Abstract
In this paper further information concerning the ecological and biogeographical aspects of scorpionism is presented. Some of the information already outlined in three previous publications(10,14,16) is assessed, and new data given concerning the pattern of distribution presented by Tityus serrulatus in Brazil. Some new ideas are also proposed regarding the possible evolutionary advantages and disadvantages of parthenogenesis versus sexuality in both the short-term and the long-term.
scorpionism; Brazil; Tityus serrulatus
Original paper
THE EVOLUTION OF SCORPIONISM IN BRAZIL IN RECENT YEARS.
W. R. LOURENÇO
 CORRESPONDENCE TO:
W.R. LOURENÇO, Laboratoire de Zoologie (Arthropodes), M.N.H.N., 61 rue de Buffon, 75005, Paris, France.
, J. L. CLOUDSLEY-THOMPSON
CORRESPONDENCE TO:
W.R. LOURENÇO, Laboratoire de Zoologie (Arthropodes), M.N.H.N., 61 rue de Buffon, 75005, Paris, France.
, J. L. CLOUDSLEY-THOMPSON , O. CUELLAR
, O. CUELLAR , V. R. D. VON EICKSTEDT
, V. R. D. VON EICKSTEDT , B. BARRAVIERA
, B. BARRAVIERA , M. B. KNOX
, M. B. KNOX
1 Laboratoire de Zoologie (Arthropodes), M.N.H.N., Paris, France, 2 Department of Biology, University College London, Gower Street, London WC1E 6BT, United Kingdom, 3 Departments of Biology, The University of Utah, Salt Lake City, Utah 84112, U.S.A., 4 Laboratory of Venomous Arthropods of the Butantan Institute, State of São Paulo, Brazil, 5 Center for the Study of Venoms and Venomous Animals - CEVAP - UNESP, State of São Paulo, Brazil, 6 Laboratory of Zoology, Brasília University (UnB), Brasília, Brazil.
ABSTRACT. In this paper further information concerning the ecological and biogeographical aspects of scorpionism is presented. Some of the information already outlined in three previous publications(10,14,16) is assessed, and new data given concerning the pattern of distribution presented by Tityus serrulatus in Brazil. Some new ideas are also proposed regarding the possible evolutionary advantages and disadvantages of parthenogenesis versus sexuality in both the short-term and the long-term.
KEY WORDS: scorpionism, Brazil, Tityus serrulatus.INTRODUCTION
A few years ago, the senior author(10) attempted, for the first time, to explain the phenomenon of scorpionism by assessing both ecological and biogeographical patterns, as well as by considering several life history traits of dangerous species. Although it did not have a great impact, this contribution was of interest, since the author tried to demonstrate incorrect concepts about the biology of scorpions, and show that the positive selection of some species, and the selection against others was not associated with the toxicity of the venom. The type of selection that operates is a consequence of life history strategies adopted by species in relation to human impact on its natural environments.
Since Lourenço's(10) paper, reports of severe envenomation by scorpion stings have shown an alarming increase in various tropical and subtropical countries including Brazil, Mexico, Tunisia and Morocco(14,16) (Figure 1). In Brazil alone, more than 30,000 scorpion stings, resulting in more than 100 deaths, have been reported over a five-year period(25) (Figure 2). Additional deaths, caused by scorpions not previously suspected of being dangerous to humans, have recently been documented in Peruvian and Brazilian Amazonia(14,16), and isolated cases of incidents, with sometimes lethal consequences, have been reported from other countries such as Colombia, Venezuela, French Guyana and Iran (Lourenço, in press). In many cases, in Brazil and elsewhere, this situation (unexpected only a few years ago) has attracted the attention of the local press, which exploits the concept of whole cities being invaded by scorpions (e.g. Jornal de Brasilia of 22 June, 1993).
In this short synthesis, we will focus on the present situation in Brazil, where the principal species responsible for incidents is Tityus serrulatus (Figure 3). Only particular traits of its ecology and life history strategies can explain its very high population densities at the present time and the large number of incidents for which it is responsible.
FIGURE 3.Tityus serrulatus. Female from Minas Gerais.
