Abstracts
The authors reviewed the influence of nutritional factors on systemic lupus erythematosus (SLE) and discussed an alternative treatment option. The autoimmunity and inflammatory process of SLE are related to the presence of dyslipidemia, obesity, systemic arterial hypertension, and metabolic syndrome, which should be properly considered to decrease cardiovascular risk. A diet with moderate protein and energy content, but rich in vitamins, minerals (especially antioxidants), and mono/polyunsaturated fatty acids can promote a beneficial protective effect against tissue damage and suppression of inflammatory activity, in addition to helping the treatment of those comorbidities. Diet therapy is a promising approach and some recommendations may offer a better quality of life to patients with SLE.
systemic lupus erythematosus; diet; nutrition assessment; nutrition programs
Os autores revisaram a influência dos fatores nutricionais sobre o lúpus eritematoso sistêmico (LES) e abordaram uma alternativa complementar em seu tratamento. A autoimunidade e o processo inflamatório do LES estão relacionados à presença de dislipidemias, obesidade, hipertensão arterial sistêmica e síndrome metabólica, que devem ser adequadamente consideradas para diminuir o risco cardiovascular. Uma alimentação com moderado teor energético e proteico, mas rica em vitaminas, minerais (principalmente os antioxidantes) e ácidos graxos mono/poli-insaturados, pode promover ação benéfica protetora contra danos tissulares e supressão da atividade inflamatória, além de auxiliar no tratamento dessas comorbidades. A dietoterapia é uma abordagem promissora, e algumas recomendações podem oferecer melhor qualidade de vida aos pacientes com LES.
lúpus eritematoso sistêmico; dieta; processos nutricionais; terapia nutricional
REVIEW ARTICLE
INutritionist of the Nutrition and Dietary Division (DND) of the Rheumatology Outpatient Clinic, Hospital das Clínicas, Medical School, Universidade de São Paulo - HC-FMUSP
IIFull Professor of the Discipline of Rheumatology, FMUSP
IIIAssociate Professor of the Discipline of Rheumatology, FMUSP; Assistant physician responsible for the Systemic Lupus Erythematosus Outpatient Clinic, HC-FMUSP
Correspondence to
ABSTRACT
The authors reviewed the influence of nutritional factors on systemic lupus erythematosus (SLE) and discussed an alternative treatment option. The autoimmunity and inflammatory process of SLE are related to the presence of dyslipidemia, obesity, systemic arterial hypertension, and metabolic syndrome, which should be properly considered to decrease cardiovascular risk. A diet with moderate protein and energy content, but rich in vitamins, minerals (especially antioxidants), and mono/polyunsaturated fatty acids can promote a beneficial protective effect against tissue damage and suppression of inflammatory activity, in addition to helping the treatment of those comorbidities. Diet therapy is a promising approach and some recommendations may offer a better quality of life to patients with SLE.
Keywords: systemic lupus erythematosus, diet, nutrition assessment, nutrition programs.
INTRODUCTION
Systemic lupus erythematosus (SLE) is a multisystemic chronic inflammatory disease of unknown cause and autoimmune nature, characterized by the presence of several autoantibodies.1 In addition to the specific aspects related to its medicamentous treatment, some supportive measures, such as instructions about the disease, psychosocial support, physical activity, and especially the dietary approach, are essential to provide comprehensive health care to patients with SLE.1 In fact, diet can help to control the inflammatory findings of the disease and the complications derived from its own therapy. Considering that the cardiovascular risk seems increased in patients with SLE due to the increased frequency of conditions associated with atherosclerosis, such as dyslipidemia, diabetes mellitus (DM), metabolic syndrome (MS), and obesity, dietary guidance is important to minimize those complications of the disease.2
The autoimmunity and the inflammatory process of SLE are directly related to changes in the lipid profile and to the metabolism of lipoproteins in SLE. The dyslipoproteinemia of the disease is characterized by higher levels of triglycerides (TG) and very-low-density lipoprotein cholesterol (VLDL-C) associated with lower levels of high-density lipoprotein cholesterol (HDL-C).3 Patients with both active and inactive disease show those lipid changes, which are aggravated by the higher inflammatory activity of the disease, demonstrating that SLE by itself promotes a proatherogenic lipoprotein profile.3 A reduction in the enzymatic activity of lipoprotein lipase is responsible for determining a dyslipoproteinemia characteristic of the disease, because it reduces the catabolism of TG-rich lipoproteins (chylomicrons and VLDL-C)4 due to either the presence of anti-lipoprotein lipase antibodies (anti-LPL)5 or the action of the tumor necrosis factor-α (TNF-α).6
Several drugs used to treat SLE determine deleterious changes in the lipid profile previously altered by the disease itself, the effect of corticosteroids being of particular importance.2,7 Their chronic use in SLE is associated with an increase in total cholesterol and its fractions and TG,2,7 which can be observed after 1-2 months of treatment.2 It is already known that, for each 10-mg/day increase in the dose of prednisone, a 7.5-mg% elevation in total cholesterol is observed.7 In addition, corticosteroids induce the appearance of other risk factors, such as obesity, systemic arterial hypertension (SAH), hyperinsulinemia, and insulin resistance.2,8
Hyperinsulinemia increases oxidative stress, which is considered an important pathophysiological mechanism for the development of atherosclerosis. Some studies have evidenced that DM is significantly more common in patients with SLE than in the general population, because of the reduced insulin sensitivity, and that approximately 18%-38% of the patients have MS.2,9,10
It is worth noting that more than half of the patients with SLE have three or more risk factors for cardiovascular disease, particularly obesity, SAH, and dyslipidemias, suggesting that they are really more susceptible to the MS.2,11 A Brazilian assessment of the nutritional status of 170 patients with SLE has reported a 1.2% prevalence of grade I thinness and a 64.2% prevalence of excessive weight (35.9% of overweight; 21.8% of grade I obesity; 4.1% of grade II obesity; 2.4% of grade III obesity). Eutrophy, according to the Body Mass Index (BMI), has been observed in only 34.7% of the patients assessed, leading to the conclusion that excessive weight is a frequent finding during the follow-up of patients with SLE.12 Thus, it is extremely important to establish strategies, such as programs to encourage the practice of physical activity and body weight reduction, in addition to nutritional counseling, to reduce the risks of MS.
In addition, the hyperlipid diet (rich in cholesterol and saturated fat) is one of the major factors for maintaining dyslipidemia in SLE, perpetuating and aggravating lipid profile changes.8,13,14 On the other hand, antioxidant nutrients, such as β-carotene, α-tocopherol, ascorbic acid, and selenium are known to protect against tissue damages by both activating macrophages, monocytes and granulocytes, and suppressing the activity of cytokines and TNF-α.15
Diet therapy is a promising way to approach SLE, with the indication of vitamin- and mineral-rich foods (mainly the antioxidant ones) and mono/polyunsaturated fatty acids, and moderate energy consumption, aiming at reducing inflammatory markers and helping in the treatment of those comorbidities and of the adverse reactions to drugs.11,13,16,17
DIET THERAPEUTIC INTERVENTIONS IN SYSTEMIC LUPUS ERYTHEMATOSUS
The dietary status refers to the intake of nutrients from food and from supplements, being part of the nutritional status. The nutritional status is extremely important to the immune system balance, the diet composition assuming a fundamental role in maintaining the health of all individuals, including those with SLE.
