Abstracts
Several studies have already shown the beneficial effects of low intensity pulsed ultrasound on osteogenesis in fracture cases. However, few reports have related the ultrasound action in bone with some injury but without fracture. Thus, we induced a rat osteopenia model by ovariectomy and the proximal third of rat femur was stimulated by ultrasound (200mus burst of 1.5 MHz sine waves repeated at 1.0 kHz, 30mW/cm², SATA) for 20 min/day, during 20 days. After the treatment period, the body weight was significantly higher in the non-treated group than the treated one. No significant difference in bone mineral content was detected among the groups (p > 0.05). Also, no significant difference was noted in the mechanical properties of the femoral diaphysis. However, histologic investigations showed that the treated femur presented less microarchitectural deterioration than the non-treated group. Moreover, it was demonstrated that the treated group did show recent bone formation which was not there in the non-treated group. These results suggest that the low intensity ultrasound can interfere in a positive way on osteoporosis.
Ultrasonics; Bone diseases, Metabolic; Calcium; Phosphorus
Neste estudo, nós analisamos a ação do ultra-som pulsado de baixa intensidade (30mW/cm², freqüência de repetição de 1Khz e ciclo de trabalho de 20%) durante 20 dias consecutivos, por 20 minutos diários, como método não invasivo, em modelo de ratas osteopênicas. A estimulação ultra-sônica foi realizada na região proximal dos fêmures, bilateralmente. Apesar dos resultados quantitativos do conteúdo mineral ósseo (cálcio e fósforo) não terem demonstrado diferenças significativas (p >0,05) entre os grupos tratado e não tratado, as investigações histológicas mostraram que o fêmur tratado apresentou uma menor deterioração microarquitetural que o grupo não tratado, além da ocorrência de neoformação óssea, a qual não foi observada no grupo não tratado. Os resultados sugerem que o ultra-som pulsado de baixa intensidade pode interferir positivamente sobre a osteoporose.
Ultrasom; Osteopatias metabólicas; Cálcio; Fósforo
ORIGINAL ARTICLE
Low intensity ultrasound effects over osteopenic female rats bones
Daniela Cristina Leite de CarvalhoI; Alberto Cliquet JrII
IPhysiotherapist. Master in Bioengineering. Doctorate Student at Post-Graduation Course in Surgery, UNICAMP
IIFull Professor EESC, USP / FCM - UNICAMP
Correspondence Correspondence to Alberto Cliquet Jr Av. Trabalhador Sãocarlense, 400 CEP 13566-590, São Carlos, SP E-mail: cliquet@sel.eesc.sc.usp.br
SUMMARY
Several studies have already shown the beneficial effects of low intensity pulsed ultrasound on osteogenesis in fracture cases. However, few reports have related the ultrasound action in bone with some injury but without fracture. Thus, we induced a rat osteopenia model by ovariectomy and the proximal third of rat femur was stimulated by ultrasound (200ms burst of 1.5 MHz sine waves repeated at 1.0 kHz, 30mW/cm2, SATA) for 20 min/day, during 20 days.
After the treatment period, the body weight was significantly higher in the non-treated group than the treated one. No significant difference in bone mineral content was detected among the groups (p > 0.05). Also, no significant difference was noted in the mechanical properties of the femoral diaphysis. However, histologic investigations showed that the treated femur presented less microarchitectural deterioration than the non-treated group. Moreover, it was demonstrated that the treated group did show recent bone formation which was not there in the non-treated group. These results suggest that the low intensity ultrasound can interfere in a positive way on osteoporosis.
Key words: Ultrasonics; Bone diseases, Metabolic;Calcium/analysis; Phosphorus/analysis
INTRODUCTION
Osteoporosis is a metabolic bone disorder affecting mainly the aged population, generally post-menopausal women due to lack of ovarian hormone. Bony mass reduction, mainly observed in cancellous bone, that is caused by increase in bony reabsorption by osteoclasts changes bones more fragile and subject to fractures(6).
Considering this, treatments preventing and stabilizing this picture are important. However, treatments currently existing for osteoporosis are predominantly pharmacological, and in some cases contra-indicated or poorly tolerated by some patients. Thus, alternative noninvasive techniques are a good choice for controlling bony mass losses.
