Abstract
The present study investigated the factors associated with anemia and micronutrient deficiencies in a cross-sectional analysis conducted with 150 children aged 11 to 14 months attending at basic health centers in Rio Branco, Acre. Venous blood samples were obtained to assess the occurrence of anemia and deficiencies of iron (ID), vitamin A (VAD), and B12 (VB12D). Multiple Poisson regression models were used to identify factors associated with anemia. Anemia, ID, VAD and B12D were observed in 23%, 76%, 18% and 20% of children, respectively. The factors associated with anemia were: do not be only child, living in households without access to cable TV or internet, stunting, late introduction of the complementary feeding (more than 240 days), VAD, VB12D, and current evidence of infection (plasma CRP > 5 mg/L). There was a lower prevalence of anemia among children with birth weight > 3,500 g. Overall, 82% of the study children had at least one of the micronutrient deficiencies (ID, VAD and VB12D). Actions with emphasis on timely and healthy feeding practices, better management of morbidities and supplementation with other micronutrients should be focused on the improvement of child care services at primary health care in this county.
Anemia; Iron deficiency; Micronutrients; Nutritional status; Infants
Resumo
O presente estudo investigou os fatores associados à anemia e deficiência de micronutrientes em análise transversal conduzida com 150 crianças de 11 a 14 meses atendidas em unidades básicas de saúde em Rio Branco, Acre. Amostras de sangue venoso foram obtidas para avaliar a ocorrência de anemia, deficiência de ferro (DF) e de vitaminas A (DVA) e B12 (DVB12). Modelos múltiplos de regressão de Poisson foram utilizados para identificar fatores associados à anemia. Anemia, DF, DVA e DVB12 foram observadas em 23%, 76%, 18% e 20% das crianças, respectivamente. Os fatores associados à anemia foram: não ser filho único, residir em domicílio sem acesso a TV a cabo ou internet, déficit de estatura para idade (E/I), introdução tardia da alimentação complementar superior a 240 dias, DVA, DVB12, e evidência de infecção vigente (proteína C reativa plasmática > 5 mg/L). Houve menor ocorrência de anemia entre crianças com peso ao nascer > 3.500g. Do total de crianças, 82% apresentaram pelo menos uma das deficiências de micronutrientes (DF, DVA, DVB12). Ações com ênfase em práticas alimentares saudáveis oportunas, melhor manejo de morbidades e suplementação com outros micronutrientes devem ser priorizadas no aprimoramento do serviço de puericultura da atenção básica à saúde deste município.
Anemia; Deficiência de ferro; Micronutrientes; Estado nutricional; Lactentes
Introduction
Anemia in infancy remains a public health problem, not only because of its prevalence, but also its severity. Estimates show that worldwide, some 273 million pre-school children suffer from this nutritional problem, or about 43% of the population in that age group11. Stevens GA, Finucane MM, De-Regil LM, Paciorek CJ, Flaxman SR, Branca F, Peña-Rosas JP, Bhutta ZA, Ezzati M. Global, regional, and national trends in haemoglobin concentration and prevalence of total and severe anaemia in children and pregnant and non-pregnant women for 1995-2011: a systematic analysis of population-representative data. Lancet Glob Health 2013; 1(1):e16-e25.. Half of these cases have been attributed to iron deficiency (ID), which is why anemia is used as an important indicator to estimate the magnitude of this deficiency in children22. World Health Organization (WHO). Center for Disease Control and Prevention. de Benoist B, McLean E, Egli I, Cogswellm M, editors. Worldwide prevalence of anaemia 1993-2005. WHO Global Database on Anaemia. Geneva: WHO; 2008..
Anemia is related to increased infant and neonatal morbimortality. The combined effects of multiple vitamin and mineral deficiencies from conception to the age of two contribute increased perinatal mortality, and reduce the individual’s physical ability to work and his/her productivity, which in turn has a negative impact on the country’s economic growth33. Lozoff B. Iron deficiency and child development. Food Nutr Bull 2007; 28(Supl. 4):S560-S571.,44. World Health Organization (WHO), United Nations Children’s Fund (UNICEF), United Nations University (UNU). Iron deficiency anaemia. Assessment, prevention and control. A guide for programme managers. Geneva: WHO; 2001..
Other factors considered in the etiology of anemia are genetic hemoglobinopathies, malaria, infections, other micronutrient deficiencies (Vitamin A, Vitamin B12 and Folate), and exposure to adverse environmental and socioeconomic conditions44. World Health Organization (WHO), United Nations Children’s Fund (UNICEF), United Nations University (UNU). Iron deficiency anaemia. Assessment, prevention and control. A guide for programme managers. Geneva: WHO; 2001..
