Abstracts
We describe the approach taken by the National Children's Study (NCS) to understanding the role of environmental factors in the development of obesity. We review the literature with regard to the two core hypotheses in the NCS that relate to environmental origins of obesity and describe strategies that will be used to test each hypothesis. Although it is clear that obesity in an individual results from an imbalance between energy intake and expenditure, control of the obesity epidemic will require understanding of factors in the modern built environment and chemical exposures that may have the capacity to disrupt the link between energy intake and expenditure. Through its embrace of the life-course approach to epidemiology, the NCS will be able to study the origins of obesity from preconception through late adolescence, including factors ranging from genetic inheritance to individual behaviors to the social, built, and natural environment and chemical exposures. It will have sufficient statistical power to examine interactions among these multiple influences, including geneenvironment and geneobesity interactions. A major secondary benefit will derive from the banking of specimens for future analysis.
Bisphenol A; Built environment; Endocrine disruptors; Diet; Obesity; Physical activity
Descrevemos a abordagem do National Children's Study (NCS) para entender o papel dos fatores ambientais no desenvolvimento da obesidade. Revisamos a literatura a respeito de duas hipóteses principais no NCS que se relacionam a origens ambientais da obesidade e descrevem estratégias que serão utilizadas para testar cada hipótese. Apesar de estar claro que a obesidade em um indivíduo é resultado de um desequilíbrio entre consumo e gasto de energia, o controle da epidemia de obesidade requer o entendimento de fatores no ambiente moderno e exposições químicas que podem ter a capacidade de interromper a ligação entre o consumo e gasto de energia. Através da aceitação da abordagem do curso de vida a epidemiologia, o NCS será capaz de estudar as origens da obesidade da preconcepção ao final da adolescência, incluindo fatores que vão da herança genética a comportamentos individuais, passando pelo ambiente social, construído e natural e exposições químicas. O estudo terá poder estatístico suficiente para examinar as interações entre essas influências múltiplas, incluindo interações entre gene-ambiente e gene-obesidade. Um benefício secundário importante será derivado do banco de espécime para análise futura.
Bisphenol A; Ambiente construído; Disruptores endócrinos; Dieta; Obesidade; Atividade física
REVISÃO REVIEW
Environment and Obesity in the National Children's Study* * This article was originally published by Environ Health Perspect 117:159166 (2009). doi:10.1289/ehp.11839 available via http://dx.doi.org/ [Online 12 September 2008] and is part of the scientific collaboration between Cien Saude Colet and EHP. The authors declare they have no competing financial interests.
Ambiente e obesidade no National Children's Study
Leonardo TrasandeI,II; Chris CronkIII; Maureen DurkinIV; Marianne WeissV; Dale SchoellerVI; Elizabeth GallIV; Jeanne HewittVII; Aaron CarrelVIII; Philip LandriganI,II; Matthew GillmanIX
IDepartment of Community and Preventive Medicine, Mount Sinai School of Medicine, One Gustave L. Levy Place, Box 1043, New York, NY 10029 USA. leo.trasande@mssm.edu
IIDepartment of Pediatrics, Mount Sinai School of Medicine
IIIMedical College of Wisconsin
IVDepartment of Population Health Sciences, University of Wisconsin
VCollege of Nursing, Marquette University
VIInterdepartmental Program in Nutritional Sciences, University of Wisconsin
VIIMarine and Freshwater Biomedical Sciences Center, University of Wisconsin-Milwaukee
VIIIDepartment of Pediatrics, University of Wisconsin
IXDepartment of Ambulatory Care and Prevention, Harvard Medical School
ABSTRACT
We describe the approach taken by the National Children's Study (NCS) to understanding the role of environmental factors in the development of obesity. We review the literature with regard to the two core hypotheses in the NCS that relate to environmental origins of obesity and describe strategies that will be used to test each hypothesis. Although it is clear that obesity in an individual results from an imbalance between energy intake and expenditure, control of the obesity epidemic will require understanding of factors in the modern built environment and chemical exposures that may have the capacity to disrupt the link between energy intake and expenditure. Through its embrace of the life-course approach to epidemiology, the NCS will be able to study the origins of obesity from preconception through late adolescence, including factors ranging from genetic inheritance to individual behaviors to the social, built, and natural environment and chemical exposures. It will have sufficient statistical power to examine interactions among these multiple influences, including geneenvironment and geneobesity interactions. A major secondary benefit will derive from the banking of specimens for future analysis.
Key words: Bisphenol A, Built environment, Endocrine disruptors, Diet, Obesity, Physical activity
RESUMO
Descrevemos a abordagem do National Children's Study (NCS) para entender o papel dos fatores ambientais no desenvolvimento da obesidade. Revisamos a literatura a respeito de duas hipóteses principais no NCS que se relacionam a origens ambientais da obesidade e descrevem estratégias que serão utilizadas para testar cada hipótese. Apesar de estar claro que a obesidade em um indivíduo é resultado de um desequilíbrio entre consumo e gasto de energia, o controle da epidemia de obesidade requer o entendimento de fatores no ambiente moderno e exposições químicas que podem ter a capacidade de interromper a ligação entre o consumo e gasto de energia. Através da aceitação da abordagem do curso de vida a epidemiologia, o NCS será capaz de estudar as origens da obesidade da preconcepção ao final da adolescência, incluindo fatores que vão da herança genética a comportamentos individuais, passando pelo ambiente social, construído e natural e exposições químicas. O estudo terá poder estatístico suficiente para examinar as interações entre essas influências múltiplas, incluindo interações entre gene-ambiente e gene-obesidade. Um benefício secundário importante será derivado do banco de espécime para análise futura.
Palavras-chave: Bisphenol A, Ambiente construído, Disruptores endócrinos, Dieta, Obesidade, Atividade física
Obesity is the consequence of a chronic net positive energy balance. The prevalence of obesity in American children has trebled in the past 30 years1-3. In 20032006, 31.9% of 2- to 19-year-olds had a body mass index (BMI) > 85th percentile for age and sex4. This great increase in obesity portends future increases in incidence of heart disease5, diabetes6, stroke, and possibly cancer7 and is therefore projected to produce the first decline in U.S. life expectancy since the Great Depression8. The recent explosive increase in prevalence of obesity reflects a complex interplay among a) changes in individual behaviors; b) changes in community structure, lifestyle, and the built environment; and c) possibly exposures to certain synthetic chemicals, such as endocrine disruptors (EDs), that may have the capacity to disrupt energy balance.
Control of the obesity epidemic will require understanding each of these factors and the interplay among them. This understanding will guide development of multipronged evidence-based strategies for obesity control. The goal of this review is to describe the approaches that the National Children's Study (NCS) will employ to develop understanding of the causes of obesity, especially with regard to environmental factors.
Background
Behavioral change is critical to the prevention and treatment of childhood obesity. Yet interventions against obesity that focus solely on modifying individual behavior to increase energy expenditure and/or reduce caloric intake in individual children have had limited success in sustaining weight loss or preventing obesity9. A successful approach to reducing obesity and its comorbidities must also embrace understanding of community-level factors including the social, built, and natural environments. These environmental influences interact with a child's diet, physical activity, genetic makeup, and metabolism10-12. An example of a multipronged approach that took careful cognizance of environmental influences is the success of the state of Arkansas in reducing obesity prevalence among school-age children. A thoughtful redesign of the school environment, with changes to school dietary options, implementation of universal physical education programs, and reduction of access to sugary soft drinks resulted in a decline in the prevalence of overweight children from 20.8% in the 20042005 school year to 20.4% in 2005200613.
Access to safe play spaces may also influence activity patterns and thus reduce risk of obesity14,15. Direct marketing to children (for example, through television ads during child-focused programming) encourages consumption of high-fat and high-sugar content foods and is a negative environmental influence16,17.
Unique windows of vulnerability have been identified for many of the environmental exposures linked to obesity12. Fetal stressors such as maternal nutritional deprivation and smoking can result in intrauterine growth restriction (IUGR) and thereby influence hypothalamicpituitary axis programming to increase future risk of obesity and diabetes10. Infants born to women with insulin-dependent diabetes are at higher risk of obesity, and milder, diet-controlled gestational diabetes may also increase risk18,19. Maternal smoking during pregnancy is an independent risk factor for the development of childhood obesity20,21. Excess gestational weight gain has been associated with increased child adiposity at 3 years of age in at least one prospective cohort22. Exposure to endocrine-disrupting chemicals during pregnancy may enhance the risk for obesity in childhood23. Rapid weight gain during the first year of life24 and fewer hours of sleep during infancy25 further enhance the risk for the development of childhood obesity.
