Abstracts
Atypical nevum (dysplastic) is considered an important factor associated with increased risk of developing cutaneous melanoma. It is believed that atypical nevi are precursor lesions of cutaneous melanoma. They may be present in patients with multiple melanocytic nevi (atypical nevus syndrome) or isolated and in small numbers in a non-familial context. The disease usually begins at puberty and predominates in young people. It has a predilection for sun-exposed areas, especially the trunk. The major challenge in relation to atypical nevi lies in the controversy of defining its nomenclature, clinical diagnosis, dermoscopic criteria, histopathological diagnosis and molecular aspects. This review aims at bringing knowledge, facilitating comprehension and clarifying doubts about atypical nevus.
Dermoscopy; Histology; Nevi and melanomas
O nevo atípico (displásico) é considerado um fator importante associado com o risco aumentado de desenvolvimento do melanoma cutâneo. Acredita-se que nevos atípicos sejam lesões precursoras do melanoma cutâneo. Podem estar presentes em pacientes com múltiplos nevos melanocíticos (síndrome do nevo atípico) ou isolados e em poucas quantidades em um contexto não familial. Aparecem, geralmente, na puberdade e prevalecem em indivíduos jovens. Têm predileção por áreas expostas ao sol, especialmente, o tronco. O grande desafio em relação ao nevo atípico reside na controvérsia em se definir sua nomenclatura, diagnóstico clínico, critérios dermatoscópicos, diagnóstico histopatológico e aspectos moleculares. Esta revisão tem por objetivo trazer o conhecimento, facilitar o entendimento e responder às questões duvidosas concernentes ao nevo atípico.
Dermoscopia; Histologia; Nevos e melanomas
REVIEW
IMSc; PhD - Oncology - Assistant Dermatologist, Department of Cutaneous Oncology, AC Camargo Hospital - São Paulo (SP), Brazil
IIPhD - Oncology - Antonio Prudente Foundation - Assistant Dermatologist, Department of Cutaneous Oncology, AC Camargo Hospital - São Paulo (SP), Brazil
IIIPhD in Surgery, University of Sao Paulo; Director, Department of Cutaneous Oncology, AC Camargo Hospital - São Paulo; Clinical Director, AC Camargo Hospital - São Paulo (SP), Brazil
Mailing address
ABSTRACT
Atypical nevum (dysplastic) is considered an important factor associated with increased risk of developing cutaneous melanoma. It is believed that atypical nevi are precursor lesions of cutaneous melanoma. They may be present in patients with multiple melanocytic nevi (atypical nevus syndrome) or isolated and in small numbers in a non-familial context. The disease usually begins at puberty and predominates in young people. It has a predilection for sun-exposed areas, especially the trunk. The major challenge in relation to atypical nevi lies in the controversy of defining its nomenclature, clinical diagnosis, dermoscopic criteria, histopathological diagnosis and molecular aspects. This review aims at bringing knowledge, facilitating comprehension and clarifying doubts about atypical nevus.
