Abstract
Propolis has been the subject of several recent studies, with the aim of elucidating its biological and pharmacological properties. Propolis has a well-known antimicrobial activity as well as antioxidant, antitumoral, antiinflammatory, and regenerative properties, but literature about its effects on the immune response is scarce. The goal of this work was to evaluate the propolis effect on macrophage activation by oxygen (H2O2) and nitrogen (NO) metabolite determination. Propolis was produced by africanized honeybees and hydroalcoholic solutions were prepared at different concentrations. Peritoneal macrophages were obtained from male BALB/c mice and culture cells were stimulated in vitro with propolis or interferon-gamma (IFN-gamma). In the in vivo assay, the animals were sacrificed after propolis treatment and cells were stimulated with IFN-gamma. We also investigated the co-stimulant action of propolis associated with IFN-gamma on macrophages. The results show that propolis induces a discreet elevation in H2O2 release and a mild inhibition of NO generation, depending on concentration. Propolis had no co-stimulant activity, diminishing IFN-gamma action on H2O2 and NO production. Data suggest that propolis acts on host non-specific immunity by macrophage activation.
propolis; macrophages; hydrogen peroxide; nitric oxide
Immunomodulatory action of propolis on macrophage activation
R.O. ORSI , S.R.C. FUNARI
, S.R.C. FUNARI , A.M.V.C. SOARES
, A.M.V.C. SOARES , S.A. CALVI
, S.A. CALVI , S.L. OLIVEIRA
, S.L. OLIVEIRA , J.M. SFORCIN
, J.M. SFORCIN
 CORRESPONDENCE TO:
J. M. SFORCIN Departamento de Microbiologia e Imunologia, Instituto de Biociências de Botucatu, UNESP, Distrito de Rubião Junior, S/N, CEP: 18618-000, Botucatu, São Paulo, Brasil.
E-mail:
sforcin@ibb.unesp.br
, V. BANKOVA
CORRESPONDENCE TO:
J. M. SFORCIN Departamento de Microbiologia e Imunologia, Instituto de Biociências de Botucatu, UNESP, Distrito de Rubião Junior, S/N, CEP: 18618-000, Botucatu, São Paulo, Brasil.
E-mail:
sforcin@ibb.unesp.br
, V. BANKOVA .
.
1 Department of Microbiology and Immunology - Institute of Biosciences UNESP, Botucatu, State of São Paulo, Brazil; 2 Department of Animal Production, School of Veterinary Medicine and Animal Husbandry FMVZ UNESP, Botucatu, State of São Paulo, Brazil and 3 Institute of Organic Chemistry with Centre of Phytochemistry, Bulgarian Academy of Sciences, 1113 Sofia, Bulgaria
ABSTRACT. Propolis has been the subject of several recent studies, with the aim of elucidating its biological and pharmacological properties. Propolis has a well-known antimicrobial activity as well as antioxidant, antitumoral, antiinflammatory, and regenerative properties, but literature about its effects on the immune response is scarce. The goal of this work was to evaluate the propolis effect on macrophage activation by oxygen (H2O2) and nitrogen (NO) metabolite determination. Propolis was produced by africanized honeybees and hydroalcoholic solutions were prepared at different concentrations. Peritoneal macrophages were obtained from male BALB/c mice and culture cells were stimulated in vitro with propolis or interferon-gamma (IFN-g). In the in vivo assay, the animals were sacrificed after propolis treatment and cells were stimulated with IFN-g. We also investigated the co-stimulant action of propolis associated with IFN-g on macrophages. The results show that propolis induces a discreet elevation in H2O2 release and a mild inhibition of NO generation, depending on concentration. Propolis had no co-stimulant activity, diminishing IFN-g action on H2O2 and NO production. Data suggest that propolis acts on host non-specific immunity by macrophage activation.
KEY WORDS: propolis, macrophages, hydrogen peroxide, nitric oxide.INTRODUCTION
Propolis is a honeybee product with a very complex chemical composition, which has been used in folk medicine since ancient times. With respect to its antimicrobial activity, propolis inhibits bacterial growth, with a major effect on Gram-positive, and a limited action on Gram-negative bacteria (8). Its antiviral activity was investigated by Amoros et al. (1). Propolis also shows a fungicidal action, mainly against superficial mycosis (4).