Although previous cohort studies have contributed greatly to identifying many individual-level factors that contribute to the development of obesity in children and its persistence into adulthood both in the United States and in other countries11,26-38, findings from those previous longitudinal studies have several limitations:
. Previous studies have not fully capitalized on the life-course approach to chronic disease epidemiology39, an approach that embraces the concept that adult disease can have its origins in early life (or even fetal) exposures. Barker and Osmond40 promulgated this concept to account for an association between low birth weight and adult ischemic heart disease in Britain and Wales. The concept has been adopted increasingly in the epidemiologic approach to understanding chronic conditions41 including obesity42-44 and neurodegenerative conditions45. The application of the life-course approach to identifying temporal relationships among risk factors for childhood obesity and their interaction is depicted in Figure 1. Multiple studies have documented unique windows of vulnerability to environmental hazards that may contribute to the causation of chronic conditions such as obesity21,46, yet few studies to date have collected the scope of data depicted in this figure at multiple points in the life span.
. Although the Centers for Children's Environmental Health and Disease Prevention have collected data on environmental exposures to pregnant women and young children, these research centers have rarely focused on child weight status as an outcome47. This weakness is especially relevant in light of new knowledge from animal studies, which suggest that endocrine-disrupting chemicals may modulate response to dietary intake48,49, disrupt the hypothalamicpituitary axis50, and possibly increase risk for childhood obesity23.
. Although some studies have collected genetic data on participants and have been able to identify polymorphisms that increase risk for obesity, they have not simultaneously collected the data on environmental exposures that are necessary to examine carefully the interactions of genetic and environmental factors with diet and physical activity.
. Recent studies also suggest that obesity develops as a chronic condition much earlier than the school-age years51. Earlier cohort studies that were first initiated when obesity in the preschool years was relatively infrequent are unlikely to provide data on exposures in early life that are essential to identify prenatal and early childhood risk factors for obesity.
. Many previous cohorts were limited in their capacity to identify risk factors for obesity that may be unique among Hispanics, a population for which obesity prevalence is increasing especially rapidly2,52.
. Previous cohorts are limited in that they have not included sufficient numbers of children to draw contrasts between risk factors specific to rural and urban environments34.
. Past studies have been unable to allow accurate assessment of the role of access to parks and other places that encourage physical activity among children living in urban areas53.
. Many cohort studies were begun before the tripling of childhood obesity prevalence occurred3,55,56 - a trend increasingly attributed to the collective effect of community-level factors for which policy changes may be the only effective means for preventing further increases in obesity prevalence9. To assess the impact of these more recent community-level factors, new cohorts in which these risk factors exist are needed.
. Although studies from other countries, such as the Avon Longitudinal Study of Parents and Children11,12 and the Danish National Birth Cohort57, will provide important insights into the etiology of childhood obesity, the environmental factors that contribute to obesity in American children are likely to be different, and the pool of genetic polymorphisms that modify risk may be much different from that of European children.
Progress of the NCS
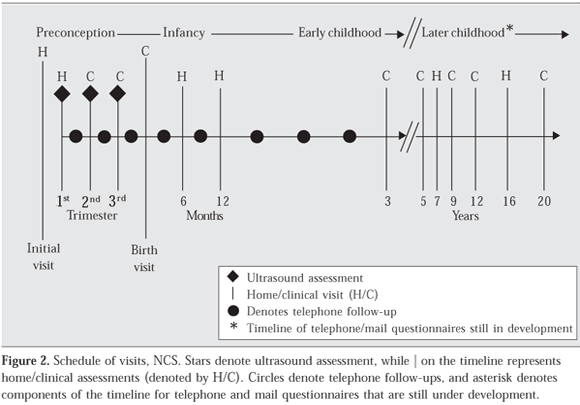
In response to increases in the prevalence of obesity and a number of other chronic conditions, the U.S. Congress, through the Children's Health Act of 2000, authorized the National Institute of Child Health and Human Development (NICHD) "to conduct a national longitudinal study of environmental influences (including physical, chemical, biological, and psychosocial) on children's health and development"58. The design of the NCS has been extensively described elsewhere59-62. With assistance from the staff of the National Center for Health Statistics at the Centers for Disease Control and Prevention, NCS staff developed a multistage clustered sampling approach to enroll a sample of 100,000 live births representative of all American children63. Families who are enrolled in the study will participate in a minimum of 13 data collection encounters: at least one visit before conception; two times during pregnancy; at birth; at 6, 12, and 18 months of age in early childhood; at 3, 5, 7, 9, and 12 years of age in childhood; and at 16 and 20 years of age in adolescence (Figure 2). Figure 2 depicts the timeline of visits across the complete study, and Tables 1 and 2 describe the measurements planned for preconception through 3 years of age for the seven Vanguard (pilot) locations. Enrollment of women will occur in 105 primary sampling units (counties or, in the case of more sparsely populated areas, clusters of counties) and began in January 2009.
The mission of the NCS is to provide the federal government with a scientifically robust guide to disease prevention, and to assure scientific rigor the study has always been hypothesis-driven. The topical working groups convened by the NCS Advisory Committee developed initial core hypotheses for the study, in consultation with thousands of scientists and representatives from community groups and professional organizations. A current list of hypotheses with supporting scientific rationales that were accepted and refined by the Interagency Coordinating Committee [composed of senior scientists from NICHD, the National Institute of Environmental Health Sciences, the Centers for Disease Control and Prevention, and the U.S. Environmental Protection Agency (EPA)] is available on the NCS website64.
Childhood obesity is a lead focus of the NCS and is addressed in 6 of 30 core hypotheses. Table 3 presents the gaps of knowledge that remain with respect to four of these core hypotheses: obesity and insulin resistance from impaired maternal glucose metabolism; obesity and insulin resistance associated with IUGR; breast-feeding associated with lower rates of obesity and lower risk of insulin resistance and fiber; and whole grains, high glycemic index, insulin resistance, and obesity.
Table 3 also presents how the NCS will address these gaps through its design. In this review, we highlight how the study will provide important new knowledge with regard to two core hypotheses that link factors in the chemical and built environments with childhood obesity.
Obesity-Related Hypotheses of the NCS
Impact of neighborhood environment on risk of obesity and insulin resistance
Built environment features such as mixed land use, increased proximity to recreational activities and green space, as well as safety (e.g., low crime rates and perceived traffic safety for pedestrian and bicyclists) have been associated in cross-sectional studies with increased physical activity65-67 and lower risk of obesity among adults68-70. Few studies have examined the impact of the built environment on younger children, and those studies have focused upon circumscribed geographic areas and/or socioeconomically advantaged and ethnically homogeneous communities71. Decreased access to healthy eating choices in low socioeconomic status neighborhoods has been documented in at least two studies72,73. Factors such as climate and topography have been taken into account infrequently74. The effect of after school and summer adult organized programs on obesity and insulin resistance is unknown. In the absence of such programs, parents living in urban areas may instruct their children to go directly home from school where indoor activities are largely limited to watching television and playing computer games in the security of the home.
A systematic review of previous studies of the built environment and childhood obesity identified inconsistencies in measurements of the built environment across studies and cross-sectional designs as major deficits of previous studies, and noted that these studies rarely studied both diet and physical activity71. Because of its focus on community characterization60, the NCS will allow more careful identification of those features of neighborhoods that affect physical activity and diet, such as proximity to play spaces, availability of healthy food stores, and neighborhood walkability.
The NCS represents a major opportunity to explore the role of specific aspects of the neighborhood environment at different periods in a child's development. Access to safe play spaces near a child's home, for example, may be especially protective against obesity during the early school years, but less so during adolescence. The design of the NCS capitalizes on the life-course approach and allows for separate analyses of the impact of certain factors on the development of obesity or increase in adiposity within certain time periods. Simultaneous collection of socioeconomic and genetic data as well as measures of diet and physical activity (Tables 1 and 2) will permit careful distinction of the role of certain environmental risk factors during each window of vulnerability.
Chemical environmental agents and the endocrine system
The impact of EDs on humans was first identified by Herbst and Bern, who observed eight cases of clear cell adenocarcinoma of the vagina in young women who had been exposed inuteroto diethylstilbestrol (DES), a synthetic estrogen prescribed to pregnant women in the 1950s, 1960s, and 1970s to prevent miscarriage75. Prenatal exposure to DES has been found subsequently to induce obesity in an animal model23. Identification of endocrine-disrupting chemicals has been limited by the lack of toxicity testing data available for many chemicals in widespread use76.
Because so few chemicals have been tested for their toxicity, the possibility exists that other chemicals besides DES influence somatic growth and obesity48,50. One potential endocrine-disrupting chemical, bisphenol A (BPA), is used to manufacture polycarbonate resin in the coatings of food and beverage containers77. Exposure to BPA, phthalates, and other EDs is widespread in American children78, and animal studies increasingly suggest the potential for toxicity at current levels of exposure79. Invitro studies have found that BPA induces fibroblast differentiation into adipocytes80. Animal studies have found that BPA affects glucose transport in fat cells81. BPA also disrupts glucagon secretion in intact Langerhans cells at nanomolar levels82. These studies raise the possibility that BPA could be a risk factor for the development of obesity, a question undergoing examination in at least one Center for Children's Environmental Health and Disease Prevention83.
