Abstract
Background:
Reduced respiratory muscle endurance (RME) contributes to increased dyspnea upon exertion in patients with cardiovascular disease.
Objective:
The objective was to characterize ventilatory and metabolic responses during RME tests in post-myocardial infarction patients without respiratory muscle weakness.
Method:
Twenty-nine subjects were allocated into three groups: recent myocardial infarction group (RG, n=9), less-recent myocardial infarction group (LRG, n=10), and control group (CG, n=10). They underwent two RME tests (incremental and constant pressure) with ventilatory and metabolic analyses. One-way ANOVA and repeated measures one-way ANOVA, both with Tukey post-hoc, were used between groups and within subjects, respectively.
Results:
Patients from the RG and LRG presented lower metabolic equivalent and ventilatory efficiency than the CG on the second (50± 06, 50± 5 vs. 42± 4) and third part (50± 11, 51± 10 vs. 43± 3) of the constant pressure RME test and lower metabolic equivalent during the incremental pressure RME test. Additionally, at the peak of the incremental RME test, RG patients had lower oxygen uptake than the CG.
Conclusions
: Post-myocardial infarction patients present lower ventilatory efficiency during respiratory muscle endurance tests, which appears to explain their inferior performance in these tests even in the presence of lower pressure overload and lower metabolic equivalent.
cardiac disease; physical therapy; exercise tolerance; respiratory muscles; muscle fatigue
Introduction
In most myocardial infarction patients, exercise limitation is manifested as dyspnea or
lower limb fatigue11. Morrison NJ, Fairbarn MS, Pardy RL. The effect of breathing frequency
on inspiratory muscle endurance during incremental threshold loading. Chest.
1989;96:85-8. PMid:2736996. http://dx.doi.org/10.1378/chest.96.1.85
http://dx.doi.org/10.1378/chest.96.1.85...
2. Jones NL, Killian KJ. Mechanisms of disease: Exercise limitation in
health and disease. N Engl J Med. 2000;342(9):632-41.
http://dx.doi.org/10.1056/NEJM200008313430907
http://dx.doi.org/10.1056/NEJM2000083134...
3. Caroci AS, Lareau SC, Linda L. Descriptors of dyspnea by patients
with chronic obstructive pulmonary disease versus congestive heart failure. Heart
Lung. 2004;33:102-10. PMid:15024375. http://dx.doi.org/10.1016/j.hrtlng.2003.11.004
http://dx.doi.org/10.1016/j.hrtlng.2003....
4. Hamilton AL, Killian KJ, Summers E, Jones NL. Muscle strength,
symptom intensity, and exercise capacity in patients with cardiorespiratory
disorders. Crit Care Med. 1995;152:2021-31. PMid:8520771.
http://dx.doi.org/10.1164/ajrccm.152.6.8520771
http://dx.doi.org/10.1164/ajrccm.152.6.8...
5. Stendardi L, Grazzini M, Gigliotti F, Lotti P, Scano G. Dyspnea and
leg effort during exercise. Respir Med. 2005;99:933-42. PMid:15950133.
http://dx.doi.org/10.1016/j.rmed.2005.02.005
http://dx.doi.org/10.1016/j.rmed.2005.02...
-
66. Ambrosino N, Serradori M. Determining the cause of dyspnoea:
linguistic and biological descriptors. Chron Respir Dis. 2006;3:117-22.
http://dx.doi.org/10.1191/1479972306cd110ra
http://dx.doi.org/10.1191/1479972306cd11...
. The dyspnea is related, among other possibilities, to respiratory muscle
strength and endurance77. Reid WD, Clarke TJ, Wallace AM. Respiratory muscle injury: evidence
to date and potential mechamisms. Can J Apl Physiol. 2001;26:356-87.
http://dx.doi.org/10.1139/h01-023
http://dx.doi.org/10.1139/h01-023...
, which are expressed as maximal inspiratory pressure (MIP) and respiratory
muscle endurance (time [Tlim] or pressure [PTHMAX]), respectively.
Respiratory muscle weakness (MIP <60% of predicted values)22. Jones NL, Killian KJ. Mechanisms of disease: Exercise limitation in
health and disease. N Engl J Med. 2000;342(9):632-41.
http://dx.doi.org/10.1056/NEJM200008313430907
http://dx.doi.org/10.1056/NEJM2000083134...
,
88. Wastford ML, Murphy A J, Pine MJ. The effects of age in on
respiratory muscle function and performance in older adults. J Sci Med Sport.
2007;10:36-44. PMid:16814604. http://dx.doi.org/10.1016/j.jsams.2006.05.002
http://dx.doi.org/10.1016/j.jsams.2006.0...
9. Ribeiro JP, Chiappa GR, Callegaro CC. The contribution of inspiratory
muscles function to exercise limitation in heart failure: pathophysiological
mechanisms. Rev Bras Fisioter. 2012;16(4):261-267. PMid:22801449.
http://dx.doi.org/10.1590/S1413-35552012005000034
http://dx.doi.org/10.1590/S1413-35552012...
-
1010. Hautmann H, Hefele S, Schotlen K, Huber RM. Maximal inspiratory
mouth pressures (PIMAX) in healthy subjects - what is lower limit of normal? Respir
Med. 2000;94:689-93. PMid:10926341. http://dx.doi.org/10.1053/rmed.2000.0802
http://dx.doi.org/10.1053/rmed.2000.0802...
and reduced respiratory muscle endurance (RME) contribute to greater dyspnea
upon exertion in patients with chronic diseases, including cardiovascular disease11. Morrison NJ, Fairbarn MS, Pardy RL. The effect of breathing frequency
on inspiratory muscle endurance during incremental threshold loading. Chest.
1989;96:85-8. PMid:2736996. http://dx.doi.org/10.1378/chest.96.1.85
http://dx.doi.org/10.1378/chest.96.1.85...
