Abstracts
The inflammatory processes in the joints of a child with juvenile idiopathic arthritis (JIA) can persist into adulthood. Inflammation has been linked to distortions of the lipid profile and accelerated atherogenesis. In the present study, we examined the lipid profiles of adults with JIA compared with those of healthy people. A lipid profile of a sample of 54 adults with JIA (57.3% with polyarticular JIA, 37.0% with oligoarticular JIA, 1.9% with enthesitis-related JIA and 3.7% with systemic onset JIA) and 54 healthy subjects were compared. In the adults with JIA, data on gender, age, age at disease onset, the presence of rheumatoid factor (RF) and antinuclear antibodies (ANA), a Health Assessment Questionnaire (HAQ) and the disease duration were collected. We found that hypercholesterolaemia, increased low-density lipoprotein (LDL) and decreased high-density lipoprotein (HDL) were more common in patients with JIA than the controls (P = 0.016, P < 0.0001 and P = 0.0008, respectively). Changes in the levels of total cholesterol (TC) and LDL were more common in the individuals who had a later onset of disease (P = 0.0017 for TC and P = 0.023 for LDL). In the entire JIA group, no other variable, such as RF, ANA, disease duration or responses to the HAQ, could be linked to dyslipidaemia (P = non-significant). We concluded that the adult patients with JIA have a lipid profile with increased TC and LDL levels and decreased levels of HDL compared to the controls. No clinical feature could be correlated with this change except for the age at disease onset.
Dyslipidemias; Cholesterol, HDL; Cholesterol, LDL; Triglycerides; Arthritis, juvenile rheumatoid
O processo inflamatório articular de uma criança com artrite idiopática juvenil (AIJ) pode persistir na vida adulta. A inflamação tem sido ligada a distorções do perfil lipídico e à aterogênese acelerada. Neste trabalho, estudou-se o perfil lipídico de indivíduos adultos com AIJ, comparando-o ao de pessoas saudáveis. Foram recrutados 54 adultos com AIJ (57,3% poliarticular, 37% oligoarticular, 1,9% do subtipo entesítica e 3,7% do subtipo sistêmica) e 54 indivíduos saudáveis para comparação de perfil lipídico. Nos adultos AIJ, coletaram-se dados sobre sexo, idade e idade de início da doença, presença de fator reumatoide (FR) e fator antinuclear (FAN), HAQ (Health Assessment Questionnaire) e tempo de doença. Observou-se que hipercolesterolemia, aumento de LDL e diminuição de HDL foram mais comuns em pacientes com AIJ do que nos controles (com P = 0,016; P < 0,0001 e P = 0,0008, respectivamente). Alterações no colesterol total (CT) e no LDL foram mais comuns em indivíduos com início de doença mais tardio (P = 0,0017 para CT e P = 0,023 para LDL). Em todo o grupo de pacientes com AIJ nenhuma outra variável pode ser ligada à dislipidemia tal como presença de FR, FAN, duração de doença e HAQ (P = ns). Concluiu-se que pacientes adultos com AIJ apresentam um perfil lipídico com aumento de CT e de LDL, e diminuição de HDL em relação aos controles. Nenhuma característica clínica pode ser ligada a essa alteração, exceto idade de início da doença.
Dislipidemias; HDL-colesterol; LDL-colesterol; Artrite reumatoide juvenil; Triglicerídeos
BRIEF COMMUNICATION
Lipid profile in adult patients with idiopathic juvenile arthritis*
Thelma Larocca Skare** ; Marilia Barreto Silva; Paulo Negreiros
Rheumatology Service, Hospital Universitário Evangélico de Curitiba, Faculdade Evangélica do Paraná, Curitiba, PR, Brazil
ABSTRACT
The inflammatory processes in the joints of a child with juvenile idiopathic arthritis (JIA) can persist into adulthood. Inflammation has been linked to distortions of the lipid profile and accelerated atherogenesis. In the present study, we examined the lipid profiles of adults with JIA compared with those of healthy people. A lipid profile of a sample of 54 adults with JIA (57.3% with polyarticular JIA, 37.0% with oligoarticular JIA, 1.9% with enthesitis-related JIA and 3.7% with systemic onset JIA) and 54 healthy subjects were compared. In the adults with JIA, data on gender, age, age at disease onset, the presence of rheumatoid factor (RF) and antinuclear antibodies (ANA), a Health Assessment Questionnaire (HAQ) and the disease duration were collected. We found that hypercholesterolaemia, increased low-density lipoprotein (LDL) and decreased high-density lipoprotein (HDL) were more common in patients with JIA than the controls (P = 0.016, P < 0.0001 and P = 0.0008, respectively). Changes in the levels of total cholesterol (TC) and LDL were more common in the individuals who had a later onset of disease (P = 0.0017 for TC and P = 0.023 for LDL). In the entire JIA group, no other variable, such as RF, ANA, disease duration or responses to the HAQ, could be linked to dyslipidaemia (P = non-significant). We concluded that the adult patients with JIA have a lipid profile with increased TC and LDL levels and decreased levels of HDL compared to the controls. No clinical feature could be correlated with this change except for the age at disease onset.