Little is known about propolis effects on the immune system. Scheller et al. (16) found that propolis ethanolic extract induces antibody production by mice spleen cells. Propolis modulates both in vivo and in vitro C1q production by macrophages as well as the action of complement system receptors on these cells (2,3).
Ivanovska et al. (11) observed that cinnamic acid, one of the propolis components, acts on host defense, stimulating lymphocyte proliferation and inducing IL-1 and IL-2 production. Sforcin (18) observed that propolis action on natural killer cell activity increased its lytic capacity against tumor cells.
Macrophages are involved in several areas of body function, such as phagocytosis, enzyme liberation, free radical generation, and as mediators of inflammatory processes.
Scheller et al. (17) suggested that propolis immunostimulant activity may be associated with macrophage activation and enhancement of macrophage phagocytic capacity. Tatefuji et al. (20) investigated the effect of six propolis compounds on macrophage mobility and spreading.
Macrophages are capable of destroying microorganisms, including oxygen (H2O2) and nitrogen (NO) intermediate metabolite mechanisms.
Ivanovska et al. (10), investigating the effects of individual propolis compounds complexed with lysine, found that cinnamic acid tends to inhibit H2O2 release by peritoneal macrophages, while caffeic acid induces increased metabolite production.
Nitric oxide is responsible for DNA synthesis inhibition in several cells, and it is also able to inhibit the active transport in membranes of fungi and bacteria (5).
The goal of this work was to evaluate the effect of propolis on macrophage activation in vitro by H2O2 and NO determination. We also evaluated the effect of in vivo treatment with propolis on macrophage activation, as well as propolis co-stimulant action associated with IFN-g.
MATERIALS AND METHODS
PROPOLIS HYDROALCOHOLIC SOLUTION. Propolis was produced by africanized honeybees from the apiary located on Lageado Farm (FMVZ - UNESP). A 30% propolis ethanolic solution was prepared. A week later, this solution was filtered and used to prepare a 10% propolis hydroalcoholic solution (PHS) (19).
ANIMALS. Thirty male BALB/c mice weighing approximately 25-30 g aged between 6 and 8 weeks old were used for propolis treatment in vivo. The animals were divided into 6 groups (G1-6). G1 was considered control and received physiological solution (NaCl 0.9%). G2, 3, 4, 5, and 6 received 250, 500, 1000, 3000, and 6000 µg/ml of PHS, respectively.
All the groups received 0.2 ml of the solutions (IP) once a day for 3 days (11). Ten male BALB/c mice were used in the in vitro assays. Cell cultures were treated with PHS (2.5, 5, 10, 20, 50, and 100 µg/ml) or IFN-g (100 U/ml) for 24 h.
PERITONEAL MACROPHAGES. Peritoneal macrophages were obtained by the same procedure for in vivo or in vitro assays, inoculating with 3-5 ml of cold PBS in the abdominal cavity. After a soft abdominal massage for 30 seconds, the peritoneal liquid was collected with a Pasteur pipette and put in plastic tubes (Falcon). This procedure was repeated 3 or 4 times for each animal and the tubes were centrifuged at 200 g for 10 minutes.
The cells were pooled for the in vitro assays, and each mouses cells were used separately for the in vivo assay. Cells were resuspended in cell culture medium (RPMI 1640 supplemented with 5% fetal calf serum, 2mM L-glutamine, 40 µg/ml gentamycine, 20 mM HEPES, 2.5x10 M 2-mercaptoethanol) and cultivated in a 96-well U-bottom plate (Corning) at a final concentration of 2x10
M 2-mercaptoethanol) and cultivated in a 96-well U-bottom plate (Corning) at a final concentration of 2x10 cells/well. After 2 h at 37ºC, non-adherent cells were removed and macrophage monolayers were reincubated at 37ºC for 24 h.
cells/well. After 2 h at 37ºC, non-adherent cells were removed and macrophage monolayers were reincubated at 37ºC for 24 h.
EVALUATION OF MACROPHAGE ACTIVATION.
Determination of Oxygen Metabolites. Hydrogen peroxide (H2O2) release by peritoneal macrophages was determined according to Pick & Mizel (15).