In fact, thinness or low weight can indicate chronic energy deficiency, being, thus, associated with greater morbidity and mortality.12,18 Overweight and obesity can also be harmful to health, depending on their duration and severity, because these factors reduce resistance, favoring infections.12,18 Because of their deleterious effects on the immune function, both disorders should be diagnosed, aiming at improving the quality of life.
In addition, there is evidence that dietary factors can contribute to the geoepidemiology of autoimmune diseases.19 An adequate diet can, thus, be an essential factor to improve the prognosis of immune diseases, in addition to helping to prevent infections and the progression of cardiovascular diseases.
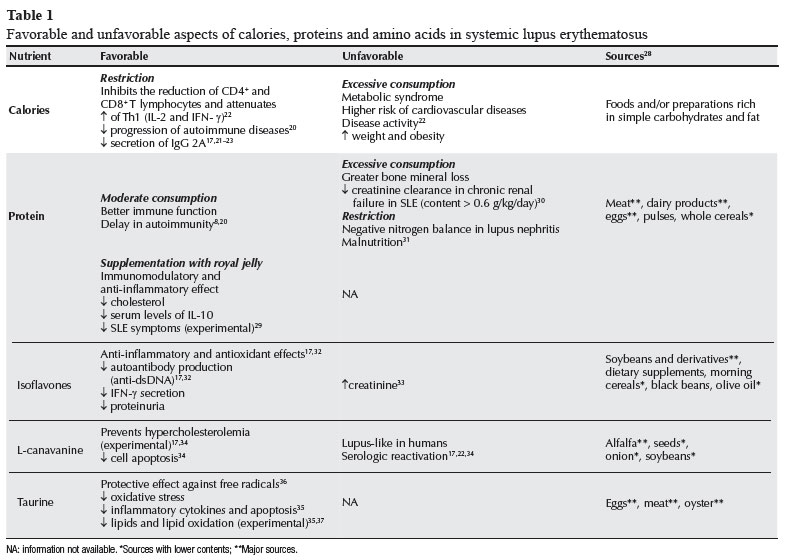
Calories
The restriction of calories in the diet alters the progression of autoimmune diseases.20 Some studies have shown that energy restriction around 30%-40% of the food intake prolongs the life of MRL/lpr mice by inhibiting the development of the lymphoproliferative syndrome, with a reduction in the secretion of IgG 2A (the major antibody of the autoimmune nephritis due to renal deposits) and of the platelet-derived growth factor (PDGF), which can reduce the glomerular lesion of NZB/ NZW mice.17,21-23
Energy restriction inhibits the decrease of CD4+ and CD8+ T lymphocytes, in addition to attenuating the increase in Th1 cytokines (IL-2 and interferon-γ [IFN-γ]) produced in NZB/NZW mice.22
The National Academy of Sciences recommends the intake of 1,800-2,000 calories/day for a sedentary eutrophic adult, and of 2,200-2,500 calories/day in the presence of minimum physical activity.24 Regarding the treatment of excessive weight, the assessment of 86 studies performed by the US National Institutes of Health25 has shown that a diet of 1,000-1,200 kcal/day results in the loss of 7-13 kg (mean of 8%) in 3-6 months, with a mean 10-cm reduction in abdominal fat in 6-24 weeks.25 That recommendation is also strongly supported by the British Nutrition Foundation.26 The National Cholesterol Education Program shares the same opinion, recommending a deficit of 500-1,000 kcal/day by use of a diet of 1,000-1,200 kcal/day for women and 1,200-1,400 kcal/day for men.27
The excessive weight particularly observed in patients with SLE on chronic corticosteroids determines a higher probability of cardiovascular diseases, generating a vicious circle, in which weight gain can maintain disease activity, requiring the continuation of corticosteroids.22
Table 1 shows the major favorable and unfavorable aspects regarding calories in the treatment of SLE. The major food sources indicated in Table 1 are found in the USDA National Nutrient Database for Standard Reference.28
Protein
Studies have shown that moderate-protein diet-fed mice had a long-lasting immune function and a delay in the autoimmunity development as compared with normal-protein diet-fed mice. A diet with restriction of the amino acids phenylalanine and tyrosine was beneficial to NZB/NZW mice.8,20
Supplementation with royal jelly (a honeybee secretion) has also been considered beneficial.29 Its composition rich in free amino acids, simple carbohydrates, proteins, short-chain fatty acids, and vitamins causes a reduction in cholesterol and has immunomodulating and anti-inflammatory activities. In fact, the royal jelly supplementation has induced a reduction in the IL-10 serum levels and has increased the life span of NZB/NZW mice, suppressing the disease symptoms.29
In human beings, the study by Caetano et al.8 has revealed that excessive protein intake causes a constant bone mineral loss in patients with juvenile SLE. On the other hand, the consumption of a protein-restricted diet (0.6 g/kg/day) has improved the glomerular filtration rate in the predialytic chronic kidney disease of patients with systemic diseases.30 It is worth noting that, in lupus nephritis, a hypoprotein diet is not recommended to prevent negative nitrogen balance and malnutrition.31
Isoflavones
Because soybean-based foods have high levels of isoflavones, whose structure is similar to that of 17β-estradiol (E2), they have estrogenic effects and reduce proteinuria and the renal lesions associated with progressive renal failure.17,32 However, potent adverse effects of isoflavones on the immune response of mice have also been reported.33
Zhao et al.33 have reported that a soybean-rich diet can exacerbate renal damages, increasing serum creatinine and reducing the creatinine clearance, which increase the severity of the glomerular disease in MPL/lpr mice. The results have shown that soybean can accelerate glomerulonephritis, but improves the proliferative function of T cells.33
On the other hand, Hong et al.32 have shown that the supplementation with isoflavones increased the survival of SLE murine models, inhibiting the production of autoantibodies (anti-dsDNA and anticardiolipin), and reducing the secretion of IFN-γ. Those authors have also reported that isoflavones have anti-inflammatory properties and antioxidant effects.17,32
L-canavanine
This non-protein amino acid can be found in grains (soybean), onion, seeds and sprouts of alfalfa (major source) and other plants. It is a natural L-arginine homologue that acts with antimetabolic activity and whose presence can result in cell apoptosis in conditions of arginine deficiency.34 Studies have demonstrated that the L-canavanine amino acid acts as a suppressor-inductor of T cells that regulate the synthesis of antibodies and the proliferation of lymphocytes.20
Alfalfa sprouts have high levels of fibers and prevent hypercholesterolemia and atherosclerosis in some SLE models.21 Hong et al.17 have concluded that supplementation with ethyl-acetate extract of alfalfa in murine models for SLE reduced significantly the secretion of IFN-γ, reducing the inflammatory risk and immune mediators. However, some studies with human volunteers and healthy cynomolgus monkeys have shown that the intake of alfalfa sprouts can induce a lupus-like autoimmune syndrome (with antinuclear antibodies, anti-dsDNA, and complement reduction), and its discontinuation induces remission.17,34
