Abstract:
Background:
Venous ulcers represent 70% of the lower limb ulcers. They are difficult to heal, requiring a correct diagnostic and therapeutic approach. Many products have been developed to healing, such as homologous platelet gel obtained from the platelet concentrate exceeding from blood transfusion.
Objective:
To evaluate the safety and efficacy of homologous platelet gel in venous ulcers compared with hydrocolloid dressing.
Method:
A pilot randomized clinical trial in patients with venous ulcers. Randomized groups (homologous platelet gel and hydrocolloid groups) were followed for 90 days and were assessed through the evolution of ulcerated area, qualitative analysis of vascularization and adverse events. Both groups used elastic compression.
Results:
We included 16 participants, with a total of 21 venous ulcers. Both treatments promoted a reduction of the areas of the ulcers in 90 days (mean 69%), there was significant difference between the groups concerning the gradual reduction of the ulcers areas, favorably to the hydrocolloid (70% vs 64%; p <0.01). There were some mild adverse events in both groups.
Study limitations:
Single-center study with a small number of patients, preventing more accurate assessment of the effects of platelet gel.
Conclusion:
The homologous platelet gel associated with the elastic compression can be an alternative to the venous ulcer treatment and is safe due to the occurrence of a few mild local adverse events and no serious adverse events. Clinical trials with larger numbers of patients must be performed to maintain the indication of this treatment for venous ulcer.
Keywords:
Wound healing, hydrocolloid bandages; Platelet-Rich plasma; Varicose ulcer
INTRODUCTION
Venous ulcers (VUs) represent 70% of lower limb ulcers, with a prevalence of 0.06 to 2% in the population.11 O'Donnell TF Jr, Passman MA, Marston WA, Ennis WJ, Dalsing M, Kistner RL, et al. Management of venous leg ulcers : Clinical practice guidelines of the Society for Vascular Surgery and the American Venous Forum. J Vasc Surg. 2014;60:3S-59S. They constitute a serious public health problem, which contributes to burden public spending in the Brazilian public healthcare system, as well as interfering in the quality of life of the population due to complications that may result in significant morbidity.22 Dias TYAF, Costa IKF, Melo MDM, Torres SMSGSO, Maia EMC, Torres GV. Avaliação da qualidade de vida de pacientes com e sem úlcera venosa. Rev Latino-Am Enferm. 2014;22:576-81. Despite the high prevalence and its importance, it is often neglected and approached inappropriately.33 Abbade LPF, Lastória S. Abordagem de pacientes com úlcera da perna de etiologia venosa. An Bras Dermatol. 2006;81:509-22. Studies aiming at rapid healing strategies are important.
Due to the difficult healing, VUs need a correct diagnostic and therapeutic approach. In this sense, many products have been developed for healing, such as the homologous platelet gel (PG), obtained from the concentrated blood platelet, exceeding from blood transfusion.44 Moroz A, Deffune E. Platelet-rich plasma and chronic wounds: remaining fibronectin may influence matrix remodeling and regeneration success. Cytotherapy. 2013;15:1436-9. This strategy aims to obtain an effective dressing by means of the release of growth factors in the wound bed, with the advantage of having low cost.
This study aimed to evaluate the safety and efficacy of homologous PG in VU comparing it with the hydrocolloid dressing.
METHODS
This is a pilot study of a randomized and prospective clinical trial design, with participants with VUs, conducted at the Department of Dermatology of the Clinics Hospital of Botucatu Medical School - Unesp between March/2010 and January/2011. It was approved by the Research Ethics Committee of the Botucatu Medical School - Unesp (nº 538/08). All participants accepted and signed the Informed Consent Form.
The inclusion criteria were: participants aged >18 years with VU, presence of good granulation tissue, defined as having a bright red color. Exclusion criteria were: presence of associated peripheral arterial disease, characterized when the ankle-brachial index was lower than 0.9, history of osteomyelitis and presence of critical colonization/ infection in the wound bed.
