Abstracts
Several dermatoses are considered idiopathic diseases. Many times lesions such as erythema nodosum or erythema multiformis, for example, cannot have their etiology defined. Human infection caused by Bartonella spp. may determine several clinical syndromic expressions. Starting with a clinically, histologically and ultrastructurally documented bacillary angiomatosis case, a review of medical literature was undertaken to evaluate the available diagnostic criteria regarding infection caused by these agents. Histological evaluations were concluded to be, practically speaking, an important and useful diagnostic method, especially when serology is not available.
Bartonella; Diagnosis; Skin
Várias dermatoses são consideradas idiopáticas. Muitas vezes, afecções como eritema nodoso ou eritema multiforme, por exemplo, não podem ter sua etiologia definida. A infecção humana por Bartonella spp. pode determinar várias expressões clínicas sindrômicas. A partir de um caso de angiomatose bacilar com documentação clínica, histológica e ultra-estrutural, foi feita a revisão da literatura médica para avaliar os critérios diagnósticos disponíveis para a infecção por esses agentes. Conclui-se que a avaliação histológica é, na prática, um importante e útil método diagnóstico, especialmente quando a sorologia não estiver disponível.
Bartonella; Diagnóstico; Pele
CASE REPORT
Diagnosis of Bartonella spp. infection: study of a bacillary angiomatosis case* * Work done at Faculdade de Ciências Médicas da Universidade Estadual de Campinas / FCM - UNICAMP - Campinas (SP), Brazil.
Paulo Eduardo Neves Ferreira VelhoI; Elemir Macedo de SouzaI; Maria Letícia CintraII; Aparecida Machado de MoraesI; Ana Maria Uthida-TanakaI
IM.D., PhD, Professor of Dermatology at the Department of Internal Medicine, Faculdade de Ciências Médicas da Universidade Estadual de Campinas / FCM - UNICAMP - Campinas (SP), Brazil
IIM.D., PhD, Professor, Head of the Departament of Pathological Anatomy at the Faculdade de Ciências Médicas da Universidade Estadual de Campinas / FCM - UNICAMP - Campinas (SP), Brazil
Mailing Address Mailing Address Paulo Eduardo Neves Ferreira Velho Departamento de Clínica Médica da Faculdade de Ciências Médicas da Universidade Estadual de Campinas FCM / Unicamp Cidade Universitária Zeferino Vaz, s/n. 13083-970 - Campinas - SP - Brazil Tel./Fax: +55 (19)3289-4107 E-mail: pvelho@unicamp.br
ABSTRACT
Several dermatoses are considered idiopathic diseases. Many times lesions such as erythema nodosum or erythema multiformis, for example, cannot have their etiology defined. Human infection caused by Bartonella spp. may determine several clinical syndromic expressions. Starting with a clinically, histologically and ultrastructurally documented bacillary angiomatosis case, a review of medical literature was undertaken to evaluate the available diagnostic criteria regarding infection caused by these agents. Histological evaluations were concluded to be, practically speaking, an important and useful diagnostic method, especially when serology is not available.
Keywords:Bartonella; Diagnosis; Skin
INTRODUCTION
Bartonela spp. form a fascinating group of pathogens and are responsible for emerging bacterial infectious diseases. They cause Carrion's disease, with its febrile (Oroya's fever) and tecidual (verruga peruana) stages; trench fever, cat-scratch disease and bacillary angiomatosis. Relapsing febrile bacteremias, endocarditis, septicemias, neurological, psychiatric, ophtalmological, osteus and hematologic manifestations are also associated with infection by these agents. Severe hemolytic anemia is a milestone of the febrile and immunosuppressing stage of Carrion's disease. Moreover, many human bartonella infections have blood-sucking ectoparasites as vectors: lutzomites, lice and ticks.1
CASE REPORT
Forty-nine year-old brown female patient, who had been diagnosed with AIDS two years before, had been presenting for one month bleeding and painful papular and nodular-papular lesions, initially in upper limbs, later disseminating through the entire body surface, including genitals, together with fever, weight loss, lack of appetite and malaise. Six months before, she had presented pruritic desquamative lesions in the entire body and mentioned having had contact with various cats.