Based on biomechanical principles, where mechanical load induces bone deformation and consequently causes stimulus for a local bony response, already presented by WOLLF in 1870, the use of low intensity ultrasound is an alternative for bone formation stimulation, since ultrasound is a mechanical energy type generating local bone micro-deformations, similarly to those occurring under external forces(2). Thus, therapy with ultrasound, by promoting beneficial effects over bony structure may be used in diseases challenging the ability to keeping bony mass, such as in osteoporosis. Many studies have already demonstrated the efficacy of low intensity ultrasound in accelerating healing of recent bone fractures, in cases of non-union and in pseudarthrosis(3,8,9,15,17). It was also demonstrated the beneficial effect of low intensity ultrasound in cases of patients with diseases impairing bony recovery, such as osteoporosis and diabetes(10,16). However, there are few studies reporting effects of low intensity ultrasound in cases of osteoporosis without fractures.
Currently, the model of ovariectomized rat is gaining wide acceptance in the study of reflexes of lack of ovarian hormone on skeletal system, as well as in evaluation of bony responses to new treatments.
This study was divided into two parts. In the first it was evaluated the efficacy of a 30-day protocol for induction of osteopenia in femoral bone of female rats, after hysterectomy and bilateral ovariectomy. In the second, it was evaluated the efficiency of pulsed low intensity ultrasound (intensity of 30 mW/cm2, frequency of 1.5 MHz, repetition frequency of 1 KHz, work cycle of 20%), over the proximal region of osteopenic femurs of female rats.
MATERIAL AND METHODS
For the first part were used 11 mature isogenic female Holstman rats (Rattus norvegicus albinus), with about 200 g of body mass. The animals were divided into two groups: control (5 animals) with non-ovarectomized animals and a group of 6 ovarectomized animals. The surgeries were performed under intramuscular anesthesia with Virbaxil 2% in a 0.04 ml/100 g of body mass and by Francotar in a 0.08 ml/100 g of body mass doses. Bilateral ovarectomy was performed in 6 female rats. After the surgery, the animals were feed ad libitum for 30 days. Body mass of the animals were evaluated in baseline and at the end of the experiment. In the 31st day the animals were sacrificed, their left femur removed, cleaned, washed in deionized water and kept in 10% formaldehyde solution for 48 hours. After this period the bones were standardized at 1.5 cm length from the great trochanter and calcined, dissolved in chloride acid at 37% and diluted in 100 ml. This phase allowed the evaluation of the bony mineral content. For quantitative analysis of calcium it was used a flame atomic absorption spectrophotometer and for phosphorus analysis a molecular absorption in an flow injection analysis (FIA) system spectrophotometer. The right femurs were decalcified by MORSE (Sodium citrate 20% and Formic acid 50% - equal parts) dehydrated and included in paraffin. For each piece seven serial longitudinal cuts were performed (6 mm thick) of proximal femur region by means of a microtome that were excluded. Sequentially were performed two serial cuts of 50 mm thick for each piece. After the cuts, these were de-paraffined, dried in stove and fixed to a support used for electronic microscopic evaluation. The cuts were gold metallized in order to facilitate electrical conduction. A LEO model 440 equipment was used.
The differences in body mass in both groups were evaluated with T test, with a 5% significance level.
In the second part of the study, 34 isogenic female Holstman rats (Rattus norvegicus albinus), with about 200 mg of body mass, were used. Se same surgical protocol of the first study part was performed. Due to the efficacy of the 30 days ovariectomy protocol for induction of osteopenia, the treatments started at the 31st day after the surgery. The rats were divided into two groups: a group treated (25 animals) and a control group (9 animals). In the treated group, the rats underwent ultrasound with a spatial and temporal intensity of 30 mW/cm2 in average. The equipment used was fixated to the support table, where the rats were immobilized. The head had normal size allowing the ultrasound application to be performed at the same time in both hind-feet involving a region from femoral head to distal third of femur. A gel was used in order to enable the ultrasound field conduction. The equipment was calibrated previously to its use in the experiment.
