Abstracts
BACKGROUND: Adult female acne (AFA) is a dermatosis of increasing incidence; hyperandrogenism is present in many cases, but there are patients without hormonal abnormalities. OBJECTIVE: To analyze the prevalent characteristics in adult women with acne without hyperandrogenism. MATERIAL AND METHODS: A survey of 226 medical records with AFA complaints was conducted; of these, 116 (51.3%) had a normal hormonal profile and were, therefore, included in the study. We investigated age, ethnicity, clinical profile of acne, complementary exams, previous treatment, prescribed treatment and evolution. RESULTS: The mean age was 33.9 years and the predominant clinical grade of acne was moderate inflammatory (grade 2). The face was the most affected area. Topical retinoids were the most prescribed drugs, and systemic medication was prescribed to 53.4% of the patients. Total regression was observed in 31 patients (26.7%) within 12 weeks of treatment. Adverse reactions to topical treatment occurred in 21.5% of the patients. CONCLUSION: The clinical manifestations of AFA in patients without hyperandrogenism are moderate, with predominance of inflammatory lesions. Treatment is similar to that of acne vulgaris; however, special attention should be taken with more irritating medication since this group appears to be more predisposed to skin irritations.
Hyperandrogenism; Menstrual cycle; Retinoids; Skin
FUNDAMENTOS: A acne da mulher adulta (AMA) é uma dermatose de incidência crescente; o hiperandrogenismo está presente em muitos casos, mas há pacientes sem anormalidades hormonais. OBJETIVO: Analisar as características prevalentes na mulher adulta com acne sem hiperandrogenismo. MATERIAL E MÉTODO: Um levantamento de 226 prontuários com queixa de AMA foi realizado; 116 pacientes (51,3%) possuíam perfil hormonal normal e, portanto, foram incluídas no estudo. Foram pesquisados idade, etnia, perfil clínico da acne, exames complementares, tratamentos anteriores, tratamentos prescritos e evolução. RESULTADOS: A média das idades foi de 33,9 anos e o grau clínico predominante foi o inflamatório moderado (grau II); a face foi a área mais acometida. Os retinoides tópicos foram as drogas mais prescritas, e a medicação sistêmica foi indicada em 53,4% das pacientes. A regressão total foi observada em 31 pacientes (26,7%) até 12 semanas de tratamento. Houve reação adversa ao tratamento tópico em 21,5% das pacientes. CONCLUSÃO: O quadro clínico da AMA em pacientes sem hiperandrogenismo é de moderada intensidade, com predomínio de lesões inflamatórias. Seu tratamento é similar ao da acne vulgar; entretanto, atenção especial deve ser tomada com medicações de maior potencial irritante, uma vez que esse grupo parece ter uma maior predisposição a irritações cutâneas.
Ciclo menstrual; Hiperandrogenismo; Pele; Retinoides
INVESTIGATION
Acne in adult women: epidemiological, diagnostic and therapeutic aspects*
Flavia Alvim Sant'Anna AddorI; Sergio SchalkaII
IMSc in Dermatology, Faculty of Medicine, University of Sao Paulo (USP); Collaborating Professor, University of Santo Amaro (UNISA); Dermatologist: MEDCIN-Private Center of Dermatology and Clinical Research - São Paulo (SP), Brazil
IIMSc in Dermatology, Faculty of Medicine, University of Sao Paulo (USP); Collaborating Professor, University of Santo Amaro (UNISA); Dermatologist: MEDCIN-Private Center of Dermatology and Clinical Research - São Paulo (SP), Brazil
Mailing address
ABSTRACT
BACKGROUND: Adult female acne (AFA) is a dermatosis of increasing incidence; hyperandrogenism is present in many cases, but there are patients without hormonal abnormalities.
OBJECTIVE: To analyze the prevalent characteristics in adult women with acne without hyperandrogenism.
MATERIAL AND METHODS: A survey of 226 medical records with AFA complaints was conducted; of these, 116 (51.3%) had a normal hormonal profile and were, therefore, included in the study. We investigated age, ethnicity, clinical profile of acne, complementary exams, previous treatment, prescribed treatment and evolution.