,
22. Jones NL, Killian KJ. Mechanisms of disease: Exercise limitation in
health and disease. N Engl J Med. 2000;342(9):632-41.
http://dx.doi.org/10.1056/NEJM200008313430907
http://dx.doi.org/10.1056/NEJM2000083134...
,
88. Wastford ML, Murphy A J, Pine MJ. The effects of age in on
respiratory muscle function and performance in older adults. J Sci Med Sport.
2007;10:36-44. PMid:16814604. http://dx.doi.org/10.1016/j.jsams.2006.05.002
http://dx.doi.org/10.1016/j.jsams.2006.0...
9. Ribeiro JP, Chiappa GR, Callegaro CC. The contribution of inspiratory
muscles function to exercise limitation in heart failure: pathophysiological
mechanisms. Rev Bras Fisioter. 2012;16(4):261-267. PMid:22801449.
http://dx.doi.org/10.1590/S1413-35552012005000034
http://dx.doi.org/10.1590/S1413-35552012...
-
1010. Hautmann H, Hefele S, Schotlen K, Huber RM. Maximal inspiratory
mouth pressures (PIMAX) in healthy subjects - what is lower limit of normal? Respir
Med. 2000;94:689-93. PMid:10926341. http://dx.doi.org/10.1053/rmed.2000.0802
http://dx.doi.org/10.1053/rmed.2000.0802...
, affecting their exercise tolerance22. Jones NL, Killian KJ. Mechanisms of disease: Exercise limitation in
health and disease. N Engl J Med. 2000;342(9):632-41.
http://dx.doi.org/10.1056/NEJM200008313430907
http://dx.doi.org/10.1056/NEJM2000083134...
,
1111. Dall'Ago P, Chiappa GR, Guths H, Stein R, Ribeiro JP. Inspiratory
muscle training in patients with heart failure and inspiratory muscle weakness: a
randomized trial. J Am Coll Cardiol. 2006 Feb 21;47(4):757-63.
PMid:16487841.
12. Wasserman K, Hansen JE, Sue DY, Whipp BJ, Casaburi R. Principles of
exercise testing and interpretation. Philadelphia: Lea & Febiger;
1999.
13. Windisch W, Hennings E, Sorichter S, Hamm H, Criée CP. Peak or
plateau maximal inspiratory mouth pressure: which is best?. Eur Respir J.
2004;23:708-13. PMid:15176684. http://dx.doi.org/10.1183/09031936.04.00136104
http://dx.doi.org/10.1183/09031936.04.00...
-
1414. Neves LMT, Karsten M, Neves VR, Beltrame T, Borghi-Silva A, Catai
AM. Relationship between inspiratory muscle capacity and peak exercise tolerance in
post-myocardial infarction patients. Heart Lung. 2012;41(2):137-45. PMid:22177761.
http://dx.doi.org/10.1016/j.hrtlng.2011.07.010
http://dx.doi.org/10.1016/j.hrtlng.2011....
.
In any muscle work, ventilatory and metabolic responses are representative of the body's ability to capture, transport, and use oxygen for energy production and eliminate carbon dioxide in order to maintain a relatively normal pH1212. Wasserman K, Hansen JE, Sue DY, Whipp BJ, Casaburi R. Principles of exercise testing and interpretation. Philadelphia: Lea & Febiger; 1999.. Several methodologies are used to evaluate RME. When assessing RME in healthy subjects, the ventilatory and metabolic responses are similar in low-intensity long-duration exercise1515. Neves LMT, Karsten M, Borghi-Silva A, Catai AM. Comportamento ventilatório e metabólico na avaliação da endurance dos músculos inspiratórios de indivíduos pós-infarto do miocárdio. Rev Bras Fisioter. 2010;14 (Supl.1):449.. We believe that reduced RME performance, proportional to the pressure used, might be a consequence of higher metabolic demand, which indicates greater effort during RME testing.
However, to our knowledge, the magnitude of ventilatory and metabolic responses during RME tests has not been well established in patients with acute or chronic myocardial infarction (MI) without respiratory muscle weakness. We hypothesize that the ventilatory efficiency of post-MI patients without respiratory muscle weakness during RME tests is less than that of healthy subjects.
The aim of the present study was to evaluate ventilatory and metabolic responses during respiratory muscle endurance tests in post-myocardial infarction patients and healthy subjects, all with preserved respiratory muscle strength.
Method
This cross-sectional study was approved by the Human Research Ethics Committee of Universidade Federal de São Carlos (UFSCar), São Carlos, SP, Brazil (protocol 353/2009) and conducted in compliance with Declaration of Helsinki guidelines at the Cardiovascular Physical Therapy Laboratory of the Exercise Research Center in the UFSCar Physical Therapy Department. All participants signed an informed consent form.