Keywords: Nevi and melanomas; dermoscopy; Histology
INTRODUCTION
The first report of families with multiple melanocytic nevi (common and atypical) and increased incidence of melanoma was published by Clark et al. in 1978. The term B-K syndrome (initials of the last name of each patient of the two families described by Clark) was used to characterize these patients with many nevi. 1 In 1980, Greene et al. used the term dysplastic nevus syndrome (DNS) for similar cases. 2 Still in 1980, Elder et al. described the presence of atypical nevi in a non-familial context and referred to it as sporadic dysplastic nevus syndrome. 3 In 1983, Lynch et al. introduced the term FAMMM ( familial atypical multiple mole melanoma) syndrome.1 Later, Ackerman and Magana Garcia named these lesions Clark's acquired melanocytic nevi. 4
Currently, several names are used in the literature to refer to dysplastic nevus, including B-K nevus, Clark's nevus, atypical nevus and nevus with architectural disorder. 5.6 The term atypical nevus was recommended by the consensus of the National Institute of Health (NIH) in 1992. 7
Many studies define the presence of atypical nevus as a major factor associated with increased risk of developing skin melanoma. 8 It is also believed that common melanocytic nevi and atypical nevi are precursor lesions of skin melanoma. The main evidence for this theory is the association between nevi and melanomas clinically and histologically observed. 9
Clark and Elder (1984) suggested a model of development and progression of melanomas, based on experimental models and clinical and histopathological observations, consisting of six steps: precursor melanocyte; common acquired or congenital nevi with the presence of normal melanocytes, dysplastic nevi with structural and architectural atypia, radial growth melanoma, nontumorigenic primary melanomas with no capacity to metastasize; vertical growth melanoma, primary melanomas that invade the dermis with potential ability to metastasize, and metastatic melanoma. In this model, atypical nevus would be considered a precursor lesion of cutaneous melanoma. 1, 10.11
The major challenge in relation to atypical nevus lies in its controversial nomenclature, clinical diagnosis, dermoscopic criteria, histopathological diagnosis and molecular aspects. 6,12,13
Epidemiologic Features
Atypical nevi have a higher prevalence among young people (those under 30-40 years) and onset usually begins at puberty. 6.14 Atypical nevi are dynamic lesions and may become progressively more or less atypical, but most regress or remain stable throughout life. New atypical nevi may appear after the age of 30, but it is not the most common occurrence.6
The predilection of atypical nevi for sunexposed areas (especially the trunk), its positive association with a history of sunburns in childhood and / or adolescence and its occurrence in people with sensitive skin to sun exposure suggest that their development may be associated with acute and intense sun exposure. 6
The incidence of clinical atypical nevus in the general population, based on studies published in the medical literature, ranges from 1.5% to 18%. In a study conducted in Pennsylvania in 1995, the incidence of melanoma was found to be 15 times higher in patients with clinical atypical nevi, as compared with members of the general population (154 versus 10 per 100,000/year). 7 Crutcher and Sagebiel observed a prevalence of 4.9% in the population of Napa Valley (California - USA), but with clinical and histological diagnosis of atypical nevus. 14 The frequency of atypical nevi in patients with a history of melanoma is higher, ranging from 34 to 59%.6
Many families are affected by a familial autosomal dominant inheritance that characterizes the atypical nevus syndrome - melanoma (FAM-M). This syndrome is defined by the occurrence of melanoma in at least one first or second-degree relative, presence of a large number of nevi (usually more than 50), some clinically atypical and with distinct histological features . 6,7,14 Individuals with this syndrome are 150 times more likely to develop melanoma compared to the general population. 6.15
The number of common and atypical nevi is an important independent risk factor to the development of melanoma. 16 If a person has 100 to 115 nevi, the risk of developing melanoma is 7 to 12 times higher than for a individual with only a maximum of 10 to 15 common nevi. 9, 17,18 The risk of an individual with 5 atypical nevi of developing melanoma is 6 times higher than that of an individual without any atypical lesion. 9.17