RESULTS: The mean age was 33.9 years and the predominant clinical grade of acne was moderate inflammatory (grade 2). The face was the most affected area. Topical retinoids were the most prescribed drugs, and systemic medication was prescribed to 53.4% of the patients. Total regression was observed in 31 patients (26.7%) within 12 weeks of treatment. Adverse reactions to topical treatment occurred in 21.5% of the patients.
CONCLUSION: The clinical manifestations of AFA in patients without hyperandrogenism are moderate, with predominance of inflammatory lesions. Treatment is similar to that of acne vulgaris; however, special attention should be taken with more irritating medication since this group appears to be more predisposed to skin irritations.
Keywords: Skin; Hyperandrogenism; Retinoids; Menstrual cycle
INTRODUCTION
Adult female acne is define as acne that develops after 25 years of age. 1.2 Hyperandrogenism in women is the main etiology associated with acne in this age group. When present, hyperandrogenism is also often characterized by seborrhea, alopecia and hirsutism, menstrual disorders and ovulatory dysfunction with infertility and metabolic syndrome, psychological disorders and virilization. 2.3
Among women with polycystic ovary syndrome, the leading cause of hyperandrogenism, 70% have acne. 4
However, in most cases of adult female acne, there is no associated endocrine disease. The most common cause is associated with an altered response of cutaneous androgen receptors to the physiological hormonal changes of menstrual cycle, related to the occurrence of inflammatory lesions and even to an increase in sebogenesis. 5 There is an increase in the number of acne lesions in up to 60-70% of women, especially in the premenstrual period.6,7
Even though there is not an underlying disease, clinical manifestations create great discomfort for the patient, not only due to the appearance of active lesions, but also due to their chronicity and scarring. 8
This study aims at investigating the particularities of adult female acne (AFA) with no correlation with endocrine diseases, as well as the response to the clinical treatments employed.
OBJECTIVES
To analyze the prevalent characteristics in adult women with acne without hyperandrogenism. Secondarily, to investigate the possible correlation with other etiological factors.
MATERIAL AND METHODS
This was an open, uncontrolled, retrospective study that surveyed the records of female patients of childbearing age, older than 25 years, from March 2006 to December 2008, diagnosed in the first visit with adult female acne (AFA).
The study included data from all the patients who had been clinically diagnosed with AFA for over a year, with negative laboratory results for endocrinopathies associated with acne, as well as patients who had been monitored for at least 3 months. This follow-up period was considered to evaluate the response to the recommended firstchoice treatment.
Laboratory tests of diagnostic value for endocrinopathies related to acne were: free testosterone, androstenidione, dihydroepiandrosterone, dihydroepiandrosterone sulphate, follicle stimulating hormone, luteinizing hormone and prolactin. Ultrasound description was also analyzed, when present.
The occurrence of concomitant diseases was also investigated; other research parameters were: age, ethnicity, clinical degree of acne, prescribed drugs, and response to the suggested treatment.
STATISTICAL METHODOLOGY
Statistical analysis was performed using frequency tables with confidence intervals to assess whether there were differences between the given proportions, or the chi-square test, to verify the association between two variables.
When the number of slots with a frequency inferior to 5 did not allow the performance of the chisquare test, Fisher's exact test was used.
RESULTS
262 medical records were collected from December 2005 to December 2008. All the records registered complaints of AFA and, of these, 226 contained information on requested complementary exams and follow-up , being therefore suitable for the study.
Of the 226 patients with complete medical records, 116 (51.3%) had normal hormonal profile and were therefore included in the study.
EPIDEMIOLOGICAL DATA
Age and ethnicity
The mean age of the sample was 33.9 years (min. 25, max. 45); Caucasians predominated in the study, with 81 patients (69.8%), followed by Asians with 22 patients (18, 9%%); Negroid ethnicity was referred by 13 patients (11.2%).
Clinical manifestations
The predominant clinical grade of acne was moderate (grade II, Sampaio & Rivitti (9) ), found in 94 patients (81.0%); 13 patients (11.0%) showed grade III; grade I was observed in 09 patients (7.7%).
There was no relationship between the degree of acne and age in the sample at a significance level of 5%, according to the chi-square test (p = 0.881); the mean age for grade 2 was 34.2 years; for grade 3, 33.6 years, and for grade 1, 31.5 years.