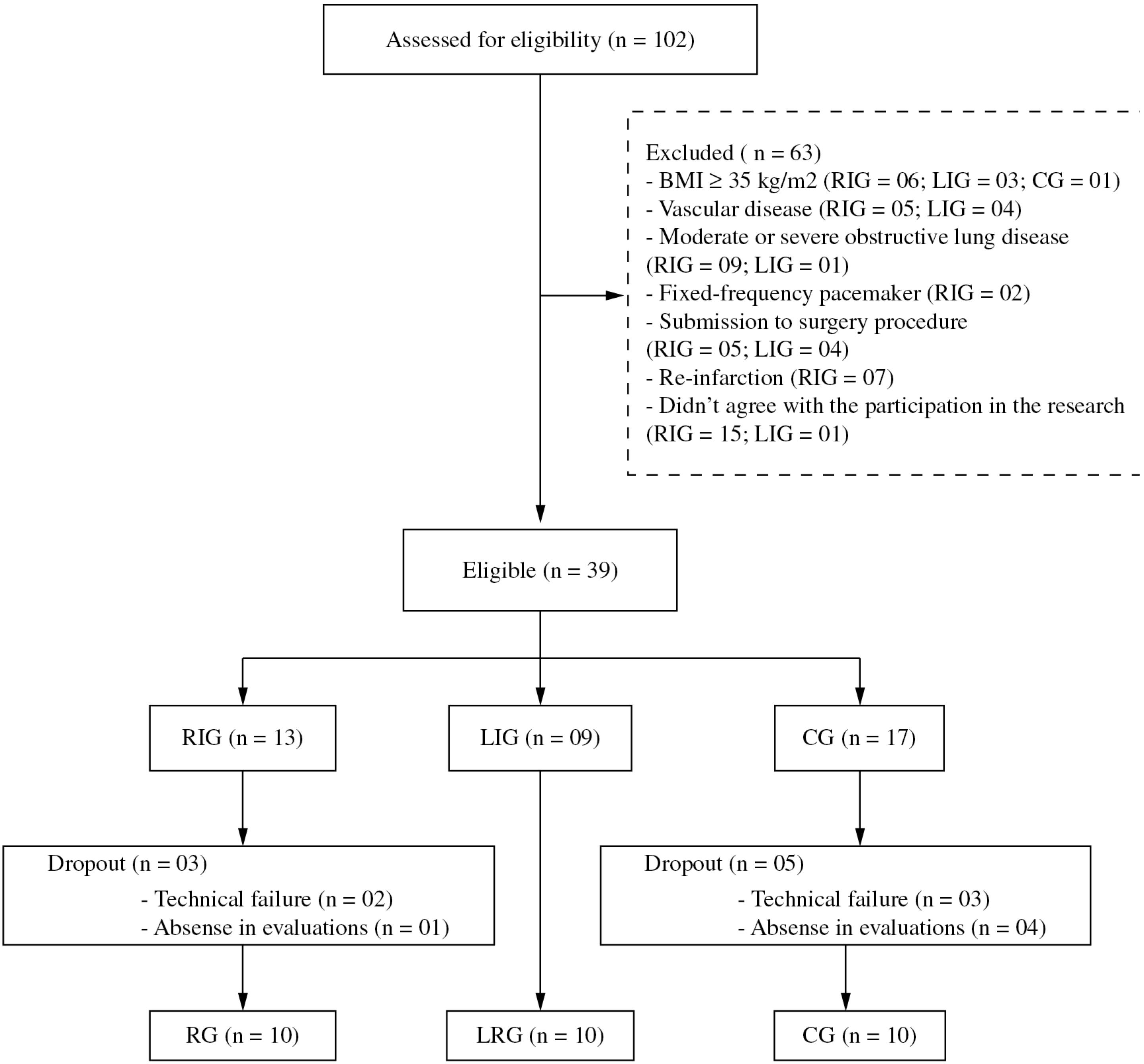
Post-myocardial infarction patients (35-65 years of age) diagnosed with a single episode of myocardial infarction (Killip I) were recruited and allocated into two groups according to the time since occurrence: recent infarction group (RG, n=09; up to 45 days post-MI) and less-recent infarction group (LRG, n=10; at least 6 months post-MI). A control group (CG, n=10) was formed from sedentary healthy subjects matched with the other groups for age, body mass, and height. The post-myocardial infarction patients were identified and recruited from the Coronary Unit of the Santa Casa de Misericórdia de São Carlos either directly during admittance (RG) or from the hospital's database (LRG). CG subjects were identified and recruited from the UFSCar School Health Unit (Figure 1). For all subjects, an adequate cognitive level and absence of musculoskeletal, joint, respiratory, neurological or vascular disorders was required for inclusion.
Diagram of sample distribution for the recent post-myocardial infarction group (RG), the less-recent post-myocardial infarction group (LRG), and the control group (CG).
Subjects were excluded if they presented at least one of the following conditions: body
mass index ≥35 kg/m2, systolic blood pressure ≥140 mmHg and/or diastolic
blood pressure ≥90 mmHg (at rest), functional capacity ≤4 metabolic equivalents;
ST-segment depression >2 mm, angina pectoris during exercise; exercise-induced
decrease in systolic blood pressure ≥15 mmHg, sustained ventricular arrhythmia,
supraventricular arrhythmia that compromised cardiac function, moderate or severe valve
disease, a fixed-frequency pacemaker, uncontrolled diabetes, obstructive lung disease,
difficulty performing a self-assessment of effort using the Borg scale or respiratory
muscle weakness (MIP<60% of the predicted)1010. Hautmann H, Hefele S, Schotlen K, Huber RM. Maximal inspiratory
mouth pressures (PIMAX) in healthy subjects - what is lower limit of normal? Respir
Med. 2000;94:689-93. PMid:10926341. http://dx.doi.org/10.1053/rmed.2000.0802
http://dx.doi.org/10.1053/rmed.2000.0802...
,
1616. Pereira CAC. Espirometria. Diretrizes para testes de função
pulmonar. J Bras Pneumol. 2002;28(3):S1-82..
Clinical and functional evaluation
The subjects underwent a) a physical examination of cardiovascular and respiratory parameters at rest, b) a pulmonary function test - slow and forced vital capacity and maximal voluntary ventilation1616. Pereira CAC. Espirometria. Diretrizes para testes de função pulmonar. J Bras Pneumol. 2002;28(3):S1-82. (CPX-D, Medical Graphics, St Paul, MN, USA), c) a 12-derivation electrocardiogram at rest (Schiller, AT1, Altgasse, Baar, Switzerland) in the supine position, and d) a clinical treadmill exercise test conducted by a cardiologist.
