Abstracts
BACKGROUND: The use of antimicrobials in the treatment of gonorrhoea started in 1930 with sulphonamides. Subsequently other drugs, such as tetracycline and its derivatives, were indicated for treating gonorrhoea. Therapeutic response to these drugs has tended to decline due to chromossomal and plasmidic resistance. However, tetracycline as a monotherapy or in association with other drugs is still prescribed for treating gonorrhea in Brazil. This justifies the need for a critical analysis in order to evaluate the sensitivity of gonococcus to this drug. OBJETIVE: Evaluate the real incidence of chromosomal resistance of Neisseria gonorrhoeae strains to tetracycline in our environment. METHOD: Analysis of the minimal inhibitory concentration in agar dilution. RESULTS: The chromosomal resistance to tetracycline was detected in 40.3% of all analyzed strains. CONCLUSIONS: This finding condemns the use of tetracycline as a mono or combined therapy for the treatment of gonorrhoea in our environment.
Neisseria gonorrhoeae; Tetracycline resistance; Microbial sensitivity tests
FUNDAMENTO: A utilização de antimicrobianos no tratamento da gonorréia iniciou-se em 1930 com as sulfonamidas. No decorrer dos anos outras drogas passaram a ser utilizadas, entre elas a tetraciclina. Embora eficaz no início, esta droga, ao longo do tempo, passou a não mais apresentar o resultado terapêutico esperado em virtude do aparecimento de quadros de resistência cromossômica e plasmidial em diversos países. Como a tetraciclina ainda continua sendo indicada, isoladamente ou associada a outras drogas antimicrobianas, para o tratamento da gonorréia no Brasil, tornou-se necessária a realização de um estudo de sensibilidade do gonococo à mesma, no intuito de se estimar a real dimensão da resistência do gonococo à tetraciclina. OBJETIVO: Avaliar a incidência de resistência cromossômica das cepas de Neisseria gonorrhoeae à tetraciclina. MÉTODO: Estudo da concentração inibitória mínima pelo método de diluição em ágar. RESULTADOS: A resistência cromossômica à tetraciclina detectada foi de 40,3% entre todas as cepas estudadas, segundo os critérios estabelecidos pelo Center for Diseases Control. CONCLUSÕES: Desaconselha-se definitivamente o uso isolado ou associado da tetraciclina e derivados, no tratamento da gonorréia no Brasil, no atual momento epidemiológico.
Neissseria gonorrhoeae; Resistência à tetraciclina; Testes de sensibilidade microbiana
CLINICAL, EPIDEMIOLOGICAL, LABORATORY AND THERAPEUTIC INVESTIGATION
Neisseria gonorrhoeae: chromosomal resistance to tetracycline in São Paulo, Brazil* * Study carried out at the Hospital das Clinicas FMUSP/University of Sao Paulo Faculty of Public Health.
Walter Belda JuniorI; Luiz Jorge FagundesII; Luis Fernando de Góes SiqueiraIII
IPhD, Professor, Department of Dermatology, Faculty of Medicine, University of Sao Paulo
IIPhD, Professor, Faculty of Public Health, University of Sao Paulo. Health Center/Geraldo de Paulo Souza School
IIIPh.D., Professor, Department of Epidemiology, Faculty of Public Health, University of Sao Paulo
Correspondence Correspondence to Walter Belda Junior Av. Açocê, 162 - Moema São Paulo SP 04075-020 Tel: (11) 5051-1921 E-mail: walterbelda@uol.com.br
ABSTRACT
BACKGROUND: The use of antimicrobials in the treatment of gonorrhoea started in 1930 with sulphonamides. Subsequently other drugs, such as tetracycline and its derivatives, were indicated for treating gonorrhoea. Therapeutic response to these drugs has tended to decline due to chromossomal and plasmidic resistance. However, tetracycline as a monotherapy or in association with other drugs is still prescribed for treating gonorrhea in Brazil. This justifies the need for a critical analysis in order to evaluate the sensitivity of gonococcus to this drug.
OBJETIVE: Evaluate the real incidence of chromosomal resistance of Neisseria gonorrhoeae strains to tetracycline in our environment.
METHOD: Analysis of the minimal inhibitory concentration in agar dilution.
RESULTS: The chromosomal resistance to tetracycline was detected in 40.3% of all analyzed strains.
CONCLUSIONS: This finding condemns the use of tetracycline as a mono or combined therapy for the treatment of gonorrhoea in our environment.
Keywords: Neisseria gonorrhoeae; Tetracycline resistance; Microbial sensitivity tests.
