Abstracts
FUNDAMENT: Psoriasis is a chronic inflammatory systemic disease mediated by immune factors. We will explore the foods that act on these factors contributing to psoriasis. As a systemic disease, which shares the same pathophysiological substrate with other comorbidities, diet also leads to worsening of comorbidities. OBJECTIVE: To indicate a group of foods that can act as a factor of manifestation and/or aggravation of psoriasis and, at the same time, enable strategies for individuals to introduce these foods to their diet. METHODS: 43 patients with various forms of psoriasis (excluding pustular and erythrodermic psoriasis) were selected and answered a questionnaire about their eating habits in the first visit, with special attention to the consumption of black coffee, black tea, chocolate, yerba mate, pepper, smoked foods, beef and flavor enhancer (monosodium glutamate). Next, the patient was instructed to suspend alcoholic drinks and tobacco. RESULTS: Beef is the most consumed food by patients followed by MSG (monosodium glutamate), which exists in processed foods, yerba matte, black coffee, chocolate, smoked foods, pepper and black tea. 88.37% noticed reduced scaling and erythema, milder outbreaks during the year and improved quality of life; 11.63% (5 patients) did not notice any effects on the skin. CONCLUSION: We found poor dietary intake in patients with psoriasis. In addition to receiving proper scientific advice, patients need to be educated regarding their eating habits for a better quality of life and as an adjuvant to the drug therapy.
Cacao; Capsicum; Glutamic acid; Meat; Psoriasis; Tea
FUNDAMENTOS: Psoríase é uma doença inflamatória crônica, sistêmica, mediada por fatores imunológicos. Serão abordados os alimentos que atuam sobre estes fatores, contribuindo para a psoríase. Como doença sistêmica, que compartilha o mesmo substrato fisiopatológico com outras comorbidades, a dieta alimentar também leva à piora das comorbidades. OBJETIVO: Destacar um grupo de alimentos que pode atuar como um fator de manifestação e/ou agravo da psoríase e ao mesmo tempo viabilizar estratégias individuais para alimentos que devam ser introduzidos. MÉTODOS: 43 pacientes foram selecionados, com diversas formas de psoríase (exceto psoríase pustulosa e eritrodérmica), e responderam a um questionário sobre seus hábitos alimentares na primeira consulta, focando a atenção no consumo de café preto, chá preto, chocolate, mate, pimenta, defumados, carne de gado e realçador de sabor (glutamato monossódico). Na sequência, o paciente foi orientado a suspender bebidas com qualquer percentagem alcoólica e o tabagismo. RESULTADO: A carne de gado é o alimento mais consumido pelos pacientes seguida por: realçador de sabor (glutamato monossódico), que existe nos alimentos industrializados, mate e café preto, chocolate, defumados, pimenta e chá preto. 88,37% notaram diminuição das escamas e eritema, surtos mais brandos durante o ano e melhora da qualidade de vida; 11,63% (5 pacientes) não notaram repercussões na pele. CONCLUSÃO: Foi observada ingestão dietética precária nos pacientes com psoríase. Além de uma orientação científica adequada, o paciente necessita de uma ação educativa frente aos seus hábitos alimentares para uma melhoria da qualidade de vida e como coadjuvante na terapia medicamentosa empregada.
Ácido glutâmico; Cacau; Capsicum; Carne; Chá; Psoríase
INVESTIGATION
Pilot study on which foods should be avoided by patients with psoriasis*
Moira Festugato
Dermatologist - Private practice - Caxias do Sul (RS), Brazil
Mailing address
ABSTRACT
FUNDAMENT: Psoriasis is a chronic inflammatory systemic disease mediated by immune factors. We will explore the foods that act on these factors contributing to psoriasis. As a systemic disease, which shares the same pathophysiological substrate with other comorbidities, diet also leads to worsening of comorbidities.
OBJECTIVE: To indicate a group of foods that can act as a factor of manifestation and/or aggravation of psoriasis and, at the same time, enable strategies for individuals to introduce these foods to their diet.