THE SELECTION OF DANGEROUS SPECIES OF SCORPIONS BY HUMAN IMPACT ON NATURAL ENVIRONMENT: Before considering the selection of noxious species of scorpions which may enter in contact with human populations, it is fundamental to define scorpions in terms of their life history strategies. Although some species tend to be associated with man-made or disturbed habitats, the majority are adapted to very specific and predictable natural environments(11,12,17,18,19,20).
Scorpions have been divided into two broad ecological categories, «equilibrium» and «opportunistic», depending on the type of environment they inhabit and their life history strategies(20). Equilibrium species occupy stable, natural environments, whereas opportunistic species, such as some Tityus spp. in Brazil, can readily invade disturbed habitats(11,12,13,20). For more specific traits, see Polis(20) and Lourenço and Cuellar(16).
Let us imagine a primary zone of distribution for a given population of scorpions. Inside this zone the species can be found under one or other of the following conditions:
a. No other species is present in the area. This rare case can be seen in certain biologically depauperate environments in which the prey potential is weak. In most cases, it is an unstable and unpredictable environment due to natural phenomena. The vicissitudes of most impoverished conditions have excluded the equilibrium species, which are not ecologically plastic, and the sole remaining species is opportunistic(11,19,20).
b. The species shares its primary area of distribution with one or more species. Many examples of 3 to 10 species in sympatry are known in the desert, natural savanna and rain forest environments(20).
In the primary area of distribution, these sympatric species do not actually live or forage together. In fact, they generally belong to different families and genera and, when they do belong to the same genus, they are quite distant phylogenetically(14). Their ecology, and the trophic levels they occupy, do not therefore overlap. If such territorial exclusion is not well observed in a spatial sense, it nevertheless exists through differing chronology of the rhythms of most functions of the different species(8).
Among the species present in the primary area, one or more may have venoms which are very toxic to mammals, including human beings. The fact that they are more or less well adapted to a given environment is not directly associated with the toxicity of their venom. Since the primary function of scorpion venom is the capture of small prey, the high toxicity of the venom of certain species is difficult to explain in terms of its evolutionary significance, especially when neither Man nor large predators were originally present in the primary distribution area. In conclusion, species presenting marked ecological plasticity (usually opportunistic species) may possess either very toxic or relatively harmless venom. Similarly, species with marked ecological requirements, may or may not be toxic to Man.
Let us now suppose that a process of human colonization begins in a given primary area in which several species of scorpions are present. Colonization can be very rapid, and the processes of destruction of the primary environment happened in only a few centuries or even a few decades. This is the situation in countries such as Brazil and Mexico.
With modifications in the primary area, environmental conditions become less and less sufficient to provide the ecological requirements of the equilibrium species. Their populations, therefore, begin a process of regression until, in some cases, they even disappear. This has already taken place in the Caribbean, where the island ecosystems are particularly fragile. Several species of the Brazilian coastal Atlantic forest and oriental Amazonia may, likewise, have been eliminated.
Selection against equilibrium species, which are incapable of readaptation to new environmental conditions, and the encouragement of opportunistic species act on both species possessing very toxic venom and those possessing innocuous venom. In the first case, the phenomenon is observed by most people, since the species are dangerous to human beings, as in the case of some Centruroides species in Mexico (e.g. Centruroides suffusus Pocock, the famous Durango scorpion), Tityus serrulatus (Lutz and Mello) in Brazil and Androctonus australis (Linnaeus) in North Africa. In the second case, however, the phenomenon has been observed only by specialists interested in the study of scorpions in general. Examples include innocuous buthid scorpions, such as Isometrus maculatus (de Geer) which lives in most tropical coastal zones of the world, and bothriurid scorpions, such as Bothriurus bonariensis (Koch). The latter inhabits urban areas in meridional South America, being common in the region of Buenos Aires, Argentina. These two species do not present any threat to human beings.
