Abstracts
Skin biopsy is a common procedure in Dermatology practice. The cost-benefit ratio, though, can be unfavorable if attention is not paid to specific details in performing a skin biopsy. For example, proper selection of anatomical site and best lesion to perform a biopsy are very important. The present article discusses those and other variables that directly influence the satisfaction level of the Dermatologist who undertakes it, the Pathologist who analysis it and the Patient of whom the biopsy is taken.
Biopsy; Scalp; Skin; Skin
A biópsia de pele é um procedimento de rotina na prática da Dermatologia. No entanto, se alguns cuidados não forem observados a relação custo/benefício será desfavorável. Decisão do local anatômico mais apropriado e da lesão com as alterações histológicas mais características da onde será colhido o fragmento de pele, por exemplo, são fundamentais. Este artigo discute estas e outras variáveis que influenciam diretamente no grau de satisfação do Dermatologista que realizou a biópsia de pele, do Patologista que a analisou e do Paciente que a sofreu.
Biópsia; Couro cabeludo; Pele; Pele
DERMATOPATHOLOGY
Skin biopsy with histopathologic analysis. Why? What for? How? Part II
Betina Werner
Specialist in dermatopathology, International Society of Dermatopathology. Master in clinical surgery, focus on surgical pathology, Universidade Federal do Paraná. Ph.D. in children and adolescent health, focus on dermatopathology, Universidade Federal do Paraná. Volunteer/ invited professor/ dermatopathologist, Service of Clinical Pathology and Dermatology, and discipline of clinical pathology, Universidade Federal do Paraná (UFPR) Curitiba (PR), Brazil
Mailing Address Mailing Address: Betina Werner Rua. Dr. Nelson de Souza Pinto, 759 82200 060 Curitiba, PR. Tel./Fax: 41 3232 1906 41 3232 3524 E-mail: betina.werner@gmail.com
ABSTRACT
Skin biopsy is a common procedure in Dermatology practice. The cost-benefit ratio, though, can be unfavorable if attention is not paid to specific details in performing a skin biopsy. For example, proper selection of anatomical site and best lesion to perform a biopsy are very important. The present article discusses those and other variables that directly influence the satisfaction level of the Dermatologist who undertakes it, the Pathologist who analysis it and the Patient of whom the biopsy is taken.
Keywords: Biopsy; Scalp; Skin; Skin/pathology
INTRODUCTION
In the first part of this article, we discussed "why" and "what for" of skin biopsy with the clinical pathology 1. We also introduced how it was made, reinforcing the valuable contribution of clinical data for the conclusion of histological diagnosis. In this second part, "how" a skin biopsy is made will be addressed with more details, including the decision in which the technique and the choice of lesion or skin region are more appropriate to the microscopic study of some specific diseases. Special situations such as the approach of biopsy of melanocytic lesions and scalp are also discussed.
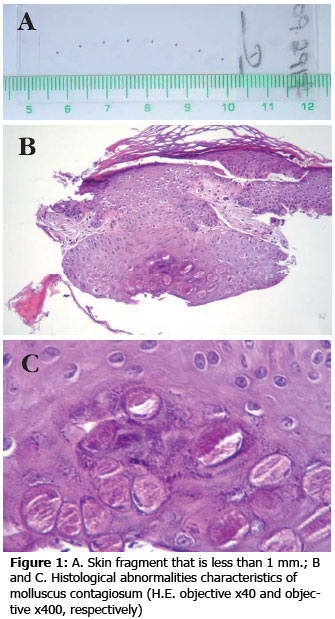
Biopsy Techniques. The purpose of this article is not to discuss how to perform punch, shaving, curettage or other skin biopsies, but rather to discuss the best indications for specific skin diseases. According to Charlton, 2 given that all skin lesions are accessible to dermatologists, there is no justification for the biopsy to be considered inappropriate. In a softer tone, we may say that the clinician should bear in mind what type of specimen the biopsy will provide and whether it is enough to clarify his diagnostic questions 3. A given fact is: the more material received by the pathologist for the analysis, the higher the likelihood of coherent diagnosis. The pathologist should also bear in mind that the dermatologist can also consider the cosmetic side of the procedure, facing the dilemma of having a sample appropriate for the disease to be studied versus the site and the size of the scar that the biopsy will leave on the patient 4. However, in some cases, histological findings are so characteristic that even tiny skin fragments may be enough for the diagnosis (Figure 1).