The Baltimore Lupus Environmental Study (BALES) has also shown a significant association between alfalfa sprout consumption and the appearance of SLE, suggesting that none of its derivatives should be used.22,34 In addition, in patients with SLE and inactive disease, the consumption of 8-15 tablets of alfalfa per day has been shown to reactivate the clinical symptoms of the disease and its serological aspects.17
Although the findings have suggested that alfalfa derivatives should not be used in SLE, it is worth noting that cooking and autoclaving apparently destroy their deleterious effects without damaging their lipid-lowering properties.16
Taurine
Taurine is the major free intracellular β-amino acid found in mammal tissues that can be synthetized through methionine and cysteine, being found mainly in foods, such as eggs, meat, oyster, and squid. Taurine exerts an important protective function, because, in addition to regulating the immune response, reduces oxidative stress, inflammatory cytokines, and apoptosis, and reduces the serum levels of lipids and their oxidation in mice.35,36
Huang et al.35 have shown that taurine supplementation in NZB/NZW mice fed a hypercholesterolemic diet has reduced cardiac abnormalities, such as histopathologic changes, increased apoptosis, and fibrosis. Taurine has been commonly indicated for the treatment of myocardial failure, hepatic abnormalities associated with SLE, and liver damages of patients with chronic hepatitis, when used at the dosage of 10 g/kg of weight in the diet of animals and 1 g/kg of weight for human beings.35,36
Several studies have shown the protective effect of taurine against free-radical damages, in addition to the inhibition of the hepatic apoptosis induced by biliary acids in mice. However, the mechanism of those effects have not been clearly established.36
Table 1 shows the major favorable and unfavorable aspects regarding proteins and amino acids in the treatment of SLE, and their major food sources according to the USDA National Nutrient Database for Standard Reference.28
Lipids
Lipids are important because they provide polyunsaturated fat to the tissues so that lymphocytes can exert their functions properly. Restriction of saturated fat and increase in the intake of unsaturated fat are recommended, because of the important role of unsaturated fat in the immune system and its response to cancer and infectious diseases.18
Dietary lipids influence the concentration and composition of plasma lipoproteins; saturated fats and omega-6 polyunsaturated fatty acid (ω-6 PUFA) can drastically affect autoimmune diseases in mice.18,20 The total, saturated and monounsaturated fats are not associated with the appearance of DM.37 The higher intake of polyunsaturated fats reduces the risk of DM, while that of trans fats increases that risk - however, its minimal consumption can reduce that risk in as much as 40%.37 Halen et al.38 have shown that a hyperlipid diet induces atherosclerosis in MRL/lpr and MRL/n mice.
On the contrary, food lipid restriction reduces the expression of immune complexes in glomerulonephritis and prolongs the life span of NZB/NZW mice.21 In addition, dietary lipids can change the balance between Th1 and Th2 cells, favoring the development of autoimmune phenomena.18,21
ω -3 and ω -6 polyunsaturated fatty acids
The eicosapentaenoic (EPA) and docosahexaenoic (DHA) unsaturated fatty acids inhibit the enzyme lipoxygenase, reducing the production of inflammatory eicosanoids derived from the arachidonic acid. The DHA has a significant inhibitory action on the nuclear factor κB (NF-κB) and TNF-α, being even more potent than EPA.39 In addition, DHA significantly reduces the serum levels of anti-dsDNA, regulates IgG renal deposits in NZB/NZW mice, and reduces IL-18.40,41 Halade GV et al.40 have reported a significant increase in the life span of NZB/NZW mice with the DHA and EPA supplementation. The α-linolenic acid (ALA), linoleic acid (LA), and gamma linolenic acid (GLA) have also shown a significant inhibitory action on TNF-α and on the IL-2 secretion.20,21,23,37,39,42,43
Some studies have reported that EPA can influence physiological processes, protecting against cardiovascular problems and inflammatory diseases, such as SLE.44 On the other hand, ω-6 PUFA can exacerbate SLE by inducing the inflammatory mediators.20,45 Fassett et al.41 have shown that ω-6 PUFA increases serum creatinine in mice with renal ischemia. The diet therapeutic intervention with EPA and DHA (at the proportion of 3:1), along with calorie restriction, has shown an important anti-inflammatory effect on NZB/NZW mice as compared with a PUFA-rich or calorie-restricted diet, in isolation.21,22,38,40,42 Other studies have demonstrated that the increase in ω-3 PUFA and calorie restriction have reduced the levels of TG, total cholesterol and LDL-C, in addition to reducing the severity of both autoimmunity and nephritis in NZB/NZW mice.38 Murine models of SLE have shown that the reduction in the consumption of ω-9 monounsaturated fatty acid and the increase in ω-3 PUFA have potentiated the therapeutic effect.46
Supplementation with primrose oil can increase the life span of MRL/1pr, NZB/NZW, and BXSB mice, mainly because of its content of GLA (19%), from which prostaglandin E1 (PGE1), which has an anti-inflammatory action and reduces lymphocytic activity, is formed.20,22
Fish oil, known as one of the major sources of ω-3 PUFA, has anti-inflammatory and anti-autoimmune (due to inhibition of T and B lymphocytes) effects. In addition, it suppresses the activity of macrophages and the production of cyclooxygenase metabolites, being significantly beneficial to the clinical, immune, and biochemical status in animal and human models of SLE.16,19,23,44,46,47 Supplementation with fish oil as the exclusive source of lipids reduces proteinuria and protects the kidneys against the deleterious effects of free radicals in NZB/NZW, BXSB, and MRL/lpr mice with lupus nephritis,39,47 by inhibiting PI3K lipid kinase (an important target for reducing glomerulonephritis).40 In addition to reducing anti-dsDNA levels, its major benefit in SLE is due to its effect on apoptosis.22,41,43 Chandrasekar et al.23 have shown that supplementation with fish oil improves glomerulonephritis in NZB/NZW females by reducing the transforming growth factor β (TGF-β), renal mRNA, and protein.
