Abstracts
Botryomycosis is a chronic, granulomatous, suppurative bacterial infection involving the skin and subcutaneous tissues. The skin lesion is characterized by multiple fistulae draining purulent secretion with white-yellowish grains PAS positive. The main etiologic agent is Staphylococcus aureus and other less frequent pathogens are Pseudomonas aeruginosa and Escherichia coli, isolated or associated. The authors report a case of this disease with exuberant lesions on the right leg and increased limb volume, fibrous consistency and multiple fistula orifices draining white-yellowish grains. Resolution was achieved with sulphamethoxazole-trimethoprim.
Bacteria; Granuloma; Staphylococcus aureus
Botriomicose é infecção bacteriana crônica, granulomatosa, supurativa, que afeta a pele e os tecidos subcutâneos. Caracteriza-se por lesão cutânea com múltiplas fístulas, que drena secreção purulenta, na qual se encontram grãos branco-amarelados, PAS-positivos. O principal agente etiológico é o Staphylococcus aureus, e outros germes menos frequentes são Pseudomonas aeruginosa, Escherichia coli, isolados ou associados. Os autores relatam um caso da doença, com lesões exuberantes de consistência fibrosa e múltiplos orifícios fistulosos dos quais drenavam grãos branco-amarelados, localizadas na perna direita e provocando aumento de volume do membro, que tiveram resolução com sulfametoxazol- trimetoprim.
Bactérias; Granuloma; Staphylococcus aureus
CASE REPORT
Cutaneous botryomycosis case report
Weber Soares CoelhoI; Lucia Martins DinizII; João Basílio de Souza FilhoIII
IDermatologist from the Brazilian Society of Dermatology. Master's degree student in Internal Medicine from the Faculdade de Medicina de Ribeirão Preto da Universidade de São Paulo (USP) - Sao Paulo (SP), Brazil
IIFull Professor of the Medical Residency Service in Dermatology, Universidade Federal do Espírito Santo (UFES)- Vitoria (ES), Brazil
IIIFull Professor of the Service of Medical Residency and Graduate Courses in Dermatology, Santa Casa de Misericórdia de Vitória (EMESCAM) - Vitoria (ES), Brazil
Mailing Address Mailing Address: Weber Soares Coelho Rua José Saretta, n. 155, bairro Nova Aliança CEP: 14026- 593 - Ribeirão Preto - SP Tel./fax: (16) 81 21 38 06 E-mail: dr.webersoares@yahoo.com.br
ABSTRACT
Botryomycosis is a chronic, granulomatous, suppurative bacterial infection involving the skin and subcutaneous tissues. The skin lesion is characterized by multiple fistulae draining purulent secretion with white-yellowish grains PAS positive. The main etiologic agent is Staphylococcus aureus and other less frequent pathogens are Pseudomonas aeruginosa and Escherichia coli, isolated or associated. The authors report a case of this disease with exuberant lesions on the right leg and increased limb volume, fibrous consistency and multiple fistula orifices draining white-yellowish grains. Resolution was achieved with sulphamethoxazole-trimethoprim.
Keywords: Bacteria; Granuloma; Staphylococcus aureus
INTRODUCTION
Botryomycosis is a rare, chronic, granulomatous, suppurative bacterial infection of the skin and organs, characterized by release of granules, such as in mycetoma and actinomycosis.1
The skin lesions may be single or multiple and pleomorphic, similar to cysts, abscesses, fistulas, nodules, plaques or ulcers. They are usually located on areas exposed to traumas (head, arms, legs) and genital regions.1,2 Muscles, aponeuroses, tendons and bones may be also affected. The general swollen and suppurative aspect of the lesion suggests local inoculation of a foreign body as the initial cause of infection and perpetuation of the disease.1
It was first considered a fungal infection, thus the term botryomycosis (from Greek, botrys bunch of grapes, mycosis fungal origin). Later, the bacterial etiology was discovered and other terms were reported, including actinophytosis, staphylococcal actinophytosis, bacterial pseudomycosis and granular bacteriosis.2,3
It was described by Bollinger, in 1870, through observation of granulomatous lesions as complication after horse castration. In 1884, Rivolta associated the disease to fungal etiology and called it botryomycosis. Opie described the first case in humans in 1913. Magrou, in 1919, reported four cases and the isolated agent was Staphylococcus aureus.2,3 Winslow, in 1959, reviewed the disease and assessed 46 cases in humans, classifying it as cutaneous and visceral forms.3,4