Cardiopulmonary exercise test
A symptom-limited cardiopulmonary exercise test (CPX) was performed on a treadmill
(Master ATL, Inbramed, Porto Alegre, RS, Brazil) using a ramp protocol. A 4-min
warm-up period (1.5 mph, no slope) was followed by an incremental increase in walking
velocity (over 3 min to achieve maximal walking velocity) and then by 0.5% increases
in elevation every 15 s. Ventilatory and metabolic parameters were monitored and
registered breath-by-breath (CPX-D/BreezeSuite 6.4.1, Medical Graphics, St Paul, MN,
USA) and were analyzed after smoothing the data by moving averages of eight
respiratory cycles1717. Balady GJ, Arena R, Sietsema K, Myers J, Coke L, Fletcher GF, et al.
Clinician's Guide to cardiopulmonary exercise testing in adults: a scientific
statement from the American Heart Association. Circulation. 2010;122:191-225.
PMid:20585013. http://dx.doi.org/10.1161/CIR.0b013e3181e52e69
http://dx.doi.org/10.1161/CIR.0b013e3181...
. The electrocardiogram was continuously monitored (Schiller, AT1, Altgasse,
Baar, Switzerland), and the heart rate was recorded with a digital telemetry system
(Polar(r) S810i; Polar Electro Oy, Kempele, Oulo, Finland). Every two
minutes, blood pressure was assessed and perceived exertion was rated with the Borg
scale. The ventilatory method was used by three independent evaluators to identify
the anaerobic threshold (AT)1717. Balady GJ, Arena R, Sietsema K, Myers J, Coke L, Fletcher GF, et al.
Clinician's Guide to cardiopulmonary exercise testing in adults: a scientific
statement from the American Heart Association. Circulation. 2010;122:191-225.
PMid:20585013. http://dx.doi.org/10.1161/CIR.0b013e3181e52e69
http://dx.doi.org/10.1161/CIR.0b013e3181...
,
1818. Higa MN, Silva E, Neves VFC, Catai AM, Gallo L Jr, Sá MFS.
Comparison of anaerobic threshold determined by visual and mathematical methods in
healthy women. Braz J Med Biol Res. 2007;40:501-8. PMid:17401493.
http://dx.doi.org/10.1590/S0100-879X2007000400008
http://dx.doi.org/10.1590/S0100-879X2007...
. Oxygen uptake at this point (VO2AT) was identified. The highest
oxygen uptake observed in the last 30 seconds of exercise was defined as
VO2peak
1818. Higa MN, Silva E, Neves VFC, Catai AM, Gallo L Jr, Sá MFS.
Comparison of anaerobic threshold determined by visual and mathematical methods in
healthy women. Braz J Med Biol Res. 2007;40:501-8. PMid:17401493.
http://dx.doi.org/10.1590/S0100-879X2007000400008
http://dx.doi.org/10.1590/S0100-879X2007...
.
Evaluation of respiratory muscle strength and endurance
Respiratory muscle strength (RMS) was assessed using a digital pressure transducer
(MVD-300, Globalmed, Porto Alegre, RS, Brazil)1919. American Thoracic Society/European Respiratory Society. ATS/ERS
Statement on respiratory muscle testing. Am J Resp Crit Care Med. 2002;166:518-624.
PMid:12186831. http://dx.doi.org/10.1164/rccm.166.4.518
http://dx.doi.org/10.1164/rccm.166.4.518...
. The values observed in the first second after peak pressure were used to
define MIP1010. Hautmann H, Hefele S, Schotlen K, Huber RM. Maximal inspiratory
mouth pressures (PIMAX) in healthy subjects - what is lower limit of normal? Respir
Med. 2000;94:689-93. PMid:10926341. http://dx.doi.org/10.1053/rmed.2000.0802
http://dx.doi.org/10.1053/rmed.2000.0802...
. RME, ventilatory, and metabolic parameters were assessed and registered
breath-by-breath while the patients breathed continuously through the mouthpiece of a
linear inspiratory load resistor (Power Breath(r), IMT Technologies Ltd.,
Birmingham, UK) connected to a ventilatory and metabolic system (CPX-D/BreezeSuite
6.4.1, Medical Graphics, St Paul, MN, USA).
Incremental and constant pressure tests were employed to assess RME, as previously
described1414. Neves LMT, Karsten M, Neves VR, Beltrame T, Borghi-Silva A, Catai
AM. Relationship between inspiratory muscle capacity and peak exercise tolerance in
post-myocardial infarction patients. Heart Lung. 2012;41(2):137-45. PMid:22177761.
http://dx.doi.org/10.1016/j.hrtlng.2011.07.010
http://dx.doi.org/10.1016/j.hrtlng.2011....
. Briefly, the incremental test was based on 3 min steps and began with a load
corresponding to 50% of the MIP, followed by load increments equivalent to 10% MIP
until the patient failed to achieve the pressure in three consecutive inspirations.
The greatest inspiratory pressure sustained at least for 1 min was taken as the
measure for inspiratory muscle endurance (Pthmax)1111. Dall'Ago P, Chiappa GR, Guths H, Stein R, Ribeiro JP. Inspiratory
muscle training in patients with heart failure and inspiratory muscle weakness: a
randomized trial. J Am Coll Cardiol. 2006 Feb 21;47(4):757-63.
PMid:16487841.
,
1919. American Thoracic Society/European Respiratory Society. ATS/ERS
Statement on respiratory muscle testing. Am J Resp Crit Care Med. 2002;166:518-624.
PMid:12186831. http://dx.doi.org/10.1164/rccm.166.4.518
http://dx.doi.org/10.1164/rccm.166.4.518...
. In the next protocol, a constant load (80% of the Pthmax) was
employed. The time elapsed to task failure was defined as respiratory muscle
endurance time (Tlim).
Statistical analysis
The data were analyzed using Statistica 7 (StatSoft Inc., Tulsa, OK, USA). Based on the results of a pilot study (n=4), we estimated that a sample size of six individuals in each group would be sufficient to detect a 10-point difference in ventilatory efficiency (statistical power of 80%). The level of significance was set at 5%. Normally-distributed data (Shapiro-Wilk test) are presented as mean± SD; Non-normally distributed data are expressed as median (minimum and maximum values). One-way ANOVA with Tukey post-hoc and repeated measures one-way ANOVA with Tukey post-hoc were used to analyze data between groups and within subjects, respectively.