Punch: Punch biopsy with 3 or 4 mm are ideal for inflammatory and infectious diseases that are manifested by rash, spots, papules, plaques, points, vesicles, blisters, purpura, exulceration and ulcers 4-8. Some authors recommend 2 mm punch for the diagnosis of any skin lesion, because they do not find statistical difference when compared to clinical pathology results obtained by elliptical biopsy 9. However, we should bear in mind that 2 mm lesion sampling is too small and a skin fragment of this size may cause problems for the histology processing. In the case of more palpable than visible inflammatory lesions, we recommend 5 mm punch or greater, or biopsy with knife, including deep reticular dermis and hypodermis, especially for the diagnosis of panniculitis 5.
Shaving: Shaving is not indicated for skin inflammatory diseases and it is well employed for the diagnosis of skin tumors that are clinically suggestive of benign course, but the deep margin will always be involved . 4, 6, 8, 10 If there is suspicion of melanoma, for example, this practice should not be applied, given that the analysis of Breslow thickness may not be correctly assessed 10. Conversely, there are authors who recommend shaving biopsy in dysplastic nevi because they believe that the technique provides appropriate specimens for the histological analysis and have the advantage of leaving a more esthetically acceptable scar than the one caused by elliptical excision 11. However, some authors recognize the potential of recurrence at the melanin lesion site and the problems of clinical pathology assessment in the differentiation between recurrent nevus and melanoma. In the specific case of basal cell carcinoma, both punch and shaving biopsy are appropriate at first for diagnosis and determination of histological type 12.
Curettage: Curettages provide the least appropriate specimens for pathologists, given that the lesion to be diagnosed comes in parts. However, it is known that this technique is widely used by dermatologists because it is at the same time diagnostic and therapeutic. Depending on the experience of the pathologist, seborrheic keratosis, actinic keratosis, basal cell carcinoma and Bowen disease are normally correctly diagnosed by this technique (Figure 2). Pigmented lesions suspected of melanoma should never undergo curettage.
Incision Biopsy: Incision biopsy (which may be performed using punch) is the partial removal of the lesion and its main objective for the diagnosis is to plan the future of the therapeutic approach determined by the results of the clinical pathology analysis of skin tumors or inflammatory lesions 7, 8.
Excision Biopsy (exeresis): Excision biopsy or exeresis is the total removal of the skin lesion and it is indicated for tumors in general and in melanocytic lesions, because they have both diagnostic and therapeutic purposes 6, 8. Excision biopsy is the only type of procedure in which the analysis of surgical margins may be safely made; free margins in punch or shaving biopsy may not correspond to the truth owing to the way the section of the specimen is made and its inclusion in paraffin 13.
Skin pigmented lesions. Owing to the great importance that a diagnosis of melanoma has in the life of a patient, the approach of pigmented lesions deserves to be a separated topic. Sometimes the histological differentiation between the melanocytic nevus and melanoma is not simple and the microscopic criteria that are considered in the scale of decision benign x malignant are conflicting, even for highly experienced dermatopathologists 14, 15. Moreover, it is estimated that 13% of all medical-legal processes in the United States involve the diagnosis of melanoma 16. Jackosn 17 reviewed all the 75 cases of medical-legal lawsuits involving melanoma in Canada from 1976 to 1993. The two most common causes of lawsuits were delay in diagnosing the melanoma by the clinician, because there was no early biopsy of the suspected lesion, and diagnostic error by the pathologist. Not all errors with melanocytic neoplasms result from negligence, lack of knowledge or imprudence, but it may start from the way the neoplasm was sampled 18. Thus, whenever possible, lesions of melanocytic origin should be completely excised 6, 8, 17-19. For the pathologist, it is much better to study it completely, because their architecture (histological profile) is more important than the cellular detail for the differentiation between nevus and melanoma (this principle also applies to keratoacanthomas, in which the lesion shape on the lateral portions is an essential diagnostic criterion). Moreover, in the case of melanoma diagnosis, excision biopsy is appropriate to assess the surgical margins and contains the whole extension, in which the pathologist may choose the best regions for the analysis of necessary diagnostic parameters for this neoplasm, such as Clark level, Breslow thickness, mitotic index, etc 19. If for some reason the dermatologist decides for an incision biopsy, it is better to perform it in the most pigmented area, to enhance the likelihood of finding enough histological findings for the diagnosis of melanoma, if that is the case. In a sense, melanocytic nevus is the most common neoplasm in human beings and melanoma is the most lethal neoplasma (a 4 mm tumor, as large as a pea, has huge metastatic potential) 18. As melanocytic nevi are so common, even if the percentage of clinical and histological difficult cases is very small, the number of problematic cases is not small 18. Overall, we should always be careful.