There are reports in the literature in which the relative risk ranges from 3.9% to 8.8%, based solely on the clinical diagnosis of atypical nevus in a nonfamilial context. Titus-Ernstoff et al. found a relative risk of 6.2% considering the clinical and histopathological diagnosis.14
Clinical Features
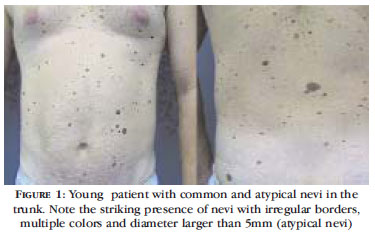
The lack of a consensus regarding the clinical diagnosis of atypical nevus led to the existence of different clinical definitions. One is based on the presence of at least three of the five following characteristics: (1) diameter larger than 5mm, (2) poorly-defined borders, (3) irregular margins, (4) multiple colors, (5) presence of macular and papular components (Figure 1). 13,19
The ABCD (E) rule can also be useful in the diagnosis of atypical nevus. The lesion is considered atypical when it is Asymmetrical (A), with uneven Borders (B), multiple Colors (C), Diameter larger than 5mm (D) and Elevation above the surface, with a maculopapular aspect (E). 12
The atypical nevus may be present in any region of the body, including double-covered areas such as breasts, buttocks and scalp, but it is most commonly found in the posterior upper trunk (Figure 1). 7
The classic atypical nevus syndrome was described by Clark et al. and is characterized by the triad: presence of 100 or more nevi, with at least one nevus with a diameter larger than 8mm and a nevus with clinical features of atypia, in patients with familial history of skin melanoma. 6 According to Kopf et al., atypical nevus syndrome can be classified into five types: type A, sporadic atypical nevus without melanoma; type B, familial atypical nevus without melanoma, Type C, sporadic atypical nevus with melanoma; type D-1, familial atypical nevus with one person in the family with melanoma, Type D-2, familial atypical nevus with two or more family members with melanoma. 20
The definition of this syndrome is also controversial, but one of the most widely used today is one suggested by the NIH in 1992 (incidence of melanoma in at least one first or second-degree relative, a large number of nevi - more than 50 - some being clinically atypical and with distinct histological features). 7
Dermoscopic criteria
The definition of dermoscopic criteria that help establish the diagnosis of atypical nevus with good accuracy is also considered a challenge. In 1993, Pehamberger et al. proposed a dermoscopic diagnostic model for melanocytic lesions called Pattern Analysis, with diagnostic accuracy of 76% compared to 59% with naked eye clinical examination.21,22,23 This model uses dermoscopic characteristics that are most commonly found in atypical nevi in comparison with those found in melanoma. 22.23 Atypical nevus is characterized by the presence of an irregular pigment network, discretly and / or focally prominent, terminating abruptly or gradually; presence of irregular diffuse pigmentation with the posibility of being severe and centrally heterogeneous, with abrupt termination at the periphery of the lesion; areas of depigmentation, when present, are usually irregular and peripheral; brown globules, when present, show varied size and form and are irregularly distributed; black dots, which are rare, are regularly distributed; radiating striae, pseudopods and blue-gray veil are often absent (Figure 2).21,22,23
The main criteria for differentiating atypical from common nevi are: the pigment network and brown globules. In atypical nevi, the pigment network is usually irregular, focally prominent and terminates abruptly at the periphery in some areas; in common nevi the pigment network is regular and fades to the periphery of the lesion. With regard to brown globules, in atypical nevi they show varied sizes and shapes and irregular distribution; in common nevi they are uniform in size and shape, usually with regular distribution in the central area of the lesion.21.22
In 2001, Hofmann-Wellenhof et al. carried out a morphological study in an attempt to determine through dermoscopy other criteria that could facilitate the diagnosis of atypical nevi and early melanomas. Eight hundred lesions clinically consistent with atypical nevi were included and four distinct subtypes were described: 13.20
Atypical nevus with central hypopigmentation: a variant of the reticular-homogeneous type, with a central hypopigmented area without the presence of other dermoscopic structures and pigment network throughout the periphery (Figure 3). 13,20