Phthalates are used in a variety of personal care products such as shampoos and in the synthesis of polyvinyl chloride84. Phthalates have been documented consistently in animal studies to have antiandrogenic effects85-87. Cohort studies have begun to assess for potential effects in humans and suggest susceptibility at lower levels of exposure than those documented to have effects in animals. It is hypothesized that the most severe effects may be associated with exposures in prenatal and early postnatal life. Decreases in anogenital distance among infant males have been associated with elevated urinary phthalate levels during pregnancy88, and breast milk levels of monoester phthalates have been associated with higher serum hormone binding globulin levels and luteinizing hormone to free testosterone ratios89. Diminished sperm motility has been identified among exposed men90-92, and low-molecular-weight phthalates have been associated with increased birth weight and longer duration of gestation in at least one birth cohort83. Although few studies have analyzed the impact of phthalate exposure on increased adiposity in children, analysis of the 1999 - 2002 National Health and Nutrition Examination Survey has identified increases in urinary phthalate levels among men with increased waist circumference and homeostatic model assessment, a measure of insulin resistance93.
Lack of accurate information on the level and timing of past exposures to EDs has been the principal limitation of most previous studies of the potential human impacts of EDs. This limitation will be directly addressed by the prospective design of the NCS. In the NCS, exposures to chemicals will be measured during pregnancy, in breast milk, and in the perinatal period before the appearance of health effects. The large sample size will facilitate investigation of possible links between low-prevalence endocrine-disruptor exposures and health outcomes, and state-of-the-art laboratory assessment of chemical exposures will further sharpen the study's ability to discern effects of exposures to EDs. The large sample size will also permit study of genetic polymorphisms and geneenvironment interactions, which may unearth individual differences in susceptibility to EDs. As new EDs are identified, specimens can be withdrawn from the NCS repository to analyze their content for appropriate biomarkers to assess whether these EDs may be risk factors in the development of obesity94.
Conclusion
The NCS presents previously unrealized opportunities for the identification of risk factors for childhood obesity, and for their subsequent elimination through prevention. Just as the Framingham Heart Study provided health care providers with hitherto novel information on risk factors for cardiovascular disease that enabled them to offer evidence-based advice to limit smoking, reduce the intake of fatty foods, and control hypertension, the NCS will suggest interventions that can be used to prevent obesity by communities, policy makers, and child health providers. A major strength of the study is that it will be representative of American children. It is anticipated, for example, that > 20,000 children in the cohort will be Hispanic, permitting examination of unique risk factors among a subgroup that has been disproportionately affected by the epidemic.
The hypotheses presented in this review cover only a small percentage of the findings likely to emerge from the NCS. The core NCS hypotheses are dynamic, and as the study is implemented, new questions will emerge and result in modifications to the study protocol. Others may be clearly answered through the NCS or other studies, or become outdated as the whole body of knowledge adjusts the direction of inquiry. For some areas of inquiry where the science is in relatively nascent stages, the major benefits to be gained from the study derive from its hypothesis-generating nature. The NCS will provide a major opportunity to confirm putative genetic links identified in other studies through the study of genetic sequences of children and their families95. As new putative EDs are identified, subsamples of biospecimens stored at the NCS Specimen Repository can be rapidly analyzed to test for associations in a large-scale cohort that represents the population of U.S. children.
Of course, no observational study by itself can demonstrate causality. The NCS will identify risk factors for which causality may be suggested on the basis of strength, consistency, temporality, biological gradient, and plausibility. Findings from the NCS will prompt further interventions such as randomized controlled trials, policy interventions, and other initiatives that will confirm or refute the role of identified risk factors in the development of obesity and its associated comorbidities.
The life-course approach underlying the design of the NCS may very well lead to delineating the duration and impact of environmental, behavioral, and social exposures on risk for obesity. No study will have followed women from preconception and subsequently followed their children at such frequent intervals early in childhood and then through adolescence and young adulthood. The NCS will collect an array of biospecimens, dietary and physical activity data, and social and chemical environmental factors on all 100,000 children for all proposed data collection time points, whereas other cohorts have collected more limited data at each time point or collected complete data on a smaller sample.
A major challenge of the NCS will be to overcome the difficulties in measuring physical activity, diet, and anthropometry in children that have bedeviled past studies. Limitations of reliability and validity do exist with food-frequency questionnaires96,97 and other instruments commonly used to measure dietary intake, although promising alternatives have been developed for populations in which past instruments have not proven reliable98. The vagaries of collecting information on physical activity by questionnaire are well documented99, but accelerometry and other measuring techniques are increasingly promising in their precision and application100,101. BMI is not a perfect measure of adiposity102, and dual-absorption X-ray absorptiometry has been strongly correlated with cardiovascular disease factors in children103. Bioimpedance analysis and skinfold thickness are increasingly used to measure adiposity104,105.
These challenges will not be easily dismissed, and the opportunity is ripe for contributions from the obesity research community to ensure that the best questionnaires and measurement approaches are utilized in an efficient and cost-effective way. At this time, the protocol has been finalized only for the seven Vanguard (pilot) locations, and even for those locations only through birth. The NCS also offers major opportunities to study the validity and reliability of alterative measurement approaches through adjunct studies in collaboration with existing study centers. These studies may use the full or a subsample of the study cohort, with the caveat that proposed new data collection not impose undue additional burden on study participants or additional financial burden on the study.
The NCS will also trigger ancillary and follow-up studies and provide the next generation of obesity researchers opportunities to apply for funding106. The NCS will make public use, deidentified data sets available in accordance with federal privacy regulations.
Previous cohort studies of cardiovascular risk have plowed the terrain to identify major risk factors and allow the NCS to close in on solutions to the epidemic of childhood obesity. However, they have also demonstrated that these relationships are complex and temporally dependent, making a large longitudinal cohort study beginning in the prenatal period essential. The NCS thus offers us great hope in combating the obesity epidemic among America's children.
Acknowledgements
The authors are investigators in the Queens, New York, and Waukesha County, Wisconsin, Vanguard Centers and the Coordinating Center of the National Children's Study. This project has been funded in whole or in part with federal funds from the National Institute of Child Health and Human Development, National Institutes of Health, under contracts NICHD HHSN 275200503411C/N01-HD-5-3411.
References
1. Ogden CL, Carroll MD, Curtin LR, McDowell MA, Tabak CJ, Flegal KM. Prevalence of overweight and obesity in the United States, 19992004. JAMA 2006; 295(13):1549-1555.
2. Strauss RS, Pollack HA. Epidemic increase in childhood overweight, 19861998. JAMA 2001; 286(22):2845-2848.
3. Troiano RP, Flegal KM, Kuczmarski RJ, Campbell SM, Johnson CL. Overweight prevalence and trends for children and adolescents. The National Health and Nutrition Examination Surveys, 1963 to 1991. Arch Pediatr Adolesc Med 1995; 149(10):1085-1091.
4. Ogden CL, Carroll MD, Flegal KM. High body mass index for age among US children and adolescents, 20032006. JAMA 2008; 299(20):2401-2405.
5. Bibbins-Domingo K, Coxson P, Pletcher MJ, Lightwood J, Goldman L. Adolescent overweight and future adult coronary heart disease. New Engl J Med 2007; 357(23):2371-2379.
6. Lee JM, Okumura MJ, Freed GL, Menon RK, Davis MM. Trends in hospitalizations for diabetes among children and young adults: United States, 1993-2004.Diabetes Care 2007; 30(12):3035-3039.
7. Bjørge T, Engeland A, Tverdal A, Smith GD. Body mass index in adolescence in relation to cause-specific mortality: a follow-up of 230,000 Norwegian adolescents. Am J Epidemiol 2008; 168:3037.
8. Olshansky SJ, Passaro D, Hershow R, Layden J, Carnes BA, Brody J, Hayflick L, Butler RN, Allison DB, Ludwig DS. A possible decline in life expectancy in the United States in the 21st century. N Engl J Med2005; 352:1138-1145.
9. Summerbell CD, Waters E, Edmunds LD, Kelly S, Brown T, Campbell KJ. Interventions for preventing obesity in children [Review]. Cochrane Database Syst Rev 2005; 3:CD001871.
10. Meaney MJ, Seckl JR. Glucocorticoid programming. Ann NY Acad Sci 2004; 1032:63-84.
11. Moll PP, Burns TL, Lauer RM. The genetic and environmental sources of body mass index variability: the Muscatine Ponderosity Family Study. Am J Hum Genet 1991; 49(6):1243-1255.
12. Ong KK, Northstone K, Wells JC, Rubin C, Ness AR, Golding J, Dunger DB. Earlier mother's age at menarche predicts rapid infancy growth and childhood obesity. PLoS Med 2007; 4(4):e132.
13. [Anonymous.] Improving individual and community health through health promotion strategies: local case study Joseph Thompson and the Body Mass Index Assessment Project in Arkansas. In: Moments in Leadership: Case Studies in Public Health Policy and Practice (Debuono B, Gonzalez AR, and Rosenbaum S, eds). New York: Pfizer Inc.; 2007. p. 127-132.
