Abstracts
Staphylococcus aureus is responsible for a broad variety of infectious diseases. The main concern is about methicillin-resistant isolates (MRSA), which are usually limited to hospitals. In recent years, community associated or acquired MRSA infections (CA-MRSA) have been frequently reported and emerged in the world. Some phenotypic and genotypic characteristics are distinct between hospital and community infection. Currently, there is reduced sensibility profile to different antimicrobials, reason why it is necessary to issue an alert to healthcare professionals, dermatologists in particular, about the importance of knowing the differences between the infections, preventing wrong and unsuccessful empirical therapy.
Bacterial Infections; Methicillin; Staphylococcus aureus
Staphylococcus aureus é uma bactéria responsável por uma ampla variedade de enfermidades infecciosas. A grande preocupação está relacionada, principalmente, com os isolados resistentes à meticilina (MRSA), que, tradicionalmente, estavam limitados aos hospitais. Nos últimos anos, infecções causadas por MRSA associadas ou adquiridas na comunidade (CA-MRSA) têm sido relatadas com frequência crescente em todo o mundo. Algumas características fenotípicas e genéticas são distintas entre a forma de infecção hospitalar e a comunitária. Atualmente, verifica-se um perfil de sensibilidade reduzido para diferentes antimicrobianos; sendo assim faz-se necessário um alerta aos profissionais da saúde, particularmente aos dermatologistas, para a importância da distinção entre as formas de infecções, evitando uma terapia empírica incorreta e sem sucesso.
Infecções bacterianas; Meticilina; Staphylococcus aureus
REVIEW
Methicillin-resistant Staphylococcus aureus: emerging community dissemination
Luciane Cristina GelattiI; Renan Rangel BonamigoII; Ana Paula BeckerIII; Pedro Alves dAzevedoIV
ISpecialist in Clinical Cytology, master studies under course in Pathology, Universidade Federal de Ciências da Saúde de Porto Alegre (UFCSPA) - Porto Alegre (RS), Brazil
IISpecialist in Dermatology, Brazilian Society of Dermatology, Ph.D. in Medical Sciences, Universidade Federal do Rio Grande do Sul (UFRGS), associate professor and Head of the Service Dermatology, Universidade Federal de Ciências da Saúde de Porto Alegre (UFCSPA) - Porto Alegre (RS), Brazil
IIIBiomedical professional, master studies under course in Medical Sciences, Universidade Federal de Ciências da Saúde de Porto Alegre (UFCSPA) - Porto Alegre (RS), Brazil
IVPh.D., Associate Professor, Department of Microbiology and Parasitology, Universidade Federal de Ciências da Saúde de Porto Alegre (UFCSPA) - Porto Alegre (RS), Brazil
Mailing Address
ABSTRACT
Staphylococcus aureus is responsible for a broad variety of infectious diseases. The main concern is about methicillin-resistant isolates (MRSA), which are usually limited to hospitals. In recent years, community associated or acquired MRSA infections (CA-MRSA) have been frequently reported and emerged in the world. Some phenotypic and genotypic characteristics are distinct between hospital and community infection. Currently, there is reduced sensibility profile to different antimicrobials, reason why it is necessary to issue an alert to healthcare professionals, dermatologists in particular, about the importance of knowing the differences between the infections, preventing wrong and unsuccessful empirical therapy.
Keywords: Bacterial Infections; Methicillin; Staphylococcus aureus
INTRODUCTION
Staphylococcus aureus can colonize the natural flora, especially of the skin, and it can become pathogenic in conditions such as the break of skin barrier or reduction of immunity. Traumas that may impair the integrity of skin barrier are the main cause of change in the behavior of this microorganism, as the most common etiological agent in skin infections 1,2,3. It is responsible for a broad variety of infections, such as skin and subcutaneous infections, post-surgical infections, osteomyelitis, pneumonias, abscesses, endocarditis and bacteremia 4. It is one of the most common hospital-related infection, as well as the community infections that may present high rates of mortality and morbidity.