Biopsy site anatomical region. Any experienced dermatopathologist agrees: skin biopsy over the waist is easier to be interpreted because we can avoid abnormalities resultant from blood stasis of the lower limbs 5. Regions of the body resulting from constant friction, such as the knees and elbows, are also unfavorable for the analysis of microscopic abnormalities, because the skin becomes thick. Similarly, areas of marked sun exposure, where elastosis is predominant, may be difficult for the pathologist. In summary, whenever possible and always sampling the lesion that is most characteristic of the disease, we should avoid biopsy of the lower limbs, extensor surfaces, elbows, knees and skin areas with marked sun damage.
Biopsy site type of lesion. Given that the purpose of performing skin biopsy in inflammatory diseases is to have the most specific diagnosis possible, ideally we should choose the lesion that provides the most frequent characteristics of the disease 20. Newer or older lesions in the evolutive phase may not contain the necessary microscopic abnormalities for the diagnostic conclusion 20. For this reason, the greater the sampling, the higher the likelihood of finding essential microscopic findings for the diagnosis. Lichenoid phthiriasis, in which lesions are normally presented in different stages of progression, is an example of a disease in which multiple biopsies enhance the likelihood of diagnostic conclusion 5. The same applies to fungoid mycosis, in which the initial phase has subtle histological abnormalities and the possibility of examining more than one skin fragments enhances the diagnostic chances. In pruriginous lesions, we should choose lesions without excoriation and secondary infection, if applicable 5. However, even if there are only lesions caused by scratching, the biopsy may be useful to exclude a primary pathological process and to confirm the hypothesis of artifact dermatitis, for example 5. In case of pigmentation disorders, it is recommended to have a biopsy of the center of the lesion, in the hyperchromic or hypochromic/ achromic region, depending on the diagnostic suspicion. The attempt to encompass in the same biopsy normal color and abnormally colored skin is not an advisable practice, because this difference is not always visualized under microscopy, because it depends on the way the skin fragment is included in paraffin 20. It is much better to perform two biopsies, preferably by punch, in a normal area and another in the abnormal area, with clear identification of both. The same practice is indicated in case of connective tissue diseases with subtle microscopic abnormalities, such as atrophodermia and anectodermia, for example. Pathologists will get familiarized with the normal aspect of the skin and will be better equipped to judge the abnormal sample by comparison. In skin diseases that have ring lesions, with desquamative crown or not, biopsy in the peripheral and most active area will most probably show the most characteristic morphology. In cases of suspicion of Hansens disease the physician should bear in mind that leprosy mycobacterium has trophism for nerves and it is in the deep dermis and hypodermis that neural branches may be commonly found. Therefore, deep biopsies with punch or knife are recommended. In bullous diseases, the lesion chosen for biopsy should be carefully selected 3. Histological assessment of blisters and vesicles with longer progression time or ruptured and with secondary infection do not necessarily bring satisfactory histological conclusions 5. Diseases that form subepidermal cleft may become intraepidermal with time, owing to the reepithelization phenomenon 5. It is better to sample a lesion of recent onset, with only the initial cleft formation; if there are only blisters and old vesicles, the blister periphery or the whole vesicle should be included 5. If there are only lesions with longer duration of evolution, sometimes characteristic samples of the lesions may be found around the lesion. If the dermatologist wants to complement the investigation of the disease using direct immunofluorescence, another sample should be made of the perilesional skin, not including the blister or vesicle. The fragment should not be placed in formalin but rather in the appropriate medium (Michel). The borders of ulcerated lesions are always preferable than the center (if the diagnostic question is whether it is infectious or not) and large ulcerated lesions with suspicion of neoplasm should be sampled in more than one region or quite generously, using a knife.