Randomization was generated using Microsoft Excel and, subsequently, matched envelopes were made, identified numerically and opened in sequential order at the time of inclusion of the patient. VUs were randomized and divided into groups using platelet gel (PG) and hydrocolloid dressing (HC). Patients from both groups received guidance for daily use of compressive elastic band (Surepress®).
Participants were followed up for 90 days, with monthly assessments (D0, D30, D60, D90), conduction of clinical consultations and measurement of evolution of the ulcer area through standardized digital photography.55 Miot HA, Mendaçolli TJ, Costa SV, Haddad GR, Abbade LPF. Úlceras crônicas dos membros inferiores: avaliação pela fotografia digital. Rev Assoc Med Bras. 2009;55:145-8.
Photographs were made with Sony Cyber-shot digital cameraTM 7.2 megapixels, at a fixed distance of approximately 15 cm from the injured area. Lesions were identified with a label (1.2 x 2.6 cm), which served as reference point in centimeters for the edge border design and area calculation. Measurement was performed using the freely available Image J software, available at: http://rsbweb.nih.gov/ij/ (Figure 1).
Vascularization was evaluated by anatomopathological examination at D0 and D90 for completely or exulcerated unhealed ulcers, with a 4 mm dermatological punch at the margin of the lesion. For histological analysis, hematoxylin-eosin (HE) staining and immunohistochemistry (IHC) were conducted. Anti-CD34 monoclonal antibody diluted 1: 150 (QBEnd 10) was used as the primary antibody for IHC reactions. Sections were washed in PBS, incubated for 30 minutes with biotinylated secondary antibody, followed by 30 minutes of incubation with streptavidin peroxidase complex (LSAB, DAKO, Carpinteria, CA, USA). Labeling was performed by incubating the slides with 3.3'-diaminobenzidine (DAB) and stained with hematoxylin. For CD34 immunostaining analysis, a comparison of the variation of the IHC scores by the chi-square test of the trend from the bootstrap simulation was performed from 10,000 simulations.
Dressing of both groups included cleaning with saline 0.9%, heated in jet application of the covering according to randomization, secondary dressing and elastic compression band (Surepress®). The secondary dressing was changed daily at home and the primary dressing of both groups was changed every four days at home or at a Basic Health Unit. For use of the hydrocolloid dressing, 10 x 10 cm plates of the brands Coloplast®, Curatec® and Casex® were used, according to availability on the Service.
PG was produced from a pool of the concentrated blood platelet group, validated as an intravenous blood product, in compliance with all legal requirements of the National Health Surveillance Agency (ANVISA). The blood components were surplus of the transfusion practice, being obtained by voluntary blood donation from the Hemotherapy service, and, therefore, presenting serological and microbiological control.
PG has a laminar shape, that is, it has the appearance of a membrane and a gelatinous consistency, which is formed when thrombin and calcium gluconate are added to the platelet rich plasma (PRP) under sterile conditions supplemented with the antioxidants agents (Figure 2). It was handled in a Laminar Flow Cabinet and then stored at 4°C in Petri dishes.
Morphology of vessels (A, B and C - general characteristic of lesions in D0). A: vascular proliferation, ++ / ++++ intensity in the papillary dermis, arrangement tending to non-cohesive (Hematoxylin & eosin X200); B: Marking by CD34, evidencing vessel in the papillary dermis (IHC X200); C: Vessels in detail with irregular lights, tending to be disposed at 90º in the epidermis, with few endothelial cells (Hematoxylin & eosin X400); D, E, F: vessel morphology of lesions of the group treated with HC in D90 (Hematoxylin & eosin X200, IHC X200 and Hematoxylin & eosin X400); G, H and I: vessel morphology of lesions of the group treated with PG in D90 (Hematoxylin & eosin X200, IHC X200 and Hematoxylin & eosin X400)
The evaluated endpoints were: safety of the use of the products through possible local adverse events, such as pain (through the Numerical Visual Scale), contact dermatitis, critical colonization/ infection and increased ulceration. Percentage of reduction of the ulcerated area (calculated from the initial area subtracted from the final area and divided by the initial area), angiogenesis and characteristics of each dressing (patient compliance, dressing changing time, ease of manipulation in both application and removal of the dressing, appearance of lesion and exudate edges, and wrapping).