INTRODUCTION
Gonorrhea is among the oldest morbid states in our civilization. Urethral secretions of venereal origin, supposedly gonococci, were already reported by the Chinese during the Huang Ti Empire in 2637 B.C. With the appearance of sulphonamides in the 1930s, a new therapeutic era began as they were indicated for the treatment of gonorrhea.1 However in spite of being extremely effective, roughly 8% of Neisseria gonorrhea strains isolated in the pre-sulphonic period were already naturally resistant to this remedy.2 Resistance to sulphonamides developed quickly.3,4 By the mid-1940s, the prevalence of sulphonamide-resistant strain has proved to be extremely high, with 15% in 1942 and 59% in 1943.4 With the appearance of penicillin in 1943, and its indication for treating gonorrhea, a new era opened for the treatment of this disease strain since the drug proved to be highly effective from the very start.5 However, expectations that Neisseria gonorrhoeae could disappear with the use of penicillin failed to materialize. By the end of the 1950s, various reports pointed to reduced sensitivity of gonococcus to penicillin.6-8 By the end of the 1970s, a therapeutic dose of 4,800,000 UI was required, which is some 16 times greater than utilized for treatments in 1943.9
From the end of the 1980s, high levels of chromosomally mediated resistance were first described involving various drugs commonly used for treating gonorrhea. Among them, spectinomycin, in which resistance is due to a single mutation site resulting in high levels of resistance to this medication, as well as cephalosporin.10,11 The latter was observed in the strain promoting an association with genetic resistance sites, and producing the so-called pleiotropic phenomenon.12
Concomitantly to the rise of chromosomal resistance to other drugs, tetracycline took the same turn. In 1972, a simple therapeutic dose of the latter or its analogues had proved to be ineffective.13
It is known today that the cultural aspects in vigor in diverse population fragments determine what is of capital importance in the installation, maintenance, propagation and resistance mechanisms of these strains. Among them, indiscriminate antibiotic use stands out. According to the World Health Organization, this is linked to social pressures relative to sexually transmitted diseases, which are still stigmatized. It is also linked to untimely action on the part of M.D.s and paramedics fomenting an excessive use of antimicrobial drugs.14
In virtue of tetracycline still being present in diverse official therapeutic manuals and guides for treating gonorrhea either mono or combined therapy with other drugs, and faced with innumerable reports of therapeutic failure associated with the absence of studies in Brazil on the real incidence of chromosomal resistance to tetracycline, the present authors have proceeded to study this phenomenon.
MATERIAL AND METHODS
This study was carried out on patients with non complicated acute gonorrhea, with or without symptomatology and in both sexes. The patients sought medical assistance spontaneously at the Sexually Transmitted Diseases Service of the Dermatology Division at the Hospital das Clinicas, linked to the University of Sao Paulo Medical Faculty and the Sexually Transmitted Diseases Ambulatory of the Health Center/Geraldo de Paula Souza School, of the Sao Paulo University Faculty of Public Health, from 1998 to 2002. The sex- and age-related characteristics of patients are expressed in chart 1. All patients in the early phase of diagnosis were subjected to a collection of urethral or cervical material, following the specific appropriate techniques.15 The material collected was processed by direct bacterioscopy after staining by the Gram modified method16 Concomitantly, a culture for Neisseria gonorrhoeae was undertaken in a selective Thayer Martin medium17 A corroborative identification was performed by sugar acidification reaction in Cistina-triptose agar medium.18 After its identification, the Neisseria gonorrhoeae strains were submitted to the minimal inhibitory concentration test (MIC) to tetracycline by the sugar dilution method, according to appropriate technical specifications.19-21 The drug tested was chlorhydrate tetracycline, which has its in vitro results applicable to chlortetracycline, oxytetracycline and other members of this group. The latter was prepared according to the tried and tested techniques in the literature. It was diluted in saline solution and incorporated to a culture medium at a temperature of 56°C,22,23 dso as to obtain the following final concentrations: 0,125; 0,25; 0,5; 1; 2; 4; 8; 16 and 32mg/ml. In the interpretation of results, the strains considered sensitive to tetracycline were those showing an inhibitory concentration at less than or equal to 0.5 mg/ml. Those considered resistant to tetracycline showed a minimal inhibitory concentration greater or equal to 1mg/ml.24
RESULTS
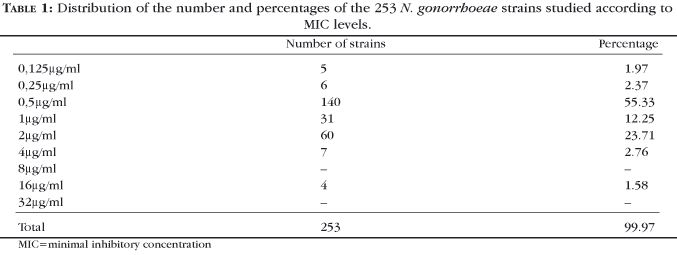
Two-hundred eighty-nine Neisseria gonorrhoeae strains were isolated. These 253 were subjected to a minimal inhibitory concentration to tetracycline tests, as 36 strains were lost during the preparation and storing process.