INVASION AND COLONIZATION OF MODIFIED ENVIRONMENTS BY OPPORTUNISTIC (PARTHENOGENETIC) SPECIES: Several authors have noted that parthenogenetic animals have a tendency to appear in environments that are different from those inhabited by their bisexual relatives(4,5,23,24). Moreover, opportunistic (parthenogenetic) species have a dispersion and colonizing potential much higher than that of equilibrium species. In this sense, Tityus serrulatus appears to combine these two characteristics and to conform to the above predictions. This species, which almost certainly occupied a restricted area in the state of Minas Gerais about 300 years ago(14), is nowadays widely distributed over a large region of the southeast of Brazil and probably reaches Bolivia via the Brazilian state of Rondonia* * TOLEDO VELARDE, Instituto Ezequiel Dias, Belo Horizonte, MG, Brasil. Personal communication. . The geographical expansion of this species is undoubtdly related to human colonization, with scorpions being introduced into newly-created cities and towns by human agency. Therefore, newly-established human communities can be invaded within a few years after their foundation, although the surrounding natural areas (e.g. Savanas/Cerrados) may be virtually devoid of this species of scorpion. The creation of new habitats suitable for colonization by T. serrulatus may be compared with the opening of clearings in dense primary forests(1). The metaclimax Cerrados of the Brazilian plateau may represent these dense primary forests and new towns are comparable with opened clearings. In both cases, the new environments are localized and occur in disclimax situations in which the process of secondary succession begins. The new towns are, in many cases, separated by several hundred kilometers. Consequently, the country between them remains almost pristine and opportunistic species do not reach and colonize it.
The process of expansion by T. serrulatus occurs in steps. When scorpions are transported by anthropic agents, via roads or rail, and reach newly-created human environments, they establish new colonies. These act as islands inside the metaclimax formation of the Cerrados. This process is greatly facilitated by the fact that T. serrulatus possesses all the necessary requirements: (i) strong dispersal ability, (ii) a very high reproductive potential, (iii) a large reserve of populations, (iv) the capacity to forage on prey resources which may be abundant, although not specific, and which are localized in a disclimax environment(1).
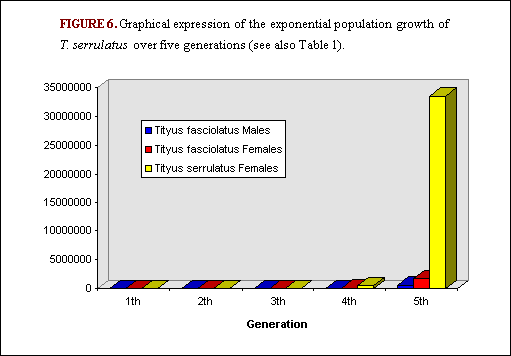
In the area of Brasilia and the Federal District, Lourenço et al. (17) studied secondary succession, with an invasion by T. serrulatus (an alien) and the consecutive regression of the population of Tityus fasciolatus (an equilibrium, sexual and autochthonous species, with a sex-ratio of 1 male to 3 females) (Figure 4 and Figure 5). This proved to be interesting because two inventories of the scorpion populations of that region were carried out within a period of 15-18 years. Comparison of the results of these inventories shows that the time necessary for the establishment of the alien species could be estimated at 10-15 years(17). Moreover, both the alien and the autochthonous species have similar biological and reproductive traits. Both average similar adult size (65 versus 67 mm), brood size (16x3 in a life time for T. fasciolatus; 16x4 in a life time for T. serrulatus), period of embryonic development (2.5 versus 3 months) and age to maturity (2.0 versus 2.5 years). The following hypothetical table indicates the potential development of the two populations over five generations (i.e. 12 years on average). The first generation consisted of two individuals for each population: 1 male and 1 female for T. fasciolatus and 2 females for T. serrulatus. Even taking into account the fact that probable survival of immature scorpions is low (in some cases 80% mortality occurs before the adult age is achieved), differences between the rates of increase of the two populations are very great. What has been observed in the region of Brasilia is certainly also taking place in other cities and towns of Brazil. For instance, a recent study(22) demonstrates that the city of Aparecida in the state of São Paulo has likewise been invaded by T. serrulatus.