The animals in control group did not underwent ultrasound treatment, however suffered the same interference stress can cause to animals. For this, they were immobilized for 20 minutes during 20 consecutive days, that is, the same as with the treated animals.
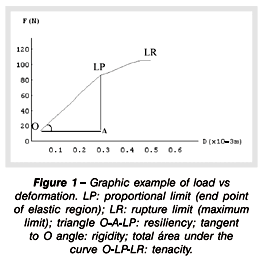
Animals were sacrificed 1 day after the treatment ended, and their left femurs removed and submitted to a three-point flexion mechanical assay in order to determine their mechanical properties. A MTS TestStart II equipment was used for the mechanical assay. The bones were laid over their extremities over two supports with 18 mm distance. The speed of load application was 5 mm/min using a 100 Kgf load cell. For each load increase applied to the bone, it was retrieved the deformation caused, allowing to plot a load versus deformation curve. Through this curve, it was possible to evaluate mechanical properties such as maximal deformation, rigidity, resiliency and tenacity (Figure 1). The parameters were calculated by means of the Mathematics 2.2 software. After the flexion assay, the femurs were standardized at 1.5 cm length from the great trochanter and prepared for a quantitative analysis of calcium and phosphorus. In each pot were placed two bones, except in some cases in it was necessary to put three bones. The right femurs were taken apart for common light microscopy and electronic microscopy. After preparation, the pieces were cut longitudinally (6 mm thick) from proximal femur region by means of a microtome, and the flakes stained with Masson Tricromion.
Sequentially, serial cuts were performed at 50 ìm thick, and prepared to be analyzed under electronic microscopy. The body mass were weighted from baseline to the end of the experiment.
In order to compare the female rats body mass it was used the T test; for comparing mechanical variables and bony mineral contents (calcium and phosphorus) between the groups, the variance analysis (ANOVA). Significance level was 5%.
RESULTS
First study part: at the time of the sacrifice it was observed a significant increase in body mass of ovarectomized female rats as shown in (Table 1).
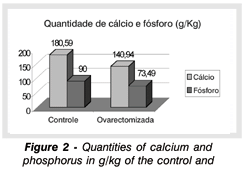
By means of a quantitative analysis of calcium and phosphorus of the femur of animals from both experimental groups, it was observed that rats from control group had a higher amount of calcium and phosphorus (average: calcium, 180.59 g/kg; phosphorus 90 g/kg) in comparison to the group of ovarectomized rats (average: calcium, 140.94 g/kg and phosphorus, 73.49 g/kg). There was a 21.956% reduction in calcium and 18.34% in phosphorus in the ovarectomized rats in comparison to the control ones (Figure 2).
By means of electronic microscopy it was possible to compare the structural differences between femur of control and ovarectomized groups ( Figures 3) shows the differences in cancellous structures between the groups. Through the comparison it is possible to observe that the bony trabeculae in ovarectomized female rats are more spaced and with reduced conectivities in comparison to the normal rats group, characterizing a larger bony reabsorption by osteoclasts in trabecullary plates.
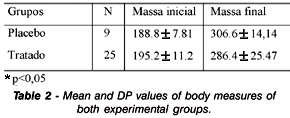
Second part of the study: (Table 2) presents the mean and SD values of the body mass measurements of female rats in control and treated groups, in baseline and at the end of the trial. Statistically significant differences (p= 0.005) in body mass gain between both groups were observed, larger in the control animals.
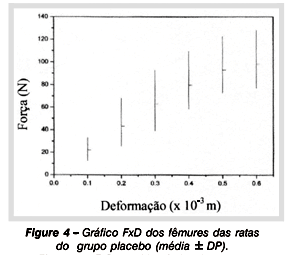
By means of the flexion assay it was possible to plot curves load versus deformation (Figures 4 and 5), so obtaining and comparing the values of mechanical properties of femoral shafts from different experimental groups. Table 3 shows the mean values and SD of mechanical properties of female rats in both groups: control and treated. It is observed that there were no statistically significant differences when mechanical variables were compared (maximum load p = 0,5603; maximum deformation p=0.1551; rigidity p=0.0546; resiliency p=0.5835; tenacity p=0.2926).