Categorical variables were represented by their percentage proportions and compared between the groups by Fisher's exact test or trend chi-square with exact probability.
Continuous variables were represented by their means and standard deviations or medians and quartiles (p25-p75), if normality was not identified by the Shapiro-Wilk test. Groups were compared by Student's t-test or Mann-Whitney, if indicated.
Longitudinal variables were evaluated according to the intention to treat population. Groups were compared through a generalized linear model of mixed effects, with unstructured covariance structure, gamma probability distribution (identity) and robust analysis. Experimental unit of analysis used was the ulcerated area in cm2.
Data were analyzed in IBM SPSS 22.0 software. Statistical significance was determined at 5%.
RESULTS
Sixteen participants were included, with a total of 21 VUs: 9 ulcers were randomized to the PG group and 12 to the HC group. Demographic and clinical characteristics of patients and characteristics of lesions are presented in table 1.
Demographic and clinical characteristics of PG and HC participants and lesion characteristics
In the studied population, women (63%) predominated, with a mean age of 62.3 years, with systemic arterial hypertension being the most frequent comorbidity (50%). The history of ulcer recurrence was 76%.
Lesions had a median of 24 months of active ulcer time, predominantly on the lateral side (62%) and thicker margins (62%). Lower limb edema was the most frequent perilesional characteristic (63%). The groups did not differ in the initial size of the ulcers (p = 0.13), nor their duration (p = 0.81).
It should be noted the homogeneity in clinical and demographic parameters (except age), the reduction of areas in both groups, the absence of effects due to treatments and the low percentage of adverse events at follow-up (Table 2).
Evolution of ulcer areas and local adverse events during follow-up of PG and HC participants
There were no ulcers at the time evaluated that presented complete healing.
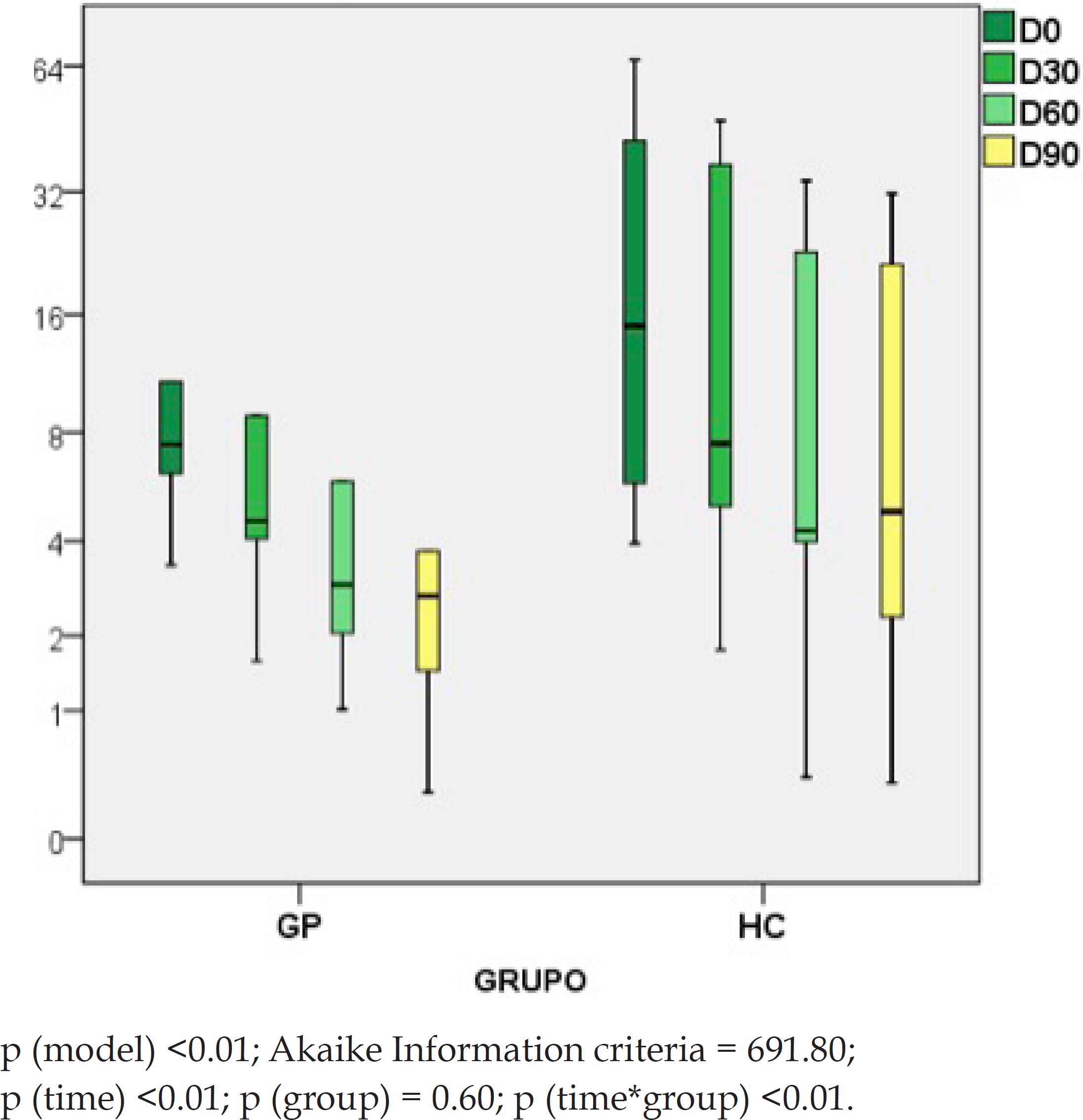