Upon physical examination, signs of malnourishment, desquamation and squamous-crusts in trunk, especially back (Figure 1A), limbs and scalp were observed. Angiomatous papular and nodular-papular lesions (Figure 1B) were observed in the whole body, ranging from few millimeters to approximately 1.5cm in diameter, besides mild hepatosplenomegaly. Direct scale examination was positive for acarides, suggesting Sarcoptes scabiei. Biopsies were taken to rule out bacillary angiomatosis and tetracycline was prescribed orally in the dose of 2g/day.
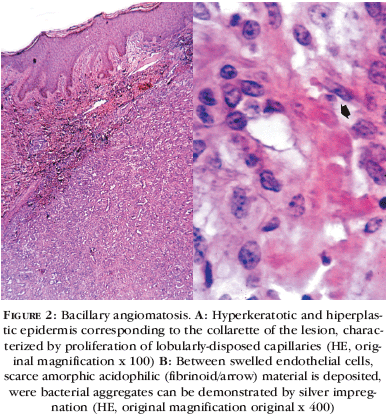
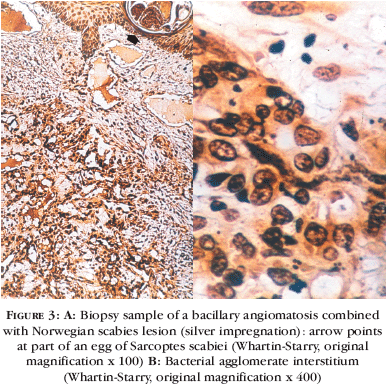
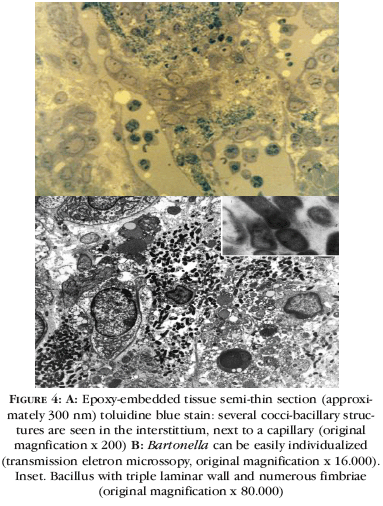
On histological examination in hematoxylin-eosin staining, thinned epidermis and numerous dermal capillaries with edematous-sometime atypical, endothelial cells were observed (Figures 2A and 2B). There were frequent endotelial mitoses and neutrophilic inflammatory infiltrate. Whartin-Starry staining showed several cocobacilli inside macrophages and free bacterial aggregates in interstitium (Figures 3A and 3B), better demonstrate in epoxy-embedded tissue sections staining with toluidine blue (Figura 4A). Tripe laminar walled bacilli were easily identified on transmission electron microscopy (Figure 4B). Improvement of fever and angiomatous lesions occurred after few days of treatment (Figures 5A and 5B). On the twentieth day of hospitalization, patient was discharged with clinical improvement of both bacillary angiomatosis and scabies. Ambulatorial follow-up was lost.
DISCUSSION
Bacillary angiomatosis is more frequent in immunocompromised patients, especially those with AIDS, but can also affect immunocompetent individuals. It is caused by Bartonella henselae and by B. quintana.2 The cat can serve as reservoir, but its role seems not to be obligatory.3 Lesions are characterized by capillary proliferation arranged in lobules. Cutaneous manifestations are the most frequent. Lesions can be papular, tumoral or nodular, multiple, single, and may be spread throughtout the entire body.4 The main differential diagnosis is Kaposi's sarcoma, with which it may coexist. Bacillary peliosis is characterized by angioproliferating lesions associated to capillary dilation, formation of cavernous spaces filled with blood in the liver, spleen or bone marrow.5,6
Prompt evolution without treatment is potentially lethal, but usually presents rapid response to antibiotic therapy with erythromycin or doxicyclin. Treatment can be prolonged, going from two to four months, in order to decrease the risk of relapse.7
Diagnosis is based on:
Direct blood examination: blood smear, stained by Giemsa method, is useful for diagnosing Oroya's fever and for follow-up of patients infected with B. bacilliformis.8
1. Febrile conditions, especially those associated with anemia, can have their etiologies defined with this diagnostic test.