METHODS: 43 patients with various forms of psoriasis (excluding pustular and erythrodermic psoriasis) were selected and answered a questionnaire about their eating habits in the first visit, with special attention to the consumption of black coffee, black tea, chocolate, yerba mate, pepper, smoked foods, beef and flavor enhancer (monosodium glutamate). Next, the patient was instructed to suspend alcoholic drinks and tobacco.
RESULTS: Beef is the most consumed food by patients followed by MSG (monosodium glutamate), which exists in processed foods, yerba matte, black coffee, chocolate, smoked foods, pepper and black tea. 88.37% noticed reduced scaling and erythema, milder outbreaks during the year and improved quality of life; 11.63% (5 patients) did not notice any effects on the skin.
CONCLUSION: We found poor dietary intake in patients with psoriasis. In addition to receiving proper scientific advice, patients need to be educated regarding their eating habits for a better quality of life and as an adjuvant to the drug therapy.
Keywords: Cacao; Capsicum; Glutamic acid; Meat; Psoriasis; Tea
INTRODUCTION
Psoriasis is a chronic inflammatory autoimmune disease of multifactorial etiology and strong genetic basis. It affects the skin, nails, mucous membranes and joints and occurs in 0.2% to 4.8% of the world population.1 There are several clinical forms of the disease (high polymorphism). It often manifests by erythematous, scaly, well defined, and occasionally pruritic patches.2 Although there is no consensus about which factor triggers the disease, it is believed that the development and maintenance of the patches are due to T lymphocytes activated by antigen-presenting cells in the epidermis (Langerhans cells) and dermis (dendritic cells).3 The antigen-presenting cells, through cell-cell interaction, present antigens via Major Histocompatibility Complex (MHC) class I or II molecules to T lymphocytes (CD8 and CD4, respectively) and, with the help of co-stimulating molecules (CD80, CD86 and CD40), cause these lymphocytes to proliferate and secrete cytokines such as IL-2, IFNgamma and TNF-alpha. 3 These cytokines promote a cascade effect that culminates in the hyperproliferation of the epidermis and blood vessels, in addition to pro-inflammatory effects.3 The secretion of TNF-alpha and IFN-gamma by T lymphocytes activates keratinocytes, which start to secrete IL-8, a cytokine with potent chemotactic action, and this promotes neutrophil recruitment to the epidermis. 3
Once in the inflammatory site, neutrophils begin to release inflammatory mediators that act on endothelial cells causing them to express a higher number of adhesion molecules, allowing an increasing influx of leukocytes to the inflammation site and perpetuation of the inflammatory process. 3
TNF-alpha plays an important role in triggering the pathogenesis of psoriasis, since this cytokine is one of the first to be secreted by a large number of cells, mediating directly or through the induction of a cascade of other cytokines, such as IL- 1, the inflammatory process that is characteristic of psoriasis. 3
There is an epidemiological association of psoriasis, especially of its severe forms, with various diseases with which it shares a common pathogenic substrate, with implications of tumor necrosis factor alpha (TNF-alpha) and different target organs (such as arthritis and Crohn's disease, for example) as well as increased incidence of metabolic syndrome. This syndrome is characterized by the association of abdominal obesity, atherogenic dyslipidemia, hypertension, insulin resistance with or without glucose intolerance and a proinflammatory and prothrombotic state as risk factor for cardiovascular disease. 4
The correlation between nutrition and the development of several chronic diseases, particularly cardiovascular diseases among adults, has been studied since 1970, providing data on risk factors in the etiology of these illnesses, in particular improper eating habits, obesity and sedentarism; however, we lack studies showing the relationship between psoriasis and nutrition, listing the foods that may aggravate the existing morbid condition and/or trigger psoriasis. 5
The international conference held in 2006 in Rhodes, Greece, redefined the magnitude of the health risks associated with severe psoriasis and its comorbidities, while revealing its association with obesity. According to the authors, psoriasis and obesity have a common underlying pathophysiology, suggesting that obesity follows but does not precede psoriasis, and implying that the psoriatic inflammation contributes to the obese condition. Both share cytokines that contribute to the metabolic syndrome pattern. 6
MATERIAL AND METHODS
We selected 43 patients from the northeast region of Rio Grande do Sul, aged 14 to 82 years, of both sexes, presenting with disseminated or more localized lesions of psoriasis, with or without joint involvement. We chose not to exclude from the study patients using antihypertensive, antilipemic, and oral hypoglycemic medication.