Atypical nevus with central hyperpigmentation: this type is also called black nevus or hypermelanocytic nevus and represents a variant composed of peripheral pigment network and central hyperpigmentation (black lamella) (Figure 4). 13,20
Atypical nevus with multifocal hypo- and hyperpigmentation: it is characterized by areas of hypopigmentation interspersed with areas of hyperpigmentation giving it a mottled appearance (Figure 5). 13,20
Atypical nevus with eccentric pigmentation: this type is the most important because it includes early melanoma as differential diagnosis. It is characterized by the presence of peripheral hyperpigmentation that reaches the border of the lesion. This area may be characterized by a hyperpigmented blotch or by reticulate hyperpigmentation (Figure 6). 13,20
However, Fikrle and Pzinger conducted a study in which Hofmann-Wellenhof classification was applied to 180 melanocytic lesions, including common melanocytic nevi, atypical nevi, melanomas in situ and thin invasive melanomas, all with histological confirmation. Unfortunately, a meaningful distinction between atypical nevi and early melanomas was not possible with this method. The authors concluded that clinical examination, together with dermoscopy, leads to increased diagnostic confidence and aid in accurate decisionmaking for difficult cases. 24
More recently, in an attempt to differentiate thin melanoma from atypical nevi, Annessi et al. arrived at a final dermoscopic model consisting of five variables: peripheral light-brown hypopigmentation areas, irregularly distributed pigmentation; regression pattern, brown globules with irregular distribution and form, and pigment network abruptly terminating at the periphery. The presence of these features suggests the diagnosis of thin melanoma (Figure 7). Lesions with light-brown hypopigmentation areas on the periphery were 30 times more likely to be diagnosed as thin melanoma compared to lesions without this dermoscopic characteristic. 25
Histopathology
The histological diagnosis of atypical nevus is based on the identification of specific architectural and cytologic abnormalities (melanocytic dysplasia ), and currently there is no criterion that is universally accepted. 1,6,7,14
According to Clark and Elder, histological melanocytic dysplasia shows the following characteristics (Figure 8):1,26,27
1. Lentiginous hyperplasia of melanocytes: characterized by the confluence of melanocytes in the basement membrane zone; melanocytic cells extending beyond the dermal papillae; "shoulder phenomenon" (peripheral extension of the junctional component beyond the dermal component).
2. Epithelioid melanocytic atypia: large melanocytic cells with large amounts of cytoplasm and finely distributed pigmentation giving it a milky aspect.
3. Lamellar fibrosis: elongated fibroblasts separated by layers of dense extracellular matrix and / or concentric eosinophilic fibrosis around the epidermal ridges.
4. Perivascular lymphocytic infiltrate in the papillary dermis.
The degeneration of melanocytic nevi (natural event that occurs over time - called "old nevi") contributes to the cytologic appearance of melanocytic dysplasia and to misdiagnosis. However, the presence of severe dysplasia may lead to the diagnosis of melanoma in situ. Thus, Mihm and Barnhill attempted to develop a gradation consisting of six different diagnoses based on histology: (1) common melanocytic nevus, (2) melanocytic nevus with features of atypical nevus, (3) atypical nevus with minimal cytologic atypia, (4) atypical nevus with moderate cytologic atypia, (5) atypical nevus with severe atypia, (6) primary melanoma. Nonetheless, in this study diagnostic agreement among experienced pathologists ranged from 35% to 58%, while among less trained pathologists it ranged from 16% to 65%. 28 In a study by Brochez et al., a group of lesions histologically diagnosed as atypical nevi by a group of experienced pathologists was diagnosed as melanoma in 21% of the cases by another group of equally experienced pathologists.Thin melanomas and melanomas in situ were diagnosed as atypical nevi in 12% of the cases. 29 These studies reinforce the fact that the histological distinction between atypical nevus and melanoma is a challenge and that histological diagnosis is far from being considered the gold standard. 7.12