Twenty-four hours after propolis treatment, the mice were sacrificed by overetherization and macrophages were plated in a final concentration of 2x10 cells/ml, incubated in a volume of 100 µl with no stimuli (control) or with recombinant IFN-g (100 U/ml). Supernatants were then collected for NO determination and 100 µl of red phenol solution, containing 140 mM NaCl, 10 mM K2HPO4 , 5.5 mM dextrose, and 5.5 mM horseradish peroxidase was added to the adherent cells for H2O2 determination. One hour later, 10 µl 1 M NaOH was added and the absorbances were measured at 620 nm, using an automatic enzyme immunoassay reader.
cells/ml, incubated in a volume of 100 µl with no stimuli (control) or with recombinant IFN-g (100 U/ml). Supernatants were then collected for NO determination and 100 µl of red phenol solution, containing 140 mM NaCl, 10 mM K2HPO4 , 5.5 mM dextrose, and 5.5 mM horseradish peroxidase was added to the adherent cells for H2O2 determination. One hour later, 10 µl 1 M NaOH was added and the absorbances were measured at 620 nm, using an automatic enzyme immunoassay reader.
H2O2 production by in vitro stimulated macrophages was also performed, where 2x10 cells/ml were incubated in a volume of 100 µl with no stimuli (control), with recombinant IFN-g (100 U/ml), or propolis hydroalcoholic solutions (2.5, 5, 10, 20, 50, and 100 µg/ml). Twenty-four hours later, supernatants were collected for NO determination and H2O2 determination was carried out with adherent cells.
cells/ml were incubated in a volume of 100 µl with no stimuli (control), with recombinant IFN-g (100 U/ml), or propolis hydroalcoholic solutions (2.5, 5, 10, 20, 50, and 100 µg/ml). Twenty-four hours later, supernatants were collected for NO determination and H2O2 determination was carried out with adherent cells.
Determination of Nitrogen Metabolites. Nitric oxide (NO) production was determined based on Griess reaction (9). As described above, supernatants of the 24-hour stimulated cells were collected and 100 µl of Griess reagent was added (N-1-naphthyl-ethyl-enediamine 0.1% + sulfanilamide 1% in H3PO4 5%). After 10 minutes, absorbances were measured at 540 nm at room temperature, using an automatic enzyme immunoassay reader.
CO-STIMULANT ACTIVITY. In order to determine a possible co-stimulant activity of propolis associated with IFN-g on H2O2 and NO production, peritoneal macrophages were stimulated in vitro with IFN-g (100 U/ml) and simultaneously with PHS (2.5, 5, 10, 20, 50, and 100 µg/ml).
STATISTICAL ANALYSIS. Statistical procedures were performed using Graph Pad Software, San Diego, Ca, USA, 1993 (7).
Analysis of variance (ANOVA) was used, followed by multiple comparison tests by Tukey-Kramer method.
Paired student t test was used to observe IFN-g effect on the same treatment for the in vivo assay. F statistics was considered significant when p<0.05.
RESULTS AND DISCUSSION
In vitro Assays
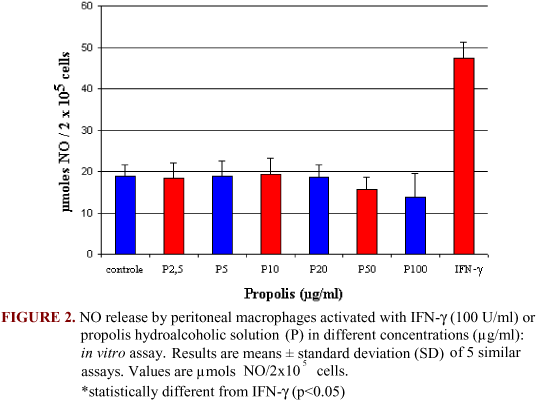
With respect to in vitro stimulated cells, it was observed that propolis (5, 10, and 20 µg/ml) induced an elevation in H2O2 production. Even if this biological effect is not statistically significant (p>0.05), these data suggest that propolis may activate macrophages with a consequent oxygen metabolite liberation (Figure 1). IFN-g was a potent stimulus for macrophage activation.
Oxygen intermediate metabolites are related to macrophage microbicidal activity (13), and propolis by enhancing the mobility and spreading of these cells, can be considered an important natural product, stimulating H2O2 production by macrophages.
Investigation of propolis isolated compounds is important for the understanding of its properties, as Ivanovska et al. (10) demonstrated that cinnamic acid inhibited and caffeic acid stimulated H2O2 release by macrophages.