EPA is considered a potent anti-inflammatory agent because it reduces the production of interleukins (IL-1β and IL-6) and of TNF-α, by changing the phospholipid composition of the cell membrane, inhibiting the production and the receptor interaction of inflammatory cytokines.16,20,21,23,39 It is worth noting that a daily dose of 6 g of ω-3 PUFA for 10 weeks can cause a decrease of 4.6 mmHg in systolic blood pressure and of 3.0 mmHg in diastolic blood pressure in patients with SAH.41
Flaxseed oil, with 70% of ω-3 PUFA in its composition and rich in ALA, is a good dietary complement, because it reduces proteinuria levels and preserves glomerular filtration, in addition to reducing anti-dsDNA and anticardiolipin antibodies in mice and suppressing the anti-β2-glycoprotein I in the experimental model of the antiphospholipid syndrome.16,21,22 That effect has not been found with the supplementation of other oils, such as those from safflower, Juniperus virginiana, fish, corn, and soybean, suggesting that the flaxseed oil has another protective component not completely identified, besides ω-3 PUFA.16,18 Flaxseed can also inhibit the platelet activating factor, commonly elevated in the inflammatory response of patients with SLE.20 It should be consumed in its whole form.20 The daily dosage of 30 g proved to be benefic to reduce serum creatinine in patients with lupus nephritis, in addition to promoting a reduction of 11% in total cholesterol levels and of 12% in LDL-C levels.16
Table 2 shows the major favorable effects regarding specific foods, sources of ω-3 PUFA, in the treatment of SLE. In healthy human beings, dietary supplementation with ω-3 PUFA causes a reduction in the production of IL-1α, IL-1β, IL-2, IL-6, NF-κB, and TNF-α.21,40 Several clinical studies have also shown that the consumption of ω-3 PUFA delays renal disease progression by reducing inflammation.39 A study with 12 patients with lupus nephritis and supplemented with fish oil (180 mg of EPA and 120 mg of DHA) has reported a reduction in the following: arachidonic acid; inflammatory status; platelet aggregation; blood viscosity; and leukotriene B4.22 It is worth noting that high doses (18 g/day) of fish oil reduce TG by 38% and increase HDL-C by 28%.16,22
Patients with SLE have a reduced concentration of GLA, ALA, EPA, and DHA in the phospholipid fraction, in addition to reduced levels of nitric oxide, which increase when those patients are supplemented with EPA/DHA.42 Mohan et al.42 and Pestka et al.39 have reported a significant increase in the levels of the antioxidant enzymes superoxide dismutase and glutathione peroxidase and an increase in the hepatic catalase levels of NZB/NZW mice with the EPA/DHA supplementation, inducing SLE remission and being beneficial in the treatment of lupus nephritis with cyclophosphamide.39,42,46In vitro studies have revealed that the supplementation with GLA or arachidonic acid inhibits the production of IL-2. On the other hand, EPA has shown less inhibition of IL-2, indicating the immunosuppressive role of the ω-3 PUFA.43
Table 3 shows the major favorable and unfavorable aspects regarding lipids and fatty acids in the treatment of SLE, and their major food sources according to the USDA National Nutrient Database for Standard Reference.28
Vitamins
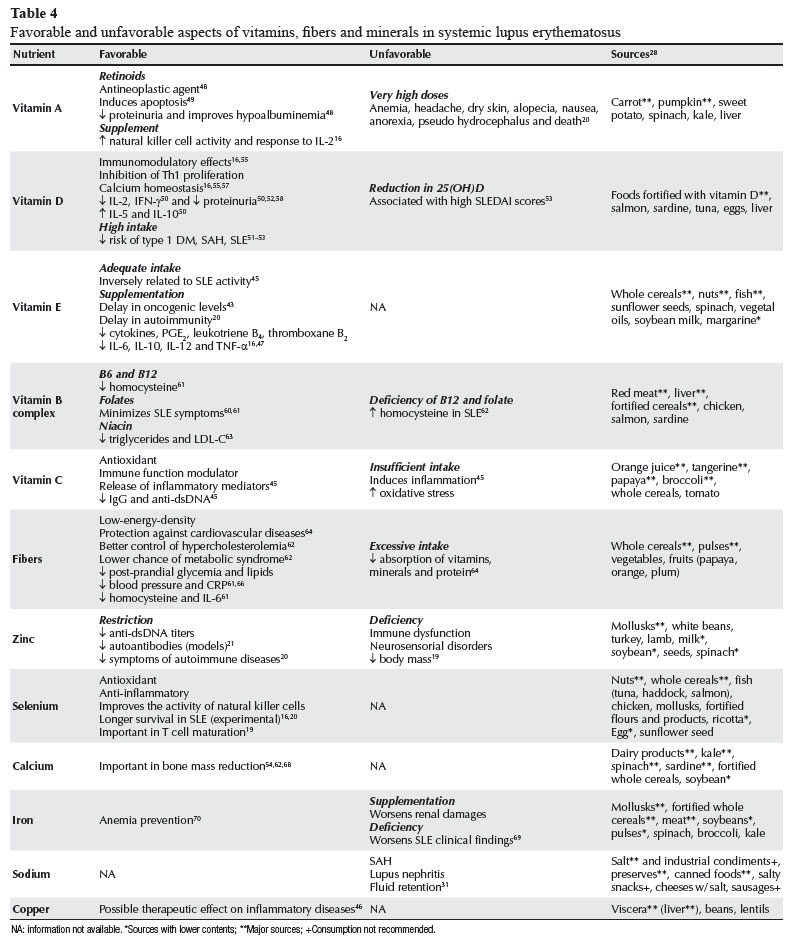
Vitamin A
The metabolites of vitamin A, such as retinoic acid, have an antineoplastic and regulatory role in cell proliferation and differentiation, in addition to increasing T cell cytotoxicity and proliferation and manifesting significant defects in Th cell activity.19,48 They also have therapeutic effects on several animal models of renal diseases, such as lupus nephritis.48
The study by Ikeda et al.49 with MRL mice has shown that vitamin A derivatives, such as etretinate (synthetic retinoic acid) and retinoids, have significantly reduced dermal thickening and proved to be therapeutic agents in cutaneous T cell lymphoma and cutaneous basal cell carcinoma, because of their apoptosis-inducing action. Those mice, treated with 5 mg/kg and 10 mg/kg of etretinate, have not even had the characteristic cutaneous and dermatological lupus-like lesions, probably due to its suppressive effect on cytokine expression.49
Other recent studies have shown that retinoids inhibit the formation of proinflammatory Th17 cells and promote the production of anti-inflammatory regulatory T cells in murine models of autoimmune diseases.15 Kinoshitak et al.48 have shown that patients treated with retinoids improved their proteinuria, their high levels of anti-dsDNA, and low titers of complements, with no side effects, suggesting that retinoids can be promising for the treatment of lupus nephritis.