TABLE 1. Comparison of the theoretical intrinsic rates of increase between a bisexual (T. fasciolatus) and a parthenogenetic (T. serrulatus) species of scorpion over 5 generations (Figure 6).
DISCUSSION
One of the characteristics of opportunistic and parthenogenetic species is the fact that they have density-independent population regulation, whereas equilibrium species show density-dependent population regulation. This is the reason why, in both old and recent literature describing scorpionism in Brazil, we find the expression of « population explosions », corresponding to the local word «surtos» (from the Latin surctu => surrectu: « to appear »), which reflects the type of population fluctuation observed in T .serrulatus (Figure 7).
Theoretical curves representing the difference between bisexual (dotted line) and unisexual (solid line) population fluctuations in nature. The beginning of each curve, up to line K (carrying capacity), represents the growth rate during the invasion of new habitats. Thereafter bisexual populations fluctuate close to K, whereas the unisexual populations fluctuate radically, overshooting K between catastrophies due to a high rate of increase, and crashing to a low level during catastrophies (adapted from Cuellar (4)).
Another interesting point is that, according to several authors, parthenogenesis would only be an advantageous mode of reproduction as a short-term strategy(4,5,15). The Red Queen hypothesis(16) for the maintenance of, or return to sexual reproduction implies that biotic factors exert selection pressure which makes asexual reproduction an unstable long-term strategy.
New data, however, indicate that this is not absolutely true. In some cases, several groups of organisms are escaping or outmanoeuvering the Red Queen scenario, sometimes for several millions of years(2,7,9). We are not in a position to say if this is the case with parthenogenetic scorpions. However, if this possibility could be applied to dangerous parthenogenetic species of scorpions, it might be rather unpleasant for human beings.
CONCLUSIONS
The mechanism of the selection of species that have low ecological plasticity acts indiscriminately on noxious and innocuous species. Ecological plasticity is not associated with the presence of toxic venom. Many noxious equilibrium species are selected against by human activities.
The positive selection of noxious opportunistic species is also directly associated with human activity. Let us suppose that, in a profoundly modified environment (as in several of the « artificial » cities of Brazil) the human population begins to increase rapidly. Within a short period of time, the three main factors needed to transform the region into an important center of scorpionism appear. These are: (i) expansion of the human population; (ii) rapid expansion of the noxious opportunistic scorpion species which soon occupy all the niches left empty by the regression or disappearence of the equilibrium species (in many cases the opportunistic species changes its behaviour and begins to live inside human dwellings); (iii) overlapping of a large human population with a large population of noxious scorpions greatly increases the probability of incidents of scorpionism. This situation is characteristic of certain regions of Brazil, Mexico and North Africa.
Curiously, however, the absolute number of noxious species appears to remain stable. Over a period of many years, several authors have suggested that only a limited number of species can be really dangerous to humans(3,14,21). We suggest that this number is, in fact, much higher in nature, but that many noxious species are never in close contact with human beings. In some large natural regions such as Amazonia, human populations have remained low for centuries, and only in recent years a rapid expansion of colonization has occurred, with dramatic destruction of the natural environment. It is quite possible that several species of the genus Tityus in Amazonia and in other areas such as the Colombian Andes may be highly toxic. Isolated cases of lethal incidents have been observed after stings by Tityus metuendus (Pocock), Tityus cambridgei (Pocock) and Tityus sp.(6,14,16)** ** OTERO, Facultad de Medicina Universidad de Medellin, Colombia. Personal communication. The available data on scorpion incidents do not provide a valid assessment of the situation. The data that figure in a 1981 WHO publication( 21) concern observations from 1930s to the 1960s. The enourmously increasing human population in many regions of the world, with consequences on the environment, urgently requires new and precise observations in the field. . Like many others, however, these are probably equilibrium species and will be partially or totally selected against by the destruction of their environment before human populations become large and vulnerable to them.
The available data on scorpion incidents do not provide a valid assessment of the situation. The data that figure in a 1981 WHO publication(21) concern observations from 1930s to the 1960s. The enourmously increasing human population in many regions of the world, with consequences on the environment, urgently requires new and precise observations in the field.