Results
Clinical and functional evaluation
The sample consisted of middle-aged men without respiratory muscle weakness and with
similar anthropometric characteristics between groups. For all groups, spirometric
values were within the range predicted for the Brazilian population, with no
difference among groups2020. Neder JA, Andreoni S, Lerario MC, Nery LE. Reference values for lung
function tests. II. Maximal respiratory pressures and voluntary ventilation. Braz J
Med Biol Res. 1999;32:719-27. PMid:10412550.
http://dx.doi.org/10.1590/S0100-879X1999000600007
http://dx.doi.org/10.1590/S0100-879X1999...
. In the post-myocardial infarction groups, the mean time elapsed between
infarction and inclusion in the study was 33 (range: 16 to 45) days in the RG and 702
(range: 219 to 923) days in the LRG. Most had an anterior or inferior infarction
(53%) with bi-arterial involvement (48%) and had undergone percutaneous transluminal
coronary angioplasty (95%). All RG and LRG subjects had preserved ventricular
function with a left ventricular ejection fraction greater than 50%, according to
ventriculography.
Cardiopulmonary exercise test
During the cardiopulmonary exercise test, VO2AT and VO2peak (absolute and corrected for body mass) were higher in the CG than in the RG or LRG (Table 1).
Ventilatory and metabolic responses during RME protocols
The majority of the ventilatory variables (tidal volume, respiratory rate, inspiratory time, expiratory time, inspiratory time relative to total respiratory cycle, minute volume, and partial CO2 pressure at the end of expiration) and metabolic variables (CO2 output and respiratory exchange ratio) remained constant during the incremental and constant pressure tests and did not differ among groups. At 70 and 80% of MIP during the incremental pressure test and during the second and third parts of the constant pressure test, the RG had lower MET and higher VE/VCO2 than the CG. At 90% MIP during the incremental pressure test, the RG had lower VO2 and MET than the CG (Tables 2 and 3).
Furthermore, at all stages of respiratory endurance tests the RG requires a higher
percentage of VO2peak (~5% above compared to CG) to maintain a lower
pressure and product pressure-time1414. Neves LMT, Karsten M, Neves VR, Beltrame T, Borghi-Silva A, Catai
AM. Relationship between inspiratory muscle capacity and peak exercise tolerance in
post-myocardial infarction patients. Heart Lung. 2012;41(2):137-45. PMid:22177761.
http://dx.doi.org/10.1016/j.hrtlng.2011.07.010
http://dx.doi.org/10.1016/j.hrtlng.2011....
(Figure 2). This behavior was not
presented at the higher stage (90% MIP) of incremental pressure test.
Illustration of the percentage of oxygen uptake at peak exercise (%VO2peak) during the incremental and constant pressure protocol endurance tests for the recent infarction group (RG-white), the less-recent infarction group (LRG-black), and the control group (CG-gray).
Discussion
The main results of this descriptive, cross-sectional study provide new evidence about
the source of reduced RME in recent post-MI patients without inspiratory muscle
weakness. The reduction seems to be due to the lower ventilatory efficiency observed
from the middle to the final part of the protocols, even when they experienced lower
pressure overload than sedentary healthy subjects. The sample consisted of middle-aged
men with similar anthropometric characteristics and similar clinical characteristics
between post-MI groups. Only subjects with normal respiratory function were included in
the present study in order to reduce the influence of obstructive or restrictive
diseases on RME performance. Impaired exercise tolerance is a characteristic of cardiac
patients who have undergone angioplasty, although their exercise tolerance tends to
increase with exercise programs2121. Gassner LA, Dunn S, Piller N. Aerobic exercise and the post
myocardial infarction patient: A review of the literature. Heart Lung.
2003;32:258-65. http://dx.doi.org/10.1016/S0147-9563(03)00039-6
http://dx.doi.org/10.1016/S0147-9563(03)...
22. Jones GL, Killian KJ, Summers E, Jones NJ. Inspiratory muscle forces
and endurance in maximum resistive loading. J Appl Physiol. 1985;58:1608-15.
PMid:3997725.
-
2323. Ainsworth BE, Haskell WL, Leon AS, Jacobs DR, Montoye JH, Sallis JF,
et al. Compendium of physical activities: classification of energy costs of human
physical activities. Med Sci Sports Exerc. 1993;25(1):71-80. PMid:8292105.
http://dx.doi.org/10.1249/00005768-199301000-00011
http://dx.doi.org/10.1249/00005768-19930...
. The exercise tolerance of the post-MI sample was greater than that found in
other studies of cardiovascular disease1111. Dall'Ago P, Chiappa GR, Guths H, Stein R, Ribeiro JP. Inspiratory
muscle training in patients with heart failure and inspiratory muscle weakness: a
randomized trial. J Am Coll Cardiol. 2006 Feb 21;47(4):757-63.
PMid:16487841.
,
2424. Chiappa GR, Roseguini BT, Vieira PJC, Alves CN, Tavares A,
Winkelmann ER, et al. Inspiratory muscle training improves blood flow to resting and
exercising limbs in patients with chronic heart failure. J Am Coll Cardiol.
2008;51(17):1663-71. PMid:18436118. http://dx.doi.org/10.1016/j.jacc.2007.12.045
http://dx.doi.org/10.1016/j.jacc.2007.12...
. This difference is likely due to the preserved left ventricular ejection
fraction observed in the present study and also to the fact that our sample was younger
and had a lower body mass index and fewer comorbidities.
The low variation in ventilatory and metabolic values in both incremental and constant
pressure RME tests may reflect a low and almost constant work rate2424. Chiappa GR, Roseguini BT, Vieira PJC, Alves CN, Tavares A,
Winkelmann ER, et al. Inspiratory muscle training improves blood flow to resting and
exercising limbs in patients with chronic heart failure. J Am Coll Cardiol.