2. Microbiological examination: isolation of different bartonella species usually requires prolonged time, from two to six weeks of culture, using enriched and recently prepared media, in a CO2-rich environment. They have been isolated from blood samples and tissue fragments, using both fresh and frozen material.1 Brenner et al., 1997, showed that freezing blood sample collected with anti-coagulants at -65ºC for 24h increased the number of seeded colony-forming units/mL, increasing sensitivity of the exam compared to direct plating in lysis tubes.9 Hemoculture is more sensitive than techniques of genetic detection. However, neither blood or tissue bartonella culture is routinily undertaken in most clinical pathology laboratories, both for the prolonged culture time and for the special conditions that are necessary for bacterial growth.
3. Serologic testing: immunoenzymatic, direct or indirect immunofluorescence assays for B. henselae and B. quintana have been the most widely used tests for the diagnosis of Bartonella spp. infection. Nevertheless, the utilization of these tests for acute phase infections has been a focus of debate in literature. For Baneth et al., 1996, it is likely that there is a great antigenic diversity among species, subspecies and strains of bartonellae.10 This can justify large differences found between serologic surveys from different regions, especially when commercially available test kits are used.11 There seems to be no cross reaction among strains of B. henselae, neither among species of bartonella.12 Velho et al., 2002, after evaluating the ultra-structure of a standard strain of B. henselae after passings in mice, postuleted that, besides genotypic differences, also phenotypic and possibly antigenic could justify the inconsistent serological findings in the diagnosis of infections by these bacteria.13
4. Molecular biology genetic detection techniques: these methods have the goal of aiding in the diagnosis of Bartonella spp. infection, and also in epidemiological studies of strains and species of this genus or in genotyping. Its identification is based on molecular characterization in tissue culture obtained from skin biopsy of .lymph node aspiration. Bacteria that grow from hemocultures of patients with endocarditis or fever can be used, as well as tissue samples or bacterial cultures stored in laboratories.1 Intergenic region 16S-23S rRNA, used by Jensen et al., 2000, was shown to be an appropriate target for containing regions with genetic sequences that are divergent enough for allowing species differentiation. The more colony-forming units/mL, the greater the sensitivity for genetic detection, reaching 100% of positivity when bacteremia levels were of 50-100 units/mL.14
5. Microscopy: it is of great usefulness for cases of bacillary angiomatosis, verruga peruana and cat-scratch disease. The former two are histologically indistinguishable. For experienced pathologists, diagnosis can be defined with basis on hematoxylin-eosin staining for most cases. It is characterized by endothelial proliferation with lobular arrangement, with more differentiated central vessels and less mature peripheral vessels, sometimes with their lumens unapparent. Lesions remind piogenic granuloma, both clinically and histologically. Acute and chronic inflammatory infiltrate, even in non-ulcerated lesions, with leukocytes and neutrophils inside the lobules, are findings that suggest the diagnosis. Inflammatory infiltrates are found surrounding bacterial aggregates, which have a fibrin-like aspect when staining. Cellular atypies can be found, especially in more solid lesions, with unapparent lumens and fusiform cells reminding Kaposi sarcoma.1 In cat-scratch disease, histological findings are similar in primary lesion and affected node, usually being constituted by granulomas with necrotic cores, surrounded by lymphocytes and histiocytes and with neutrophilic infiltrate, forming microabscesses. In the nodes, findings can be mistaken with those of Hodgkin's disease.15 Bacilli are seen within the initial neutrophilic inflammatory infiltrate and in recently formed granulomas, being rare within mature ones. The same findings are obtained in epoxy-embedded tissue, semi-thin sections staining with toluidine blue. Transmission electronic microscopy allows individualization of triple walled agents. Immunofluorescence and immunohistochemical techniques allow differentiation among bartonella species.1
There is no 'golden standard' for the diagnosis of bartonella infections. Serology is commercialized only for research, and genetic detection techniques are not available for clinical use in most medical centers in the world.