Biopsy of the scalp. The histology study of the scalp for diagnosing alopecia has a number of peculiarities. The two main facts that differentiate it from the analysis of other skin lesions are: the inclusion of the hypodermis in the sample is a must, because hair bulbs are located in it and the recommendation is to use transversal histological sections in relation to the cutaneous surface for the hair follicle examination 21-23. Concerning the first item, it is not necessary to perform the biopsy with knife to sample the fat. Moreover, sometimes the cut with knife provides too wide but not too deep fragments, which do not have enough subcutaneous cell tissue and may cause difficulties to include it in paraffin because of the difference in thickness of the epidermal superficial portion and the deep dermal/ hypodermal portion. The 4 mm and 5 mm punch biopsies are ideal for sampling the hypodermis and provide a more regular specimen, which facilitates the inclusion. Concerning the need for non-conventional inclusion (transversal or horizontal), it is advisable to have only one biopsy to that end 21-23. The assessment of transversal histological sections is complementary to the conventional study with longitudinal or vertical sections and it is really important in non-scarring alopecia in which the total number of hair follicles present in the sample may be studied 21-23. Therefore, for the analysis of alopecia, one punch biopsy is indicated for conventional study and another one for special histological sections.
Handling and Fixation. Any skin fragment removed from the body should be handled with care, because instruments such as clamps may cause severe crushing of the tissue, impairing the microscopic analysis (Figure 3) 5. Transfixing the fragment removed by punch with a needle (to hold it before cutting it with the scissors) makes a central hole in the specimen that may compromise the histological assessment (Figure 4) 5. The liquid for fixation of a specimen for anatomical study is formol at 10% concentration and preferably buffered 5, 20. Moreover, it is recommended that the liquid volume is 10-20 times greater than the size/ volume of the tissue fragment 5. For specimens below 1 cm in the largest axis, 30 ml of fixation is enough 18.
CONCLUSION
Skin biopsy is important and has to be well made. It involves its own difficulties, requiring dedication of the dermatologist and suffering from the patient. It carries much expectation and anxiety both from the physician and the patient, anxious for the diagnosis. It takes time and study by the pathologist that makes the macro and microscopic analysis of the specimen. It depends on the interpretation of histological findings by the dermatologist. It defines treatment and management of the skin disease. Finally, skin biopsy and its anatomical opinion may be reason for lawsuits against the dermatologist or the pathologist. Above all, the dermatologist should know the answers for the questions why, what for and how. The most careful the clinician is, the higher the likelihood of the three stakeholders of the process, to wit, the patient, the dermatologist and the pathologist, become satisfied with the outcomes. There is nothing more frustrating for a dermatopathologist than an inappropriate skin sample or total absence of clinical information;
In the case of the dermatologist, a not very specific clinical anatomy report is very disappointing. The worst part of the equation lies in the hands of the patient, who realizes that the minor surgery that he underwent, which sometimes leaves scars behind, have contributed nothing or very little to relieving his discomfort.
ACKNOWLEDGEMENT
To Dr José Fillus Neto, Dermatopathologist, and Dr Sergio Z. Serafini, Dermatologist, my masters and professional role models.
REFERENCES
Conflict of interest: None
Financial funding: None
How to cite this article: Werner B. Biópsia de pele e seu estudo histológico. Por quê? Para quê? Como? Parte II. An Bras Dermatol. 2009;84(5):507-13.