The creation of a diagnostic model based on major and minor criteria facilitated the histological diagnosis of atypical nevus and is, therefore, widely used. The major criteria are: 1. atypical nevomelanocytic proliferation in the basament membrane zone extending over three epidermal ridges in relation to the intradermal component; 2. intraepidermal melanocytic proliferation (lentiginous or epithelioid). The minor criteria are: 1. concentric eosinophilic fibrosis involving epidermal ridges or lamellar fibroplasia; 2. neovascularization; 3. dermal inflammatory response; 4. crest fusion. Diagnosis is established with two major and at least two minor criteria. 6
Clinicopathological Correlation
With regard to atypical nevi, the presence of clinical features of atypia is imperfectly correlated with the presence of histologic dysplasia. In a study with 101 patients with sporadic melanoma, the most atypical clinical lesion was surgically removed and the presence of dysplasia was histologically detected in 7% of the cases in which the lesion showed only one clinical feature of dysplasia, in 23% of the cases with two clinical characteristics of atypia and in 62% of the cases with three characteristics. 6.29
In a population study in Utah (Caucasians), Piepkorn et al. removed one or two atypical nevi of each patient and there was a 53% prevalence of histological dysplasia. They found a statistically significant relationship between the total number of melanocytic lesions and histological dysplasia. This suggests that melanocytic dysplasia is not randomly distributed, but that some people are predisposed to the appearance of these lesions. 6.30
In a series of 940 benign melanocytic nevi, Annessi et al. classified 53% as clinically benign and found 78% of histological atypicality, showing discrepancy between clinical and histopathological diagnosis. 25 In the same study, they divided the nevi into two groups: group A, consisting of nevi with a diameter between 3 and 5mm, and group B, with nevi larger than 5 mm. Comparing the two groups, a greater clinicopathological diagnostic discrepancy was observed (group A: 21% of clinical dysplasia and 68.3% of histological dysplasia, and group B: 67.4% of clinical dysplasia and 83.2% of histological dysplasia) in the group of lesions with smaller diameter (group A). They also established the sensitivity and specificity of the clinical diagnosis of atypical nevus: 58.4% and 66.6% respectively. 31
Molecular Biology
The occurrence of atypical nevi in patients with personal and family history of melanoma is frequent, a clinical syndrome known as familial melanoma. Due to this strong correlation, it has been admitted that the CDKN2A gene is nevogenic. 32 The characteristic phenotype described in these families is related to a high count of benign melanocytic nevi and atypical nevi. 33 In this syndrome the CDKN2A gene is the most involved. It is possible to detect germline mutations in affected patients at a frequency ranging from 20% to 40%, depending on the number of affected patients in the family, the average age at diagnosis, the occurrence of pancreatic carcinoma, tumors of the central nervous system and multiple primary melanoma. 34 The CDKN2A gene is considered a tumor suppressor gene, acting in the two checkpoints of the cell cycle through its primary transcripts, p16 and p14. p16 acts in the G1 restriction point and is encoded by exons 1a, 2 and 3, and its expression is regulated by an independent promoter. The second product of this gene, p14, acts in the G2 restriction point, is encoded by an alternative exon 1, exon1 b and exon 2, but in a different reading window, generating a protein without any similarity with p16. 35 In families studied in North America, Europe and Australia, the germline mutations responsible for the occurrence of melanoma and the phenotype of atypical nevus syndrome mainly affect exon 2, which is common to both transcripts of this gene, p16 and p14. 36 In a study by Leon et al. (2008), conducted at AC Camargo Hospital in Sao Paulo, Brazil, with 40 families, a mutation detection rate of 25%, was observed, and these mutations were found in similar frequencies in different regions of the gene (unpublished data). Another gene, CDK4, may also be associated with familial melanoma syndrome and, consequently, with the occurrence of atypical nevi, but of much less importance, involving about 1% of affected families. 37 In these cases, mutations are restricted to codon 24, with a substitution of histidine or cysteine for arginine, leading to inactivation of CDK4. 38 Germline mutations in this gene were not detected in Brazilian families (unpublished data).