Patients answered a questionnaire about their eating habits and described what they ate for breakfast, lunch, dinner and snacks with special attention to the consumption of black coffee, black tea, yerba mate, chocolate, pepper, smoked food, beef and food with flavor enhancers such as monosodium glutamate. In their first visit, patients were given a list of the most common drugs that should be avoided in psoriasis, such as acetylsalicylic acid, beta-blockers, diclofenac, omeprazole, etc, and were referred for possible drug therapy readjustment. Emulsions containing emollients with glyceryl monostearate only or in combination with sunflower oil 4% and shampoos (formulated with salicylic acid 3%, and coal tar - liquor carbonis detergens - 5%), when the scalp was affected, were prescribed.
Patients with more localized patches could apply liquor carbonis detergens cream 10% at night and an emollient lotion with glyceryl monostearate in the morning. Laboratory tests were required for patients with disseminated psoriasis and they were asked to undergo PUVA phototherapy.
Patients were initially assessed and advised on which foods should be avoided, with the recommendation for consumption of foods rich in beta carotene (provitamin A carotenoid that transforms into retinol). They were also advised to increase consumption of fruit, white meat and whole grains. Patients would then return six weeks later to report their impressions of the changes in their eating habits. They answered whether the new diet had contributed to lesion and quality of life improvement.
RESULTS
In this study, we observed an inadequate diet in patients with psoriasis. Ignorance of the importance of food and of having regular meals, ease of ready meals with excess calories and carbohydrates and lack of determination to reconcile work schedules and diet are factors that contribute to obesity, nutritional deficiency and worsening of psoriasis in these patients.
It was initially difficult for patients to break with the tradition of coffee, but they accepted this dietary restriction when they noticed the gradual improvement of lesions, even if associated with drug therapy. Another upside was greater physical and emotional energy. Decreased consumption of yerba mate represented by "chimarrão" and of all drinks containing caffeine contributed to the general well-being of patients, since these drinks were replaced by natural fruit juices. The introduction of beta-carotene-rich food changed not only the patients' diet but also that of their families, who started worrying about having vegetables and legumes at home. The patients, who were company employees and therefore had to eat what they were offered by the company, began to choose better foods and eat morning and afternoon snacks, such as cereal bars and fruit.
Beef and barbecue were not prohibited, but their frequency was decreased. Patients were advised to choose stews that preserved most of the cooking water during preparation. The consumption of chicken and fish was encouraged since their fat content and calories are lower.
As for age 4.65% were between 10-19 years, 16.28% were between 20-29 years, 27.91% were between 30-39 years, 20.93% were between 40-49 years, 20.93% were between 50-59 years, 6.98% were between 60-69 years, and 2.32% were between 80 to 89 years (Graph 1). We saw more male (60.46%) than female (39.54%) patients.
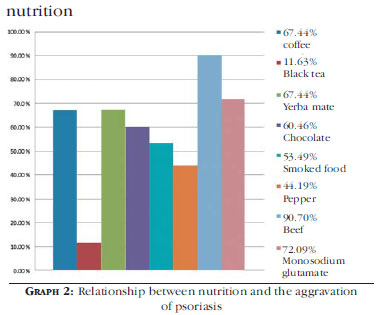
The most consumed foods were beef (90.70%), followed by monosodium glutamate (72.09%), yerba mate (67.44%), black coffee (67.44%), chocolate (62.79 %), smoked food (53.49%), pepper (44.19%) and black tea (11.63%) (Graph 2).
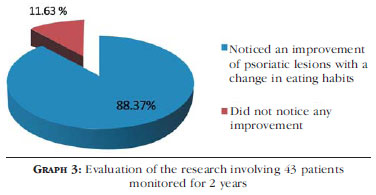
Of the 43 patients who were evaluated over 2 years, 88.37% had positive results with the change of eating habits. The positive aspects were reduction of erythema and scaling, milder outbreaks, with further delay in the onset of lesions during the year, and improved quality of life; 11.63% (5 patients) did not notice any effects on the skin, although one patient was using acetylsalicylic acid and enalapril (Graph 3).