The antimicrobial therapy for infections caused by this microorganism was simple at first. The first time that an antimicrobial agent was clinically used was against a sample of Staphylococcus aureus, after the discovery of penicillin, which worked fine up to the 1960s, when resistant isolates to this antimicrobial started to appear. To overcome this problem, a synthetic beta-lactam drug, methicillin, which was resistant to the beta-lactam action that Staphylococcus aureus produced.
However, right after the creation of methicillin, there were reports of resistant samples to this antimicrobial agent as well, in addition to the expression of multi-resistance. These strains were named MRSA (Methicillin-resistant Staphylococcus aureus) and are resistant to all beta-lactam antimicrobials.4
Beta-lactam antimicrobials bind to proteins that participate in the synthesis of cell wall, named Penicillin-Binding Proteins, preventing the formation of cell wall, releasing bacterial lysis. The resistance mechanism of methicillin is related with the development of additional PBP, PBP2a, which is fully functional, but has no affinity for beta-lactam antimicrobials. The codification of these new PBPs, transforming these pathogens as resistant to oxacillin, is related with the acquisition of mecA gene, which is part of a mobile genetic element detected in MRSA isolates. This gene is an integral part of a element genomically named "staphylococcal chromosome cassette MEC (SCCmed)5.
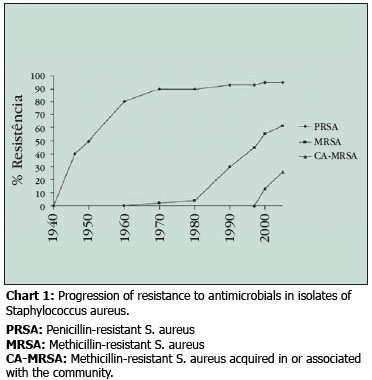
Traditionally, MRSA infections were limited to hospitals (HA-MRSA), but in recent years infections associated or acquired in the community (CA-MRSA) are been increasingly documented all over the world (Chart 1) 6. Epidemiologically, clinical isolates of MRSA are defined as CA-MRSA if collected from outpatients, or collected within up to 48 hours after hospital admission. Risk factors such as: recent hospitalization, surgical procedures, use of venous catheter, or long-term intravascular or cutaneous devices, and long-term stay in nursing home should be excluded 7.
COMMUNITY-ACQUIRED METHICILLIN-RESISTANT S. AUREUS
In addition to representing one of the main pathogens associated with hospital infection, S. aureus is also one of the main isolated agents in patients with skin and subcutaneous infections acquired in the community, including folliculitis, impetigo, cellulitis and erysipela 8. Differently from HA-MRSA, that has the mobile genetic element, named SCCmec of types I, II and III, CA-MRSA carry preferably SCCmec types IV and sometimes type V. This type of chromosome cassette is smaller than the other types and do not have coupled genes that codify the resistance to other non-beta lactam antimicrobials. Thus, in general, CA-MRSA is susceptible to most non-beta lactam antimicrobial agents 9. This phenotypic characteristic is expressed most of the times in the antibiogram that presents resistance only to oxacillin or cefoxitine disk, which are markers of beta-lactam resistance (Figure 1).
Other significant genetic characteristics associated with CA-MRSA strains are the genes of cytotoxin Panton-Valentine Leucocidin (PVL). PVL is codified by genes lukF and lukS and its presence in isolates of S. aureus is associated with tissue necrosis and destruction of leukocytes, by the formation of pores on the cell membrane 10. The presence of this endotoxin may be noticed by investigating specific genes, by polymerase chain reaction (PCR) (Figure 2). In a study carried out in the United States, 98% of the isolates presented PVL codifying genes 11. Infections frequently associated with these strains are furuncles, folliculitis, impetigo, septic arthritis, necrotizing fasciitis, pneumonia and severe sepsis 12,13,14.