The results obtained in the sensitivity test are illustrated in table 1. The chromosomal resistance detected was 40.3% within all of the strains tested, observing the criteria established by the Center for Disease Control.24
DISCUSSION
The United States National Committee for Clinical Laboratory Standards25 (NCCLS) has adopted four categories of sensitivity to antibiotics, classifying them as sensitive, moderately sensitive, intermediate and resistant. In exchange, the Center for Disease Control (CDC) in Atlanta, which is the most effective epidemiological watchdog on the American continent, has simplified the interpretive categories regarding susceptibility to antimicrobials. As such, in accordance with chart 2, tetracycline resistant strains were considered, according to NCCLS,25 which presented with a MIC greater or equal to 2mg/ml; On the other hand, according to CDC, strains with a MIC greater or equal to 1mg/ml. are to be considerable resistant.
Therefore, this study detected a Neisseria gonorrhoeae tetracycline resistance rate of 28.05% among the strain studied, when analyzed according to NCCLS. Still according to NCCLS, 67.58% of isolated strain was moderately sensitive to tetracycline with the possibility of a therapeutic failure oscillating between 5 and 15%, and only 4.34% of the strain was considered sensitive. This category divisions proposed by NCCLS, albeit scientific, might still be considered risky if and when adopted in a system of epidemiological surveillance since the strain classified as moderately sensitive shows a potentially greater probability of experiencing modifications to its sensitivity by selective pressure mechanisms. In relation to the CDC-adopted criteria, which are more adequate for an epidemiological surveillance program, 40.3% of the Neisseria gonorrhoeae strain were found to be tetracycline resistant.
The percentages in tetracycline resistance findings were first emphasized at the beginning of the 1970s. Ever since then, resistance to tetracycline has shown a rising tendency.26-29
In relation to the results obtained in this study, the authors believe that the problem of chromosomal resistance to tetracycline in Brazil, from the percentages found (40.3%), must be as out-dated as what may be observed in the literature. The high levels of resistance found are thought to be linked to selective pressure phenomena, which act in such a way as to favor the installation, maintenance and propagation of these resistance characteristics.
In a study of risk factors in the installation of tetracycline resistance, Telzak30 indicates that indiscriminate use of this drug will select microorganisms that go on to colonize the genitourinary tract. With the strength of a gonococcus infection, these could potentially have the capacity to transfer such genic characteristics to gonococci through conjugation and recombination
CONCLUSIONS
Based on the results obtained in this study, the authors believe that installing an efficient epidemiological surveillance system is indispensable. This must be done in relation to the sensitivity of etiological agents to antimicrobials, as well as through a reevaluation of the indications for the use of tetracycline and derivatives either in mono- or combined therapy with other drugs for treating gonorrhea in Brazil. Faced with the current state of chromosomal resistance detected, the authors advise against the use of tetracycline and derivatives given the possibility of severe clinical consequences for the patients. q
REFERENCES
Received on August 25, 2003.
Approved by the Consultive Council and accepted for publication on December 12, 2004.
- 1. Cokkins AJ, McElligott GLM. Sulphanilamide in Gonorrhoeae. Lancet. 1938;2:355-62.
- 2. Reyn A. Sensitivity of N. Gonorrhoeae to antibiotics. Br J Vener Dis. 1961;37:145-57.
- 3. Goodale WT, Gould RG. Laboratory identification of sulfonamide-resitant gonococcic infections. JAMA. 1943;123:547-49.
- 4. Carpenter CM, Ackerman H. Correlation of in vitro sulfonamide resistance of the gonococcus with results sulfonamide therapy. Am J Public Health. 1944;34:250-4.
- 5. Mahoney JF, Ferguson C, Buchholtz M. The use of penicillin sodium in the treatment of sulfonamide-resistant gonorrhea in men:a preliminary report. Am J Syph Gonor Vener Dis. 1943;27:525-8.
- 6. Thayer JD, Field FN, Magnusos HJ. The sensitivity of gonococci to penicillin and its relationship to penicillin failures. Antibiot Chemother. 1957;7:306-10.
- 7. Curtis FR, Wilkinson AE. A comparison of the in vitro sensitivity of gonococci to penicillin with the results of treatment. Br J Vener Dis. 1958;34:70-8; discussion 78-82.
- 8. Willcox RR. Treatment problems of gonorrhoea. Bull Word Health Organ.1961;24:307-19
- 9. Kauffman RE, Johnson RE, Jaffe HW, Thornsberry C, wiesner PJ. National gonorrhoeae therapy monitoring study. Treatment results. N Engl J Med. 1976;294:1-4.