Keywords: Dyslipidaemias; Cholesterol HDL; Cholesterol LDL; Triglycerides; Arthritis juvenile rheumatoid
Introduction
Juvenile idiopathic arthritis (JIA) is the most common arthropathy in childhood, and this disease involves several subtypes of arthritis that appear before 16 years.1 In affected children, JIA has the potential to cause serious impacts on performance in adulthood. Nevertheless, there are few studies on adults who developed JIA.
It is a well-known fact that the survival of individuals subject to long-term inflammation is altered by the risk of accelerated atherogenesis.2,3 There are many studies on rheumatoid arthritis (RA) in adults that clearly demonstrate this effect, which causes an increased risk of death from cardiovascular complications.4-7 Changes in the lipid profile due to exposure to pro-inflammatory cytokines, such as interleukin (IL)-1β, IL-6 and tumour necrosis factor-α (TNF-α), are among the causes associated with the increased risk of atherogenesis.7 The chronic use of glucocorticoids for the treatment of this condition has been associated with hypercholesterolaemia.8 In patients with RA, decreased high-density lipoprotein (HDL)-cholesterol levels have been found, as well as less statistically significant changes in total cholesterol (TC) and low-density lipoprotein (LDL)-cholesterol levels and a change in the relationship between the different components of the lipid profile.9
JIA is a disease with a much more heterogeneous clinical profile than RA, and manifestations with inflammatory processes of different intensity are also expected.1 A study that compared 25 children with JIA with 15 healthy children revealed increased levels of TC and LDL-cholesterol and a decreased level of HDL-cholesterol.10 Another study demonstrated the presence of increased triglycerides (TG) and decreased HDL-cholesterol levels, and this study also indicated that these changes were proportional to the duration of the disease and the level of inflammatory activity.11
The present study evaluated the lipid profiles of adult patients with JIA, compared these profiles with the lipid profile of a healthy population and established the relationship of the lipid profiles to the clinical variables of JIA.
Method
The present study was an analytical cross-sectional study of the lipid profiles of adult patients with JIA that was duly approved by the local research ethics committee. All the participants signed an informed consent form. This sample consists of all the adult patients with JIA treated at the outpatient rheumatology clinic of a university hospital in the last five years who agreed to participate in the study. The participants were classified according to the different JIA subtypes described by the International League of Associations for Rheumatology (ILAR)1, and the following data were collected: the demographic data, the autoantibodies profile (antinuclear antibodies, ANA and IgM rheumatoid factor, RF), the Health Assessment Questionnaire (HAQ)12 values and the drugs being used. Pregnant and nursing women, as well as individuals with uncorrected hypothyroidism (n = 3), were excluded from the study.
For comparison, subjects without an inflammatory process, who were paired according to age and sex and who sought preventive care at the gynaecology outpatient clinic or refraction correction at the ophthalmology service, were studied.
The levels of TC, TG and LDL- and HDL-cholesterol were obtained from the population of adults with JIA and from the controls using the enzymatic colorimetric method. The following values were considered normal: TC values up to 200 mg/dL, HDL-cholesterol levels above 40 mg/dL, LDL-cholesterol levels up to 110 mg/dL and TG levels up to 150 mg/dL.13
The data obtained were analysed with frequency and contingency tables. For the comparative analysis of the nominal data, the Fisher's test and the chi-square test were used. According to the sample distribution, the Student's t-test and the Mann-Whitney test were used to study the numerical values. The Spearman test was used for the correlation studies. A significance level of 5% was specified.