Oral antimicrobials that are normally active against staphylococcus infections by CA-MRSA include tetracyclines, trimethoprim/ sulfamethoxazole, fluoroquinolones and clindamycin. For potentially severe clinical forms, there are linezolide, tigecycline, daptomycin, teociplamine and vancomycin. Clindamycin is a frequently used antimicrobial agent that is used as therapeutic strategy in managing skin and tissue staphylococcal infections because it has good penetration and potential antitoxin effect. However, most of the concern of this type of empirical therapy lies in the inducible resistance of clindamycin, which may be presented in the antibiogram as a resistance to erythromycin and false sensitivity to clindamycin. This phenotype of resistance may be identified by the microbiology laboratory through the susceptibility test with erythromycin and clindamycin disks, the disk test 15-16.
The emergency of CA-MRSA isolates is considered of concern given that the empirical treatment of a variety of skin and soft tissue community infections and even pneumonias is made with beta-lactam antimicrobials 17.
In a study carried out in the USA it was shown that 80% of the patients with skin and soft tissue infections received empirical treatment; out of the total, 57% did not respond to treatment because the microorganism presented resistance. These findings suggested the need to reconsider the choice of empirical treatments for skin and soft tissue infections where MRSA is prevalent in the community 11. In a teaching hospital in California, USA, a study showed the presence of 87% of MRSA among the isolated S. aureus of patients with dermatological infections seen in the emergency department. This study advocated for drainage of cutaneous abscesses as preferential management approach, as opposed to the use of antimicrobials 18.
There is controversy in choosing the treatment and even the antimicrobial agents used to manage skin infections caused by S. aureus. Cephalosporins are among the most used ones.
CLINICAL ASPECTS OF CA-MRSA INFECTION
The first report of a form of CA-MRSA was made in 1993 in Australia, detected in a local indigenous population 19 and in 2002, it gained attention after an outbreak of skin infections in Los Angeles athletes 20. In 2003, in Missouri, USA, in a study carried out among Professional American football players, the presence of MRSA on the skin lesions was present in 5 out of 58 players 21.
In South America, infections caused by MRSA in non-hospitalized patients were observed initially in 2001 in Uruguay. At first, there were occasional causes, but as a result of the subsequent increase in number of skin infections in patients in the community caused by this pathogen, it was characterized as an outbreak 22. In Brazil, the first isolated characterized as CA-MRSA had clinical presentation of furunculosis and septic arthritis 17.
In a multicenter study carried out in Argentina there was high prevalence of methicillin-resistance SA isolated in infections in children coming from the community. Among the 447 samples analyzed in the years 2006 and 2007, 281 complied with the criteria set for CA-MRSA. This investigation showed that 62% of isolates came from skin and soft part infections. The study emphasized the importance of having an urgent review of the empirical antimicrobial management approach to treat skin lesions in children coming from the community 23.
The elucidation of infections caused by CA-MRSA strains is of special interest to dermatologists, given that there is high likelihood of having skin and soft tissue infections. In a comparative study between CA-MRSA and HA-MRSA it was shown that skin and subcutaneous tissue infections are more prevalent in patients isolated with CA-MRSA (75%) than those by HA-MRSA (37%) 9.
Epidemiologically, infections related with CA-MRSA strains are normally associated with children, young people and healthy people, especially those who live in agglomerates or who have close physical contact. Most literature reports related collective sport athletes, military service members, inmates, injecting drug users, homeless, and day care center children, who are the subjects with increased risk of developing CA-MRSA infections. The typical presentation is in an young athlete with abscess and surrounding cellulitis which is probably caused by physical contact, skin damage and sharing of contaminated equipment.