The quantitative calcium and phosphorus analysis showed that the group of ovarectomized female rats that underwent low intensity ultrasound treatment reached non-statistically significant values in comparison to those found in control animals (calcium, p=0.734; phosphorus, p=0.396). (Table 4) shows calcium and phosphorus values in both groups.
Figures 6-AB and 6-CD compare the femoral head region between the control and treated groups. Masson Tricromium staining allows studying the bony age. Mature bone is stained in blue and immature ones in red. It is possible to observe that, in control group, bony matrix is predominantly formed by mature bone, without indication of recent bony formation. In the treated group, there are neo-bony formation zones, indicating active osteoblasts. The increase in the osteoblastic activity was subjectively observed, without the use of morphometric methods.
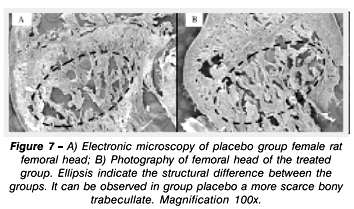
Images obtained by electronic microscopy show structural differences of the femoral head region between control and treated group. It is possible to observe structural differences between the experimental groups, with a larger loss of trabecullar bone in control group (Figures 7 and 8).
DISCUSSION
Ovarectomized female rats is an useful model due to presenting biological mechanisms that are similar to those occurring in osteoporotic women(11,14). This study showed that the ovarian hormone deficiency for 30 days leads to bony loss in mature female rats, and a body mass increase that is statistically significant in comparison to the control group (p < 0.05), supporting the literature reports showing that ovariectomy causes metabolic changes responsible for a pronounced increase in food intake and body mass (as a consequence of increase of body fat)(5,18,19). The body mass increase is higher in ovarectomized rats in comparison to intact female rats in similar food-intake conditions(5,13).
By means of electronic microscopy analysis it was possible to observe structural differences between the femurs of control and ovarectomized groups. The findings support the results of studies that also found trabecullar bone loss in consequence of lack of ovarian hormone(1,6,7,18).
After the analysis of the efficiency of the ovariectomy protocol, it was studied the effect of low intensity ultrasound on osteopenic bones. Our results showed an increase in body mass that was statistically significant in the control group (p = 0.005) in comparison to the treated group, establishing a correlation between ultrasound and a lower body mass increase. However, there were not found in the literature works correlating ultrasound to body mass in ovarectomized animals.
Flexion test results had no statistically significant differences (p > 0.05) in relation to the several studied mechanic variables. The treatment did not interfere in femoral shaft mechanic properties. Ultrasound may have not stimulated an increase in the bony mineral content, and consequently in femoral shaft bony endurance, due to a lower change in bony metabolism in regions with predominance of cortical bone, making them less sensitive to ultrasound stimuli.
Quantitative results of mineral content did not show a significant increase of calcium and phosphorus in osteopenic bone of female rats treated with low intensity ultrasound in comparison to non-treated control female rats. Nevertheless this quantitative analysis failed to show statistically significant differences (p > 0.05) in comparison of the groups, qualitative findings pointed to structural differences between the groups.
The histological analysis (stained with Masson Tricromium) showed bony neoformation in the treated group which was not observed in the control group.
By means of electronic microscopy it could be noticed that in the group treated with low intensity ultrasound, bony micro-architecture appears to be more preserved than the one in the control group, with less bony trabecullar loss, making it to appear as more dense.
From the quantitative and qualitative obtained data, it can be presumed that treatment with low intensity ultrasound in osteopenic bone lead to an improvement in the bony trabecullate. Qualitative results showed a slight improvement of the architecture of treated bone. However, this increase and/or preservation of the mineral content was not quantitatively observed. Thus, it can be supposed that the quantitative analysis did not demonstrate any difference due to the presence of large quantities of new formed not yet mineralized bone 4, 12. Perhaps the duration of the low intensity ultrasound treatment was not sufficient for allowing the total mineralization of the stimulated regions, preventing these changes to be quantitatively observed.
AKNOWLEGEMENTS
To FAPESP (#96/12198-2) and to CAPES for financial support.