The supplementation of vitamins A and D in patients with SLE can be indicated. The dose of 100,000 IU of vitamin A for two weeks has proved to be beneficial for the increased antibody-dependent cell cytotoxicity, activity of natural killer cells, and IL-2 response.16 However, the consumption of extremely high doses of vitamin A (> 100,000 IU) can result in symptoms such as anemia, headache, dry skin, alopecia, nausea, anorexia, pseudo hydrocephalus, and death. On the other hand, the deficiency of vitamin A in experimental models of SLE has shown greater severity of symptoms. The researchers have attributed that fact to the increase in hypergammaglobulinemia and presence of autoantibodies.20
Vitamin D
Vitamin D, an important nutrient due to its multiple immunomodulating effects, is produced in the skin and obtained from food. The effects of its active form [1,25(OH)2D3] on immune response occur due to the inhibition of the proliferation of T lymphocytes (Th1).47,50 The treatment of CD4 T cells with 1,25(OH)2D3 inhibits the proliferation of Th1 cells and the production of cytokines, reduces the secretion of IL-2 and IFN-γ by the CD4 T cells, and promotes the production of IL-5 and IL-10, determining a shift toward a Th-2 response.50
The high consumption of vitamin D (> 37 ng⁄mL) has been associated with the reduction in the risks for type I DM, autoimmune encephalomyelitis, SAH, hypertriglyceridemia, MS, inflammatory bowel disease, SLE, and multiple sclerosis.51-53 A recent prospective study carried out with 18,000 women during 22 years has found no association between vitamin D intake and risk for SLE, disagreeing with the hypothesis that the high vitamin D intake would be associated with protection against SLE.51 However, other epidemiological evidence supports the association between vitamin D and the severity of those autoimmune diseases.53
Patients with SLE have been shown to have several factors that reduce vitamin D levels (< 20 ng∕mL), which does not occur with patients with rheumatoid arthritis (RA) and osteoarthritis.16 One possible explanation for the reduction in vitamin D levels in SLE is the intense photoprotection of those patients,16,50,51,53,54 in addition to the relative hypoparathyroidism caused by the high IL-6 levels (mainly in disease activity) and the chronic use of steroids, which changes its metabolism leading to the formation of biologically inactive metabolites and decreasing calcium absorption.50,55,56 Some studies have also suggested that excessive weight is an important risk factor for vitamin D deficiency in SLE.50,55,56
In addition to those factors, hydroxychloroquine seems to reduce the conversion of vitamin D2 into D3, its biologically more active form. Antibodies antivitamin D have also been described in patients with SLE, being associated with anti-dsDNA antibodies, present during disease activity.50,53 It is worth noting that low levels of 25(OH)D are related to the highest scores of inflammatory activity in SLE (SLEDAI);53 on the other hand, its high levels (> 36.8 ng/mL) are associated with greater bone mineral density (both in young and elderly individuals of both genders), according to data of the Third National Health and Nutrition Examination Survey.57
Recent reviews have confirmed that patients with SLE have significantly low serum levels of 25(OH)D (close to 25.5 ± 12.1 nmol/L), while the minimum serum concentration recommended is 50-80 nmol/L.50,52,55,57 Supplementation of that vitamin is appropriate, because its better indicator [1,25(OH)2D3] also plays a role in calcium homeostasis and immune regulation.16,54,55,57 Supplementation with vitamin D3 in MRL/lpr mice has yielded longevity, a reduction in proteinuria, improvement in bone health and a positive impact on immunity.50,52,58
Vitamin E
The combination of fish oil and vitamin E has an impact on several SLE mediators. Mice fed with fish oil and 75 IU of vitamin E showed a reduction in inflammatory cytokines, PGE2, leukotriene B4, and thromboxane B2, to which a reduction in the following factors was added with the increase in vitamin E offer to 500 IU: IL-6; IL-10; IL-12; and TNF-α.16,47 The significant effects on IL-2, IL-4, and TNF-α, obtained through the supplementation with vitamin E and ω-3 PUFA, have suggested that oncogenic levels can be delayed.43
Some studies with MRL/lpr mice have shown that treatment with vitamin E supplementation modulates the levels of inflammatory cytokines, delays the appearance of autoimmunity, and increases survival,15 but the treatment in patients with SLE is still controversial.20 Other studies have found effects of vitamin E supplementation on neither endothelial dysfunction nor lipid peroxidation.59
Another factor to be considered regarding not only vitamin E, but also vitamin A and β-carotene, supplementation relates to the reduction in the levels of the antioxidants α-tocopherol, β-carotene and retinol found in patients with SLE and RA, suggesting important damage to the inflammatory process caused by free radicals.15,16 The adequate consumption of vitamins A and E is inversely related to the SLE activity, according to Minami et al.45 However, the recent study by Costenbader et al.15 has not supported the existence of a relationship between consumption of food antioxidants or supplements and the risk for developing RA or SLE in women.
Vitamin B complex
The study by Varghese et al.60 in mice has shown that immune therapy with folate minimizes the symptoms of SLE and prolongs life span.
The higher plasma levels of homocysteine might be associated with atherosclerosis in SLE, which reinforces the need for a higher consumption of vitamins B6 and B12 (in addition to folate), which are important cofactors in its metabolism and promote a reduction in homocysteine levels.61 In addition, those vitamins also influence the serum levels of some inflammatory markers, such as cytokines and C-reactive protein (CRP).61
Some studies have shown that the consumption of vitamin B12- and folate-deficient diets has caused a plasma increase in homocysteine in patients with SLE. Thus, it has been suggested that patients on a hypolipid diet (indicated for SLE) should increase their consumption of cereals fortified with those nutrients, in addition to vegetables and fruits. The possibility of supplementation should also be considered.62
The study by Minami et al.61 has shown the association of high doses of vitamin B6 and folate with a lower severity of SLE in Japanese patients, regardless of the non-dietary factors. The studyby Ardoin et al.63 has shown that niacin reduces TG and LDL-C levels by 23% and 30%, respectively, in children with dyslipidemia, although it does not significantly affect HDL-C levels.
Vitamin C
Studies with mice have suggested that vitamin C reduces IgG and anti-dsDNA levels and that its insufficient consumption can maintain oxidative stress and induce inflammation in the active phase of disease.45
According to the study by Minami et al.,45 carried out with 279 patients with SLE, vitamin C consumption is inversely associated with the risk of SLE inflammatory activity. The antioxidant properties of that vitamin modulate the immune functions and release of inflammatory mediators.45 Tam et al.59 have shown that the supplementation with antioxidants is a potent therapy to prevent cardiovascular complications. In fact, monthly vitamin C supplementation has determined a significant improvement in the mediation flow of vasodilation in patients with coronary artery disease.59 The safe maximum dose is 1 g/day, because the consumption of higher doses determines the appearance of ascorbate in urine. The combination of daily supplementation of vitamin C (500 mg) with vitamin E (800 IU) for three months was associated with a small reduction in lipid peroxidation, without affecting other markers of oxidative stress or endothelial function in patients with SLE.59
Table 4 shows the major favorable and unfavorable aspects regarding vitamins for the treatment of SLE, and their major food sources according to the USDA National Nutrient Database for Standard Reference.28
Fibers
Adequate intake of dietary fibers is recommended, because it lowers post-prandial glycemia and lipids, yielding lowenergy- density nutrients. In addition, dietary fibers improve intestinal constipation, promoting satiety due to the longer chewing time.
Epidemiological studies have reported that fibers protect against cardiovascular diseases.64,65 In fact, 10 cohort studies in the United States and Europe, with a six-to-ten-year follow-up, have concluded that fibers were associated with a risk reduction of 14% and 27% in coronary events and coronary death, respectively.65 Those results can be explained by the effect of fibers on blood pressure and on CRP levels. The intake of fibers has been inversely associated with CRP in the National Health and Nutrition Examination Survey 1999-2000.66
As foods are digested and absorbed in the small intestine, the fibers increase the viscosity in the intestinal lumen, interfering with the biliary acid absorption from the ileum. The LDL-C is removed from the blood and converted to biliary acids to replace those eliminated with defecation. This change in the pool of biliary acids, along with the intake of viscous fibers, depress cholesterol synthesis.61,64 Simultaneously, inulin, oligosaccharides, resistant starch, and other fibers increase mineral absorption, especially that of calcium.67
The diet therapeutic intervention to control hypercholesterolemia and MS in SLE should emphasize the importance of consuming fiber-rich foods, especially the soluble ones (found in oat, fruits and pulses) to control dyslipidemia.62 The recommendation is 14 g of fibers per 1,000 kcal consumed (or 38 g for men and 25 g for women), adequate fluid ingestion being required.