Individual measurements of VU area at each visit are shown in graph 1. Both treatments promoted a significant reduction (p<0.01) in the areas of the ulcers in 90 days (mean 69%) and there was a significant difference between the groups regarding progressive reduction of ulcer areas favoring HC (70% vs. 64%, p<0.01).
When adjusted for age, varicose veins and recurrence of ulcers, variables in which the groups were different at p<0.20, the statistical difference between the groups (p <0.01) was maintained, favoring treatment with HC.
Regarding neovascularization, biopsies of 9 participants were analyzed, totaling 10 VUs, 7 of the HC group and 3 of the PG group, who remained in the study until D90 (Figure 2). There was no difference between the groups in the evaluation of the scores between D0 and D90 (p = 0.67).
Observed characteristics of both dressings were described in chart 1.
DISCUSSION
This pilot study, although with a small number of patients, was randomized, with a control group and obeying the methodology of clinical trials to evaluate the efficacy and safety of VU treatment.
PG showed to be an alternative in the treatment of VUs. Although the area reduction percentage of the PG group was lower than that of the HC group, PG also presented a good percentage of area reduction, approximately 64%, which has been reported in other studies in the treatment of VUs. A study by Scotton et al.66 Scotton MF, Miot HA, Abbade LP. Factors that influence healing of chronic venous leg ulcers : a retrospective cohort. An Bras Dermatol. 2014;89:414-22. with 137 VUs verified an area reduction of 50% or more using different dressings associated with the use of elastic compression.
HC group presented an area reduction of 70%. In a systematic review of 42 studies of VU treatment, no statistically significant difference was found in relation to ulcer healing regardless of the dressing used, and the use of the hydrocolloid associated with elastic compression was not beneficial when compared with simple dressing with low adherence.77 Palfreyman S, Nelson EA, Michaels JA. Dressings for venous leg ulcers : systematic review and meta-analysis. BMJ. 2007;335:244.