- 1. Werner B. Biópsia de pele e seu estudo histológico. Por quê? Para quê? Como? Parte I. An Bras Dermatol. 2009;84:391-5
- 2. Charlton R. Diagnosis and biopsy specimens. Am J Dermatopathol. 1981;3:234
- 3. Kak BK, Azulay DR. Exames complementares em dermatologia. In: Azulay RD, Azulay DR, editors. Dermatologia. Rio de Janeiro: Guanabara Koogan; 2006. p.61
- 4. Scope A, Halpern AC. Diagnostic procedures and devices. In: Wolff K, Goldsmith LA, Katz SI, Gilchrest BA, Paller AS, Leffell DJ, editors. Fitzpatrick's Dermatology in General Medicine. New York: McGraw-Hill; 2007. p. 42
- 5. Ackerman AB, Böer A, Bennin B, Gottlieb G. Proper biopsy. In: Histologic Diagnosis of Inflammatory Skin Diseases. An Algorithmic Method Based on Pattern Analysis. Nova York: Ardor Scribendi; 2005. pp. 171-9
- 6. Boyd AS, Neldner KH. How to submit a specimen for cutaneous pathology analysis. Using the '5 D's' to get the most from biopsies. Arch Fam Med. 1997;6:64-6
- 7. Zuber TJ. Punch biopsy of the skin. Am Fam Physician. 2002;65:1155-8,1161-2,1164
- 8. Alguire PC, Mathes BM. Skin biopsy techniques for the internist. J Gen Intern Med. 1998;13:46-54
- 9. Todd P, Garioch JJ, Humphreys S, Seywright M, Thomson J, du Vivier AW. Evaluation of the 2-mm punch biopsy in dermatological diagnosis. Clin Exp Dermatol. 1996;21:11-13
- 10. Ackerman AB. Shave biopsies: the good and right, the bad and wrong. Am J Dermatopathol. 1983;5:211-2
- 11. Armour K, Mann S, Lee S. Dysplastic naevi: to shave, or not to shave? A retrospective study of the use of the shave biopsy technique in the initial management of dysplastic naevi. Australas J Dermatol. 2005;46:70-5
- 12. Russell EB, Carrington PR, Smoller BR. Basal cell carcinoma: a comparison of shave biopsy versus punch biopsy techniques in subtype diagnosis. J Am Acad Dermatol. 1999;41:69-71
- 13. Sellheyer K, Bergfeld WF. When to ask your dermatopathologist to evaluate for surgical margins and when not: a matter of confusion for dermatologists and dermatopathologists alike. J Am Acad Dermatol. 2005;52:1095-7
- 14. Ackerman AB. Readers of dermatopathology: practical & conceptual at 7 years of age: old enough to be told a secret. Dermatopathol Prac Concept. 2001;7:1
- 15. Werner B, Radevic M, Ackerman AB. Discordance among experts in diagnosis of unconventional melanocytic neoplasms: facts and lessons derived from them. Dermatopathol Prac Concept. 2003;9:4
- 16. Crowson AN. Medicolegal aspects of neoplastic dermatology. Mod Pathol. 2006;19Suppl 2:S148-54
- 17. Jackson R. Malignant melanoma: a review of 75 malpractice cases. Int J Dermatol. 1997;36:497-8
- 18. Glusac EJ. Under the microscope: doctors, lawyers, and melanocytic neoplasms. J Cutan Pathol. 2003;30:287-93
- 19. Stell VH, Norton JH, Smith KS, Salo JC, White Jr RL. Method of Biopsy and Incidence of Positive Margins in Primary Melanoma. Ann Surg Oncol. 2007;14: 893-8
- 20. Sampaio SAP, Rivitti EA. Exame histopatológico, glossário e padrões hitopatológicos. In: Dermatologia. São Paulo: Artes Médicas; 2007. p. 123
- 21. Madani S, Shapiro J. The scalp biopsy: making it more efficient. Dermatol Surg. 1999;25:537-8
- 22. Sperling LC. Specimen acquisition handling and processing. In: An Atlas of Hair Pathology with Clinical Correlations. New York: The Parthenon Publishing Group, 2003. p. 15-8
- 23. Elston DM, McCollough ML, Angeloni VL. Vertical and transverse sections of alopecia biopsy specimens: combining the two to maximize diagnostic yield. J Am Acad Dermatol. 1995;32:4547
Publication Dates
-
Publication in this collection
07 Jan 2010 -
Date of issue
Oct 2009
History
-
Accepted
29 May 2009 -
Received
05 May 2009