The distribution of lesions was also investigated. The area with the greatest number of lesions was the face (100%), followed by the presternal area in 37 patients (31.8%) and dorsum in 12 patients (10.3%).
An association between the development of acne and menstrual cycle was referred by 54 patients (46.5%), who reported aggravation of lesions in the premenstrual period.
Ultrasound pattern
Ultrasound was requested of 21 (18.1%) of the 116 patients who comprised the sample. Of these patients, 11 had follicular pattern, 8, homogeneous pattern and 2, microcystic pattern.
Based on Fisher's exact test, there was no evidence of a relationship between the clinical grade of acne and any ulrasound finding in the sample of patients studied (p = 0.8978) at a significance level of 5%, as shown in Table 1.
Concomitant diseases and drugs
Concomitant diseases were found in 5 patients (4.3%). One patient had diabetes mellitus type 2 and two patients had hypertension; one had a diagnosis of hyperthyroidism; one patient was suffering from chronic lupus, posterior to the appearance of acne.
Use of topical products
75 patients (64.6%) had a history of aggravation of the clinical manifestations of acne with the use of cosmetics such as makeup, moisturizers and sunscreens. With regard to self-prescribed topical treatments, 58 (50%) had used drugs and cosmetics with no improvement or partial and temporary improvement of their condition.
TREATMENT REGIMENS INDICATED
Systemic therapy
Systemic treatment was recommended to 62 patients (53.4%). With regard to the systemic drugs prescribed, contraceptives predominated and were used by 35 (30.17%) patients.
The association of contraceptives with other systemic drugs was recommended to 9 out of 35 patients (35.7%); 5 used azithromycin and 4, isotretinoin.
Systemic antibiotics were prescribed to some of the patients; 15 used limecycline and 9 used azithromycin; minocycline was prescribed to 5 patients.
Isotretinoin was prescribed for 2 patients without oral contraceptives, since both had undergone tubal ligation and did not want to take hormones. The percentage distribution of systemic drug use is illustrated in Graph 1.
There was statistical significant relationship between the indication of systemic drugs and the clinical grade of acne, based on Fisher's exact test (p = 0.0916)
Topical therapy
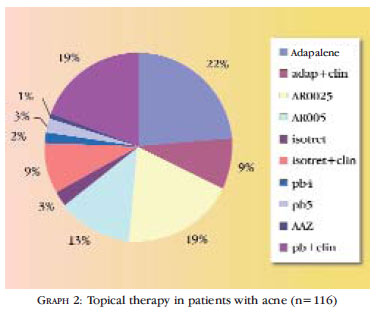
All the patients were instructed to use topical medication. The most commonly prescribed drugs were retinoids, used by 66 patients (56.9%); among retinoids, tretinoin was prescribed to 37 patients, adapalene to 27 patients and isotretinoin to 2 patients. Formulations associated with antibiotics were used by 43 patients (37%): benzoyl peroxide + clindamycin were prescribed to 22 patients, isotretinoin-erythromycin, to 11 patients, adapalene and clindamycin, to 10 patients. Benzoyl peroxide was prescribed to 7 patients. The percentage distribution of prescribed topical drugs is illustrated in Graph 2.
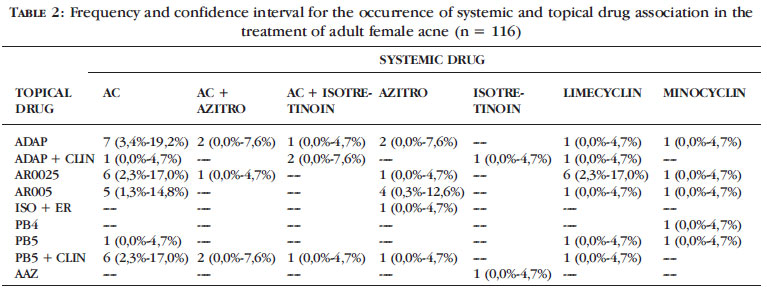
We also investigated whether there was a more frequent association profile of topical and systemic drugs by analyzing relative frequencies and confidence intervals (Table 2).
Although the most used association was ADAP with AC, there were no significant statistical differences between the association profile of the drugs due to the intersection of confidence intervals (ICI).