DISCUSSION
Pepper is a capsaicinoid that adds spicy flavor, aroma and heat to food. There are many analogous substances, but capsaicin and nonivamide are the most powerful and most spicy. 7 In the form of oleoresin capsicum, they are classified as GRAS (Generally Regarded As Safe ) by the FDA and approved as food additives or topical analgesics without a toxicity profile; however, the metabolism of capsaicinoids raises questions about toxicity and the potential to increase the risk of certain cancers.7 The metabolism of capsaicin is done by the microsomal enzyme system (cytochrome P450) and produces several metabolites that bind to macromolecules in the body, and different pathological effects may occur. 7
An experimental study showed that consumption of red pepper produced exfoliation and nitrosation of the intestinal epithelium of rats because it has nitrophenols, resulting in chronic inflammatory bowel disease with neutrophil infiltrate and transepithelial neutrophil migration as markers. 8.9 Evidence shows that induction of IL8 in the intestinal epithelium acts as a trigger factor for neutrophil recruitment in several types of inflammation, including colitis and psoriasis. 9
Studies of the colonic mucosa in rats showed that, under the action of capsaicin, there was increased expression of transforming growth factor (TGF).10 The transforming growth factor TGF-alpha is a polypeptide produced by keratinocytes whereas the epidermal growth factor is produced anywhere in the body. Both bind to the same active receptor tyrosine kinase in the basal and immediately suprabasal epidermis to stimulate cell proliferation. 11.12 In psoriasis, the keratinocyte intensely participates in the multicellular and multimolecular activation of the network coordinated by cytokines. 11
Caffeine, theophylline and theobromine are three alkaloids found in plants distributed throughout the world. They are called methylxanthines or simply xanthines and are important constituents of coffee, tea, and cocoa. 13
Caffeine (1,2,7 trimethylxanthine) is one of the three methylxanthines present in coffee - in addition to theophylline and theobromine. They are associated with a number of chemicals, among them diterpenes (cafestol and kahweol) and phenolic compounds (chlorogenic acid). 13.14
Caffeine is quickly and completely absorbed in the digestive tract, being distributed to all body tissues. It has a half-life of 2.5 to 10 hours. Plasma concentrations are reached within one hour. Metabolism occurs mainly in the liver; 95% of its transformation occurs in cytochrome P 450, originating more than 25 metabolites, while the remaining 5% are excreted in urine. 14
The main mechanism of action of caffeine is due to its structural similarity with the adenosine molecule, being able to bind to its receptors (A1, A2A), blocking them and, therefore, having a stimulating action. 15
In addition to being an antagonist of adenosine receptors, caffeine in high concentrations can act in vivo as an inhibitor of cAMP phosphodiesterase and can increase immunosuppressive cAMP content in immune cells, as demonstrated in a study by Sitkovsky et al. 13.16 Caffeine has pro-inflammatory effects when administered in the presence of an acute inflammatory process in rats, increasing tissue damage evidenced by increased mRNA levels of TNF-alpha, TNF-beta, lymphotoxin-beta, IL-6 and IFN-gamma in the spleen and increased IFN-gamma in peripheral blood. 16 Cytokines such as IFN-gamma act as mitogens for keratinocytes in psoriasis. 2
Cafestol and kahmeol with hypercholesterolemic activity were identified in the lipid portion of coffee. This action may contribute to increase LDL cholesterol (low density lipoprotein). 14-15
In the basal cells of the epidermis there are endocytic receptors that capture the cholesterol that is systemically supplied. In psoriasis LDL receptors are not only present in basal cells but also in suprabasal cells that express hyperproliferation markers. 17 Caffeine is highly soluble in lipids 13.