Infections such as necrotizing fasciitis caused by Staphylococcus aureus are uncommon, but the increasing number of cases caused by CA-MRSA is a concern. In a US study from 2005, 14 out of 843 patients had necrotizing fasciitis during a period of 15 months. The main risk factor associated with these cases was the use of injectable drugs and all S. aureus isolates showed similar genotype and the presence of PVL codifying genes 13. There are no prevalence studies in Brazil yet, but data from other countries, such as the United States, show that there is great variation of frequency related with geography. In a study carried out in 11 emergency departments from different US cities during August 2004, the rate of global prevalence was 59%, ranging from 15 to 74% 11.
TYPES OF CLONES OF CIRCULATING CA-MRSA
To present, molecular typing has enabled updated data, giving rise to molecular epidemiology. In the case of S. aureus, after analyzing a broad variety of strains that circulate in different regions of the world and in different periods, it was found that MRSA strains have a preserved clonal structure and count on a reduced number of clones that have global dissemination capability 6.
The first isolates of CA-MRSA were detected in the West of Australia, in the end of 1980, causing infection in the local indigenous population, reason why they were named WA-MRSA (Western Australia MRSA) 19. After that, two other CA-MRSA lines emerged in Australia and New Zealand: clone Queensland and Oceania Southwest Pacific" (SWP), also known as OSPC (Oceania South Pacific Clone). 24,25 The USA clones were described for the first time in the United States clone USA 400 was reported in the center-west region of the United States in neonatal units and in puerperal infections. USA 300 was reported in regions of the United States, especially among American football players and inmates 26. In a study by Huang et al. it was demonstrated that isolates of CA-MRSA in skin and soft tissue infections in California amounted to 87% clones of USA 300 27. Recently, there was a report of about 50% MRSA collected from skin and soft tissue infections in the USA, beings USA 300 clones 11 and, less frequently, USA 400 clones 28.
In Brazil, the first isolates characterized as CA-MRSA were similar to clone OSPC and came from one single city in the south (Porto Alegre) 17. Later, a study showed the presence of the same clone in isolates from the Southeast region (Rio de Janeiro) 29. These two geographical areas are separated by about 1,500 km, showing the dissemination of OSPC clone into the different regions of the country. The study demonstrated also the presence of other SCCmec IV lines which is classically community-acquired, such as USA300 and USA400 recovered from skin infections and pneumonia.
However, the origin of CA-MRSA strains is subject to debates. One of the possibilities is the wild descendant aspect of hospital strains, which occurred due to vertical transformation. However, in a study in which hospital strains and community strains were compared, there was no correlation between HA-MRSA and CA-MRSA. Another possibility is that community strains start as a consequence of vertical transference of genes resistant to methicillin. Horizontal transference is enabled by the complex of ccr gene that is contained in the SCCmec, which codifies recombinases responsible for their mobility. It is thought that this transfer occurs very little, and that CA-MRSA strains are consequence of one of these rare events of gene MEC transfer, from a susceptible donor to a recipient. All these hypotheses are still under analysis 6.
CLOSING REMARKS
Infections caused by MRSA are no longer a problem associated only with hospital environment and have become an emerging problem in the community. To present, risk factors associated with community MRSA infections are not completely defined and the inflow of outpatients to healthcare units may have an impact on the intra-hospital epidemiology of this pathogen. It is possible that patients who are colonized or infected by MRSA SCCmec IV remain free from the measures adopted to control the dissemination of MRSA, leading to the possibility of intra-hospital dissemination of this strain. Conversely, the patients that colonized with MRSA SCCmec IV may undergo surgeries or procedures where prophylaxis for MRSA may be considered, increasing the risk of infections in these patients.
It is suggested that in outpatients in which MRSA is isolated and they have susceptibility to many antimicrobial agents, we should consider for diagnostic purposes as a possible CA-MRSA infection. Genetic investigation including gene SCCmec typing, PFGE (pulsed-field gel electrophoresis), multiloci sequence typing for PVL and MLST, may be used to better characterize these bacteria.