Iron deficiency is the main cause of anemia in Brazilian children under the age of two, the result of inadequate nutritional practices (resulting in decreased iron consumption and bioavailability)55. Hadler MC, Juliano Y, Sigulem DM. Anemia do lactente: etiologia e prevalência. J Pediatr 2002; 78(4):321-326.,66. Carvalho AGC, Lira PIC, Barro MFA, Aléssio MLM, Lima MC, Carbonneau MA, Berger J, Léger CL. Diagnóstico por deficiência de ferro em crianças do Nordeste do Brasil. Rev Saude Publica 2010; 44(3):513-519., fast growth and recurring infections, especially in children in the poorer regions of the country44. World Health Organization (WHO), United Nations Children’s Fund (UNICEF), United Nations University (UNU). Iron deficiency anaemia. Assessment, prevention and control. A guide for programme managers. Geneva: WHO; 2001.. However, recent studies in several regions of the country77. Castro TG, Silva-Nunes M, Conde WL, Muniz PT, Cardoso MA. Anemia e deficiência de ferro em pré-escolares da Amazônia Ocidental brasileira: prevalência e fatores associados. Cad Saude Publica 2011; 27(1):131-142.,88. Bortolini GA, Vitolo MR.Relação entre deficiência de ferro e anemia em crianças de até 4 anos de idade. J Pediatr 2010; 86(6):488-492. have also taken into account the contribution of other micronutrient deficiencies, confirming international evidence found among children in Mexico99. Villalpando S, Pérez-Expósito A, Shamah-Levy T, Rivera JA. Distribution of anemia associated with micronutrient deficiencies other than iron: a probabilistic sample of Mexican children. Ann Nutr Metab 2006; 50(6):506-511., and schoolchildren in Colombia1010. Arsenault JE, Mora-Plazas M, ForeroY, Lopez-Arana S, Baylin A, Villamor E. Hemoglobin concentration is inversely associated with erythrocyte folate concentrations in Colombian school-age children, especially among children with low vitamin B12 status. Eur J Clin Nutr 2009; 63(7):842-849..
Data from the National Demographics and Health Survey1111. Brasil. Ministério da Saúde (MS). Pesquisa Nacional de Demografia e Saúde da Criança e da Mulher – PNDS 2006: dimensões do processo reprodutivo e da saúde da criança. Brasília: MS; 2009. shows that Brazilian children have an inadequate nutritional standard, with the introduction of semi-solid foods of low nutritional value at a very early age (47.1% of children under five months use a bottle), and delayed introduction of important food groups: 37% of the children between the ages of 6 and 11 months did not eat fruits and vegetables daily, 47% did not eat beans and 83% did not eat meat.
Different strategies have been used to prevent and control anemia and ID, however few have been successful in reducing their incidence1212. World Health Organization (WHO). Use of multiple micronutrient powders for home fortification of foods consumed by infants and children 6-23 months of age. Geneva: WHO; 2011.. Global data shows a very slow downward trend in prevalence - around 4% a year over the past two decades (1995-2011)11. Stevens GA, Finucane MM, De-Regil LM, Paciorek CJ, Flaxman SR, Branca F, Peña-Rosas JP, Bhutta ZA, Ezzati M. Global, regional, and national trends in haemoglobin concentration and prevalence of total and severe anaemia in children and pregnant and non-pregnant women for 1995-2011: a systematic analysis of population-representative data. Lancet Glob Health 2013; 1(1):e16-e25..
While Brazil’s development indicators have improved significantly, with better income distribution, easier access to goods and public services, and declining gaps between regions, some inequalities remain. and the North of the country has one of the highest social vulnerabilities, which reflects in infant health and nutrition. 80.1% of the households in the North do not have access to sanitation services, and the area has the highest rate of hospitalizations for oral fecal transmissible diseases (693.8 per 100 thousand inhabitants, or 80.1% in 2010)1313. Instituto Brasileiro de Geografia e Estatística (IBGE). Estudos e Pesquisas. Informação Demográfica e Socioeconômica, n. 32. Síntese de Indicadores Sociais: uma análise das condições de vida da população brasileira. 2013. [acessado 2014 jun 23]. Disponível em: ftp://ftp.ibge.gov.br/Indicadores_Sociais/Sintese_de_Indicadores_Sociais_2013/SIS_2013.pdf.
