O. Notes on the geography of parthenogenetic scorpions. Biogeographica, 1994, 70, 19-23.
- 15 LOURENÇO WR., KNOX MB., YOSHIZAWA MAC. L'invasion d'une communauté à le stade initial d'une succession secondaire par une espèce parthénogénétique de Scorpion. Biogeographica, 1994, 70, 77-91.
- 16 MATTHIESEN FA. Parthenogenesis in scorpions. Evolution, 1962,16, 255-6.
- 17 MAURANO HR. O escorpionismo These, Rio de Janeiro: Faculdade Medicina. 1915
- 18 MAYNARD SMITH J. The evolution of sex Cambridge: Cambridge University Press. 1978
- 19 MAYNARD SMITH J. Evolutionary Genetics Oxford: Oxford University Press. 1989
- 20 MAYR E. Animal species and evolution Cambridge: Belknap. 1963
- 21 MULLER HJ. Some genetic aspects of sex. Am. Nat., 1932,66: 118-38.
- 22 PIANKA ER. On r-and K-selection. Am. Nat., 1970, 104, 592-7.
- 24 POLIS GA. Ecology. In: POLIS, GA. Ed. The biology of Scorpions. Stanford: Stanford University Press, 1990: 247-93.
- 25 POLIS GA., FARLEY RD. Characteristics and environmental determinants of natality, growth and maturity in a natural population of the desert scorpion Paruroctonus mesaensis (Scorpionida, Vaejovidae). J.Zool., 1979, 187, 517-42.
- 26 POLIS GA., FARLEY RD. Population biology of a desert scorpion: survivorship, microhabitat, and the evolution of life history strategy. Ecology, 1980,61, 620-9.
- 27 POLIS GA., SISSOM WD. Life History. In: POLIS, GA. Ed. The Biology of Scorpions Stanford: Stanford University Press, 1990: 161-223.
- 28 SPIRANDELLI CRUZ EF., YASSUDA CRW., JIM J., BARRAVIERA B. Programa de controle de surto de escorpião Tityus serrulatus Lutz e Mello, 1922, no Município de Aparecida (SP), (Scorpiones, Buthidae). Rev. Soc. Bras. Med. Trop , 1995, 28, 123-8.
- 29 STUTZ WH. Ocorrência de escorpionídeos no municipio de Uberlândia-MG-Brasil. Tese, Uberlandia: Universidade Federal de Uberlândia, 1990
- 30 UDVARDY MDF. Dynamic zoogeography With special reference to land animals. New York: R. V. Nostrand, 1969
- 31 VANDEL A. La parthénogenèse géographique: Contribution à l'étude biologique et cytologique de la parthénogenèse naturelle. Bull. Biol. France Belg., 1928, 62, 164-281.
- 32 VAN DER HAMMEN T. The age of the Mondonedo formation and the Mastodon Fauna of Mosquera (Sabana de Bogota). Geologie Mijnbouw, 1965, 44, 384-90.
- 33 VAN VALEN L. A new evolutionary law. Evol. Theory, 1973, 1, 1-30.
- 34 WILLIAMS GC. Sex and evolution Princeton: Princeton University Press, 1975
- 35 WHITE MJD. Animal Cytology and Evolution Cambridge: Cambridge University Press, 1954, 454.
- 36 WHITE MJD. Animal Cytology and Evolution New York: Cambridge University Press, 1973, 931.
 CORRESPONDENCE TO:
CORRESPONDENCE TO:Publication Dates
-
Publication in this collection
08 Jan 1999 -
Date of issue
1995