Results
Characterisation of the sample studied
Of the 54 individuals of the control group, 40 (74.1%) were women and 14 (25.9%) were men, with a mean age of 20.6 ± 1.8 years; the mean body mass index (BMI) was 22.6 ± 3.5. None of the subjects were smokers. There were no cases of diabetes mellitus or hypertension. In the group of adults with JIA, there were 54 patients, and 72.2% (39/54) of the JIA patients were women and 27.8% (15/54) were men (P = 0.8 compared to the control group), with a mean age of 22.0 ± 5.2 years (P = 0.1 compared to the control group); the mean disease time was 15.0 ± 7.8 years, and the mean age at diagnosis was 9.4 ± 4.1 years. The mean BMI was 23.4 ± 4.0 (P = 0.3 compared to the control group). Among these 54 patients, 3 were smokers (P = 0.2 compared to the control group). Among the patients with JIA, 1 had hypertension and 1 had diabetes mellitus (P = 1.0 for both variables compared to the control group).
The systemic subgroup of JIA was present in 2/54 (3.7%) of the patients; 1/54 JIA patients (1.8%) had the enthesitisrelated subtype, 20/54 (37.0%) had the oligoarticular subtype, and 31/54 (57.4%) had the polyarticular subtype. Among the JIA patients, 28.0% tested positive for ANA, and 18.0% were positive for RF. There was a history of uveitis in 13.0% of the patients with JIA. Approximately 6.0% of the patients used sulfasalazine; 10.0% used leflunomide; 31.3% used chloroquine; 33.3% used anti TNF-α; 33.3% used prednisone (at a mean dose of 9.5 mg/day); and 50.9% used methotrexate. The HAQ had a median value of 0.3.
Lipid profile study
The following values were altered in the population of adults with JIA: TC, in 16.6% of the adult JIA patients; HDL-cholesterol, in 23% of the patients; LDL-cholesterol, in 20.3% of the JIA group; and TG, in 16.6% of this group. In the control sample, the following values were altered: TC, in 3.7% of the control patients; HDL-cholesterol in 1.8% of the controls; LDLcholesterol in 1.8% of the control group; and TG, in 18.5% of this group. A comparison between the two populations can be seen in Fig. 1.
The mean values obtained for TC, HDL-cholesterol and TG in the 2 groups are presented in Table 1, in which higher levels of LDL-cholesterol and TC can be observed in the group of adults with JIA and higher levels of HDL-cholesterol and TG can be observed in the control group.
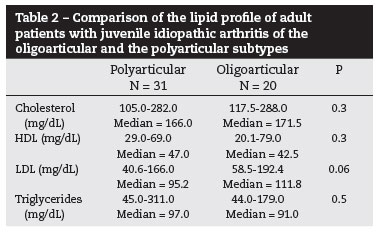
When studying the lipid profiles of the adult patients with JIA according to the subtype at the onset, there were no differences between the oligoarticular and the polyarticular subtypes (Table 2).
The analysis of the association between the lipid profiles and the presence of RF (RF and TC, P = 0.7; RF and HDL, P = 0.3; RF and LDL, P = 0.5; and RF and TG, P = 0.3) or the presence of ANA (ANA and TC, P = 0.8; ANA and HDL, P = 0.8; ANA and LDL, P = 0.6; and ANA and TG, P = 0.7) indicated that the association was not significant. The same was observed for the correlation between the lipid profiles and the length of time of the disease (TC with P = 0.1 and Spearman's R = 0.19; HDL with P = 0.1 and Spearman's R = 0.19; LDL with P = 0.2 and Spearman's R = 0.15; and TG with P = 0.6 and Spearman's R = -0.06).