- 10. Zienilman JM, Nims LJ, Menegus MA, Knapp JS. Spectinomycin-resistant gonococcal infections in the United States, 1985-86. J Infect Dis. 1987;156:1002-4.
- 11. Gollow MM, Blums M, Ismail A. Penicillin-sensitive spectinomycin resistant- Neisserea gonorrhoeae. Med J Aust. 1986;144:651-2.
- 12. Ison CA, Gedney J, Easmon CSF. Chromossomal resistance of gonococci to antibiotics. Genitourin Med. 1987; 63:239-43.
- 13. Wiesner PJ, Holmes KK, Sparling PF, Karney WW. Single doses of methacycline and doxycycline for gonorrhoea: A cooperative study of the frequency and cause of treatment failure. J Infect Dis. 1973;127:461-6.
- 14. Organizacion Panamericana de la Salud. Neisseria gonorrhoeae. Resistência à multiples antibióticos. Bol Epidemiol. 1985;6:7-10.
- 15. Siqueira LFG. O laboratório nas Doenças Sexualmente Transmissíveis. Bol Inform Union. 1983;8:1-2.
- 16. Siqueira LFG. O laboratório nas doenças sexualmente Transmissíveis II. Bol Inform Union. 1984;9:6-8.
- 17. Martin JE, Lester A. Transgrow,a medium for transport and growth of Neisseria gonorrhoeae and Neisseria meningitides. Public Health. 1971;86:30-3.
- 18. Shtibel R, Toma S. Neisseria gonorrhoeae:evaluation of some methods used for carbohydrate utilization. Can J Microbiol. 1978;24:177-81.
-
19National Commitee for Clinical Laboratory Standards. Performance standards antimicrobial susceptibility testing: second informational supplement. M100-52 National Commitee for Clinical Laboratory Standards. 1987.
-
20National Commitee for Clinical Laboratory Standards. Methods for diluition antimicrobial susceptibility tests for bacteria that grow aerobically:tentative standard. M7-T2. National Commitee for Clinical Laboratory Standards. 1988.
- 21. Washington II JA .Susceptibility tests:Agar diluition. In: Lennette EH, Balows A, Hausler WJ, Shadomy HJ, editors. Manual of clinical microbiology. 4th. Washington, American Society for Microbiology; 1985. p. 967-71.
- 22. Anhalt JP, Washington II JA. Preparation and storage of antimicrobial solutions.In: Lennette EH, Balows A, Hausler WJ, Shadomy HJ, editors. Manual of clinical microbiology. 4th. Washington: American Society for Microbiology; 1985. p.1019-20.
- 23. Finegolg SM, Baron EJ. Metodos para evaluar la efectividad antimicrobiana.In: Bailey-Scott. Diagnostico microbiologico. 7Ş ed. Buenos Aires: Editora Medica Panamericana; 1989. p.190-210.
- 24. Center for Disease Control and Prevention. Antibiotic resistant strains of Neisseria gonorrhoeae. Policy guidelines for detection, management and control. MMWR. 1987; 36:1-14.
-
25National Commitee for clinical Laboratory Standards. MIC interpretive standards of three categories of susceptibility for Neisseria gonorrhoeae. M7-10. National Commitee for Clinical Laboratory Standards. 1990.
- 26. Piot P, Dyck EV, Colaert J, Ursi JP, Meheus A. Antibiotic susceptibility of N.gonorrhoeae strains from Europe and Africa. Antimicrob Agents Chemother. 1974;15:535-39
- 27. Fernandez H, Otth L. Sensibilidade in vitro de N. gonorrhoeae a seis drogas antimicrobianas. Rev Microbiol. 1983;14:273-5.
- 28. King SD, Brathwaite AR, Dillon JR. Treatment of gonorrhoea and susceptibility to antimicrobials of PPNG and non-PPNG strains in Jamaica.Genitourin Med. 1987;63:365-70.
- 29. Belda Jr W, Siqueira LFG, Nico M, Fagundes LJ. Atividade in vitro de cinco drogas antimicrobianas contra Neisseria gonorrhoeae. An Brás Dermatol. 2002;77:661-7.
- 30. Telzak EE, Spitalny KC, Faur YC, Knapp JS, Schultz S, Gunn RA, Blum S. Risk factors for infection with plasmid mediated high-level tetracycline resistant Neisseria gonorrhoeae. Sex transm Dis. 1989;16:132-6.
Publication Dates
-
Publication in this collection
25 Nov 2005 -
Date of issue
Feb 2005
History
-
Received
25 Sept 2003 -
Accepted
04 Dec 2004