Evolution in 4-6 weeks
All the patients were evaluated between 4 and 6 weeks of treatment.
Total regression of symptoms was described by 6 patients (5.17%) during this period of treatment. The other patients reported some degree of regression, but 9 patients (7.75%) did not observe any degree of improvement; lack of improvement appears to have no correlation with the clinical grade of acne, since all of the patients were classified as having acne grade II; mean age, although slightly higher than that of the sample (35.3 years) showed no correlation with the clinical evolution of the disease.
Of these 9 patients, only 1 was on systemic antibiotics and two had systemic diseases (diabetes and lupus, respectively).
There was no predominance of any topical drug used in this group. Of these, 3 were taking antibiotics and one was using oral contraceptive.
With respect to adverse effects, they were reported as mild by 5 patients, all in use of retinoids (4 using tretinoin 0.05% and 1, tretinoin 0.025%), but treatment was not discontinued. The patients were reoriented on how to use the drugs.
Evolution in 10-12 weeks
Total regression was observed in 31 patients (26.7%) Of these, 18 (58%) were using oral medication: 03 were taking oral isotretinoin, 8, systemic antibiotics and 7, oral contraceptives alone. All these patients were included in the group who had reported some improvement in the first assessment. These results do not confirm whether there is a significant statistical relationship between the use of associated therapy and patient's outcome based on the chi-square test (P = 0.494).
The possible association between the evolution of the disease and the clinical grade of acne was then investigated based on Fisher's exact test, as shown in Table 3.
There was no statistical significance between the evolution of the disease and the clinical grade of acne.
Medication was changed in 25 patients (21.5%) due to lack of significant improvement, reported for 15 patients (12.9%). Adverse effects were the reason for change of medication in 06 patients; of these, 3 were using tretinoin 0.05%, 2 were using adapalene with clindamycin and 1 patient was using benzoyl peroxide + clindamycin.
Adverse effects
Adverse effects were observed in 27 patients; 24 were skin reactions associated with the use of topical products, such as erythema, dryness and scaling; 02 patients reported gastrointestinal symptoms related to antibiotic therapy, namely epigastric pain (with limecycline) and diarrhea (azithromycin).
Table 4 describes the number of reactions related to each topical treatment regimen.
By comparing confidence intervals, it appears that patients who used ISO + ER presented fewer adverse reactions than patients who used AR0025.
Differences were not identified for the remaining patients.
The following products - ISO, PB4, PB5 and PB8-are included in the table, but they were not considered in the analysis due to low usage rates.
DISCUSSION
Adult female acne (AFA) is a frequent medical complaint requiring proper investigation of hormonal diseases because these may trigger acne and must be treated. 9 However, the development of acne with no hormonal imbalances associated is becoming increasingly frequent in this age group.10 The findings of a study with 200 adult women with acne found that only 37% had hyperandrogenism.
An epidemiological study conducted in France with 4000 women showed that 41% of the adult women population had acne. 12
Two different groups of adult acne can be identified: the so-called 'persistent acne', which is more common and an evolution of adolescent acne, and 'acne of late onset', starting only after 25 years of age.
Perhaps these patient have incipient abnormalities in ovarian and adrenal functions, which require investigation. 11, 2
Most studies on AFA consider patients with associated hormonal changes. In this sample of patients without hormonal imbalances, the predominant clinical grade (grade 2, inflammatory acne) was the same as that of patients with hormonal diseases. 13,14
Some authors suggest that predominant lesions of female adult acne are inflammatory, without many comedones 15
The age group most frequently affected by AFA appears to be 25-35 years. In a study by França and Teixeira, 16 of 60 women with acne, the mean age obtained was 26.5 years; in the present study, the mean age was 33.9 years, but it is important to note that this number refers only to patients without hormonal diseases.
In the sample of patients studied, with a normal hormone profile, the expected ultrasound findings would also be normal. None of the patients presented a polycystic pattern or increased ovarian stroma, which would be linked to hormonal alterations. However, normal ultrasound findings do not rule out ovary diseases, so hormone level investigation is always necessary. 17, 18,19
Although our sample size did not allow the study of systemic diseases related to AFA, the clinical grade of acne did not appear to suffer any influence of the underlying diseases found in the sample. The therapeutic response, also unaffected, was slower in two of the five patients with systemic diseases in the sample .