14. Ewing R, Schmid T, Killingsworth R, Zlot A, Raudenbush S. Relationship between urban sprawl and physical activity, obesity, and morbidity. Am J Health Promot 2003; 18(1):47-57.
15. Frumkin H, Frank L, Jackson R. Urban Sprawl and Public Health: Designing, Planning, and Building for Healthy Communities. Washington, D.C.: Island Press; 2004.
16. Gortmaker SL, Must A, Sobol AM, Peterson K, Colditz GA, Dietz WH. Television viewing as a cause of increasing obesity among children in the United States, 1986-1990. Arch Pediatr Adolesc Med 1996; 150(4):356362.
17. Lobstein T, Dibb S. Evidence of a possible link between obesogenic food advertising and child overweight. Obes Rev 2005; 6(3):203-208.
18. Bo S, Menato G, Gallo ML, Bardelli C, Lezo A, Signorile A, Gambino R, Cassader M, Massobrio M, Pagano G. Mild gestational hyperglycemia, the metabolic syndrome and adverse neonatal outcomes. Acta Obstet Gynecol Scand 2004; 83(4):335-340.
19. Dabelea D, Hanson RL, Lindsay RS, Pettitt DJ, Imperatore G, Gabir MM, Roumain J, Bennett PH, Knowler WC. Intrauterine exposure to diabetes conveys risks for type 2 diabetes and obesity: a study of discordant sibships. Diabetes 2000; 49(12):2208-2211.
20. Bergmann KE, Bergmann RL, von Kries R, Böhm O, Richter R, Dudenhausen JW, Wahn U. Early determinants of childhood overweight and adiposity in a birth cohort study: role of breast-feeding. Int J Obes 2003; 27:162-172.
21. Oken E, Levitan EB, Gillman MW. Maternal smoking during pregnancy and child overweight: systematic review and meta-analysis. Int J Obes 2008; 32:201-210.
22. Oken E, Taveras EM, Kleinman KP, Rich-Edwards JW, Gillman MW. Gestational weight gain and child adiposity at age 3 years. Am J Obstet Gynecol 2007; 196(4):322-322.
23. Newbold RR, Padilla-Banks E, Snyder RJ, Phillips TM, Jefferson WN. Developmental exposure to endocrine disruptors and the obesity epidemic. Reprod Toxicol 2007; 23(3):290-296.
24. Reilly JJ, Armstrong J, Dorosty AR, Emmett PM, Ness A, Rogers I, Steer C, Sherriff A; Avon Longitudinal Study of Parents and Children Study Team. Early life risk factors for obesity in childhood: cohort study. BMJ 2005; 330:1357.
25. Taveras EM, Rifas-Shiman SL, Oken E, Gunderson EP, Gillman MW. Short sleep duration in infancy and risk of childhood overweight. Arch Pediatr Adolesc Med 2008; 162(4):305-311.
26. Berkey CS, Rockett HRH, Field AE, Gillman MW, Frazier AL, Camargo CA, Colditz GA. Activity, dietary intake, and weight changes in a longitudinal study of preadolescent and adolescent boys and girls. Pediatrics 2000; 105:e56.
27. Demerath EW, Li J, Sun SS, Chumlea WC, Remsberg KE, Czerwinski SA, Towne B, Siervogel RM. Fifty-year trends in serial body mass index during adolescence in girls: the Fels Longitudinal Study. Am J Clin Nutr 2004; 80(2):441-446.
28. Freedman DS, Khan LK, Serdula MK, Dietz WH, Srinivasan SR, Berenson GS. The relation of childhood BMI to adult adiposity: the Bogalusa Heart Study. Pediatrics 2005; 115(1):22-27.
29. Gordon-Larsen P, Nelson MC, Page P, Popkin BM. Inequality in the built environment underlies key health disparities in physical activity and obesity. Pediatrics 2006; 117(2):417-424.
30. Guo SS, Wu W, Chumlea WC, Roche AF. Predicting overweight and obesity in adulthood from body mass index values in childhood and adolescence. Am J Clin Nutr 2002; 76(3):653-658.
31. Lake JK, Power C, Cole TJ. Child to adult body mass index in the 1958 British birth cohort: associations with parental obesity. Arch Dis Child 1997; 77(5):376-381.
32. Lauer RM, Clarke WR, Burns TL. Obesity in childhood: the Muscatine Study. Zhonghua Min Guo Xiao Er Ke Yi Xue Hui Za Zhi 1997; 38(6):432-437.
33. Nader PR, O'Brien M, Houts R, Bradley R, Belsky J, Crosnoe R, Friedman S, Mei Z, Susman EJ; National Institute of Child Health and Human Development Early Child Care Research Network. Identifying risk for obesity in early childhood. Pediatrics 2006; 118(3):e594-601.
34. Nelson MC, Gordon-Larsen P, Song Y, Popkin BM. Built and social environments associations with adolescent overweight and activity. Am J Prev Med 2006; 31(2):109-117.
35. Parsons TJ. Fetal and early life growth and body mass index from birth to early adulthood in 1958 British cohort: longitudinal study. BMJ 2001; 323(7325):1331-1335.
36. Siervogel RM, Wisemandle W, Maynard LM, Guo SS, Chumlea WC, Towne B. Lifetime overweight status in relation to serial changes in body composition and risk factors for cardiovascular disease: The Fels Longitudinal Study. Obes Res 2000; 8(6):422-430.
37. Strauss RS, Knight J. Influence of the home environment on the development of obesity in children. Pediatrics 1999; 103(6):e85.
38. Thompson DR, Obarzanek E, Franko DL, Barton BA, Morrison J, Biro FM, Daniels SR, Striegel-Moore RH. Childhood overweight and cardiovascular disease risk factors: The National Heart, Lung, and Blood Institute Growth and Health Study. J Pediatr 2007; 150(1):18-25.
39. Ben-Shlomo Y, Kuh D. A life course approach to chronic disease epidemiology: conceptual models, empirical challenges and interdisciplinary perspectives. Int J Epidemiol 2002; 31:285-293.
40. Barker DJ, Osmond C. Infant mortality, childhood nutrition, and ischaemic heart disease in England and Wales. Lancet 1986; 1(8489):1077-1081.
41. Lynch J, Smith GD. A life course approach to chronic disease epidemiology. Annu Rev Public Health 2005; 26(1):1-35.
42. Gillman MW. A life course approach to overweight and obesity. In: A Life Course Approach to Chronic Diseases Epidemiology (Kuh D, Ben-Shlomo Y, eds). Oxford, UK: Oxford University Press; 2004.
43. James SA, Fowler-Brown A, Raghunathan TE, Van Hoewyk J. Life-course socioeconomic position and obesity in African American women: The Pitt County Study. Am J Public Health 2006; 96:554-560.
44. Novak M, Ahlgren C, Hammarström A. A life-course approach in explaining social inequity in obesity among young adult men and women. Int J Obes 2006; 30:191-200.
45. Landrigan PJ, Sonawane B, Butler RN, Trasande L, Callan R, Droller D. Early environmental origins of neurodegenerative disease in later life. Environ Health Perspect 2005; 113:1230-1233.
46. National Research Council. Pesticides in the Diets of Infants and Children. Washington, D.C.: National Academy Press; 1993.
47. Wolff MS, Britton JA, Boguski L, Hochman S, Maloney N, Serra N, Liu Z, Berkowitz G, Larson S, Forman J. Environmental exposures and puberty in innercity girls. Environ Res 2008; 107(3):393-400.
48. Bhathena SJ, Velasquez MT. Beneficial role of dietary phytoestrogens in obesity and diabetes. Am J Clin Nutr 2002; 76(6):1191-1201.
49. Enan E, Lasley B, Stewart D, Overstreet J, Vandevoort CA. 2, 3, 7, 8-Tetrachlorodibenzo-p-dioxin (TCDD) modulates function of human luteinizing granulosa cells via cAMP signaling and early reduction of glucose transporting activity. Reprod Toxicol 1996; 10(3):191-198.
50. Rubin BS, Murray MK, Damassa DA, King JC, Soto AM. Perinatal exposure to low doses of bisphenol A affects body weight, patterns of estrous cyclicity, and plasma LH levels. Environ Health Perspect 2001; 109:675-680.
51. Kim J, Peterson KE, Scanlon KS, Fitzmaurice GM, Must A, Oken E, Rifas-Shiman SL, Rich-Edwards JW, Gillman MW. Trends in overweight from 1980 through 2001 among preschool-aged children enrolled in a health maintenance organization. Obesity (Silver Spring) 2006; 14(7):1107-1112.
52. Freedman DS, Khan LK, Serdula MK, Ogden CL, Dietz WH. Racial and ethnic differences in secular trends for childhood BMI, weight, and height. Obesity (Silver Spring) 2006; 14(2):301-308.
53. Kipke MD, Iverson E, Moore D, Booker C, Ruelas V, Peters AL, Kaufman F. Food and park environments: neighborhood-level risks for childhood obesity in East Los Angeles. J Adolesc Health 2007; 40(4):325-333.