2008;51(17):1663-71. PMid:18436118. http://dx.doi.org/10.1016/j.jacc.2007.12.045
http://dx.doi.org/10.1016/j.jacc.2007.12...
. Analyzing other similar studies2222. Jones GL, Killian KJ, Summers E, Jones NJ. Inspiratory muscle forces
and endurance in maximum resistive loading. J Appl Physiol. 1985;58:1608-15.
PMid:3997725.
,
2525. Martyn JB, Moreno RH, Paré PD, Pardy RL. Measurement of inspiratory
muscle performance with threshold loading. Am Rev Respir Dis. 1987;135:919-23.
PMid:3565939.
26. Eastwood PR, Hillman DR, Finucane KF. Inspiratory muscles
performance in endurance athletes and sedentary subjects. Respirology. 2001;6:95-104.
PMid:11422888. http://dx.doi.org/10.1046/j.1440-1843.2001.00314.x
http://dx.doi.org/10.1046/j.1440-1843.20...
27. Freedman S, Campbell EJM. The ability of normal subjects to tolerate
added inspiratory loads. Respir Physiol. 1970;10:213-35.
http://dx.doi.org/10.1016/0034-5687(70)90084-8
http://dx.doi.org/10.1016/0034-5687(70)9...
28. Jederlinic P, Muspratt JA, Miller MJ. Inspiratory muscle training in
clinical practice: physiologic conditioning or habituation to suffocation?. Chest.
1984;86:870-3. PMid:6499549. http://dx.doi.org/10.1378/chest.86.6.870
http://dx.doi.org/10.1378/chest.86.6.870...
29. McElvaney G, Fairban MS, Wilcox PG, Pardy RL. Comparison of
two-minute incremental threshold loading and maximal loading as measures of
respiratory muscles endurance. Chest. 1989;96:557-63. PMid:2766814.
http://dx.doi.org/10.1378/chest.96.3.557
http://dx.doi.org/10.1378/chest.96.3.557...
30. Yan S, Sliwinski P, Gauthier AP, Lichros I, Zakynthinos S, Macklem
PT. Effect of global inspiratory muscle fatigue on ventilatory and respiratory muscle
responses to CO2. J Appl Physiol. 1993;75(3):1371-7. PMid:8226553.
-
3131. Clanton TL, Diaz PT. Clinical assessment of the respiratory muscles.
Phys Ther. 1995;75(11):983-95. PMid:7480128., we inferred that the RME assessment methods involved are the most probable
contributors to the variation in results. The differences include the type of applied
load (elastic or resistive), the mode of load application (constant or incremental), the
degree of motivation and the respiratory pattern (free or controlled)2626. Eastwood PR, Hillman DR, Finucane KF. Inspiratory muscles
performance in endurance athletes and sedentary subjects. Respirology. 2001;6:95-104.
PMid:11422888. http://dx.doi.org/10.1046/j.1440-1843.2001.00314.x
http://dx.doi.org/10.1046/j.1440-1843.20...
. The assessment of oxygen uptake percentage at peak exercise in the RME tests
was an attempt to classify the intensity level of the achieved work rate, and we
observed similar values between groups. However, the fact that the RG performed the RME
tests with a 20% lower pressure than the CG could indicate that the RME tests
represented a proportionally higher intensity for the RG. The consensus for respiratory
tests is that the greater the pressure imposed with higher tidal volume, inspiratory
time or inspiratory flow, the lower the test tolerance is3232. Callegaro CC, Martinez D, Ribeiro PAB, Brod M, Ribeiro JP. Augmented
peripheral chemoreflex in patients with heart failure and inspiratory muscle
weakness. Respir Physiol Neurobiol. 2010;171:31-5. PMid:20096381.
http://dx.doi.org/10.1016/j.resp.2010.01.009
http://dx.doi.org/10.1016/j.resp.2010.01...
. Furthermore, the influence of other factors such as gender, age, and height on
RME test performance is poorly understood1313. Windisch W, Hennings E, Sorichter S, Hamm H, Criée CP. Peak or
plateau maximal inspiratory mouth pressure: which is best?. Eur Respir J.
2004;23:708-13. PMid:15176684. http://dx.doi.org/10.1183/09031936.04.00136104
http://dx.doi.org/10.1183/09031936.04.00...
. We observed that in post-MI patients, lower ventilatory efficiency was not
compensated with superficial breathing and a consequent increase in respiratory
rate2828. Jederlinic P, Muspratt JA, Miller MJ. Inspiratory muscle training in
clinical practice: physiologic conditioning or habituation to suffocation?. Chest.
1984;86:870-3. PMid:6499549. http://dx.doi.org/10.1378/chest.86.6.870
http://dx.doi.org/10.1378/chest.86.6.870...
but, rather, increased recruitment and contraction of respiratory muscles was
the means of generating enough inspiratory flow to deal with the imposed pressure1414. Neves LMT, Karsten M, Neves VR, Beltrame T, Borghi-Silva A, Catai
AM. Relationship between inspiratory muscle capacity and peak exercise tolerance in
post-myocardial infarction patients. Heart Lung. 2012;41(2):137-45. PMid:22177761.
http://dx.doi.org/10.1016/j.hrtlng.2011.07.010
http://dx.doi.org/10.1016/j.hrtlng.2011....
. Considering that the variables were kept constant, other dynamic factors such
as the recruitment of respiratory muscles and changes in airway resistance28 28. Jederlinic P, Muspratt JA, Miller MJ. Inspiratory muscle training in
clinical practice: physiologic conditioning or habituation to suffocation?. Chest.
1984;86:870-3. PMid:6499549. http://dx.doi.org/10.1378/chest.86.6.870
http://dx.doi.org/10.1378/chest.86.6.870...
appear to influence respiratory muscle function and interfere with RME test
performance in post-MI patients.