The caffeine and chlorogenic acid present in coffee contribute to increased homocysteine, which is a risk factor for cardiovascular disease. 14 Homocysteine requires folic acid, vitamin B12 and vitamin B6, which act as cofactors for its metabolism.14-15 In psoriasis, plasma homocysteine is increased and correlates directly with the PASI score and inversely with folic acid levels; there is probably an increase in consumption or a decrease in absorption.4
Other drinks containing caffeine include guarana, yerba mate and cola-flavored drinks. 13 Cocoa and chocolate contain theobromine and some caffeine. 13
At least half of the world population consumes tea. 13 Prepared from the leaves of Thea sinensis, tea contains caffeine, theobromine and theophylline. 13 Theophylline has the ability to enhance epidermal keratinization, possibly by restricting the activity of the gene associated with proliferation, and to strengthen activities related to the maturation and cell differentiation of normal or psoriatic epidermis. 18
Monosodium glutamate (MSG) is the sodium salt of glutamic acid. Together with aspartic acid, it is particularly important in the synthesis of purines as nitrogen donors.19.20
Glutamic acid is one of the most common amino acids found in nature. 19 It is produced in many countries today by a fermentation process of molasses from sugar cane or sugar beet, as well as corn starch and corn syrup. It is used as flavor enhancer. 19 In 1958 the U.S. Food and Drug Administration (FDA) designated MSG as a GRAS (Generally Recognized As Safe) substance. 19
Its use is permitted by the Brazilian legislation (RDC Resolution No. 1, January 2, 2001, of ANVISA). Its use in food corresponds to "quantum satis", ie, the amount required to give food the desired flavor. 21
There are not many scientific studies that emphasize the health benefits or harmful effects of MSG, but it is related to heat production. There are no studies that relate it to psoriasis, except for the fact that it contributes to the formation of adenine derivatives (AMP), hypoxanthine derivatives (ADP and ATP) with energy production. 20
Smoked food and barbecue are impregnated with tar from coal smoke. Tar is also found in cigarettes and has known carcinogenic action. 22 Tar is the name given to a complex mixture of about 4,000 chemical compounds that are formed during burning, due to the incomplete combustion of organic materials present in cigarettes, cigars, cigarillos or any other smoking product. 22
Cigarette smoking leads to functional and morphological alterations in polymorphonuclear leukocytes and it can also cause a higher release of chemotactic factors, with an increase of interleukin 1 B, TNF-alpha and transforming growth factor-beta, which have been associated with the severity of psoriasis. The severity of the disease is related to the number of cigarettes smoked per day. 23 In this study, Fortes emphasizes the importance of advising the patient to stop smoking not to worsen psoriasis. The same could be done in relation to smoked food.
According to the study, beef was the most consumed food by patients with psoriasis. What differentiates beef from other meats is the quantity of iron, which would be involved in the synthesis of hydroxyl radicals that cause damage to intestinal cells, its caloric value, water content and the amount of fat. 24.25
Meat, when cooked or roasted, loses water during preparation, increasing its fat content, protein and caloric value. 25 Water reduction is more pronounced in dried beef and the caloric value greatly increases.25
Cooking methods that use high temperatures and low humidity (frying, roasting, grilling), especially food high in lipids, contribute to the high dietary content of AGEs-Advanced glycated-End Products, which damage cells and bind to specific receptors, causing the production of inflammatory cytokines and growth factors. 26
The study by Vlassara demonstrates the effects of AGEs in the vascular pathology of diabetes and the formation of inflammatory mediators (TNF-alpha, IL1B, IL6 and vascular adhesion molecules). 27 Further studies that address the implications of AGEs in skin diseases such as psoriasis are needed, but we can infer that cooked or roasted meat loses water during preparation, increasing its fat and protein content and becoming a source of proinflammatory AGEs. 25.27