REFERENCES
01 BLONDEL J. Stratégies démographiques et successions écologiques.
Bull. Soc. Zool. France,
1976, 101, 695-718.
02 CHAPLIN JA., HAVEL EH., HEBERT PDN. Sex and Ostracods. Trends Ecol. Evol., 1994, 9, 435-9.
03 CLOUDSLEY-THOMPSON JL. Spiders and scorpions (Araneae and Scorpiones). In: LANE RP., CROSSKEY RW. Eds. Medical insects and arachnids. London:Chapman & Hall, 1993: 659-82.
04 CUELLAR O. Animal parthenogenesis. Science, 1977, 197, 837-43.
05 GLESENER RR., TILMAN D. Sexuality and the components of environmental uncertainty: clues from geographic parthenogenesis in terrestrial animals. Am. Nat., 1978, 112, 659-73.
06 GONZALEZ-SPONGA MA. Escorpiones de Venezuela. Caracas:Cuadernos Lagoven, 1984: 126.
07 GRIFFITHS HI., BUTLIN RK. A timescale for sex versus parthenogenesis: evidence from subfossil ostracods. Proc. R. Soc. Lond. [Biol.], 1995, 260, 65-71.
08 HADLEY NF., WILLIAMS SC. Surface activities of some North American scorpions in relation to feeding. Ecology, 1968, 49, 726-34.
09 LADLE RJ., JOHNSTONE RA., JUDSON OP. Coevolutionary dynamics of sex in a metapopulation: escaping the Red Queen. Proc. R. Soc. Lond. [Biol.], 1993, 253, 155-60.
10 LOURENÇO WR. Peut-on parler d'une biogéographie du scorpionisme? C. R. Soc. Biogéogr., 1988, 64, 139-45.
11 LOURENÇO WR. Biogéographie évolutive, écologie et les stratégies biodémographiques chez les Scorpions néotropicaux. C.R. Soc. Biogéogr., 1991, 67, 171-90.
12 LOURENÇO WR. La province biogéographique guyanaise; étude de la biodiversité et des centres d'endémisme en vue de la conservation des patrimoines génétiques. C.R. Soc. Biogéogr., 1991, 67, 113-31.
13 LOURENÇO WR. Diversity and endemism in tropical versus temperate scorpion communities. Biogeographica, 1994, 70, 155-60.
14 LOURENÇO WR., CLOUDSLEY-THOMPSON JL. Effects of human activities on the environment and their influence on the distribution of dangerous species of scorpions. In: INTERNATIONAL CONGRESS ON ENVENOMATIONS AND THEIR TREATMENTS, 1, Paris. [in press].
15 LOURENÇO WR., CUELLAR O. Notes on the geography of parthenogenetic scorpions. Biogeographica, 1994, 70, 19-23.
16 LOURENÇO WR., CUELLAR O. Scorpions, scorpionism, life history strategies and parthenogenesis. J. Venom. Anim. Toxins, 1995, 1, 50-64.
17 LOURENÇO WR., KNOX MB., YOSHIZAWA MAC. L'invasion d'une communauté à le stade initial d'une succession secondaire par une espèce parthénogénétique de Scorpion. Biogeographica, 1994, 70, 77-91.
18 PIANKA ER. On r-and K-selection. Am. Nat., 1970, 104, 592-97.
19 PIANKA ER. Evolutionary ecology. New York: Harper & Row, 1988.
20 POLIS GA. Ecology. In: POLIS GA. Ed.. The biology of scorpions. Stanford: Stanford University Press, 1990: 247-93.
21 SIMARD JM., WATT DD. Venoms and toxins. In: POLIS GA. Ed. The biology of scorpions. Stanford: Stanford University Press, 1990: 414-44.
22 SPIRANDELLI CRUZ EF., YASSUDA WINTHER CR., JIM J., BARRAVIERA B. Programa de controle de surto de escorpião Tityus serrulatus (Lutz e Mello, 1922), no Município de Aparecida (SP), (Scorpiones, Buthidae). Rev. Soc. Bras. Med. Trop., 1995, 28, 123-8.