The analysis of the correlation between the HAQ values and the lipid profiles did not reveal any positive correlation (HAQ and CT, P = 0.1 and Spearman's R = 0.20; HAQ and HDL, P = 0.1 and Spearman's R = 0.20; HAQ and LDL, P = 0.6 and Spearman's R = 0.05; and HAQ and TG, P = 0.9 and Spearman's R = -0.01). However, the correlation between the age at the onset of the disease and the level of TC was positive [P = 0.001; Spearman's R = 0.4; 95% Confidence Interval (CI), 0.1 to 0.6], as was the correlation between the age at disease onset and LDL (P = 0.02; Spearman's R = 0.3; 95% CI, 0.03 to 0.54), while there was no association of this parameter with HDL (P = 0.3; Spearman's R = 0.1) or TG (P = 0.2; Spearman's R = 0.1).
Finally, when comparing the lipid profiles of the patients who were using corticoids (n = 18/54) to those of the patients not using corticoids, we found the following: for the patients using and not using corticoids, the mean values for cholesterol were 173.0 mg/dL and 169.0 mg/dL (P = 0.2); the mean values for LDL-cholesterol were 102.8 mg/dL and 100.6 mg/dL (P = 0.2); the mean values for HDL-cholesterol were 52.1 mg/dL and 44.5 mg/dL (P = 0.03); and the levels of TG were 95.0 mg/dL and 96.5 mg/dL (P = 0.3), respectively.
Discussion
A few results stand out from the data of the present study. The first is, undoubtedly, the changes found in the lipid profiles of the adult individuals with JIA, which displayed proatherogenic characteristics. We observed increases in the TC and LDL-cholesterol levels as well as decreases in the HDLcholesterol levels in the JIA group compared to the control group. None of the characteristics of the JIA, such as the subtype of the disease or the presence of autoantibodies, identified a group with a higher risk for these changes, although the patients with a late onset of the disease had higher values of TC and LDL-cholesterol.
A study by Gonçalves et al.14 conducted with 51 children with JIA (mean age of 11 years) also found decreased HDL, but, unlike the present study, these authors also found increased TG. In our study, the prevalence of hypertriglyceridaemia was the same in the 2 groups; however, the mean level of TG was higher in the control group. Bakkaloglu et al.,15 in a study of 19 Turkish children with JIA, did not find increased TG in the sample.
In the present study, the patients with the oligoarticular subtype of JIA had a tendency to have increased LDL levels. This aspect is interesting because the patients with the polyarticular subtype, i.e., with more intense inflammation, would be expected to be more affected. However, all of these patients were being treated, and thus, the intensity of the inflammatory process may have been equalised by the use of disease modifying drugs.
The use of corticoids is another interesting aspect. In the present sample, the levels of HDL-cholesterol were higher in the patients that were using corticoids. The effect of glucocorticoids on the lipid profiles of rheumatoid patients may be considered to be ambiguous, at best. While, on the one hand, the direct effect of these agents on lipid metabolism suggests that glucocorticoids are pro-atherogenic,8 on the other hand, the anti-inflammatory effects of these corticoids must also be considered.16 The anti-inflammatory action of the corticoids may counteract the lipid changes caused by the inflammation, thus resulting in a more benign lipid profile. Boers et al.16 demonstrated that, in individuals with RA, the dyslipidaemia caused by the activity of the disease was reversed by a treatment that included glucocorticoids. Unfortunately, in the present study, there were no data on the previous use of corticoids, which would have allowed for a study of the effects of cumulative doses of these drugs. Many of the patients analysed herein came from different paediatric clinics, which made it difficult to obtain data on previous treatments.
Another fact that stands out is the existence of lipid abnormalities in a very young population (mean age of 25 years). This last observation is very important when considering the risks of cardiovascular disease and its high morbidity and mortality. The life expectancy in patients with JIA is decreased. French et al.17 found a mortality rate 4 times higher in this population compared to the general population. Thomas et al.18, in a study of the standardized mortality ratio (SMR) in JIA, found an increase of 3-fold for men and 5-fold for women, values that were found to be higher than those obtained for early RA in the adult. However, the reasons for this high mortality are not well known.18,19
Finally, we conclude that adult patients with JIA have an increased risk of dyslipidaemia and that all efforts towards controlling this dyslipidaemia should be undertaken.
REFERENCES
-
1Lowell DJ. Juvenile Idiopathic arthritis: clinical features. In Klippel JH, Stone JH, Crofford LJ, White PH. (eds) Primer on the Rheumatic diseases. New York: Springer, 2008; p.142-53.