Minami et al.61 have also reported that fiber intake was inversely proportional to the SLE severity risk. Some studies has already shown that fiber intake is inversely associated with the plasma levels of homocysteine and of the inflammatory markers IL-6 and CRP.61 However, excessive fiber intake reduces the absorption of vitamins, minerals, protein and energy.64
Table 4 shows major favorable and unfavorable aspects regarding dietary fibers for the treatment of SLE, and their major food sources according to the USDA National Nutrient Database for Standard Reference.28
Minerals
Zinc
MRL/lpr mice on a zinc-restricted diet have shown a reduction in lymphoproliferation and in anti-dsDNA titers, and an improvement in glomerulonephritis, as well as a reduction in the production of autoantibodies in NZB/NZW models.21
A zinc-restricted diet determines an increase in the serum levels of corticosteroids, which can contribute to control SLE.20 On the other hand, a study in human beings has shown that zinc deficiency causes an immune dysfunction that affects mainly Th cells, and can cause neurosensorial disorders and body mass reduction.19
Selenium
A diet rich in selenium, a natural antioxidant, increases anti-inflammatory properties, with a reduction in anti-dsDNA antibodies, improving the activity of natural killer cells and survival in murine models of SLE.16,20,47 It can have a significant effect on the maturation of T cells and on the response of T cell-dependent autoantibodies.19
Calcium
An adequate consumption of calcium is extremely important in SLE, particularly in patients with a reduction in bone mineral density either associated or not with corticotherapy and regardless of disease duration.62,68 The risk of osteoporosis is greater due to disease activity, vitamin D deficiency, non-exposure to sunlight, and early menopause caused by cytotoxic agents.54,68 In fact, women with SLE are five times more likely to undergo fractures as compared with healthy women of the same age.62
The American College of Rheumatology (ACR) has issued recommendations to reduce bone mass loss in patients with SLE treated with corticosteroids. In addition, ACR has suggested that patients receiving more than 5 mg of prednisone daily, for three months, should begin to receive calcium and vitamin D prophylactically, and undergo assessment of bone density and of the use of other medications. Changes in life style and a calcium-rich diet have also been suggested.20,62,68 Supplementation of calcium (> 1,500 mg) and vitamin D (20 µg or 800 UI) is indicated in cases of difficulty in obtaining those nutrients from the diet.20,58
Iron
Some studies have suggested that iron can cause cell damages and that the use of chelating agents has shown benefits in experimental models for autoimmune diseases. More severe renal lesions were more prevalent in mice being supplemented with iron as compared with controls; however, animals with mineral deficiency developed more severe clinical signs of the disease. That study has suggested that a dietary restriction would reduce mortality in those models.69
On the other hand, some studies with patients with SLE have shown that anemia can be detected in up to 70% of them during the course of disease. The most often found type of anemia is that of chronic disease (characterized by deficient mobilization of iron to the bone marrow, despite the normal or increased values of iron reserves).70 In the case of iron-deficiency anemia, the consumption of the following is indicated: lean meat (mainly white); dark-green leafy vegetables; whole cereals; iron-fortified foods; and, in more severe cases, medicamentous supplementation.62
Sodium
Patients with lupus nephritis and those with SAH, either secondary or not to corticotherapy, need to follow a sodium-restricted diet. The daily amount considered adequate and safe for those conditions is of 3 g sodium/day, and should be also followed by a restriction of fluid intake (maximum of 1.5 L/day).31
Copper
High serum concentrations of copper have been observed in patients with SLE and RA, and such levels are directly related to disease activity and probable inflammatory response. Copper is believed to exert a therapeutic effect on the treatment of chronic diseases because its liver storage is insufficient to meet the demands of the inflammatory response. Exogenous copper reduces cell formation in mice, but its supplementation has not produced significant serum changes in the study by Duffy et al.46
Table 4 shows major favorable and unfavorable aspects regarding minerals for the treatment of SLE, and their major food sources according to the USDA National Nutrient Database for Standard Reference.28
CONCLUSION
Considering all favorable and unfavorable aspects of the major nutrients, it is safe to state that the adequate diet for the treatment of SLE is mainly aimed at reducing the risk for cardiovascular and atherosclerotic diseases, in addition to reducing the inflammatory factors and improving the immune function. Patients with SLE can benefit from a nutritionally balanced diet for maintaining the ideal body weight, with effective calorie control to avoid insulin resistance, increasing HDL-C (protective) levels and reducing TG levels.
The patient with SLE should be instructed to follow a calorie-restricted diet to prevent and/or treat excessive weight, in addition to a diet with a moderate protein content, isoflavones being indicated for protein supplementation, but L-canavanine, which aggravates SLE symptoms, being contraindicated. Regarding the lipids in the diet, a greater offer of mono- and polyunsaturated fatty acids is recommended in daily meals, due to their anti-inflammatory and cardiovascular protective functions. Supplementation with ω-3 PUFA can be suggested due to its countless benefits. On the other hand, the restriction of ω-6 PUFA and other sources of saturated and trans fatty acids in the diet should be enforced, because of their pro-inflammatory effects and aggravating effects for cardiovascular diseases, respectively.
It is worth noting the importance of offering vitamins, mainly vitamin D, fibers and minerals, such as calcium and selenium. On the other hand, the sources of zinc and sodium should be restricted to prevent possible disease aggravation. Thus, the major recommendations of diet therapy (Table 5) are intended to provide better quality of life to patients with SLE and more safety in the course of their treatment.
REFERENCES
-
1Borba EF, Latorre LC, Brenol JC, Kayser C, Silva NA, Zimmermann AF et al Consenso de lúpus eritematoso sistêmico. Rev Bras Reumatol 2008;48(4):196-207.
-
2Bruce IN. Not only...but also: factors that contribute to accelerated atherosclerosis and premature coronary heart disease in systemic lupus erythematosus. Rheumatology (Oxford) 2005;44(12):1492-502.
-
3Borba EF, Bonfá E. Dyslipoproteinemias in systemic lupus erythematosus: influence of disease, activity, and anticardiolipin antibodies. Lupus 1997;6(6):533-9.
-
4Borba EF, Bonfá E, Vinagre CG, Ramires JA, Maranhão RC. Chylomicron metabolism is markedly altered in systemic lupus erythematosus. Arthritis Rheum 2000;43(5):1033-40.
-
5Carvalho JF, Borba EF, Viana VS, Bueno C, Leon EP, Bonfá E. Anti-lipoprotein lipase antibodies: a new player in the complex atherosclerotic process in systemic lupus erythematosus? Arthritis Rheum 2004;50(11):3610-5.
-
6Svenungsson E, Gunnarsson I, Fei GZ, Lundberg IE, Klareskog L, Frostegård J. Elevated triglycerides and low levels of high-density lipoprotein as markers of disease activity in association with up-regulation of the tumor necrosis factor alpha/tumor necrosis factor receptor system in systemic lupus erythematosus. Arthritis Rheum 2003;48(9):2533-40.
-
7Petri M, Lakata C, Magder L, Goldman D. Effect of prednisone and hydroxychloroquine on coronary artery disease risk factors in systemic lupus erythematosus: a longitudinal data analysis. Am J Med 1994;96(3):254-9.
-
8Caetano MC, Ortiz TT, Terreri MT, Sarni RO, Silva SG, Souza FI et al Inadequate dietary intake of children and adolescents with juvenile idiopathic arthritis and systemic lupus erithematosus. J Pediatr 2009;85(6):509-15.