In this study, we observed that propolis did not show a significant increase in H2O2 production by macrophages. Also, since in propolis chemical composition there are both stimulant and inhibitory compounds, we suggest that this final stimulant action may occur due to its constituent concentration in the hydroalcoholic solution, depending either on the predominance of one compound or synergism of several propolis constituents.
Nitric oxide has been reported as an important mechanism for macrophages against microorganisms (6). Propolis showed no significant alterations in NO production, with a mild inhibition in the concentrations of 50 and 100 µg/ml (Figure 2). Moriyasu et al. (14) observed that propolis extracts inhibit NO production by LPS-stimulated macrophages. This inhibitory effect is very important because NO is an inflammatory mediator in several pathologies, such as neuroinflammatory diseases (12).
In Vivo Assays
Propolis-treated mice showed a reduction in H2O2 production by macrophages in a dose-dependent way (p<0.001) (Figure 3).
In mice treated with 250 and 500 µg/ml of PHS, cell cultures activated with IFN-g showed higher H2O2 release than non-activated cells (control). This suggests that the treatment makes macrophages more responsive to stimuli such as IFN-g, which could be important to body defense. Cells of animals treated with 1000, 3000, and 6000 µg/ml of PHS and stimulated with IFN-g showed a significant inhibition in H2O2 generation compared to IFN-g stimulated control (p<0.001).
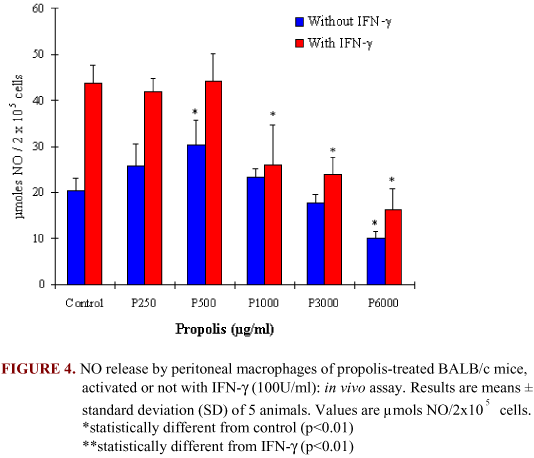
Propolis stimulated NO generation by macrophages when the animals were treated with the following concentrations of PHS: 250, 500, and 1000 µg/ml. Inhibition was seen in 3000 and 6000 µg/ml (Figure 4).
IFN-g-stimulated cells showed a similar NO liberation for control and 250 and 500 µg/ml of PHS. No liberation was inhibited for 1000, 3000, and 6000 µg/ml of PHS, even after IFN-g treatment (p<0.01).
We suggest that propolis induces NO production in a dose-dependent way, as high concentrations inhibit it. The understanding of propolis concentration in the solution as well as its administration time can provide further information about its biological effects in the body.
CO-STIMULANT ACTIVITY. Since IFN-g stimulates macrophage activation and propolis may show an immunomodulatory action, we investigated the co-stimulant activity of propolis associated with IFN-g. Figure 5 and Figure 6 shows propolis inhibition of IFN-g action.
These data may suggest an antagonistic effect of propolis on IFN-g stimulant action. Further investigation is necessary to identify whether it is one substance or a group of substances in propolis that induces this effect, maybe due to receptor competition or IFN-g inactivation. New assays can provide further information about propolis action in vivo and its relation with cytokines.
From these results, one can observe that propolis induces a discreet elevation in H2O2 release and an inhibition in NO generation, depending on concentration. These data suggest that propolis has an important role on the immune systems action on host non-specific immunity by macrophage activation.
ACKNOWLEDGEMENTS
The authors thank FAPESP for the grant.
REFERENCES
Received 13 August 1999
Accepted 28 October 1999
- 01 AMOROS M., SAUVAGER F., GIRRE L., CORMIER M. In vitro antiviral activity of propolis. Apidologie, 1992, 23, 231-40.
- 02 DIMOV V., IVANOVSKA N., MANOLOVA N., BANKOVA V., NIKOLOV N., POPOV S. Immunomodulatory action of propolis. Influence on anti-infectious protection and macrophage function. Apologie, 1991, 22, 155-62.
- 03 DIMOV V., IVANOVSKA N., BANKOVA V., POPOV, S. Immunomodulatory action of propolis: IV. Prophylactic activity against Gram-negative infections and adjuvant effect of the water-soluble derivative. Vaccine, 1992, 10, 817-23.