Conduct
According to evidence, the clinical diagnosis of atypical nevi does not require histological confirmation. Despite the recognized association between atypical nevi and the risk of developing melanoma, most atypical nevi do not evolve to melanoma. Prophylactic excision of these nevi appears not to be cost-effective and may bring a false sense of security to the patient, since the risk remains. Some professionals justify excision of atypical nevi in difficult-to-monitor sites, but there are no published data that validate this conduct. 6Recommendations of how to approach the patient with atypical nevi were published by the NIH and by the Melanoma Working Group, the Netherlands (Table 1) and are widely used. 6.7
These recommendations include: 1. detailed personal history (information about any skin lesion, prior melanoma (s), non-melanoma skin cancer, excision of previous lesions and histopathological diagnosis, episodes of sunburn in childhood and / or adolescence); 2. family history of melanoma or atypical nevi (detailing the degree of kinship); 3. complete clinical examination of the skin including double-covered areas and scalp; 4. dermoscopic examination of all melanocytic lesions (total body mapping and digital dermoscopy monitoring, if available); 5. Guidelines for patients on sun exposure-avoid exposure during periods of peak intensity of UV rays, wear protective clothing, sunglasses and sunscreen with an SPF of 15 or more. 6
Patients with multiple atypical nevi are at increased risk of developing ocular melanoma. Thus, some professionals recommend that patients with atypical nevus syndrome undergo a complete eye examination once a year. 7 In the evaluation of patients with atypical nevi, it is also recommended that special attention be paid to family history of melanoma. When there is evidence of two or more cases of the disease in first or second degree relatives, the performance of genetic testing of the CDKN2A gene is indicated when available. 6
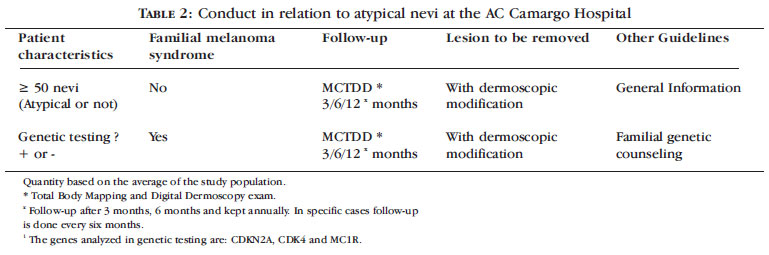
Rezze and Soares de Sá et al performed the follow-up of 254 patients at high risk of developing melanoma through total body mapping and digital dermoscopy at the Department of Cutaneous Oncology, AC Camargo Hospital, São Paulo, from 05/2003 to 11/2007 (unpublished data). A total of 13,038 melanocytic lesions was mapped (average of 50 lesions per patient) and, during follow-up, dermoscopic changes were found in 289 lesions. Of these, 225 were common melanocytic nevi, 46 were atypical nevi, 13 were non-melanocytic lesions and 5 were cutaneous melanoma lesions. The incidence of melanoma in the study population was 1.96 per 100 patients and in modified lesions, 1.73%. In this study it was possible to suggest that digital dermoscopy is a useful tool to monitor patients at high risk for developing cutaneous melanoma (among these, patients with atypical nevi), allowing the early diagnosis of this neoplasm, which is critical to the cure and prolonged survival of patients. Based on data obtained in this same study, a guide was developed for the monitoring of patients at high risk of developing melanoma (Table 2).39
REFERENCIAS
- 1. Clark Jr WH, Elder DE, Guerry Dt, Epstein MN, Greene MH, Van Horn M. A study of tumor progression: the precursor lesions of the superficila spreading and nodular melanoma. Hum Pathol. 1984;15:1147-65.
- 2. Greene MH, Clark WH Jr, Tucker MA, Elder DE, Kraemer KH, Fraser MC, et al. Precursor naevi in cutaneous malignant melanoma: a proposed nomenclature. Lancet. 1980;2:1024.
- 3. Elder DE, Goldman LI, Goldman SC, Greene MH, Clark WH Jr. Dysplastic nevus syndrome: a phenotypic association of sporadic cutaneous melanoma. Cancer. 1980;46:1787-94.
- 4. Ackerman AB, Magana-Garcia M. Naming acquired melanocytic nevi. Unna's, Miescher's, Spitz's Clark's. Am J Dermatopathol. 1990;12:193-209.
- 5. Fung MA. Terminology and management of dysplasticnevi: responses from 145 dermatologists. Arch Dermatol. 2003;139:1374-5.
- 6. Naeyaert JM, Brochez L. Clinical practice. Dysplastic nevi. N Engl J Med. 2003;349:2233-40.
- 7. NHI consensus Conference: diagnosis and treatment of early melanoma. JAMA. 1992;251:1314-09.
- 8. Hussein MR. Melanocytic dysplastic naevi occupy the middle ground between benign melanocytic naevi and cutaneous malignant melanomas: emerging clues. J Clin Pathol. 2005;58:453-6.