Study limitations
In this small descriptive cross-sectional study, we showed that there is lower ventilatory efficiency in the second part of test protocols in recent infarction patients without respiratory muscle weakness, even in the presence of lower pressure overload. Our findings were limited by the absence of a breath-by-breath respiratory muscle strength evaluation and patient follow-up over the recent and less recent myocardial infarction periods. Another limitation is related to the lack of predicted values for respiratory muscle endurance in each population.
Clinical implications
Our findings indicate that the reduced RME with increased respiratory effort observed in recent post-MI patients during RME tests could be influenced by their lower ventilatory efficiency. This is the first time that a possible limiting factor for dynamic exercise capacity in post-myocardial infarction has been associated with ventilatory efficiency due to a respiratory muscle component. Our results also raise the question of whether the addition of inspiratory muscle training in cardiac rehabilitation can improve RME performance and ventilatory efficiency in recent post-MI patients without respiratory muscle weakness.
Conclusion
In this descriptive, cross-sectional study with recent and less-recent post-MI patients without inspiratory muscles weakness, the lower ventilatory efficiency observed during respiratory muscle endurance tests appears to clarify the inferior performance on these tests, even in the presence of lower pressure overload.
References
-
1Morrison NJ, Fairbarn MS, Pardy RL. The effect of breathing frequency on inspiratory muscle endurance during incremental threshold loading. Chest. 1989;96:85-8. PMid:2736996. http://dx.doi.org/10.1378/chest.96.1.85
» http://dx.doi.org/10.1378/chest.96.1.85 -
2Jones NL, Killian KJ. Mechanisms of disease: Exercise limitation in health and disease. N Engl J Med. 2000;342(9):632-41. http://dx.doi.org/10.1056/NEJM200008313430907
» http://dx.doi.org/10.1056/NEJM200008313430907 -
3Caroci AS, Lareau SC, Linda L. Descriptors of dyspnea by patients with chronic obstructive pulmonary disease versus congestive heart failure. Heart Lung. 2004;33:102-10. PMid:15024375. http://dx.doi.org/10.1016/j.hrtlng.2003.11.004
» http://dx.doi.org/10.1016/j.hrtlng.2003.11.004 -
4Hamilton AL, Killian KJ, Summers E, Jones NL. Muscle strength, symptom intensity, and exercise capacity in patients with cardiorespiratory disorders. Crit Care Med. 1995;152:2021-31. PMid:8520771. http://dx.doi.org/10.1164/ajrccm.152.6.8520771
» http://dx.doi.org/10.1164/ajrccm.152.6.8520771 -
5Stendardi L, Grazzini M, Gigliotti F, Lotti P, Scano G. Dyspnea and leg effort during exercise. Respir Med. 2005;99:933-42. PMid:15950133. http://dx.doi.org/10.1016/j.rmed.2005.02.005
» http://dx.doi.org/10.1016/j.rmed.2005.02.005 -
6Ambrosino N, Serradori M. Determining the cause of dyspnoea: linguistic and biological descriptors. Chron Respir Dis. 2006;3:117-22. http://dx.doi.org/10.1191/1479972306cd110ra
» http://dx.doi.org/10.1191/1479972306cd110ra -
7Reid WD, Clarke TJ, Wallace AM. Respiratory muscle injury: evidence to date and potential mechamisms. Can J Apl Physiol. 2001;26:356-87. http://dx.doi.org/10.1139/h01-023
» http://dx.doi.org/10.1139/h01-023 -
8Wastford ML, Murphy A J, Pine MJ. The effects of age in on respiratory muscle function and performance in older adults. J Sci Med Sport. 2007;10:36-44. PMid:16814604. http://dx.doi.org/10.1016/j.jsams.2006.05.002
» http://dx.doi.org/10.1016/j.jsams.2006.05.002 -
9Ribeiro JP, Chiappa GR, Callegaro CC. The contribution of inspiratory muscles function to exercise limitation in heart failure: pathophysiological mechanisms. Rev Bras Fisioter. 2012;16(4):261-267. PMid:22801449. http://dx.doi.org/10.1590/S1413-35552012005000034
» http://dx.doi.org/10.1590/S1413-35552012005000034 -
10Hautmann H, Hefele S, Schotlen K, Huber RM. Maximal inspiratory mouth pressures (PIMAX) in healthy subjects - what is lower limit of normal? Respir Med. 2000;94:689-93. PMid:10926341. http://dx.doi.org/10.1053/rmed.2000.0802
» http://dx.doi.org/10.1053/rmed.2000.0802 -
11Dall'Ago P, Chiappa GR, Guths H, Stein R, Ribeiro JP. Inspiratory muscle training in patients with heart failure and inspiratory muscle weakness: a randomized trial. J Am Coll Cardiol. 2006 Feb 21;47(4):757-63. PMid:16487841.
-
12Wasserman K, Hansen JE, Sue DY, Whipp BJ, Casaburi R. Principles of exercise testing and interpretation. Philadelphia: Lea & Febiger; 1999.
-
13Windisch W, Hennings E, Sorichter S, Hamm H, Criée CP. Peak or plateau maximal inspiratory mouth pressure: which is best?. Eur Respir J. 2004;23:708-13. PMid:15176684. http://dx.doi.org/10.1183/09031936.04.00136104
» http://dx.doi.org/10.1183/09031936.04.00136104 -
14Neves LMT, Karsten M, Neves VR, Beltrame T, Borghi-Silva A, Catai AM. Relationship between inspiratory muscle capacity and peak exercise tolerance in post-myocardial infarction patients. Heart Lung. 2012;41(2):137-45. PMid:22177761. http://dx.doi.org/10.1016/j.hrtlng.2011.07.010
» http://dx.doi.org/10.1016/j.hrtlng.2011.07.010 -
15Neves LMT, Karsten M, Borghi-Silva A, Catai AM. Comportamento ventilatório e metabólico na avaliação da endurance dos músculos inspiratórios de indivíduos pós-infarto do miocárdio. Rev Bras Fisioter. 2010;14 (Supl.1):449.