- 04 DOBROWOSDKI JW., VOHORA S.B., SHARMA K., SHAH SA., NAQVI SAH., DANDIYA PC. Antibacterial, antifungal, antiamoebic, antiinflammatory and antipiryretic studies on propolis bee products. J. Etnopharmacol., 1991, 35, 77-82.
- 05 DRAPIER JC., HIBBS JR. Murine cytotoxic activation macrophages inhibit aconitase in tumor cells. Inhibition involves the iron-sulfur prostetic group and is reversible. J. Clin. Invest., 1986, 78, 790-5.
- 06 GAUTHIER YP., BURCKHART MF., VIDAL DR. Role of nitric oxide in antibacterial non-specific immunity: in vivo and in vitro study. Trav. Sci. Chercheurs Serv. Sante Armees, 1994, 15, 57-8.
- 07 GODFREY K. Statistics in practice. Comparing the means of several groups. N. Engl. J. Med., 1985, 313, 1450-6.
- 08 GRANGE JM., DAVEY RW. Antibacterial properties of propolis (bee glue). J. R. Soc. Med., 1990, 83, 159-60.
- 09 GREEN LC., LUZURIAGA KR., WAGNER DA., RAND W., ISTFAN N., YOUNG VR., TANNENBAUM SR. Nitrate biosynthesis in man. Proc. Natl Acad. Sci. USA, 1981, 78, 7764-8.
-
10IVANOVSKA N., STEFANOVA Z., VALEVA V., NEYCHEV H. Immunomodulatory action of propolis: VII. A comparative study on cinnamic and caffeic acid lysine derivatives. Biol. Immun., 1993, 46, 115-7.
-
11IVANOVSKA N., NEYCHEV H., STEFANOVA Z., BANKOVA V., POPOV S. Influence of cinnamic acid on lymphocyte proliferation, cytokine release and Klebsiella infection in mice. Apidologie, 1995, 26, 73-81.
-
12KORYTKO PJ., BOJE KMK. Pharmacological characterization of nitric oxide production in a rat model of meningitis. Neuropharmacology, 1996, 35, 231-7.
-
13MOONIS M., AHMAD I., BACHHAWAT BK. Macrophage in host defense: an overview. Indian J. Biochem. Biophys., 1992, 29, 115-22.
-
14MORIYASU J., ARAI S., MOTODA R., KURIMOTO M. In vitro activation of mouse macrophage by propolis extract powder. Biotherapy, 1994, 8, 364-5.
-
15PICK E., MIZEL D. Rapid microassays for the measurement of superoxide and hydrogen peroxide production by macrophages in culture using an automatic enzyme immunoassay reader. J. Immunol. Methods, 1981, 46, 2111-26.
-
16SCHELLER S., GADZA G., PIETSZ G., GABRYS J., SZUMLAS J., ECKERT L., SHANI J. The ability of ethanol extract of propolis to stimulate plaque formation in immunized mouse spleen cells. Pharmacol. Res. Commun., 1998, 20, 323-8.
-
17SCHELLER S., GAZDA G., KROL W., CZUBA Z., ZAJUSZ A., GABRYS J., SHANI J. The ability of ethanolic extract of propolis (EEP) to protect mice against gamma irradiation. Z. Naturforsch. Sect. C. Biosc., 1989, 44, 1040-52.
-
18SFORCIN J.M. Efeito da sazonalidade sobre as propriedades imunomoduladora e antibacteriana da própolis e perfil bioquímico de ratos Botucatu: Universidade Estadual Paulista, Faculdade de Medicina Veterinária e Zootecnia, 1996. 59p. [Tese Doutorado].
-
19SFORCIN J.M., FUNARI S.R.C., NOVELLI E.L.B. Serum biochemical determinations of propolis-treated rats. J. Venom. Anim. Toxins, 1995, 1, 31-7.
-
20TATEFUJI T., IZUMI N., OHTA T., ARAI S., IKEDA M., KURIMOTO M. Isolation and identification of compounds from Brazilian propolis which enhance macrophage spreading and mobility. Biol. Pharm. Bull., 1996, 19, 966-70.
 CORRESPONDENCE TO:
CORRESPONDENCE TO:Publication Dates
-
Publication in this collection
22 Sept 2000 -
Date of issue
2000
History
-
Accepted
28 Oct 1999 -
Received
13 Aug 1999