- 9. Skender-Kalnenas TM, English DR, Heenan PJ. Benign melanocytic lesions: risk markers or precursors of cutaneous melanoma? J Am Acad Dermatol. 1995;33:1000-7.
- 10. Li G, Herlyn M. Dynamics of intercellular communication during melanoma development. Mol Med Today. 2000;6:163-9.
- 11. Chammas R, Duarte APM, Otake AH, Costa M. Genética e biologia molecular do melanoma e de sua progressão. In: Oliveira Filho RS, editor. Melanoma cutâneo localizado e linfonodo sentinela. São Paulo: Lemar; 2003. p.1-19.
- 12. Roesch A, Burgdorf W, Stolz W, Landthaler M, Vogt T. Dermatoscopy of "dysplastic nevi": a beacon in diagnos tic darkness. Eur J Dermatol. 2006;16:479-93.
- 13. Hofmann-Wellenhof R, Blum A, Wolf IH, Piccolo D, Kerl H, Garbe C, Soyer HP. Dermoscopic classification of atypical melanocytic nevi (Clark nevi). Arch Dermatol. 2001;137:1575-80.
- 14. Arumi-Uria M. Dysplastic nevus: the eye of the hurricane. J Cutan Pathol. 2008;35 Suppl 2:16-9.
- 15. Platz A, Ringborg U, Hansson J. Hereditary cutaneous melanoma. Semin Cancer Biol. 2000;10:319-26.
- 16. Thomas NE, Edmiston SN, Alexander A, Millikan RC, Groben PA, Hao H, et al. Number of nevi and early-life ambient UV exposure are associated with BRAF-mutant melanoma. Cancer Epidemiol Biomarkers Prev. 2007;16:991-7.
- 17. Gandini S, Sera F, Cattaruzza MS, Pasquini P, Abeni D, Boyle P, et al. Meta-analysis of risk factors for cutaneous melanoma: I. Common and atypical naevi. Eur J Cancer. 2005;41:28-44.
- 18. Grulich AE, Bataille V, Swerdlow AJ, Newton-Bishop JA, Cuzick J, Hersey P, McCarthy WH. Naevi and pigmentary characteristics as risk factors for melanoma in a high-risk population: a case-control study in New South Wales, Australia. Int J Cancer. 1996;67:485-91.
- 19. Garbe C, Büttner P, Weiss J, Soyer HP, Stocker U, Krüger S, et al. Associated factors in the prevalence of more than 50 common melanocytic nevi, atypical melanocytic nevi, and actinic lentigines: multicenter case-control study of the Central Malignant Melanoma Registry of the German Dermatological Society. J Invest Dermatol. 1994;102:700-5.
- 20. Hofmann-Wellenhof R, Soyer PH. Atypical (Dysplastic) Nevus. In: Soyer HP, Argenziano G, Hofmann-Wellenhof R, Johr RH, ed. Color Atlas of Melanocytic Lesions of the Skin. v.1. Berlin: Springer-Verlag Berling Heidelberg; 2007. p87-96.
- 21. Pehamberger H, Steiner A, Wolff K. In vivo epiluminescence microscopy of pigmented skin lesions. I. Pattern analysis of pigmented skin lesions. J Am Acad Dermatol. 1987;17:571-83.
- 22. Soares de Sa BC, Rezze GG. Dermatoscopia dos nevos atípicos. In: Rezze GG, Soares de Sa BC, Neves RI, eds. Atlas de dermatoscopia Aplicada ("Atlas on Dermoscopy"). São Paulo: Lemar; 2004. v. 1. p. 103-7.
- 23. Rezze GG, Sá BCS, Neves RI. Dermatoscopia: o método de análise de padrões. An Bras Dermatol. 2006;81:261-8.
- 24. Fikrle T, Pizinger K. Dermatoscopic differences between atypical melanocytic naevi and thin melanomas. Melanoma Res. 2006;16:45-50.