54. Glass TA, McAtee MJ. Behavioral science at the crossroads in public health: extending horizons, envisioning the future. Soc Sci Med 2006; 62(7):1650-1671.
55. Kroke A, Hahn S, Buyken AE, Liese AD. A comparative evaluation of two different approaches to estimating age at adiposity rebound. Int J Obes 2006; 30:261-266.
56. Wisemandle W, Maynard LM, Guo SS, Siervogel RM. Childhood weight, stature, and body mass index among never overweight, early-onset overweight, and late-onset overweight groups. Pediatrics 2000; 106(1):e14.
57. Olsen J, Melbye M, Olsen SF, Sorensen TIA, Aaby P, Nybo Andersen AM, . The Danish National Birth Cohortits background, structure and aim. Scand J Public Health 2001; 29(4):300-307.
58. Children's Health Act of 2000. Public Law 106-310.
59. Branum AM, Collman GW, Correa A, Keim SA, Kessel W, Kimmel CA, Taxbøl D, Hansen KD, Juhl M, Schow TB, Sørensen HT, Andresen J, Mortensen EL, Olesen AW, Søndergaard C. The National Children's Study of environmental effects on child health and development. Environ Health Perspect 2003; 111:642-646.
60. Landrigan PJ, Trasande L, Thorpe LE, Gwynn C, Lioy PJ, D'Alton ME, Lipkind HS, Swanson J, Wadhwa PD, Clark EB, Rauh VA, Perera FP, Susser E. The National Children's Study: a 21-year prospective study of 100,000 American children. Pediatrics 2006; 118(5):2173-2186.
61. Trasande L, Landrigan PJ. The National Children's Study: a critical national investment. Environ Health Perspect2004; 112:A789-A790.
62. Trasande L, Cronk CE, Leuthner SR, Hewitt JB, Durkin MS, McElroy JA, Anderson HA, Landrigan PJ. The National Children's Study and the children of Wisconsin. WMJ 2006; 105(2):50-54.
63. Strauss W LJ, Menkedick J, Ryan L, Pivetz T, McMillan N, Pierce B, Rust S. White Paper on Evaluation of Sampling Design Options for the National Children's Study. [accessed 1 May 2007]. Available from: http://www.nationalchildrensstudy.gov/research/analytic_reports/upload/Executive-Summary-for-the-White-Paper-on-Evaluation-of-Sampling-Design-Options-for-the-National-Children-s-Study.pdf
64. NCS. National Children's Study Home Page. [accessed 1 July 2008]. Available from: www.national childrensstudy.gov
65. Cervero R, Duncan M. Walking, bicycling, and urban landscapes: evidence from the San Francisco Bay Area. Am J Public Health2003; 93(9):1478-1483.
66. Ellaway A, Macintyre S, Bonnefoy X. Graffiti, greenery, and obesity in adults: secondary analysis of European cross sectional survey. BMJ2005; 331(7517): 611-612.
67. Li F, Fisher KJ, Brownson RC, Bosworth M. Multilevel modelling of built environment characteristics related to neighbourhood walking activity in older adults. BMJ 2005; 59(7):558-564.
68. Ewing R, Brownson RC, Berrigan D. Relationship between urban sprawl and weight of United States youth. Am J Prev Med2006; 31(6):464-474.
69. Frank LD, Andresen MA, Schmid TL. Obesity relationships with community design, physical activity, and time spent in cars. Am J Prev Med2004; 27(2):87-96.
70. Lopez R. Urban sprawl and risk for being overweight or obese. Am J Public Health2004; 94:1574-1579.
71. Papas MA, Alberg AJ, Ewing R, Helzlsouer KJ, Gary TL, Klassen AC. The built environment and obesity. Epidemiol Rev2007; 29(1):129143.
72. Galvez MP, Morland K, Raines C, Kobil J, Siskind J, Godbold J, Brenner B. Race and food store availability in an inner-city neighbourhood. Public Health Nutr2008; 11(6):624-631.
73. Morland K, Diez Roux AV, Wing S. Supermarkets, other food stores, and obesity: the Atherosclerosis Risk in Communities Study. Am J Prev Med 2006; 30(4):333-339.
74. Timperio A, Ball K, Salmon J, Roberts R, Giles-Corti B, Simmons D, Baur LA, Crawford D. Personal, family, social, and environmental correlates of active commuting to school. Am J Prev Med2006; 30(1):45-51.
75. Bern H. The fragile fetus. In: Chemically-Induced Alteration in Sexual and Functional Development: The Wildlife/Human Connection (Colborn T, Clement C, eds). Princeton, NJ: Princeton Scientific Publishing; 1992. p. 915.
76. U.S. EPA. Chemical Hazard Data Availability Study: What Do We Really Know About the Safety of High Production Volume Chemicals? Washington, D.C.: Office of Pollution Prevention and Toxics, U.S. Environmental Protection Agency; 1998.
77. Brotons J. Xenoestrogens released from lacquer coatings in food cans. Environ Health Perspect1995; 103:608-612.
78. Centers for Disease Control and Prevention. Third National Report on Human Exposure to Environmental Chemicals. Atlanta, GA: Centers for Disease Control and Prevention; 2005.
79. Vom Saal FS, Hughes C. An extensive new literature concerning low-dose effects of bisphenol A shows the need for a new risk assessment. Environ Health Perspect2005; 113:926-934.
80. Masuno H, Kidani T, Sekiya K, Sakayama K, Shiosaka T, Yamamoto H, Honda K. Bisphenol A in combination with insulin can accelerate the conversion of 3T3-L1 fibroblasts to adipocytes. J Lipid Res 2002; 43(5):676-684.
81. Sakurai K, Kawazuma M, Adachi T, Harigaya T, Saito Y, Hashimoto N, Mori C. Bisphenol A affects glucose transport in mouse 3T3-F442A adipocytes. Br J Pharmacol 2004; 141:209-214.
82. Alonso-Magdalena P, Laribi O, Ropero AB, Fuentes E, Ripoll C, Soria B, Nadal A. Low doses of bisphenol A and diethylstilbestrol impair Ca2+ signals in pancreatic a-cells through a nonclassical membrane estrogen receptor within intact islets of Langerhans. Environ Health Perspect2005; 113:969-967.
83. Wolff MS, Engel SM, Berkowitz GS, Ye X, Silva MJ, Zhu C, Wetmur J, Calafat AM. Prenatal phenol and phthalate exposures and birth outcomes. Environ Health Perspect2008; 116:1092-1097.
84. Sathyanarayana S. Phthalates and children's health. Curr Probl Pediatr Adolesc Health Care 2008; 38(2):34-49.
85. Bell FP. Effects of phthalate esters on lipid metabolism in various tissues, cells and organelles in mammals. Environ Health Perspect1982; 45:41-50.
86. Fisher JS. Environmental anti-androgens and male reproductive health: focus on phthalates and testicular dysgenesis syndrome. Reproduction 2004; 127(3):305-315.
87. Parks LG, Ostby JS, Lambright CR, Abbott BD, Klinefelter GR, Barlow NJ, Gray Jr. LE. The plasticizer diethylhexyl phthalate induces malformations by decreasing fetal testosterone synthesis during sexual differentiation in the male rat. Toxicol Sci 2000; 58:339-349.
88. Swan SH, Main KM, Liu F, Stewart SL, Kruse RL, Calafat AM, Mao CS, Redmon JB, Ternand CL, Sullivan S, Teague JL, Study for Future Families Research Team. Decrease in anogenital distance among male infants with prenatal phthalate exposure. Environ Health Perspect2005; 113:1056-1061.
89. Main KM, Mortensen GK, Kaleva MM, Boisen KA, Damgaard IN, Chellakooty M, Schmidt IM, Suomi AM, Virtanen HE, Petersen DV, Andersson AM, Toppari J, Skakkebaek NE. Human breast milk contamination with phthalates and alterations of endogenous reproductive hormones in infants three months of age. Environ Health Perspect2006; 114:270276.
90. Duty SM, Silva MJ, Barr DB, Brock JW, Ryan L, Chen Z, Herrick RF, Christiani DC, Hauser R. Phthalate exposure and human semen parameters. Epidemiology 2003; 14(3):269-277.
91. Hauser R. The environment and male fertility: recent research on emerging chemicals and semen quality. Semin Reprod Med 2006; 24(3):156-167.
92. Hauser R, Meeker JD, Duty S, Silva MJ, Calafat AM. Altered semen quality in relation to urinary concentrations of phthalate monoester and oxidative metabolites. Epidemiology 2006; 17(6):682-691.
93. Stahlhut RW, van Wijngaarden E, Dye TD, Cook S, Swan SH. Concentrations of urinary phthalate metabolites are associated with increased waist circumference and insulin resistance in adult US males. Environ Health Perspect2007; 115:876-882.
94. Landrigan P, Garg A, Droller DBJ. Assessing the effects of endocrine disruptors in the National Children's Study. Environ Health Perspect2003; 111:1678-1682.