On the use of homologous PG, there are few studies on venous ulcers. Bernuzzi et al.88 Bernuzzi G, Tardito S, Bussolati O, Adorni D, Cantarelli S, Fagnoni F, et al. Platelet gel in the treatment of cutaneous ulcers: the experience of the Immunohaematology and Transfusion Centre of Parma. Blood Transfus. 2010;8:237-47., using autologous and homologous PG in ulcers of different etiologies, verified area reduction of >50% in the period of 96 days. Frykberg et al.99 Frykberg RG, Driver VR, Carman D, Lucero B, Borris-Hale C, Fylling CP, et al. Chronic wounds treated with a physiologically relevant concentration of platelet-rich plasma gel: a prospective case series. Ostomy Wound Manage. 2010;56:36-44., using autologous platelet-rich plasma in ulcers of different etiologies, found improvement in 97% of the lesions, with a mean area reduction of 39.5%. In another similar study, with the use of autologous platelet-rich plasma, the area reduction found was 47.5% in 86.3% of lesions. 1010 de Leon JM, Driver VR, Fylling CP, Carter MJ, Anderson C, Wilson J, et al. The clinical relevance of treating chronic wounds with an enhanced near-physiological concentration of platelet-rich plasma gel. Adv Skin Wound Care. 2011;24:357-68.
The healing effects of PG can be attributed to the release of innumerable growth factors after platelet activation, which suggests that the therapy is similar to natural healing by the application of multiple growth factors in a biological way.1111 Shan GQ, Zhang YN, Ma J, Li YH, Zuo DM, Qiu JL, et al. Evaluation of the effects of homologous platelet gel on healing lower extremity wounds in patients with diabetes. Int J Low Extrem Wounds. 2013;12:22-9. A variety of growth factors have been reported as participant in the tissue repair process, including EGF, PDGF, FGF, TGF-β1 and IGF-1. 1212 Boateng JS, Matthews KH, Stevens HN, Eccleston GM. Wound healing dressings and drug delivery systems: a review. J Pharm Sci. 2008;97:2892-923.
Choosing to use the homologous PG is due to its low cost, since it uses surplus platelet of blood transfusion, and also due to the comfort to the patient. Shan et al.1111 Shan GQ, Zhang YN, Ma J, Li YH, Zuo DM, Qiu JL, et al. Evaluation of the effects of homologous platelet gel on healing lower extremity wounds in patients with diabetes. Int J Low Extrem Wounds. 2013;12:22-9. reported that the preparation and storage of autologous PG limit its clinical use, because in addition to being uncomfortable to the patient, for each application it is necessary a new blood collection, which stimulates the homologous PG proposal.
Demographic and clinical characteristics were homogeneous between the groups, except for age, and agreed with those reported in previous studies, such as women predominance, age around 60 years and hypertension as the more frequent comorbidity. 1313 de Almeida Medeiros AB, de Queiroz Frazão CM, de Sá Tinôco JD, Nunes de Paiva Md, de Oliveira Lopes MV, Brandão de Carvalho Lira AL. Venous ulcer: risk factors and the Nursing Outcomes Classification. Invest Educ Enferm. 2014;32:252-9.
Lesion and perilesional characteristics, as well as the history of ulcer recurrence, are those commonly found in patients with VUs. In general, VUs have an irregular shape, with well-defined borders, which can become deep with yellowish exudate, and present eczema, ocher dermatitis and perilesional lipodermosclerosis.33 Abbade LPF, Lastória S. Abordagem de pacientes com úlcera da perna de etiologia venosa. An Bras Dermatol. 2006;81:509-22. Prevalence of recurrence was 76%; a study conducted by Finlayson et al.1414 Finlayson K, Wu ML, Edwards HE. Identifying risk factors and protective factors for venous leg ulcer recurrence using a theoretical approach: A longitudinal study. Int J Nurs Stud. 2015;52:1042-51. found 78% of VU recurrence in patients followed up for three years, having as risk factors history of deep vein thrombosis, multiple anterior leg ulcers and a longer duration of anterior ulcer.
It was observed that PG presented adverse events, such as critical colonization and increased area, but without statistical difference in relation to HC group. This is probably due to the fact that PG contains a large amount of proteins and tissue growth factors that also become excellent means of culturing bacteria and, in its preparation, there is no incorporation of antibacterial agents. Other characteristics observed in relation to the use of dressings were the difficulties of handling PG, which evidences the need for improvements of the product to facilitate its use, as well as the addition of antimicrobial agents to its formula to avoid bacterial proliferation and, thus, improve the healing rates that can be obtained with this product.
Our study has limitations: it is unicentric, with a small number of patients, which made impossible a more precise evaluation of PG.