- 25. Annessi G, Bono R, Sampogna F, Faraggiana T, Abeni D. Sesitivity, specificity and diagnostic accuracy of three dermoscopic algorithmic methods in the diagnosis of doubtful melanocytic lesions. J Am Acad Dermatol. 2007;56:759-67.
- 26. Elder DE. The dysplastic nevus. Phatology. 1985; 17: 291-7.
- 27. Clark Jr. WH, Reimer RR, Greene M, Ainsworth AM, Mastrangelo MJ. Origin of familial malignant-melanomas from heritable melanocytic lesions. The B-K mole syndrome. Arch Dermatol. 1978;114:732-8.
- 28. Duncan LM, Berwick M, Bruijn JA, Byers HR, Mihm MC, Barnhill RL. Histopathologic recognition and grading of dysplastic melanocytic nevi: an interobserver agreement study. J Invest Dermatol. 1993;100:S318-321.
- 29. Brochez L, Verhaeghe E, Grosshans E, Haneke E, Piérard G, Ruiter D, Naeyaert JM. Inter-observer variation in the histopathological diagnosis of clinically suspicious pigmented skin lesions. J Pathol. 2002;196:459-66.
- 30. Piepkorn M, Meyer LJ, Goldgar D, Seuchter SA, Cannon-Albright LA, Skolnick MH, Zone JJ. The dysplastic melanocytic nevus: a prevalent lesion that correlates poorly with clinical phenotype. J Am Acad Dermatol. 1989;20:407-15.
- 31. Annessi G, Cattaruzza MS, Abeni D, Baliva G, Laurenza M, Macchini V, et al. Correlation between clinical atypia and histologic dysplasia in acquired melanocytic nevi. Correlation between clinical atypia and histologic dysplasia in acquired melanocytic nevi. J Am Acad Dermatol. 2001;45:77-85.
- 32. Bertram CG, Gaut RM, Barrett JH, Pinney E, Whitaker L, Turner F, et al. An assessment of the CDKN2A variant Ala148Thr as a nevus/melanoma susceptibility allele. J Invest Dermatol. 2002;119:961-5.
- 33. Bataille V, Bishop JA, Sasieni P, Swerdlow AJ, Pinney E, Griffiths K, Cuzick J. Risk of cutaneous melanoma in relation to the numbers, types and sites of naevi: a case-control study. Br J Cancer. 1996;73:1605-11.
- 34. Goldstein AM, Chan M, Harland M, Gillanders EM, Hayward NK, Avril MF, et al. High-risk melanoma susceptibility genes and pancreatic cancer, neural system tumors, and uveal melanoma across GenoMEL. Cancer Res. 2006;66:9818-28.
- 35. Pho L, Grossman D, Leachman SA. Melanoma genetics: a review of genetic factors and clinical phenotypes in familial melanoma. Curr Opin Oncol. 2006;18:173-9.
- 36. Goldstein AM, Chan M, Harland M, Hayward NK, Demenais F, Bishop DT, et al. Features associated with germline CDKN2A mutations: a GenoMEL study of melanoma-prone families from three continents. J Med Genet. 2007;44:99-106.
- 37. Goldstein AM, Chidambaram A, Halpern A, Holly EA, Guerry IV D, Sagebiel R, et al. Rarity of CDK4 germline mutations in familial melanoma. Melanoma Res. 2002;12:51-5.
- 38. Majore S, De Simone P, Crisi A, Eibenschutz L, Binni F, Antigoni I, et al. CDKN2A/CDK4 molecular study on 155 Italian subjects with familial and/or primary multiple melanoma. Pigment Cell Res. 2008;21:209-11.
- 39. Rezze GG, Leon A. Nevo Displasico. In: Belfort FA, Wainstain AJA, eds. Melanoma diagnostico e tratamento. São Paulo: Lemar; 2009. v. 1. p. 99-110.
Dysplastic nevus (atypical nevus)
Publication Dates
-
Publication in this collection
27 Jan 2011 -
Date of issue
Dec 2010
History
-
Received
08 Nov 2009 -
Accepted
17 June 2010