Psoriasis and obesity share similar mediators of inflammation such as TNF-alpha and IL-6, and the generators of adipocytes and psoriatic inflammation the adipocyte and the macrophage, respectively - have a common mesothelial origin. 6 Serum TNF-alpha increases with a higher total body mass index (BMI), induces an increase in free fatty acids and a further increase in insulin resistance. It also increases PAI-1 levels, resulting in stimulation of coagulation. Similarly, IL-6 can induce insulin resistance, increased endothelial adhesion molecules, hepatic release of fibrinogen and C-reactive protein, and increased procoagulant effects on platelets leading to atherosclerosis.6
The level of eicosapentaenoic acid (EPA), 20:5, omega-3 in meat and lactic lipids is negligible and contains higher amounts of arachidonic acid (AA), 20:4, omega-6. 28 AA, an omega-6 polyunsaturated fatty acid, is responsible for the production of eicosanoids (prostaglandins and leukotrienes), which are mediators of the inflammatory process through the lipoxygenase and cyclooxygenase pathways. 29 Leukotrienes, especially leukotriene B4 (LTB4), derived from AA, induces the stimulation of DNA synthesis in cultured human keratinocytes. Studies show that in plaque psoriasis there are increased lipoxygenase products of AA, particularly LTB4. 28
Fish oil contains large amounts of EPA and docosahexaenoic acid (DCHA), 22:6, omega-3, which inhibits prostaglandin synthesis, but it has a negligible effect on the leukotriene pathway. 28 Eicosapentaenoic acid (EPA) is responsible for the production of leukotriene B5, which is a less potent mediator for neutrophil chemotaxis and less potent stimulator of keratinocyte proliferation, inhibiting the conversion of AA to LTB4 by human neutrophils. 28
EPA competes with AA at the substrate for incorporation into phospholipids in the membrane, thus decreasing the metabolism of AA into eicosanoids. 28 A diet rich in omega-3 polyunsaturated fatty acid, EPA and DCHA, and low in omega-6 polyunsaturated fatty acid , AA, may improve lesions of psoriasis. 28
CONCLUSION
No study to date has addressed thermogenic food as a factor that worsens psoriasis and that, therefore, should be avoided. It is recommended that the dermatologist guide the patient's diet so that the family can get involved and, together, change their eating habits. Although there is no way to quantify the reduction of erythema and scales and prove the correlation between dietary change and psoriasis, it became clear that the suspension of these food products and the introduction of healthy eating habits contributed to a better treatment outcome.
REFERENCES
- 1. Miot HA. Genética da Psoríase. In: Romiti R, editor. Novos Conceitos em Psoríase. Rio de Janeiro: Elsevier; 2009. p.32-38.
- 2. Sociedade Brasileira de Dermatologia. Consenso Brasileiro de Psoríase e Guias de Tratamento. Rio de Janeiro: Sociedade Brasileira de Dermatologia; 2006. p.3.
- 3. Bernard G, Silva LCR. Imunologia da Psoríase. In: Romiti R, editor. Novos Conceitos em Psoríase. Rio de Janeiro: Elsevier; 2009. p. 17-27.
- 4. Puig-Sanz L. La psoriasis, una efermedad sistémica? Actas Dermosifiliogr. 2007;98: 396-402.
- 5. Rodrigues UTFM. Revisão sistemática sobre a ação do chocolate, chá, vinho tinto e café na saúde cardiovascular. Revista Brasileira de Obesidade, Nutrição e Emagrecimento. 2007;1:36-46.
- 6. Sterry W, Strober BE, Menter A; International Psoriasis Council. Obesity in psoriasis: the metabolic, clinical and therapeutic implications. Report of an interdisciplinary conference and review. Br J Dermatol. 2007;157: 649-55.
- 7. Reilly CA, Yost GS. Metabolism of capsaicinoids by P450 enzymes. Drug Metab Rev. 2006;38:685-706.
- 8. Gupta PJ. Efeito da pimenta vermelha nos sintomas de pacientes com fissuras anais agudas. Arq Gatroenterol. 2008;45:124-7.
- 9. Kucharzik T, Hudson JT, Lügering A, Abbas JA, Bettini M, Lake JG, et al. Acute induction of human IL-8 production by intestinal epithelium triggers neutrophil infiltration without mucosae injury. Gut. 2005;54:1565-72.
- 10. Hoffmann P, Mazurkiewicz J, Holtmann G, Gerken G, Eysselein VE, Goebell H. Capsaicin - sensitive nerve fibres induce epithelial cell proliferation, inflamatory cell immigration and transforming growth factor- alpha expression in the rat colonic mucosa in vivo. Scand J Gastroenterol. 2002;37:414-22.
- 11. Nickoloff JB. The cytokine network in psoriasis. Arch Dermatol. 1991;127:871-84.