In spite of strategies to prevent and control anemia, with compulsory fortifying of wheat and corn flours, and prophylactic supplementation for high-risk groups, the incidence of anemia in children remains a public health problem in all regions of the country1414. Spinelli MGN, Marchioni DML, Souza JMP, Souza SB, Szarfac SC. Fatores de risco para anemia em crianças de 6 a 12 meses no Brasil. Rev Panam Salud Pública 2005; 17(2):84-91.
15. Jordão RE, Bernardi JLD, Barros Filho A. Prevalência de anemia ferropriva no Brasil: uma revisão sistemática. Rev Paul Pediatr 2009; 27(1):90-98.-1616. Vieira RCS, Ferreira HS. Prevalência de anemia em crianças brasileiras, segundo diferentes cenários epidemiológicos. Rev Nutrição 2010; 23(3):433-444.. Studies indicate the prevalence of anemia in children under the age of two in the Brazilian Amazon ranges from 40 to 70%1717. Garcia MT, Granado FS, Cardoso MA. Alimentação complementar e estado nutricional de crianças menores de dois anos atendidas no Programa Saúde da Família em Acrelândia, Acre, Amazônia Ocidental Brasileira. Cad Saude Publica 2011; 27(2):305-316.
18. Castro TG, Baraldi L, Muniz PT, Cardoso MA. Dietary practices and nutritional status of 0-24 month-old children from Brazilian Amazonia. Publ Health 2009; 12(12):2335-2342.
19. De Souza OF, Macedo, LFM, Oliveira CSM, Araújo TS, Muniz PT. Prevalence and associated factors to anaemia in children. J Hum Growth Dev 2012; 22(3):307-313.
20. Cardoso MA, Scopel KKG, Muniz PT, Villamor E, Ferreira MU. Underlying factors associated with anemia in Amazonian children: a population-based, cross-sectional study. PLoS One 2012; 7:e36341.-2121. Oliveira CSM, Cardoso MA, Araujo TS, Muniz PT. Anemia em crianças de 6 a 59 meses e fatores associados no Município de Jordão, Estado do Acre, Brasil. Cad Saude Publica 2011; 27(5):1008-1020.. The literature has few studies that analyze the factors associated with anemia in children in developing nations that also consider their nutritional status in terms of micronutrients. For this reason, this study looked at the nutritional status of children under the age of two seen in basic healthcare units of the Unified Healthcare System (SUS) in Rio Branco, Acre.
We hope to contribute with information on the magnitude and factors associated with anemia and micronutrient deficiency in these children, thus fostering improved infant care services, within those already provided in primary child healthcare.
Methodology
Study design, area and population
This is a cross-sectional study that is part of ENFAC (Estudo Nacional de Fortificação Caseira da Alimentação Complementar), aimed at home supplementation of complementary feeding. This is a pragmatic, multi-center clinical trial performed in four Brazilian cities (Rio Branco, Olinda, Goiânia and Porto Alegre). ENFAC is comprised of two study groups, a control with children 12 to 14 months routinely seen by the childcare services of basic healthcare units (UBS) participating in the trial, and the intervention group, made up of children receiving fortified nutrition at home with a variety of powdered micronutrients from the age of 6 - 8 months.
For this analysis, we used data from children in the control group, ranging in age from 11 to 14 months, seen by the childcare units of the UBS in the city of Rio Branco. Inclusion criteria were: a) approval to participate in the trial given by the mother or guardian; b) not being treated for anemia when the trial started. Exclusion criteria were premature birth (children born at under 37 weeks gestation), twins, children with HIV, malaria, tuberculosis, hemoglobinopathies or fever (> 39°) on the day blood was drawn.
In Rio Branco the trial included six basic care units (UBS) between June 2012 and February 2013. Rio Branco is the capital of the Brazilian state of Acre, located in the north of the country. Based on the 2010 Demographic Census, the city’s Human Development Index (HDI-M) is 0.727, which is considered high. The city has a total population of 336,038, with children under the age of four making up 9% of the total. The infant mortality rate (IMR) in Rio Branco in 2010 was 20 per thousand live births, higher than the country average of 16.7 per TLB2222. Instituto Brasileiro de Geografia e Estatística (IBGE). Cidades. Resultados da amostra do Censo Demográfico 2010. [acessado 2014 maio 23]. Disponível em: http://cidades.ibge.gov.br/xtras/perfil.php?codmun=120040
http://cidades.ibge.gov.br/xtras/perfil....