-
9Azevedo GD, Gadelha RG, Vilar MJ. Metabolic syndrome in systemic lupus erythematosus: lower prevalence in Brazil than in the USA. Ann Rheum Dis 2007;66(11):1542.
-
10Cunha BM, Klumb EM, Moura JQ, Santos A, Pinheiro GRC, Albuquerque EMN et al Frequência de síndrome metabólica em pacientes com nefrite lúpica. Rev Bras Reumatol 2008;48(5):267-73.
-
11Chung CP, Avalos I, Oeser A, Gebretsadik T, Shintane A, Raggi P et al High prevalence of the metabolic syndrome in patients with systemic lupus erythematosus: association with disease characteristics and cardiovascular risk factors. Ann Rheum Dis 2007;66(2):208-14.
-
12dos Santos F de M, Borges MC, Correia MI, Telles RW, Lanna CC. Assessment of nutritional status and physical activity in systemic lupus erythematosus patients. Rev Bras Reumatol 2010;50(6):631-8.
-
13Shah M, Coyle Y, Kavanaugh A, Adams-Huet B, Lipsky PE. Development and initial evaluation of a culturally sensitive cholesterol-lowering diet program for Mexican and African American patients with systemic lupus erythematosus. Arthritis Care Res 2000;13(4):205-12.
-
14Shah M, Kavanaugh A, Coyle Y, Adams-Huet B, Lipsky PE. Effect of a culturally sensitive cholesterol lowering diet program on lipid and lipoproteins, body weight, nutrient intakes, and quality of life in patients with systemic lupus erythematosus. J Rheumatol 2002;29(10):2122-8.
-
15Costenbader KH, Kang JH, Karlson EW. Antioxidant intake and risks of rheumatoid arthritis and systemic lupus erythematosus in women. Am J Epidemiol 2010;172(2):205-16.
-
16Patavino T, Brady DM. Natural medicine and nutritional therapy as an alternative treatment in systemic lupus erythematosus. Altern Med Rev 2001;6(5):460-71.
-
17Hong YH, Huang CJ, Wang SC, Lin BF. The ethyl acetate extract of alfalfa sprout ameliorates disease severity of autoimmune-prone MRL-lpr/lpr mice. Lupus 2009;18(3):206-15.
-
18Maki PA, Newberne PM. Dietary lipids and immune function. J Nutr 1992;122(3 Suppl):610-4.
-
19Selmi C, Tsuneyama K. Nutrition, geoepidemiology, and autoimmunity. Autoimmun Rev 2010;9(5):A267-70.
-
20Brown AC. Lupus erythematosus and nutrition: a review of the literature. J Ren Nutr 2000;10(4):170-83.
-
21Leiba A, Amital H, Gershwin ME, Shoenfeld Y. Diet and lupus. Lupus 2001;10(3):246-8.
-
22Petri M. Diet and systemic lupus erythematosus: from mouse and monkey to woman? Lupus 2001;10(11):775-7.
-
23Chandrasekar B, Troyer DA, Venkatraman JT, Fernandes G. Dietary omega-3 lipids delay the onset and progression of autoimmune lupus nephritis by inhibiting transforming growth factor beta mRNA and protein expression. J Autoimmun 1995;8(3):381-93.
-
24Trumbo P, Schlicker S, Yates AA, Poos M; Food and Nutrition Board of the Institute of Medicine, The National Academies. Dietary reference intakes for energy, carbohydrate, fiber, fat, fatty acids, cholesterol, protein, and amino acids. J Am Diet Assoc 2002;102(11):1621-30. [Erratum in: J Am Diet Assoc 2003;103(5):563]
-
25US National Institutes of Health. Clinical Guidelines on the Identification, Evaluation, and Treatment of Overweight and Obesity in Adults. Washington, DC: National Heart, Lung and Blood Institute, 1998.
-
26British Nutrition Foundation. Obesity. London: Blackwell Science, 1999.
-
27National Institutes of Health, National Heart, Lung, and Blood Institute. Obesity education initiative. Clinical guidelines on the identification, evaluation, and treatment of overweight and obesity in adults: the evidence report. Obes Res 1998;6(Suppl 2):51S-209S. [Erratum in: Obes Res 1998;6(6):464]
-
28United States Department of Agriculture. USDA National Nutrient Database For Standard Reference Release 18. Agricultural Research Service. [Available from: http://www.nal.usda.gov/fnic/foodcomp/Data/SR18/sr18.html]
-
29Mannoor MK, Shimabukuro I, Tsukamotoa M, Watanabe H, Yamaguchi K, Sato Y. Honeybee royal jelly inhibits autoimmunity in SLE-prone NZB x NZW F1 mice. Lupus 2009;18(1):44-52.
-
30Milovanov IuS, Lysenko LV, Milovanova Llu, Dobrosmyslov IA. The role of balanced low-protein diet in inhibition of predialysis chronic kidney disease progression in patients with systemic diseases. Ter Arkh 2009;81(8):52-7.
-
31Hull RP, Goldsmith DJ. Nephrotic syndrome in adults. BMJ 2008;336(7654):1185-9.
-
32Hong Y, Wang T, Huang C, Cheng W, Lin B. Soy isoflavones supplementation alleviates disease severity in autoimmune-prone MRL-lpr/lpr mice. Lupus 2008;17(9):814-21.
-
33Zhao JH, Sun SJ, Horiguchi H, Arao Y, Kanamori N, Kikuchi A et al A soy diet accelerates renal damage in autoimune MRL/Mp-lpr/lpr mice. Int Immunopharmacol 2005;5(11):1601-10.
-
34Akaogi J, Barker T, Kuroda Y, Nacionales DC, Yamasaki Y, Stevens BR et al Role of non-protein amino acid L-canavanine in autoimmunity. Autoimmun Rev 2006;5(6):429-35.
-
35Huang CY, Hsu TC, Kuo WW, Wu SP, Lin YM, Yen CY et al Beneficial effects of taurine on cardiac abnormality in NZB/W F1 mice fed with a high-cholesterol diet. J Agric Food Chem 2009;57(18): 8635-42.
-
36Hsu TC, Chiang SY, Wu JH, Tsai CC, Huang CY, Chen YC et al Treatment with taurine attenuates hepatic apoptosis in NZB/W F1 mice fed with a high-cholesterol diet. J Agric Food Chem 2008;56(20):9685-91.
-
37Kien CL. Dietary interventions for metabolic syndrome: role of modifying dietary fats. Curr Diab Rep 2009;9(1):43-50.
-
38Halen BH, Lourenço EV, McMahon M, Skaggs B, Le E, Anderson M et al Pro-inflammatory high density lipoproteins and atherosclerosis are induced in lupus-prone mice by a high-fat diet and leptin. Lupus 2010;19(8):913-7.
-
39Pestka JJ. N-3 polyunsaturated fatty acids and autoimmune-mediated glomerulonephritis. Prostaglandins Leukot Essent Fatty Acids 2010;82(4-6):251-8.
-
40Halade GV, Rahman MM, Bhattacharya A, Barnes JL, Chandrasekar B, Fernandes G. Docosahexaenoic acid-enriched fish oil attenuates kidney disease and prolongs median and maximal life span of autoimmune lupus-prone mice. J Immunol 2010;184(9):5280-6.