The most frequent etiological agent is Staphylococcus aureus (40%), followed by Pseudomonas sp (20%).3 Other microorganisms already reported are Escherichia coli, Proteus vulgaris, Bacillus spp, Actinobacillus lignieresii, etc.1,2
The pathogenesis of the disease has not been well established. It may be related to low virulence of agents, large local bacterial inoculum, change in specific cellular immunity (decreased number of T lymphocytes, like in agammaglobulinemia, aplastic anemia, agranulocytosis and AIDS), or in humoral immune response (reduced IgA or increased IgE levels).3,5
The granule formation process remains undefined, and alteration in the host-parasite balance has been postulated, such as in Splendore-Hoeppli phenomenon, considered as an antigen-antibody reaction in which the granules are involved by eosinophilic matrix.3,6
The predisposing factors associated to the disease are skin trauma, complicated postoperative period, diabetes mellitus, lung diseases, corticotherapy in high doses or for a prolonged period, alcohol abuse and cystic fibrosis.1,3,7 Less frequently, malnutrition, glomerulonephritis, AIDS, asthma, Job syndrome and follicular mucinosis are associated to the condition.3,7,8
The treatment of botryomycosis should be based on identification of the etiological agent by means of direct examination, culture and antibiogram of secretion of the lesion. Antibiotic therapy is often used for weeks and, in some cases, surgical excision or drainage of lesions are required.3,9
CLINICAL CASE
A 51 year-old male patient, driver, living in the State of Espirito Santo. Four years ago he presented a gummy lesion that evolved to an extensive tumor, with fistulas and spontaneous drainage of purulent material, in which granules and some areas of cicatritial retraction were observed on the right leg. During progression of the skin condition, he used benzhatine penicillin 1200000 units/week for three months, cephalexin 2 grams/day for 14 days, repeating after 30 days and amoxicillin 1.5 grams/day for three weeks, with no improvement. Hence he had received no medication for six months.
Upon dermatological examination, he presented nodules with multiple fistulous orifices and some cicatritial areas of fibrotic consistency, on the right leg (Figure 1), local suppuration and drainage of white-yellowish granules. Absence of local or systemic inflammatory signs. The patient suffered from no systemic diseases associated to the skin condition.
The following tests were ordered based on the presumptive diagnoses of botryomycosis and exogenous actinomycosis: complete blood count, fasting glucose, urea, creatinine, TGO, TGP, antiHIV serology, chest radiograph, right leg radiograph and complete abdominal ultrasonography. Purulent secretion was collected from the fistulas for culture using the media chocolate agar, blood agar and Sabouraud agar, and antibiograms. Incisional biopsy was performed. The complementary tests were within normal range. Staphylococcus aureus grew in chocolate and blood agar and no colony grew in Sabouraud agar.
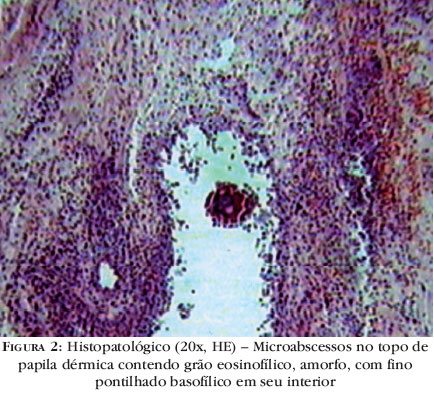
The histopathological examination showed moderately acanthotic epidermis, dermis with discreet lymphoplasmocytary infiltrate, with rare polymorphonuclear cells and microabscesses, with eosinophilic and amorphous granules, with fine basophilic stippling in its interior, in hematoxylin-eosin staining (Figure 2) and Grocott method, associated to surrounding radiated intense eosinophilic substance, denominated Splendore-Hoeppli phenomenon (Figure 3).
Pathological examination associated to isolation of Staphylococcus aureus in culture for bacteria confirmed the diagnosis of cutaneous botryomycosis.
The antibiogram analysis showed resistance of the agent to the antibiotics cephalosporin, penicillin, quinolone and sensitivity to sulphametoxazole-trimethoprim, which was prescribed at 800mg/160mg, twice a day, per oris, for eight weeks, with regression of the dermatological condition and scarring with fibrosis.