With respect to ethnicity, although the disease predominated among Caucasians, this finding may be related to the profile of the patients and cannot be considered relevant. In the medical literature there is no evidence of predominance of AFA in any ethnic group.
Although there is no clear association of the use of cosmetics in the etiology of adult female acne, 15 the use of acnegenic cosmetics, as well as that of drugs, appears to be relevant to the aggravation of acne in women older than 25 years. 20.21
The analyzed sample confirms this association: 64.6% of patients reported onset of lesions after the use of cosmetics. Self-medication also seems to be a frequent practice, always attempted before visiting a dermatologist: 50% of the sample used some product to reduce acne lesions, without obtaining the expected improvement.
The literature also describes stress, heredity and smoking as factors associated with acne aggravation, but in the sample studied these data were not collected. Since this is a retrospective study based on data from medical records, these pieces of information are not systematically investigated. Nonetheless, they should not be neglected, since up to 67% of the cases are associated with family history of the disease. 22
Gouden et al., in a demographic study of 236 patients, showed that the risk of someone who has a first-degree relative with adult acne to develop the same clinical manifestations of the disease is up to three times higher than the general population. 23
As for emotional stress, the authors suggest that about 50% of patients report worsening of symptoms.(11.12) And there is evidence that acne is more prevalent and severe in smokers than in nonsmokers.24
Therapy
The results suggest that systemic therapy appears to have a positive effect: of the 10 patients who showed no improvement, only 01 was using systemic medication. Although not a statistically significant result, of the 31 patients with total regression in the last evaluation, 18 (58%) were taking systemic medication. Even though the treatment of AFA does not differ significantly from that of adolescent acne, published data suggest that the association of systemic drugs such as contraceptives and antibiotics should be considered, and isotretinoin should be used in more resistant cases .19
Important factors to consider in the use of systemic antibiotic therapy are bacterial resistance (it has been shown that 80% of women with AFA do not respond to systemic antibiotics because of this phenomenon) 25 and the possible reduction of the contraceptive effect of antiandrogens when associated with cyclins. 26
The adverse reactions observed were in the order of 21.5%, of which 88% were skin reactions with an irritating pattern. This finding suggests greater susceptibility to irritation in the sample, which is confirmed by some authors. This pattern of irritation seems more frequent in older age groups. 27
There are no studies that investigate the possible differences between the levels of sebaceous gland activity and skin barrier in AFA compared to adolescent acne. A finding that suggests greater predisposition to irritation is the higher percentage of reactions with tretinoin gel 0.025% than with tretinoin cream 0.05%; in other words, the form and strength of the product appear to play a larger role in irritation than retinoid itself.
Finally, it should be noted that data from this study allowed these interpretations; however, there is a methodological limitation to the study due to its retrospective and not cohort nature. Prospective studies with larger samples that allow comparisons should be encouraged as they provide greater scientific evidence.
CONCLUSION
Patients with adult female acne have a high probability of having hormonal abnormalities, which should be dismissed at the start of treatment. The treatment of AFA in patients without hormonal diseases is similar to that of acne vulgaris, producing the same results within a similar period of time. However, special attention should be taken with more irritating medication, since this group appears to be more prone to skin irritations.
REFERENCES
- 1. Goodman NF, Bledsoe MB, Cobin RH, Futterweit W, Goldzieher JW, Petak SM, et al. Hyperandrogenic Disorders Task Force. American Association of Clinical Endocrinologists medical guidelines for the clinical practice for the diagnosis and treatment of hyperandrogenic disorders. Endocr Pract. 2001;7:120-34.
- 2. Rivera R, Guerra A. Management of acne in women over 25 years of age. Actas Dermosifiliogr. 2009;100:33-7.
- 3. Yarak S, Bagatin E Hassun KM, Parada MOAB, Talarico Filho S. Hiperandrogenismo e pele: sindrome do ovario policistico e resistencia periferica a insulina. An Bras Dermatol. 2005;80:395-410.