95. Landrigan PJ, Trasande L, Swanson JM. Genetics, altruism, and the National Children's Study. Am J Med Genet A 2008; 146(3):294-296.
96. Coates RJ, Monteilh CP. Assessments of food-frequency questionnaires in minority populations. Am J Clin Nutr 1997; 65(suppl 4):1108S-1115S.
97. Teufel NI. Development of culturally competent food-frequency questionnaires. Am J Clin Nutr 1997; 65(4 suppl): 1173S-1178S.
98. Yaroch A, Resnicow KEN, Davis M, Davis A, Smith M, Khan LK. Development of a modified picture-sort food frequency questionnaire administered to low-income, overweight, African-American adolescent girls. J Am Dietetic Assoc 2000; 100(9):1050-1056.
99. Kohl HW, Fulton JE, Caspersen CJ. Assessment of physical activity among children and adolescents: a review and synthesis. Prev Med 2000; 31(2):54-76.
100. Ekelund ULF, Sjöström M, Yngve A, Poortvliet E, Nilsson A, Froberg K, Wedderkopp N, Westerterp K. Physical activity assessed by activity monitor and doubly labeled water in children.Med Sci Sports Exerc 2001; 33(2):275-281.
101. Janz KF, Levy SM, Burns TL, Torner JC, Willing MC, Warren JJ. Fatness, physical activity, and television viewing in children during the adiposity rebound period: The Iowa Bone Development Study. Prev Med 2002; 35(6):563-571.
102. Pietrobelli A, Faith MS, Allison DB, Gallagher D, Chiumello G, Heymsfield SB. Body mass index as a measure of adiposity among children and adolescents: a validation study. J Pediatr 1998; 132(2):204-210.
103. Lindsay RS, Hanson RL, Roumain J, Ravussin E, Knowler WC, Tataranni PA. Body mass index as a measure of adiposity in children and adolescents: relationship to adiposity by dual energy x-ray absorptiometry and to cardiovascular risk factors. J Clin Endocrinol Metab 2001; 86:4061-4067.
104. Gutin B, Litaker M, Islam S, Manos T, Smith C, Treiber F. Body-composition measurement in 9-11-y-old children by dual-energy X-ray absorptiometry, skinfold-thickness measurements, and bioimpedance analysis. Am J Clin Nutr 1996; 63(3):287-292.
105. Kettaneh A, Heude B, Lommez A, Borys JM, Ducimeticre P, Charles MA. Reliability of bioimpedance analysis compared with other adiposity measurements in children: The FLVS II Study. Diabetes Metab 2005; 31(6):534-541.
106. Lyman WD, Barone C, Castle V, Davies HD, Stanton B, Paneth N.Making the National Children's Study a real partnership with academic Pediatrics. J Pediatr 2005; 147(5):563-564.
Received 21 June 2008
Accepted 11 September 2008
- 1. Ogden CL, Carroll MD, Curtin LR, McDowell MA, Tabak CJ, Flegal KM. Prevalence of overweight and obesity in the United States, 19992004. JAMA 2006; 295(13):1549-1555.
- 2. Strauss RS, Pollack HA. Epidemic increase in childhood overweight, 19861998. JAMA 2001; 286(22):2845-2848.
- 3. Troiano RP, Flegal KM, Kuczmarski RJ, Campbell SM, Johnson CL. Overweight prevalence and trends for children and adolescents. The National Health and Nutrition Examination Surveys, 1963 to 1991. Arch Pediatr Adolesc Med 1995; 149(10):1085-1091.
- 4. Ogden CL, Carroll MD, Flegal KM. High body mass index for age among US children and adolescents, 20032006. JAMA 2008; 299(20):2401-2405.
- 5. Bibbins-Domingo K, Coxson P, Pletcher MJ, Lightwood J, Goldman L. Adolescent overweight and future adult coronary heart disease. New Engl J Med 2007; 357(23):2371-2379.
- 6. Lee JM, Okumura MJ, Freed GL, Menon RK, Davis MM. Trends in hospitalizations for diabetes among children and young adults: United States, 1993-2004.Diabetes Care 2007; 30(12):3035-3039.
- 7. Bjørge T, Engeland A, Tverdal A, Smith GD. Body mass index in adolescence in relation to cause-specific mortality: a follow-up of 230,000 Norwegian adolescents. Am J Epidemiol 2008; 168:3037.
- 8. Olshansky SJ, Passaro D, Hershow R, Layden J, Carnes BA, Brody J, Hayflick L, Butler RN, Allison DB, Ludwig DS. A possible decline in life expectancy in the United States in the 21st century. N Engl J Med2005; 352:1138-1145.
- 10. Meaney MJ, Seckl JR. Glucocorticoid programming. Ann NY Acad Sci 2004; 1032:63-84.
- 11. Moll PP, Burns TL, Lauer RM. The genetic and environmental sources of body mass index variability: the Muscatine Ponderosity Family Study. Am J Hum Genet 1991; 49(6):1243-1255.
- 12. Ong KK, Northstone K, Wells JC, Rubin C, Ness AR, Golding J, Dunger DB. Earlier mother's age at menarche predicts rapid infancy growth and childhood obesity. PLoS Med 2007; 4(4):e132.
- 13. [Anonymous.] Improving individual and community health through health promotion strategies: local case study Joseph Thompson and the Body Mass Index Assessment Project in Arkansas. In: Moments in Leadership: Case Studies in Public Health Policy and Practice (Debuono B, Gonzalez AR, and Rosenbaum S, eds). New York: Pfizer Inc.; 2007. p. 127-132.
- 14. Ewing R, Schmid T, Killingsworth R, Zlot A, Raudenbush S. Relationship between urban sprawl and physical activity, obesity, and morbidity. Am J Health Promot 2003; 18(1):47-57.
- 15. Frumkin H, Frank L, Jackson R. Urban Sprawl and Public Health: Designing, Planning, and Building for Healthy Communities. Washington, D.C.: Island Press; 2004.
- 16. Gortmaker SL, Must A, Sobol AM, Peterson K, Colditz GA, Dietz WH. Television viewing as a cause of increasing obesity among children in the United States, 1986-1990. Arch Pediatr Adolesc Med 1996; 150(4):356362.
- 17. Lobstein T, Dibb S. Evidence of a possible link between obesogenic food advertising and child overweight. Obes Rev 2005; 6(3):203-208.
- 18. Bo S, Menato G, Gallo ML, Bardelli C, Lezo A, Signorile A, Gambino R, Cassader M, Massobrio M, Pagano G. Mild gestational hyperglycemia, the metabolic syndrome and adverse neonatal outcomes. Acta Obstet Gynecol Scand 2004; 83(4):335-340.
- 19. Dabelea D, Hanson RL, Lindsay RS, Pettitt DJ, Imperatore G, Gabir MM, Roumain J, Bennett PH, Knowler WC. Intrauterine exposure to diabetes conveys risks for type 2 diabetes and obesity: a study of discordant sibships. Diabetes 2000; 49(12):2208-2211.
- 20. Bergmann KE, Bergmann RL, von Kries R, Böhm O, Richter R, Dudenhausen JW, Wahn U. Early determinants of childhood overweight and adiposity in a birth cohort study: role of breast-feeding. Int J Obes 2003; 27:162-172.
- 21. Oken E, Levitan EB, Gillman MW. Maternal smoking during pregnancy and child overweight: systematic review and meta-analysis. Int J Obes 2008; 32:201-210.
- 22. Oken E, Taveras EM, Kleinman KP, Rich-Edwards JW, Gillman MW. Gestational weight gain and child adiposity at age 3 years. Am J Obstet Gynecol 2007; 196(4):322-322.
- 23. Newbold RR, Padilla-Banks E, Snyder RJ, Phillips TM, Jefferson WN. Developmental exposure to endocrine disruptors and the obesity epidemic. Reprod Toxicol 2007; 23(3):290-296.
- 24. Reilly JJ, Armstrong J, Dorosty AR, Emmett PM, Ness A, Rogers I, Steer C, Sherriff A; Avon Longitudinal Study of Parents and Children Study Team. Early life risk factors for obesity in childhood: cohort study. BMJ 2005; 330:1357.
- 25. Taveras EM, Rifas-Shiman SL, Oken E, Gunderson EP, Gillman MW. Short sleep duration in infancy and risk of childhood overweight. Arch Pediatr Adolesc Med 2008; 162(4):305-311.
- 26. Berkey CS, Rockett HRH, Field AE, Gillman MW, Frazier AL, Camargo CA, Colditz GA. Activity, dietary intake, and weight changes in a longitudinal study of preadolescent and adolescent boys and girls. Pediatrics 2000; 105:e56.
- 27. Demerath EW, Li J, Sun SS, Chumlea WC, Remsberg KE, Czerwinski SA, Towne B, Siervogel RM. Fifty-year trends in serial body mass index during adolescence in girls: the Fels Longitudinal Study. Am J Clin Nutr 2004; 80(2):441-446.