The shortage of prevalence studies is a limiting factor in the knowledge about local epidemiology related with CM-MRSA. An important aspect that can facilitate the confirmation of these circulating strains is laboratory cultivation of skin and soft tissue lesions in primary care centers, such as the emergency departments in hospitals and dermatology centers. Thus, the identification of the microorganism and the performance of a susceptibility test may lead to appropriate treatment and guide the control measures against this pathogen.
REFERENCES
- 1. Altemeier WA, Culbertson WR, Veto M. Prophylactic antibiotic therapy. AMA Arch Surg. 1955;71:2-6
- 2. Barraviera B. Estudo clínico das estafilococcias. J Bras Med. 1994;67:160-92
- 3. Ferreira MS, Gonçalves EG, Assis VP. Como diagnosticar e tratar infecções estafilocócicas . Rev Bras Med. 1985;42:179-89
- 4. Lowy FD. Staphylococcus aureus infections. N Engl J Med. 1998;339:520-32
- 5. Chambers HF. Methicillin resistance in Staphylococci: molecular and biochemical basis and clinical implications. Clin Microbiol Rev. 1997;10:781-91
- 6. Bustos-Martínez JA, Hamdan-Partida A, Gutiérrez- -Cárdenas M. Staphylococcus aureus: la reemergencia de un patógeno en la comunidad. Rev Biomed. 2006;17:287-305
- 7. Salgado CD, Farr BM, Calfee DP. Community-acquired methicillin-resistant Staphylococcus aureus: a meta-analysis of prevalence and risk factors. Clin Infect Dis. 2003;36:131-9
- 8. Forbes BA, Sahm DF, Weissfeld AS. Staphylococcus, Micrococcus, and similar organisms. In: Forbes BA, Sahm DF, Weissfeld AS, editors. Bailey & Scott's Diagnostic Microbiology. St Louis: Mosby; 1998. p.607-18
- 9. Naimi TS, LeDell KH, Como-Sabetti K, Borchardt SM, Boxrud DJ, Etienne J, et al Comparison of communityand health care-associated methicillin-resistant Staphylococcus aureus infection. JAMA. 2003; 290: 2976-84
- 10. Dufour P, Gillet Y, Bes M, Lina G, Vandenesch F, Floret D, et al Community-acquired methicillin-resistant infections in France: emergence of a single clone that produces Panton-Valentine leukocidin. Clin Infect Dis. 2002;35:819-24
- 11. Moran GJ, Krishnadasan A, Gorwitz RJ, Fosheim GE, Mcdougal LK, Carey RB, et al Methicillin-resistant S. aureus infections among patients in the emergency department. N Engl J Med. 2006; 355:666-74
- 12. Lina G, Piemont Y, Godail-Gamot F, Bes M, Peter MO, Gauduchon V, et al Involvement of Panton-Valentine leukocidin-producing Staphylococcus aureus in primary skin infections and pneumonia. Clin Infect Dis. 1999;29:1128-32
- 13. Miller LG, Perdreau-Remington F, Rieg G, Mehdi S, Perlroth J, Bayer AS, et al Necrotizing fasciitis caused by community-associated methicillin-resistant Staphylococcus aureus in Los Angeles. N Engl J Med. 2005;352:1445-53
- 14. Gillet Y, Issartel B, Vanhems P, Fournet JC, Lina G, Bes M, et al Association between Staphylococcus aureus strains carrying gene for Panton-Valentine leukocidin and highly lethal necrotizing pneumonia in young immunocompetent patients. Lancet. 2002;359:753-9
- 15. Chambers HF. MRSA: the San Francisco experience. Program and abstracts of the 45th Interscience Conference on Antimicrobial Agents and Chemotherapy; December 16-19, 2005; Washington, DC. Abstract 1324
- 16. Patel M, Waites KB, Moser SA, Cloud GA, Hoesley CJ. Prevalence of inducible clindamycin resistance among community and hospital-associated Staphylococcus aureus isolates. J Clin Microbiol. 2006;44:2481-4
- 17. Ribeiro A, Dias C, Silva-Carvalho MC, Berquo L, Ferreira FA, Santos RN, et al First report of infection with community-acquired methicillin-resistant Staphylococcus aureus in South America. J Clin Microbiol. 2005;43:1985-88
- 18. Rajendran PM, Young D, Maurer T, Chambers H, Perdreau-Remington F, Ro P, et al Randomized, double-blind, placebo-controlled trial of Cephalexin for treatment of uncomplicated skin abscesses in a population at risk for community-acquired methicillinresistant Staphylococcus aureus infection. Antimicrob Agents Chemother. 2007;51:4044-48.