- 4. Azziz R, Woods KS, Reyna R, Key TJ, Knochenhauer ES, Yildiz BO. The prevalence and features of the polycystic ovary syndrome in an unselected population. J Clin Endocrinol Metabol. 2004;89:2745-9.
- 5. Strauss JS, Thiboutot DM. Diseases of the sebaceous glands. In: Freedberg IM, Eisen AZ, Wolff K, Austen KF, Goldsmith LA, Katz SI, Fitzpatrick TB, eds. Fitzpatrick's Dermatology in General Medicine. New York: Mc Graw-Hill; 1999. p.769-84.
- 6. Williams M, Cunliffe WJ. Explanation of premenstrual acne. Lancet. 1973;2:1055-1057.
- 7. Lucky AW. Quantitative documentation of a premenstrual flare of facial acne in adult women. Arch Dermatol. 2004;140:423-4.
- 8. Bergfeld WF. A lifetime of healthy skin: implications for women. Int J Fertil Womens Med. 1999;44:83-95.
- 9. Sampaio S, Rivitti E. Dermatologia. São Paulo: Artes Médicas; 2001. p.291-94.
- 10. Goulden V, Stables GI, Cunliffe WJ. The prevalence of facial acne in adults. J Am Acad Dermatol. 1999;41:577-80.
- 11. Goulden V, Clark SM, Cunliffe WJ. Post-adolescent acne: a review of clinical features. Br J Dermatol. 1997;136:66-70.
- 12. Poli F, Dreno B, Verschoore M. An epidemiological study of acne in female adults: results of a survey conducted in France. J Eur Acad Dermatol Venereol. 2001;15:541-5.
- 13. White GM. Recent findings in the epidemiologic evidence, classification, and subtypes of acne vulgaris. J Am Acad Dermatol. 1998;39: S34-7.
- 14. Reingold SB, Rosenfield RL. The relationship of mild hirsutism or acne in women to androgens. Arch Dermatol. 1987;123:209-12.
- 15. Parish Lc. Brenner S, Ramos e Silva M. Women's Dermatology. From infancy to maturity. London: Parthenon Publishing; 200. p 394;112.
- 16. Teixeira MAG, França ER Acne in adult women: behavior, hormone and ovarian ultrasonographic profile Rev Bras Saúde Matern Infant. 2007;7:39-44.
- 17. Teixeira RJ, Silva VCG, Freitas JR. Ovários policísticos em meninas e adolescentes normais: uma avaliação ultra-sonográfica. Radiol Bras. 2001;34: 217-20.
- 18. Slayden SM, Moran C, Sams Jr. WM. Hyperandrogenemia in patients presenting with acne. Fertil Steril. 2001;75:889-92.
- 19. Held BL, Nader S, Rodriguez-Rigau LJ. Acne and hyperandrogenism. J Am Acad Dermatol. 1984;10: 223-5.
- 20. Kligman AM, Mills Jr OH. 'Acne cosmetica'. Arch Dermatol 1972;106:843-50.
- 21. Williams C, Layton AM. Persistent Acne in Women: Implications for the Patient and for Therapy. Am J Clin Dermatol. 2006;7:281-290.
- 22. Dumont-Wallon G, Dréno B. Specificity of acne in women older than 25 years. Presse Med. 2008;37(Pt 1):585-91.
- 23. Goulden V, Mcgeown CH, Cunliffe. WJ. The familial risk of adult acne: a comparison between first-degree relatives of affected and unaffected individuals. Br J Dermatol. 1999;141:297ą300.
- 24. Schäfer T, Nienhaus A, Vieluf D, Berger J, Ring J. Epidemiology of acne in the general population: the risk of smoking. Br J Dermatol. 2001;145:100-4.
- 25. Shaw JC, White LE. Persistent acne in adult women. Arch Dermatol. 2001;137:1252-3.
- 26. DeRossi SS, Hersh EV. Antibiotics and oral contraceptives. Dent Clin North Am. 2002;46:653-4.
- 27. Marks R. Acne and its management beyond the age of 35 years. Am J Clin Dermatol. 2004;5:459-62.
Publication Dates
-
Publication in this collection
27 Jan 2011 -
Date of issue
Dec 2010
History
-
Accepted
30 May 2010 -
Received
24 Sept 2009