- 28. Freedman DS, Khan LK, Serdula MK, Dietz WH, Srinivasan SR, Berenson GS. The relation of childhood BMI to adult adiposity: the Bogalusa Heart Study. Pediatrics 2005; 115(1):22-27.
- 29. Gordon-Larsen P, Nelson MC, Page P, Popkin BM. Inequality in the built environment underlies key health disparities in physical activity and obesity. Pediatrics 2006; 117(2):417-424.
- 30. Guo SS, Wu W, Chumlea WC, Roche AF. Predicting overweight and obesity in adulthood from body mass index values in childhood and adolescence. Am J Clin Nutr 2002; 76(3):653-658.
- 31. Lake JK, Power C, Cole TJ. Child to adult body mass index in the 1958 British birth cohort: associations with parental obesity. Arch Dis Child 1997; 77(5):376-381.
- 32. Lauer RM, Clarke WR, Burns TL. Obesity in childhood: the Muscatine Study. Zhonghua Min Guo Xiao Er Ke Yi Xue Hui Za Zhi 1997; 38(6):432-437.
- 33. Nader PR, O'Brien M, Houts R, Bradley R, Belsky J, Crosnoe R, Friedman S, Mei Z, Susman EJ; National Institute of Child Health and Human Development Early Child Care Research Network. Identifying risk for obesity in early childhood. Pediatrics 2006; 118(3):e594-601.
- 34. Nelson MC, Gordon-Larsen P, Song Y, Popkin BM. Built and social environments associations with adolescent overweight and activity. Am J Prev Med 2006; 31(2):109-117.
- 35. Parsons TJ. Fetal and early life growth and body mass index from birth to early adulthood in 1958 British cohort: longitudinal study. BMJ 2001; 323(7325):1331-1335.
- 36. Siervogel RM, Wisemandle W, Maynard LM, Guo SS, Chumlea WC, Towne B. Lifetime overweight status in relation to serial changes in body composition and risk factors for cardiovascular disease: The Fels Longitudinal Study. Obes Res 2000; 8(6):422-430.
- 37. Strauss RS, Knight J. Influence of the home environment on the development of obesity in children. Pediatrics 1999; 103(6):e85.
- 38. Thompson DR, Obarzanek E, Franko DL, Barton BA, Morrison J, Biro FM, Daniels SR, Striegel-Moore RH. Childhood overweight and cardiovascular disease risk factors: The National Heart, Lung, and Blood Institute Growth and Health Study. J Pediatr 2007; 150(1):18-25.
- 39. Ben-Shlomo Y, Kuh D. A life course approach to chronic disease epidemiology: conceptual models, empirical challenges and interdisciplinary perspectives. Int J Epidemiol 2002; 31:285-293.
- 40. Barker DJ, Osmond C. Infant mortality, childhood nutrition, and ischaemic heart disease in England and Wales. Lancet 1986; 1(8489):1077-1081.
- 41. Lynch J, Smith GD. A life course approach to chronic disease epidemiology. Annu Rev Public Health 2005; 26(1):1-35.
- 42. Gillman MW. A life course approach to overweight and obesity. In: A Life Course Approach to Chronic Diseases Epidemiology (Kuh D, Ben-Shlomo Y, eds). Oxford, UK: Oxford University Press; 2004.
- 43. James SA, Fowler-Brown A, Raghunathan TE, Van Hoewyk J. Life-course socioeconomic position and obesity in African American women: The Pitt County Study. Am J Public Health 2006; 96:554-560.
- 44. Novak M, Ahlgren C, Hammarström A. A life-course approach in explaining social inequity in obesity among young adult men and women. Int J Obes 2006; 30:191-200.
- 45. Landrigan PJ, Sonawane B, Butler RN, Trasande L, Callan R, Droller D. Early environmental origins of neurodegenerative disease in later life. Environ Health Perspect 2005; 113:1230-1233.
-
46National Research Council. Pesticides in the Diets of Infants and Children. Washington, D.C.: National Academy Press; 1993.
- 47. Wolff MS, Britton JA, Boguski L, Hochman S, Maloney N, Serra N, Liu Z, Berkowitz G, Larson S, Forman J. Environmental exposures and puberty in innercity girls. Environ Res 2008; 107(3):393-400.
- 48. Bhathena SJ, Velasquez MT. Beneficial role of dietary phytoestrogens in obesity and diabetes. Am J Clin Nutr 2002; 76(6):1191-1201.
- 49. Enan E, Lasley B, Stewart D, Overstreet J, Vandevoort CA. 2, 3, 7, 8-Tetrachlorodibenzo-p-dioxin (TCDD) modulates function of human luteinizing granulosa cells via cAMP signaling and early reduction of glucose transporting activity. Reprod Toxicol 1996; 10(3):191-198.
- 50. Rubin BS, Murray MK, Damassa DA, King JC, Soto AM. Perinatal exposure to low doses of bisphenol A affects body weight, patterns of estrous cyclicity, and plasma LH levels. Environ Health Perspect 2001; 109:675-680.
- 51. Kim J, Peterson KE, Scanlon KS, Fitzmaurice GM, Must A, Oken E, Rifas-Shiman SL, Rich-Edwards JW, Gillman MW. Trends in overweight from 1980 through 2001 among preschool-aged children enrolled in a health maintenance organization. Obesity (Silver Spring) 2006; 14(7):1107-1112.
- 52. Freedman DS, Khan LK, Serdula MK, Ogden CL, Dietz WH. Racial and ethnic differences in secular trends for childhood BMI, weight, and height. Obesity (Silver Spring) 2006; 14(2):301-308.
- 53. Kipke MD, Iverson E, Moore D, Booker C, Ruelas V, Peters AL, Kaufman F. Food and park environments: neighborhood-level risks for childhood obesity in East Los Angeles. J Adolesc Health 2007; 40(4):325-333.
- 54. Glass TA, McAtee MJ. Behavioral science at the crossroads in public health: extending horizons, envisioning the future. Soc Sci Med 2006; 62(7):1650-1671.
- 55. Kroke A, Hahn S, Buyken AE, Liese AD. A comparative evaluation of two different approaches to estimating age at adiposity rebound. Int J Obes 2006; 30:261-266.
- 56. Wisemandle W, Maynard LM, Guo SS, Siervogel RM. Childhood weight, stature, and body mass index among never overweight, early-onset overweight, and late-onset overweight groups. Pediatrics 2000; 106(1):e14.
- 57. Olsen J, Melbye M, Olsen SF, Sorensen TIA, Aaby P, Nybo Andersen AM, . The Danish National Birth Cohortits background, structure and aim. Scand J Public Health 2001; 29(4):300-307.
- 58. Children's Health Act of 2000. Public Law 106-310.
- 59. Branum AM, Collman GW, Correa A, Keim SA, Kessel W, Kimmel CA, Taxbøl D, Hansen KD, Juhl M, Schow TB, Sørensen HT, Andresen J, Mortensen EL, Olesen AW, Søndergaard C. The National Children's Study of environmental effects on child health and development. Environ Health Perspect 2003; 111:642-646.
- 60. Landrigan PJ, Trasande L, Thorpe LE, Gwynn C, Lioy PJ, D'Alton ME, Lipkind HS, Swanson J, Wadhwa PD, Clark EB, Rauh VA, Perera FP, Susser E. The National Children's Study: a 21-year prospective study of 100,000 American children. Pediatrics 2006; 118(5):2173-2186.
- 61. Trasande L, Landrigan PJ. The National Children's Study: a critical national investment. Environ Health Perspect2004; 112:A789-A790.
- 62. Trasande L, Cronk CE, Leuthner SR, Hewitt JB, Durkin MS, McElroy JA, Anderson HA, Landrigan PJ. The National Children's Study and the children of Wisconsin. WMJ 2006; 105(2):50-54.
- 63. Strauss W LJ, Menkedick J, Ryan L, Pivetz T, McMillan N, Pierce B, Rust S. White Paper on Evaluation of Sampling Design Options for the National Children's Study [accessed 1 May 2007]. Available from: http://www.nationalchildrensstudy.gov/research/analytic_reports/upload/Executive-Summary-for-the-White-Paper-on-Evaluation-of-Sampling-Design-Options-for-the-National-Children-s-Study.pdf
-
64NCS. National Children's Study Home Page. [accessed 1 July 2008]. Available from: www.national childrensstudy.gov
» link - 65. Cervero R, Duncan M. Walking, bicycling, and urban landscapes: evidence from the San Francisco Bay Area. Am J Public Health2003; 93(9):1478-1483.
- 66. Ellaway A, Macintyre S, Bonnefoy X. Graffiti, greenery, and obesity in adults: secondary analysis of European cross sectional survey. BMJ2005; 331(7517): 611-612.
- 67. Li F, Fisher KJ, Brownson RC, Bosworth M. Multilevel modelling of built environment characteristics related to neighbourhood walking activity in older adults. BMJ 2005; 59(7):558-564.