23 UDVARDY MDF. Dynamic zoogeography. With special reference to land animals. New York: Van Nostrand Reinhold, 1969.
24 VANDEL A. La parthénogenèse géographique: Contribution à l'étude biologique et cytologique de la parthénogenèse naturelle. Bull. Biol. France Belg., 1928, 62, 164-281.
25 WEN FH., CARDOSO JLC., ARAÚJO FAA. Epidemiology of scorpion stings in Brazil. In: INTERNATIONAL CONGRESS ON ENVENOMATIONS AND THEIR TREATMENTS), 1, Paris, 1995. [Poster presented].
- 01 BLONDEL J. Stratégies démographiques et successions écologiques. Bull. Soc. Zool. France, 1976, 101, 695-718.
- 02 CHAPLIN JA., HAVEL EH., HEBERT PDN. Sex and Ostracods. Trends Ecol. Evol., 1994, 9, 435-9.
- 04 CUELLAR O. Animal parthenogenesis. Science, 1977, 197, 837-43.
- 05 GLESENER RR., TILMAN D. Sexuality and the components of environmental uncertainty: clues from geographic parthenogenesis in terrestrial animals. Am. Nat., 1978, 112, 659-73.
- 06 GONZALEZ-SPONGA MA. Escorpiones de Venezuela. Caracas:Cuadernos Lagoven, 1984: 126.
- 08 HADLEY NF., WILLIAMS SC. Surface activities of some North American scorpions in relation to feeding. Ecology, 1968, 49, 726-34.
- 10 LOURENÇO WR. Peut-on parler d'une biogéographie du scorpionisme? C. R. Soc. Biogéogr., 1988, 64, 139-45.
- 11 LOURENÇO WR. Biogéographie évolutive, écologie et les stratégies biodémographiques chez les Scorpions néotropicaux. C.R. Soc. Biogéogr., 1991, 67, 171-90.
- 12 LOURENÇO WR. La province biogéographique guyanaise; étude de la biodiversité et des centres d'endémisme en vue de la conservation des patrimoines génétiques. C.R. Soc. Biogéogr., 1991, 67, 113-31.
- 13 LOURENÇO WR. Diversity and endemism in tropical versus temperate scorpion communities. Biogeographica, 1994, 70, 155-60.
- 15 LOURENÇO WR., CUELLAR O. Notes on the geography of parthenogenetic scorpions. Biogeographica, 1994, 70, 19-23.
- 16 LOURENÇO WR., CUELLAR O. Scorpions, scorpionism, life history strategies and parthenogenesis. J. Venom. Anim. Toxins, 1995, 1, 50-64.
- 17 LOURENÇO WR., KNOX MB., YOSHIZAWA MAC. L'invasion d'une communauté à le stade initial d'une succession secondaire par une espèce parthénogénétique de Scorpion. Biogeographica, 1994, 70, 77-91.
- 18 PIANKA ER. On r-and K-selection. Am. Nat., 1970, 104, 592-97.
-
20POLIS GA. Ecology. In: POLIS GA. Ed.. The biology of scorpions. Stanford: Stanford University Press, 1990: 247-93.
- 21 SIMARD JM., WATT DD. Venoms and toxins. In: POLIS GA. Ed. The biology of scorpions. Stanford: Stanford University Press, 1990: 414-44.
- 22 SPIRANDELLI CRUZ EF., YASSUDA WINTHER CR., JIM J., BARRAVIERA B. Programa de controle de surto de escorpião Tityus serrulatus (Lutz e Mello, 1922), no Município de Aparecida (SP), (Scorpiones, Buthidae). Rev. Soc. Bras. Med. Trop., 1995, 28, 123-8.
- 23 UDVARDY MDF. Dynamic zoogeography. With special reference to land animals. New York: Van Nostrand Reinhold, 1969.
-
24VANDEL A. La parthénogenèse géographique: Contribution à l'étude biologique et cytologique de la parthénogenèse naturelle. Bull. Biol. France Belg., 1928, 62, 164-281.
Publication Dates
-
Publication in this collection
08 Jan 1999 -
Date of issue
1996