-
16Pereira CAC. Espirometria. Diretrizes para testes de função pulmonar. J Bras Pneumol. 2002;28(3):S1-82.
-
17Balady GJ, Arena R, Sietsema K, Myers J, Coke L, Fletcher GF, et al. Clinician's Guide to cardiopulmonary exercise testing in adults: a scientific statement from the American Heart Association. Circulation. 2010;122:191-225. PMid:20585013. http://dx.doi.org/10.1161/CIR.0b013e3181e52e69
» http://dx.doi.org/10.1161/CIR.0b013e3181e52e69 -
18Higa MN, Silva E, Neves VFC, Catai AM, Gallo L Jr, Sá MFS. Comparison of anaerobic threshold determined by visual and mathematical methods in healthy women. Braz J Med Biol Res. 2007;40:501-8. PMid:17401493. http://dx.doi.org/10.1590/S0100-879X2007000400008
» http://dx.doi.org/10.1590/S0100-879X2007000400008 -
19American Thoracic Society/European Respiratory Society. ATS/ERS Statement on respiratory muscle testing. Am J Resp Crit Care Med. 2002;166:518-624. PMid:12186831. http://dx.doi.org/10.1164/rccm.166.4.518
» http://dx.doi.org/10.1164/rccm.166.4.518 -
20Neder JA, Andreoni S, Lerario MC, Nery LE. Reference values for lung function tests. II. Maximal respiratory pressures and voluntary ventilation. Braz J Med Biol Res. 1999;32:719-27. PMid:10412550. http://dx.doi.org/10.1590/S0100-879X1999000600007
» http://dx.doi.org/10.1590/S0100-879X1999000600007 -
21Gassner LA, Dunn S, Piller N. Aerobic exercise and the post myocardial infarction patient: A review of the literature. Heart Lung. 2003;32:258-65. http://dx.doi.org/10.1016/S0147-9563(03)00039-6
» http://dx.doi.org/10.1016/S0147-9563(03)00039-6 -
22Jones GL, Killian KJ, Summers E, Jones NJ. Inspiratory muscle forces and endurance in maximum resistive loading. J Appl Physiol. 1985;58:1608-15. PMid:3997725.
-
23Ainsworth BE, Haskell WL, Leon AS, Jacobs DR, Montoye JH, Sallis JF, et al. Compendium of physical activities: classification of energy costs of human physical activities. Med Sci Sports Exerc. 1993;25(1):71-80. PMid:8292105. http://dx.doi.org/10.1249/00005768-199301000-00011
» http://dx.doi.org/10.1249/00005768-199301000-00011 -
24Chiappa GR, Roseguini BT, Vieira PJC, Alves CN, Tavares A, Winkelmann ER, et al. Inspiratory muscle training improves blood flow to resting and exercising limbs in patients with chronic heart failure. J Am Coll Cardiol. 2008;51(17):1663-71. PMid:18436118. http://dx.doi.org/10.1016/j.jacc.2007.12.045
» http://dx.doi.org/10.1016/j.jacc.2007.12.045 -
25Martyn JB, Moreno RH, Paré PD, Pardy RL. Measurement of inspiratory muscle performance with threshold loading. Am Rev Respir Dis. 1987;135:919-23. PMid:3565939.
-
26Eastwood PR, Hillman DR, Finucane KF. Inspiratory muscles performance in endurance athletes and sedentary subjects. Respirology. 2001;6:95-104. PMid:11422888. http://dx.doi.org/10.1046/j.1440-1843.2001.00314.x
» http://dx.doi.org/10.1046/j.1440-1843.2001.00314.x -
27Freedman S, Campbell EJM. The ability of normal subjects to tolerate added inspiratory loads. Respir Physiol. 1970;10:213-35. http://dx.doi.org/10.1016/0034-5687(70)90084-8
» http://dx.doi.org/10.1016/0034-5687(70)90084-8 -
28Jederlinic P, Muspratt JA, Miller MJ. Inspiratory muscle training in clinical practice: physiologic conditioning or habituation to suffocation?. Chest. 1984;86:870-3. PMid:6499549. http://dx.doi.org/10.1378/chest.86.6.870
» http://dx.doi.org/10.1378/chest.86.6.870 -
29McElvaney G, Fairban MS, Wilcox PG, Pardy RL. Comparison of two-minute incremental threshold loading and maximal loading as measures of respiratory muscles endurance. Chest. 1989;96:557-63. PMid:2766814. http://dx.doi.org/10.1378/chest.96.3.557
» http://dx.doi.org/10.1378/chest.96.3.557 -
30Yan S, Sliwinski P, Gauthier AP, Lichros I, Zakynthinos S, Macklem PT. Effect of global inspiratory muscle fatigue on ventilatory and respiratory muscle responses to CO2. J Appl Physiol. 1993;75(3):1371-7. PMid:8226553.
-
31Clanton TL, Diaz PT. Clinical assessment of the respiratory muscles. Phys Ther. 1995;75(11):983-95. PMid:7480128.
-
32Callegaro CC, Martinez D, Ribeiro PAB, Brod M, Ribeiro JP. Augmented peripheral chemoreflex in patients with heart failure and inspiratory muscle weakness. Respir Physiol Neurobiol. 2010;171:31-5. PMid:20096381. http://dx.doi.org/10.1016/j.resp.2010.01.009
» http://dx.doi.org/10.1016/j.resp.2010.01.009
Publication Dates
-
Publication in this collection
2014 -
Date of issue
Mar 2014
History
-
Received
01 Oct 2012 -
Reviewed
08 Apr 2013 -
Accepted
18 June 2013