DISCUSSION
Cutaneous botryomycosis is a rare disease, scarcely described in the international literature.1
The decisive factors in the pathogenesis of botryomycosis remain unclear. In a case reported in Brazil, cellular immunity demonstrated deficient response assessing by PPD skin test, streptokinase-streptodornase, oidiomycin and vaccinia. 5 The increased IgG, IgM and gammaglobulin levels suggest a possible change in humoral defense of the host.1,5 Hence, existence of immunological defect was postulated, or a deficient tissue response to tackle Staphylococcus. On the other hand, there is eosinofilic amorphous material involving the microorganisms and hindering action of antimicrobial agents.1,3,5
In the case reported, intense eosinofilic material around a botryomycotic granule was observed, in crown- or radiated shape, PAS positive; it is not pathognomonic of the disease, but has been frequently reported in the international literature.7
The main differential diagnoses of botryomycosis are exogenous actinomycosis and eumycetoma, which clinically present enlarged affected area, fistulas and drainage of granules, differing as to etiology, location and consistency of the lesions.1-10
The assessment of size, consistency, color and shape of the granule may suggest the etiological agent. However, since multiple agents may determine formation of granules, varying from fungi to bacteria, direct mycological examinations, cultures in media for fungi to bacteria, and histopathology of the material with special stainings are essential.1,5
The eumycotic granules contain hyphae in their interior and large and globous edematous cells, measuring up to 15 κm in the border; the actinomycotic granules have fine and ramified filaments in the periphery, diameter varying from 0.5 to 1 milimicron; the granule of botryomycosis present as coccoidal or bacillary structures with no fine filaments in their interior.1,3,5 In this case, there was drainage of granules, and the histopathological examination showed coccoidal structures in the interior and the culture was positive for Staphylococcus aureus.
The authors found 140 cases published on botryomycosis, and 28 articles addressed the cutaneous form of the disease. Three cases of cutaneous botryomycosis were published in Brazil.
As to differential diagnoses of botryomycosis, the authors found 124 published cases of cutaneous exogenous actinomycosis and 75 cases of eumycetoma on the skin. In face of these data, cutaneous botryomycosis represented the skin condition less frequent out of three diagnoses.
In face of a clinical picture similar to that presented by our patient, the authors draw attention to the need to make laboratory evaluation for accurate diagnosis and for perfect management of the case. It is a rare condition, thus justifying publication of the case.
REFERENCES
Conflict of interest: None
Financial funding: None
How to cite this article: Coelho WS, Diniz LM, Souza Filho JB. Botriomicose cutânea Relato de caso. An Bras Dermatol. 2009;84(4):396-9.
- 1. Fernandes NC, Maceira JP, Knackfuss IG, Fernandes N. Botriomicose cutânea. An Bras Dermatol. 2002;77:65-70
- 2. Mehregan DA, Su WPD, Anhalt JP. Cutaneous botryomycosis. J Am Acad Dermatol. 1991; 24:393-6
- 3. Bonifaz A, Carrasco E. Botryomycosis. Int J Dermatol. 1996;35:381-8
- 4. Oliveira ZNP, Cuce LC, Salebian A. Botriomicose. Med Cut ILA.1986;14:49-54
- 5. Towersey L, Estrella RR, Mendonça AMN, Conceição MM, Arnez R. Botriomicose em paciente com testes sugestivos de função imunológica alterada. An Bras Dermatol. 1986;61:49-52
- 6. Conti-Diaz IA, Almeida EA, Rondan M, Algorta G, Otero M. Cutaneous botryomycosis. Report of two first uruguayan cases. Rev Inst Med Trop Sao Paulo.1996;38:375-8
- 7. de Vries HJ, van Noesel CJ, Hoekzema R, Hulsebosch HJ. Botryomycosis in an HIV-positive subject. J Eur Acad Dermatol Venereol. 2003;17:87-90
- 8. Harman RR, English MP, Halford M, Saihan EM, Greenham LW. Botryomycosis: a complication of extensive follicular mucinosis. Br J Dermatol. 1980;102:215-21
- 9. Brunken RC, Linchon-Chao N, van der Broek H. Immunologic abnormalities in botryomycosis. J Am Acad Dermatol. 1983;9:428-34
- 10. Simantov A, Chosidow O, Fraitaq S, Feuilhade de Chauvin M, Prost C, Ravisse P, et al. Disseminated cutaneous botryomycosis--an unexpected diagnosis after 20-years' duration. Clin Exp Dermatol. 1994;19:259-61
Publication Dates
-
Publication in this collection
19 Oct 2009 -
Date of issue
Aug 2009
History
-
Received
01 Feb 2007 -
Accepted
18 Dec 2008