Trabalho recebido em 29/09/2002
Aprovado em 27/11/2002
Work performed at Escola de Engenharia de São Carlos - (USP) at the Post-Graduate Course in Interunidades Bioengenharia and at the Ambulatory of the Orthopedics and Traumatology Department of Faculdade de Ciencias Médicas - UNICAMP.
- 1- Canotilho MM. Osteoporose experimental em ratas. Efeitos da administraçăo crônica de ácido acetilsalicílico. [Dissertaçăo], Săo Carlos: Universidade Federal de Săo Carlos; 1996.
- 2- Carvalho DCL, Carvalho MM, Cliquet A Jr., Osteoporose por desuso: aplicaçăo na reabilitaçăo do lesado medular. Acta Ortop Bras 9:34-43, 2001.
- 3- Colombo SJM. Efeitos da variaçăo da intensidade acústica da consolidaçăo ultra-sônica de fraturas experimentais. [Dissertaçăo], Săo Carlos: Campus de Săo Carlos, Universidade de Săo Paulo; 1992.
- 4- Cowin SC. Cortical bone. In: Cowin SC. Bone Mechanics. Flórida: CRC Press, 1989. p.98-142.
- 5- Danielsen CC, Mosekilde L, Svenstrup B. Cortical bone mass, composition, and mechanical properties in female rats in relation to age, long-term ovariectomy and estrogen substitution. Calcif Tissue Int 52:26-33, 1993.
- 6- Dempster DW. Bone remodeling. In: Riggs BL, Melton LJ. Osteoporosis: etiology, diagnosis, and management. 2nded. Philadelphia: Lippincott-Raven Publishers, 1995. p.67-91.
- 7- Devlin H, Ferguson MW, Carter DH. Cancellous bone resorption in the proximal ilium of the ovariectomized rat. Calcif Tissue Int 46:395-400, 1990.
- 8- Duarte LR. The Stimulation of Bone Growth by Ultrasound. Arch Orthop Trauma Surg 101: 153-159, 1983.
- 9- Dyson M. Brookes M. Stimulation of Bone Repair by Ultrasound. Ultrasound Med Biol 2(Suppl.):61-66, 1983.
- 10- Frankel VH. Results of prescription use of pulse ultrasound therapy in fracture management. In: Szabo Z et al., eds. Surgical Technology international. Washington: Universal Medical Press, 1998. p. 389-393. (VII Orthopaedic Surgery).
- 11- Frost HM, Jee WS. On the rat model of human osteopenias and osteoporosis. Bone Miner 18: 227-236, 1992.
- 12- Hadjidakis D, Lempert UG, Minne HW, Ziegler R. Bone loss in experimental diabetes. Comparison with the model of inflammation mediated osteopenia. Hormon Metab Res 25: 77-81, 1993.
- 13- Kalu DN, Liu CC, Hardin RR, Hollis BW. The aged rat model of ovarian hormone deficiency bone loss. Endocrinology 124:7-16, 1989.
- 14- Kalu DN. The ovarectomized rat model of postmenopausal bone loss. Bone Miner 15: 175-192, 1991.
- 15- Kristiansen TK, Ryaby JP, McCabe J, Frey JJ, Roe LR. Accelerated healing of distal radial fractures with the use of specific, low-intensity ultrasound. J Bone Joint Surg Am 79:961-973, 1997.
- 16- Mayr E, Frankel V, Rüter A. Ultrasound - an alternative healing method nonunions? Arch Orthop Trauma Surg 120:1-8, 2000.
- 17- Pilla AA, Mont MA, Nasser PR, et al. Non-invasive low-intensity pulsed ultrasound accelerates bone healing in the rabbit. J Orthop Trauma 4:246-253, 1990.
- 18- Thompson DD, Simmons HA, Pirie CM, Ke HZ. FDA Guidelines and animal models for osteoporosis. Bone 17(suppl):125-133, 1995.
- 19- Wronski TJ, Schench PA, Cintron M, Walsh CC. Effect of body weight on osteopenia in ovariectomized rats. Calcif Tissue Inter 40:155-159, 1987.
Publication Dates
-
Publication in this collection
16 Apr 2003 -
Date of issue
Jan 2003
History
-
Received
29 Sept 2002 -
Accepted
27 Nov 2002