- 19. Udo EE, Pearman JW, Grubb WB. Genetic analysis of community isolates of methicillin-resistant Staphylococcus aureus in Western Australia. J Hosp Infect. 1993; 25:97-108.
-
20Centers of Disease Control and Prevention. Outbreaks of community-associated methicillin-resistant Staphylococcus aureus skin infections – Los Angeles County, California, 2002-3. MMWR Morb Mortal Wkly Rep. 2003;52:88
- 21. Kazakova SV, Hageman JC, Matava M, Srinivasan A, Phelan L, Garfinkel B, et al A clone of methicillin-resistant Staphylococcus aureus among football players. N Engl J Med. 2005; 352:468-475
- 22. Ma XX, Galiana A, Pedreira W, Mowszowicz M, Chrisphersen I, Machiavello S, et alCommunityacquired methicillin-resistant Staphylococcus aureus, Uruguay. Emerg Infect Dis. 2005;11:973-976
- 23. Paganini H, Della Latta MP, Opet BM, Ezcurra G, Uranga M, Aguirre C, et al Estudio multicéntrico sobre las infecciones pediátricas por Staphylococcus aureus meticilino-resistente provenientes de la comunidad en la Argentina. Arc Argen Pediatr. 2008;106:397-403.
- 24. Nimmo GR, Schooneveldt J, OKane G, McCall B, Vickery A. Community acquisition of gentamicin-sensitive methicillin-resistant Staphylococcus aureus in Southeast Queensland, Australia. J Clin Microbiol. 2000;38:3926-31.
- 25. Munckhof WJ, Schooneveldt J, Coombs GW, Hoare J, Nimmo GR . Emergence of community-acquired methicillin-resistant Staphylococcus aureus (MRSA) infection in Queensland, Australia. Int Infect Dis. 2003;7:259-67.
- 26. Bratu S, Landmand D, Gupta J, Irehan M, Panwar M, Quale J. A population-based study examining the emergente of community-associated methicillin-resistant Staphylococcus aureus USA300 in New York City. Ann Clin Microbiol Antimicrob. 2006;5:29.
- 27. Huang H, Flynn NM, King JH, Monchaud C, Morita M, Cohen SH. Comparisons of community-associated methicillin resistant Staphylococcus aureus (MRSA) and hospital-associated MRSA infections in Sacramento, California. J Clin Microbiol.2006;44:2423-27.
- 28. Maree CL, Daum RS, Boyle-Vavra S, Matayoshi K, Miller LG. Community-associated methicillin-resistant Staphylococcus aureus isolates causing healthcareassociated infections. Emerg Infect Dis. 2007;13:236-42.
- 29. Ribeiro A, Coronado AZ, Silva-Carvalho MC, Ferreira- Carvalho BT, Dias C, Rozenbaum R, et al Detection and characterization of international community-acquired infections by methicillin-resistant Staphylococcus aureus clones in Rio de Janeiro and Porto Alegre cities causing both community-and hospital-associated diseases. Diagn Microbiol Infect Dis. 2007;59:339-45.
Publication Dates
-
Publication in this collection
07 Jan 2010 -
Date of issue
Oct 2009