- 12. Haak AR, Holbrook K. The Structure and Development of Skin. In: Fitzpatrick TB, Freedberg IM, Eisen AZ, Wolff K, Austen KF, Goldsmith LA et al. Dermatology in General Medicine. New York: McGraw-Hill; 1999. p. 70-113.
- 13. Rall TW. Excitantes do Sistema Nervoso Central. In: Gilman AG, Goodman LS, Gilman. As Bases Farmacológicas da Terapêutica. 6 ed. Rio de Janeiro. Guanabara Koogan; 1983. p.519-32.
- 14. Gotteland M, Pablo VS. Algunas verdades sobre el café . Rev Chil Nutr. 2007; 34:105-15.
- 15. Alves RC, Casa S, Oliveira B. Benefícios do café na saúde: mito ou realidade? Quim Nova. 2009;32:2169-80.
- 16. Ohta A, Lukashev D, Jackson EK, Fredholm BB, Sitkovsky M. 1,3,7- Trimethylxantine (caffeine) may exacerbate acute inflammatory liver injury by weakening the physiological immunosupressive mechanism. J Immunol. 2007;179: 7431-8.
- 17. Taube MBP, Taib A. Metabolismo lipídico na cultura de queratinócitos. An Bras Dermatol. 2000;75:75-84.
- 18. Chopra DP. Effects of theophyline and dibutyryl cyclic AMP on proliferation and keratinization of human keratinocytes. Br J Dermatol. 1977;96:255-62.
- 19. International Food Information Council Foundation - IFIC Review on monosodium glutamate: Examining the myths. 1994. [cited 2010 Sept 23]. Available from: http://www.foodinsight.org/Content/76/Glutamate-and-Monosodium-Glutamate.pdf
- 20. Harper HA. Manual de Química Fisiológica. Rio de Janeiro: Atheneu; 1968. p.299-322.
-
21Ministério da Saúde. Agência Nacional de Vigilância Sanitária. Legislação. Resolução -RDC n.1, de 2 janeiro de 2001.
-
22International Agency for Reserch on Cancer - IARC. [Internet]. International Agency for reserch on cancer onographs on the evaluation of the carcinogenic risk of chemicals to humans - tobacco smoking - 1986; vol 38. [cited 2010 May 10]. Available from: Disponível em: http://monographs.iarc.fr/ENG/Monographs/PDFs/index.php
- 23. Fortes C. Relationship between smoking and the clinical severity of psoriasis. Arch Dermatol. 2005;141:1580-84.
- 24. Inca.gov. Instituto Nacional do Câncer [Internet]. Falando sobre o câncer do intestino. [acesso 13 Abr. 2010]. Disponível em: http://www.inca.gov.br/publicaçőes/Falando_sobre_Cancer_de_Intestino.pdf
- 25. Barbosa JHP, Oliveira SL, Seara LT. O papel dos produtos finais da glicação avançada (AGEs) no desencadeamento das complicações vasculares do diabetes. Arq Bras Endocrinol Metabol. 2008;52:940-50.
- 26. Vlassara H, Cai W, Crandall J, Goldberg T, Oberstein R, Dardaine V, et al. Inflamatory mediators are induced by dietary glycotoxins, a major risk factor for diabetic angiopathy. Proc Natl Acad Sci U S A. 2002;99:15596-601.
- 27. Roça RO. Composição química da carne. In: Universidade Estadual Paulista "Júlio de Mesquita Filho" - UNESP. Departamento de Gestão e Tecnologia Agroindustrial. Artigos técnicos, científicos e teses. [acesso 20 Jun. 2010]. Disponível em: http://dgta.fca.unesp.br/carnes/Artigos%20Tecnicos/Rocal02.pdf .
- 28. Ziboh VA, Cohen KA, Ellis CN, Miller C. Effects of dietary supplementation of fish oil on neutrophil and epidermal fatty acids. Arch Dermatol. 1986;122: 1277-82.
- 29. Isseroff RR, Davis CA. Fish again for dinner! The role of fish and other dietary oils in the therapy of skin disease. J AM Acad Dermatol. 1988;19:1073-80.
Publication Dates
-
Publication in this collection
23 Jan 2012 -
Date of issue
Dec 2011
History
-
Received
25 Oct 2010 -
Accepted
03 Dec 2010