Sensitivity for a given method is a function of both clinical manifestation and sample origin.4,9
Histopathological examination is important for cases of bacillary angiomatosis, verruga peruana and cat-scratch disease.
In granulomatous cases of undefined etiology, this differential diagnosis should be considered, especially when microabscesses are present.
Actually, little is known about infection by these bacteria. However, it is possible that many idiopathic dermatoses are, in fact, caused by these agents. Man, cat, rat, mouse and rabbit can all live asymptomatically with bacteria from this genus. Their true importance for the development of diseases is unknown; it is certain, nevertheless, that bartonella infections are potentially lethal, which justifies larger efforts in their study.
REFERENCES
Received on January 28, 2003.
Approved by the Consultive Council and accepted for publication on October 17, 2003.
Conflict of interests: None
- 1. Velho PENF. Estudo das bartoneloses humanas e da Bartonellahenselae: infecção experimental, microbiologia e microscopia de luz e eletrônica de transmissão [dissertação]. Campinas (SP): Faculdade de Ciências Médicas da Universidade Estadual de Campinas; 2001.
- 2. Tappero JW, MohleBoetani J, Koehler JE, Swaminathan B, Berger TG, Leboit PE, et al. The epidemiology of bacillary angiomatosis and bacillary peliosis. J Am Med Assoc. 1993;269:770-5.
- 3. Koelhler JE, Glaser CA, Tappero JW. Rochalimaea henselae infecion: a new zoonosis with the domestic cat as reservoir. J Am Med Assoc. 1994;71:531-5.
- 4. Levell NJ, Bewley AP, Chopra S, Churchill D, French P, Miller R, et al. Bacillary angiomatosis with cutaneous and oral lesions in an HIV-infected patient from the UK. Brit J Dermatol. 1995;132:113-5.
- 5. Loutit JS. Bartonella infections. Cur Clin Top Infec Dis. 1997;17:269-90.
- 6. Raoult D. Infections humaines à Bartonella Presse Med. 1999;28:429-34.
- 7. Maurin M, Raoult D. Bartonella (Rochalimaea) quintana infections. Clin Microbiol Rev. 1996;9:273-92.
- 8. Garcia-Caceres U, Garcia FU. Bartonellosis: an immunodepressive disease and the life of Daniel Alcides Carrión. Am J Clin Pathol. 1991;95(Suppl 1):S58-66.
- 9. Brenner SA, Rooney JA, Manzewitsch P, Regnery RL. Isolation of Bartonella (Rochalimae) henselae: effects of methods of blood collection and handling. J Clin Microbiol. 1997;35:544-7.
- 10. Baneth, G, Kordick DL, Hergarty BC, Breitschwerdt EB. Comparative seroreactivity to Bartonella henselae and Bartonella quintana among cats from Israel and North Carolina. Vet Microbiol. 1996;50:95-103.
- 11. Dehio C, Sander A. Bartonella as emerging pathogens. Trends Microbiol. 1999;7:226-8.
- 12. Regnery RL, Rooney JA, Johnson AM, Nesby SL, Manzewitsch P, Beaver K, et al. Experimentally induced Bartonella henselae infections followed by challenge exposure and antimicrobial therapy in cats. Am J Vet Res. 1996;57:1714-9.
- 13. Velho PENF, Moraes AM, Uthida-Tanaka AM, Cintra ML, Gilioli R. Ultrastructural changes in a standard strain of Bartonella henselae after passages through BALB/cAn mice. Ultrastruct Pathol. 2002;26:161-9.
- 14. Jensen WA, Fall MZ, Rooney J, Kordick DL, Breitschwerdt EB. Rapid identification and differentiation of Bartonella species using a single-step PCR assay. J Clin Microbiol. 2000;38:1717-22.
- 15. Guccion JG, Gilbert CL, Ortega LG, Hadfield TL. Cat scratch disease and acquired immunodeficiency disease: diagnosis by transmission electron microscopy. Ultrastruct Pathol. 1996;20:195-202.
Publication Dates
-
Publication in this collection
21 June 2007 -
Date of issue
Aug 2006
History
-
Accepted
17 Oct 2003 -
Received
28 Jan 2003