CONCLUSION
Homologous platelet gel associated with compressive treatment showed favorable results for its indication in the treatment of venous ulcers, being safe due to the occurrence of few mild local adverse events and the absence of serious adverse events. Clinical trials with a larger number of patients and allowing the exploration of subgroups should substantiate the proper indication of this treatment.
-
*
Study conducted at Clinics Hospital of Botucatu Medical School - São Paulo State University "Júlio de Mesquita Filho" (UNESP) - Botucatu (SP), Brazil.
-
Financial support: FAPESP financial grant (nº 2009/03559-3).
REFERENCES
-
1O'Donnell TF Jr, Passman MA, Marston WA, Ennis WJ, Dalsing M, Kistner RL, et al. Management of venous leg ulcers : Clinical practice guidelines of the Society for Vascular Surgery and the American Venous Forum. J Vasc Surg. 2014;60:3S-59S.
-
2Dias TYAF, Costa IKF, Melo MDM, Torres SMSGSO, Maia EMC, Torres GV. Avaliação da qualidade de vida de pacientes com e sem úlcera venosa. Rev Latino-Am Enferm. 2014;22:576-81.
-
3Abbade LPF, Lastória S. Abordagem de pacientes com úlcera da perna de etiologia venosa. An Bras Dermatol. 2006;81:509-22.
-
4Moroz A, Deffune E. Platelet-rich plasma and chronic wounds: remaining fibronectin may influence matrix remodeling and regeneration success. Cytotherapy. 2013;15:1436-9.
-
5Miot HA, Mendaçolli TJ, Costa SV, Haddad GR, Abbade LPF. Úlceras crônicas dos membros inferiores: avaliação pela fotografia digital. Rev Assoc Med Bras. 2009;55:145-8.
-
6Scotton MF, Miot HA, Abbade LP. Factors that influence healing of chronic venous leg ulcers : a retrospective cohort. An Bras Dermatol. 2014;89:414-22.
-
7Palfreyman S, Nelson EA, Michaels JA. Dressings for venous leg ulcers : systematic review and meta-analysis. BMJ. 2007;335:244.
-
8Bernuzzi G, Tardito S, Bussolati O, Adorni D, Cantarelli S, Fagnoni F, et al. Platelet gel in the treatment of cutaneous ulcers: the experience of the Immunohaematology and Transfusion Centre of Parma. Blood Transfus. 2010;8:237-47.
-
9Frykberg RG, Driver VR, Carman D, Lucero B, Borris-Hale C, Fylling CP, et al. Chronic wounds treated with a physiologically relevant concentration of platelet-rich plasma gel: a prospective case series. Ostomy Wound Manage. 2010;56:36-44.
-
10de Leon JM, Driver VR, Fylling CP, Carter MJ, Anderson C, Wilson J, et al. The clinical relevance of treating chronic wounds with an enhanced near-physiological concentration of platelet-rich plasma gel. Adv Skin Wound Care. 2011;24:357-68.
-
11Shan GQ, Zhang YN, Ma J, Li YH, Zuo DM, Qiu JL, et al. Evaluation of the effects of homologous platelet gel on healing lower extremity wounds in patients with diabetes. Int J Low Extrem Wounds. 2013;12:22-9.
-
12Boateng JS, Matthews KH, Stevens HN, Eccleston GM. Wound healing dressings and drug delivery systems: a review. J Pharm Sci. 2008;97:2892-923.
-
13de Almeida Medeiros AB, de Queiroz Frazão CM, de Sá Tinôco JD, Nunes de Paiva Md, de Oliveira Lopes MV, Brandão de Carvalho Lira AL. Venous ulcer: risk factors and the Nursing Outcomes Classification. Invest Educ Enferm. 2014;32:252-9.
-
14Finlayson K, Wu ML, Edwards HE. Identifying risk factors and protective factors for venous leg ulcer recurrence using a theoretical approach: A longitudinal study. Int J Nurs Stud. 2015;52:1042-51.
Publication Dates
-
Publication in this collection
Jul-Aug 2017
History
-
Received
10 Dec 2015 -
Accepted
20 June 2016