-
41Fassett RG, Gobe GC, Peake JM, Coombes JS. Omega-3 polyunsaturated fatty acids in the treatment of kidney disease. Am J Kidney Dis 2010;56(4):728-42.
-
42Mohan IK, Das UN. Oxidant stress, anti-oxidants and essential fatty acids in systemic lupus erythematosus. Prostaglandins Leukot Essent Fatty Acids 1997;56(3):193-8.
-
43Venkatraman JT, Chu WC. Effects of dietary omega-3 and omega-6 lipids and vitamin E on proliferative response, lymphoid cell subsets, production of cytokines by spleen cells, and splenic protein levels for cytokines and oncogenes in MRL/MpJ-lpr/lpr mice. J Nutr Biochem 1999;10(10):582-97.
-
44MacLean CH, Mojica WA, Morton SC, Pencharz J, Hasenfeld Garland R, Tu W et al Effects of omega-3 fatty acids on lipids and glycemic control in type II diabetes and the metabolic syndrome and on inflammatory bowel disease, rheumatoid arthritis, renal disease, systemic lupus erythematosus, and osteoporosis. Evid Rep Technol Assess (Summ) 2004;(89):1-4.
-
45Minami Y, Sasaki T, Arai Y, Kurisu Y, Hisamichi S. Diet and systemic lupus erythematosus: a 4 year prospective study of Japanese patients. J Rheumatol 2003;30(4):747-54.
-
46Duffy EM, Meenagh GK, McMillan SA, Strain JJ, Hannigan BM, Bell AL. The clinical effect of dietary supplementation with omega-3 fish oil and/or copper in systemic lupus erythematosus. J Rheumatol 2004;31(8):1551-6.
-
47Chou CT. Alternative therapies: what role do they have in the management of lupus? Lupus 2010;19(12):1425-9.
-
48Kinoshita K, Kishimoto K, Shimazu H, Nozaki Y, Sugiyama M, Ikoma S et al Successful treatment with retinoids in patients with lupus nephritis. Am J Kidney Dis 2010;55(2):344-7.
-
49Ikeda T, Nishide T, Ohtani T, Furukawa F. The effects of vitamin A derivative etretinate on the skin of MRL mice. Lupus 2005;14(7):510-6.
-
50Cutolo M, Otsa K. Vitamin D, immunity and lupus. Lupus 2008;17(1):6-10.
-
51Costenbader KH, Feskanich D, Holmes M, Karlson EW, Benito-Garcia E. Vitamin D intake and risks of systemic lupus erythematosus and rheumatoid arthritis in women. Ann Rheum Dis 2008;67(4):530-5.
-
52Kamen DL, Cooper GS, Bouali H, Shaftman SR, Hollis BW, Gilkeson GS. Vitamin D deficiency in systemic lupus erythematosus. Autoimmun Rev 2006;5(2):114-7.
-
53Wu PW, Rhew EY, Dyer AR, Dunlop DD, Langman CB, Price H et al. 25-hydroxyvitamin D and cardiovascular risk factors in women with systemic lupus erythematosus. Arthritis Rheum 2009;61(10):1387-95.
-
54Knott HM, Martínez JD. Innovative management of lupus erythematosus. Dermatol Clin 2010;28(3):489-99.
-
55Wright TB, Shults J, Leonard MB, Zemel BS, Burnham JM. Hypovitaminosis D is associated with greater body mass index and disease activity in pediatric systemic lupus erythematosus. J Pediatr 2009;155(2):260-5.
-
56Toloza SM, Cole DE, Gladman DD, Ibañez D, Urowitz MB. Vitamin D insufficiency in a large female SLE cohort. Lupus 2010;19(1):13-9.
-
57Lane NE. Vitamin D and systemic lupus erythematosus: bones, muscles, and joints. Curr Rheumatol Rep 2010;12(4):259-63.
-
58Aghdassi E, Morrison S, Landolt-Marticorena C, Su J, Pineau C, Gladman D et al. The use of micronutrient supplements is not associated with better quality of life and disease activity in Canadian patients with systemic lupus erythematosus. J Rheumatol 2010;37(1):87-90.
-
59Tam LS, Li EK, Leung VY, Griffith JF, Benzie IF, Lim PL et al Effects of vitamins C and E on oxidative stress markers and endothelial function in patients with systemic lupus erythematosus: a double blind placebo controlled pilot study. J Rheumatol 2005;32(2):275-82.
-
60Varghese B, Haase N, Low PS. Depletion of folate-receptor-positive macrophages leads to alleviation of symptoms and prolonged survival in two murine models of systemic lupus erythematosus. Mol Pharm 2007;4(5):679-85.
-
61Minami Y, Hirabayashi Y, Nagata C, Ishii T, Harigae H, Sasaki T. Intakes of vitamin B6 and dietary fiber and clinical course of systemic lupus erythematosus: a prospective study of Japanese female patients. J Epidemiol 2011. [Epub 2011 Apr 23]
-
62Shah M, Adams-Huet B, Kavanaugh A, Coyle Y, Lipsky P. Nutrient intake and diet quality in patients with systemic lupus erythematosus on a culturally sensitive cholesterol lowering dietary program. J Rheumatol 2004;31(1):71-5.
-
63Ardoin SP, Sandborg C, Schanberg LE. Management of dyslipidemia in children and adolescents with systemic lupus erythematosus. Lupus 2007;16(8):618-26.
-
64US Department of Health and Human Services, US Department of Agriculture. Dietary Guidelines for Americans, 2005. [Available from: http://www.health.gov/DietaryGuidelines/dga2005/document]
-
65Pereira MA, O'Reilly E, Augustsson K, Fraser GE, Goldbourt U, Heitmann BL et al Dietary fiber and risk of coronary heart disease: a pooled analysis of cohort studies. Arch Intern Med 2004;164(4):370-6.
-
66Ajani UA, Ford ES, Mokdad AH. Dietary fiber and C-reactive protein: findings from National Health and Nutrition Examination Survey data. J Nutr 2004;134(5):1181-5.
-
67Abrams SA, Griffin IJ, Hawthorne KM, Liang L, Gunn SK, Darlington G et al A combination of prebiotic short- and long-chain inulin-type fructans enhances calcium absorption and bone mineralization in young adolescents. Am J Clin Nutr 2005;82(2):471-6.
-
68Schmajuk G, Yelin E, Chakravarty E, Nelson LM, Panapolis P, Yazdany J. Osteoporosis screening, prevention, and treatment in systemic lupus erythematosus: application of the systemic lupus erythematosus quality indicators. Arthritis Care Res (Hoboken) 2010;62(7):993-1001.
-
69Leiter LM, Reuhl KR, Racis SP Jr, Sherman AR. Iron status alters murine systemic lupus erythematosus. J Nutr 1995;125(3):474-84.
-
70Falcão S, Barros R, Mateus M, Nero P, de Matos AA, Pimentão JB et al. Lúpus eritematoso sistêmico e anemia. Acta Reumatol Port 2007;32(1):73-9.
Diet and nutritional aspects in systemic lupus erythematosus
Publication Dates
-
Publication in this collection
25 May 2012 -
Date of issue
June 2012
History
-
Received
25 Apr 2011 -
Accepted
05 Mar 2012