-
2Kitas GD, Erb N. Tackling ischaemic heart disease in rheumatoid arthritis. Rheumatology. 2003;42:607-13.
-
3Brenol CV, Monticielo OA, Xavier RM, Brenol JCT. Artrite reumatoide e aterosclerose. Rev Assoc Med Bras. 2007;53:465-70.
-
4Van Doomum S, Mc Coll G, Wicks IP. Accelerated atherosclerosis: an extra articular feature of rheumatoid arthritis? Arthritis Rheum. 2002;46:862-73.
-
5Assous N, Touze E, Meune C, Kahan A, Allanore Y. Cardiovascular disease in rheumatoid arthritis: single center hospital based cohort study in France. Joint Bone Spine. 2007;74:66-72.
-
6Turesson C, Jarenros A, Jacobsson L. Increased incidence of cardiovascular disease in patients with rheumatoid arthritis: result from a community based study. Ann Rheum Dis. 2004;63:952-5.
-
7Avelar AB, de Melo AKG, Souza BD, Avaliação prospectiva do perfil lipídico na artrite reumatoide. Rev Bras Reumatol. 2008;48:213-7.
-
8Maxwell SR, Moot RJ, Kendall MJ. Corticosteroids: do they damage the cardiovascular system? Postgrad Med J. 1994;70:863-70.
-
9Steiner G, Urowitz MB. Lipid profile in patients with rheumatoid arthritis: mechanisms and the impact of treatment. Sem Arthitis Rheum. 2009;38:372-81.
-
10Urban M, Pietrewicz E, Gorska A, Glowinska B. Lipid and homocysteine levels in juvenile idiopathic arthritis. Pol Merkur Lekarski. 2004;17:235-8.
-
11Ilowite NT, Samuel P, Beseler L, Jacobson MS. Dyslipoproteinemia in juvenile rheumatoid arthritis. J Pediatr. 1989;114:823-6.
-
12Ferraz MB, Oliveira LM, Araujo PM, Atra E, Tugwell P. Crosscultural reliability of the physical ability dimension of the health assessment questionnaire. J Rheumatol. 1990;17:813-7.
-
13Santos RD, Giannini SD, Fonseca FH, Moriguchi EH, Maranhão RC, da Luz PL, et al. Sociedade Brasileira de Cardiologia. III Diretrizes Brasileiras Sobre Dislipidemias e Diretriz de Prevenção da Aterosclerose. Arq Bras Cardiol. 2001;77(supl 3):1-48.
-
14Gonçalves M, D'Almeida V, Guerra-Shinohara EM, Galdieri L, Len CA, Hilário MOE. Homocysteine and lipid profile in children with juvenile idiopathic arthritis. Pediatr Rheumatol Online J. 2007;5:2. [Available from: http://www.ped-rheum.com/content/5/1/2]
-
15Bakkaloglu A, Kirel B, Ozen S, Saatçi U, Topaloğlu Beşbaşç N. Plasma lipid and lipoproteins in juvenile chronic arthritis. Clin Rheumatol. 1996;15:341-5.
-
16Boers M, Nurmohamed MT, Doelman CJA, Lard LR, Verhoeven AC, Vaskuyl AE, et al. Influence of glucocorticoids and disease activity on total and high density lipoprotein cholesterol in patients with rheumatoid arthritis. Ann Rheum Dis. 2003;62:842-5.
-
17French AR, Mason T, Nelson AM, O'Fallon WM, Gabriel SE. Increased mortality in adults with a history of juvenile rheumatoid arthritis: a population-based study. Arthritis Rheum. 2001;44:523-7.
-
18Thomas E, Symmons DP, Brewster DH, Black RJ, Macfarlane GJ. National study of cause-specific mortality in rheumatoid arthritis, juvenile chronic arthritis, and other rheumatic conditions: a 20 year follow up study. J Rheumatol. 2003;30:958-65.
-
19Minden K. Adult Outcomes of Patients with Juvenile Idiopathic Arthritis. Horm Res 2009;72(S1):20-5.
Publication Dates
-
Publication in this collection
06 Nov 2013 -
Date of issue
Aug 2013
History
-
Received
22 Dec 2011 -
Accepted
14 May 2013