- 68. Ewing R, Brownson RC, Berrigan D. Relationship between urban sprawl and weight of United States youth. Am J Prev Med2006; 31(6):464-474.
- 69. Frank LD, Andresen MA, Schmid TL. Obesity relationships with community design, physical activity, and time spent in cars. Am J Prev Med2004; 27(2):87-96.
- 70. Lopez R. Urban sprawl and risk for being overweight or obese. Am J Public Health2004; 94:1574-1579.
- 71. Papas MA, Alberg AJ, Ewing R, Helzlsouer KJ, Gary TL, Klassen AC. The built environment and obesity. Epidemiol Rev2007; 29(1):129143.
- 72. Galvez MP, Morland K, Raines C, Kobil J, Siskind J, Godbold J, Brenner B. Race and food store availability in an inner-city neighbourhood. Public Health Nutr2008; 11(6):624-631.
- 73. Morland K, Diez Roux AV, Wing S. Supermarkets, other food stores, and obesity: the Atherosclerosis Risk in Communities Study. Am J Prev Med 2006; 30(4):333-339.
- 74. Timperio A, Ball K, Salmon J, Roberts R, Giles-Corti B, Simmons D, Baur LA, Crawford D. Personal, family, social, and environmental correlates of active commuting to school. Am J Prev Med2006; 30(1):45-51.
- 75. Bern H. The fragile fetus. In: Chemically-Induced Alteration in Sexual and Functional Development: The Wildlife/Human Connection (Colborn T, Clement C, eds). Princeton, NJ: Princeton Scientific Publishing; 1992. p. 915.
- 76. U.S. EPA. Chemical Hazard Data Availability Study: What Do We Really Know About the Safety of High Production Volume Chemicals? Washington, D.C.: Office of Pollution Prevention and Toxics, U.S. Environmental Protection Agency; 1998.
- 77. Brotons J. Xenoestrogens released from lacquer coatings in food cans. Environ Health Perspect1995; 103:608-612.
- 78. Centers for Disease Control and Prevention. Third National Report on Human Exposure to Environmental Chemicals. Atlanta, GA: Centers for Disease Control and Prevention; 2005.
- 79. Vom Saal FS, Hughes C. An extensive new literature concerning low-dose effects of bisphenol A shows the need for a new risk assessment. Environ Health Perspect2005; 113:926-934.
- 80. Masuno H, Kidani T, Sekiya K, Sakayama K, Shiosaka T, Yamamoto H, Honda K. Bisphenol A in combination with insulin can accelerate the conversion of 3T3-L1 fibroblasts to adipocytes. J Lipid Res 2002; 43(5):676-684.
- 81. Sakurai K, Kawazuma M, Adachi T, Harigaya T, Saito Y, Hashimoto N, Mori C. Bisphenol A affects glucose transport in mouse 3T3-F442A adipocytes. Br J Pharmacol 2004; 141:209-214.
- 82. Alonso-Magdalena P, Laribi O, Ropero AB, Fuentes E, Ripoll C, Soria B, Nadal A. Low doses of bisphenol A and diethylstilbestrol impair Ca2+ signals in pancreatic a-cells through a nonclassical membrane estrogen receptor within intact islets of Langerhans. Environ Health Perspect2005; 113:969-967.
- 83. Wolff MS, Engel SM, Berkowitz GS, Ye X, Silva MJ, Zhu C, Wetmur J, Calafat AM. Prenatal phenol and phthalate exposures and birth outcomes. Environ Health Perspect2008; 116:1092-1097.
- 84. Sathyanarayana S. Phthalates and children's health. Curr Probl Pediatr Adolesc Health Care 2008; 38(2):34-49.
- 85. Bell FP. Effects of phthalate esters on lipid metabolism in various tissues, cells and organelles in mammals. Environ Health Perspect1982; 45:41-50.
- 86. Fisher JS. Environmental anti-androgens and male reproductive health: focus on phthalates and testicular dysgenesis syndrome. Reproduction 2004; 127(3):305-315.
- 87. Parks LG, Ostby JS, Lambright CR, Abbott BD, Klinefelter GR, Barlow NJ, Gray Jr. LE. The plasticizer diethylhexyl phthalate induces malformations by decreasing fetal testosterone synthesis during sexual differentiation in the male rat. Toxicol Sci 2000; 58:339-349.
- 88. Swan SH, Main KM, Liu F, Stewart SL, Kruse RL, Calafat AM, Mao CS, Redmon JB, Ternand CL, Sullivan S, Teague JL, Study for Future Families Research Team. Decrease in anogenital distance among male infants with prenatal phthalate exposure. Environ Health Perspect2005; 113:1056-1061.
- 89. Main KM, Mortensen GK, Kaleva MM, Boisen KA, Damgaard IN, Chellakooty M, Schmidt IM, Suomi AM, Virtanen HE, Petersen DV, Andersson AM, Toppari J, Skakkebaek NE. Human breast milk contamination with phthalates and alterations of endogenous reproductive hormones in infants three months of age. Environ Health Perspect2006; 114:270276.
- 90. Duty SM, Silva MJ, Barr DB, Brock JW, Ryan L, Chen Z, Herrick RF, Christiani DC, Hauser R. Phthalate exposure and human semen parameters. Epidemiology 2003; 14(3):269-277.
- 91. Hauser R. The environment and male fertility: recent research on emerging chemicals and semen quality. Semin Reprod Med 2006; 24(3):156-167.
- 92. Hauser R, Meeker JD, Duty S, Silva MJ, Calafat AM. Altered semen quality in relation to urinary concentrations of phthalate monoester and oxidative metabolites. Epidemiology 2006; 17(6):682-691.
- 93. Stahlhut RW, van Wijngaarden E, Dye TD, Cook S, Swan SH. Concentrations of urinary phthalate metabolites are associated with increased waist circumference and insulin resistance in adult US males. Environ Health Perspect2007; 115:876-882.
- 94. Landrigan P, Garg A, Droller DBJ. Assessing the effects of endocrine disruptors in the National Children's Study. Environ Health Perspect2003; 111:1678-1682.
- 95. Landrigan PJ, Trasande L, Swanson JM. Genetics, altruism, and the National Children's Study. Am J Med Genet A 2008; 146(3):294-296.
- 96. Coates RJ, Monteilh CP. Assessments of food-frequency questionnaires in minority populations. Am J Clin Nutr 1997; 65(suppl 4):1108S-1115S.
- 97. Teufel NI. Development of culturally competent food-frequency questionnaires. Am J Clin Nutr 1997; 65(4 suppl): 1173S-1178S.
- 98. Yaroch A, Resnicow KEN, Davis M, Davis A, Smith M, Khan LK. Development of a modified picture-sort food frequency questionnaire administered to low-income, overweight, African-American adolescent girls. J Am Dietetic Assoc 2000; 100(9):1050-1056.
- 99. Kohl HW, Fulton JE, Caspersen CJ. Assessment of physical activity among children and adolescents: a review and synthesis. Prev Med 2000; 31(2):54-76.
- 100. Ekelund ULF, Sjöström M, Yngve A, Poortvliet E, Nilsson A, Froberg K, Wedderkopp N, Westerterp K. Physical activity assessed by activity monitor and doubly labeled water in children.Med Sci Sports Exerc 2001; 33(2):275-281.
- 101. Janz KF, Levy SM, Burns TL, Torner JC, Willing MC, Warren JJ. Fatness, physical activity, and television viewing in children during the adiposity rebound period: The Iowa Bone Development Study. Prev Med 2002; 35(6):563-571.
- 102. Pietrobelli A, Faith MS, Allison DB, Gallagher D, Chiumello G, Heymsfield SB. Body mass index as a measure of adiposity among children and adolescents: a validation study. J Pediatr 1998; 132(2):204-210.
- 103. Lindsay RS, Hanson RL, Roumain J, Ravussin E, Knowler WC, Tataranni PA. Body mass index as a measure of adiposity in children and adolescents: relationship to adiposity by dual energy x-ray absorptiometry and to cardiovascular risk factors. J Clin Endocrinol Metab 2001; 86:4061-4067.
- 104. Gutin B, Litaker M, Islam S, Manos T, Smith C, Treiber F. Body-composition measurement in 9-11-y-old children by dual-energy X-ray absorptiometry, skinfold-thickness measurements, and bioimpedance analysis. Am J Clin Nutr 1996; 63(3):287-292.
- 105. Kettaneh A, Heude B, Lommez A, Borys JM, Ducimeticre P, Charles MA. Reliability of bioimpedance analysis compared with other adiposity measurements in children: The FLVS II Study. Diabetes Metab 2005; 31(6):534-541.
- 106. Lyman WD, Barone C, Castle V, Davies HD, Stanton B, Paneth N.Making the National Children's Study a real partnership with academic Pediatrics. J Pediatr 2005; 147(5):563-564.
Publication Dates
-
Publication in this collection
08 Feb 2010 -
Date of issue
Jan 2010
History
-
Accepted
11 Sept 2008 -
Received
21 July 2008