,2323. Programa das nações unidas para o desenvolvimento (PNUD). Atlas do desenvolvimento humano no Brasil: atlas brasil 2013. [acessado 2014 jun 23]. Disponível em:http://www.pnud.org.br/idh/atlas2013.aspx?indiceaccordion=1&li=li_atlas2013
http://www.pnud.org.br/idh/atlas2013.asp...
. Over the course of the past decade, coverage of the Family Health Program in Rio Branco has varied quite widely (25.9% in 2004, jumping to 50.1% in 2009)2424. Brasil. Ministério da Saúde (MS). Caderno de Informações em Saúde. [acessado 2014 maio 23]. Disponível em: http://tabnet.datasus.gov.br/tabdata/cadernos/ac.htm
http://tabnet.datasus.gov.br/tabdata/cad...
. By 2012, the year this study was conducted, it had dropped again to 38.7%2525. Secretaria Municipal de Saúde de Rio Branco. Departamento de Atenção Básica (DAB). DATASUS. Fonte: SIAB - Sistema de Informação de Atenção Básica. Relatório do Consolidado da Cobertura do Programa Saúde da Família. Rio Branco: Secretaria Municipal de Saúde de Rio Branco; 2013.
The sample anticipated for Rio Branco was 105 children per group (control and intervention), plus 30% to cover withdrawals and refusals, thus a total sample of 135 children [per group]. We were able to recruit 155 children who met the inclusion criteria for the control group. Of these, five refused to participate in the study. The final sample was made up of 150 children. This sample size resulted in a testing power of 80% and a two-tail significance level of 0.05 for detecting 6 g/L differences in mean hemoglobin assays, estimating a standard deviation of 12 g/L for the main study (ENFAC). Children included in the study were no different from those who were not in terms of gender, age, socioeconomic, maternal and early childhood conditions.
Field teams were comprised of nurses and undergraduate students in nutrition at Universidade Federal do Acre. Teams were trained and allocated to the UBSs participating in the study. A Free and Informed Consent Form was signed by the mothers or guardians once we had explained the goals and advantages of the study. The Consent Form stated they were voluntarily participating in the study, and were assured that all of the information provided would be kept as confidential. The study was approved by the Research Ethics Committee of the Public Health School at the University of São Paulo. Data collection was preceded by consent from the Rio Branco, Acre City Health Department.
Data collection
Interviewers applied a structured questionnaire to mothers or guardians going to the healthcare service, or actively sought out mothers and guardians living in the area served by the participating UBSs. The following data was collected: a) demographics (gender, age of child, race/color), b) socioeconomic data (per capita household income, parent (mother and father) years of schooling, registration in the Programa Bolsa Família (cash transfer trogramme), cable TV and Internet in the home, number of children under the age of 5 in the household, number of children of same mother); c) environmental (treated water); d) maternal characteristics (age, type of delivery, number of pre-natal visits); e) characteristics and morbidities at birth (weight at birth, hospitalizations in the past 12 months, diarrhea, fever and cough in the past 15 days); f) nutritional status and infant nutrition habits (malnutrition, nursed in the first hour after birth, how long child nursed exclusively, age at which complementary feeding is introduced); g) use of nutritional supplements by the child (iron and vitamins A and D).
Trained researchers measured the length and weight of the children using standard procedures and calibrated equipment2626. World Health Organization (WHO). Expert Committee on Physical Status. Physical status: the use and interpretation of anthropometry: report of a WHO Expert Committee. Geneva: WHO; 1995.. Each measurement was repeated and the mean calculated. To measure weight
We used an electronic Tanita UM-061 scale with capacity of 150 kg and sensitive to 100 g. Length was measured using a Sanny ES-2000 portable infantometer with 1 meter extension and 1 mm precision. Children were measured lying on their backs on a flat service. Length was measured in centimeters. To analyze their nutritional status we used the Length/Age Z-score obtained using WHO Anthro version 3.2.2 software, using as a reference the WHO growth standards2727. World Health Organization (WHO).WHO Anthro (version 3.2.2, January 2011b) and macros. [acessado 2014 jun 23]. Disponível em: http://www.who.int/childgrowth/software/es/index.html
http://www.who.int/childgrowth/software/...
. Malnutrition was defined as a Z score of less than -2.
Blood sample collections were scheduled for the week following the interview, based on the availability of the caretakers. Blood was drawn by a nurse technician experienced in infant venipuncture. Blood samples were drawn after a fast of at least three hours.
The samples of blood were placed in 2 test tubes - a) a dry tube, protected from light for obtaining serum, kept at ambient temperature and centrifuged within 1 hour of collection; b) a tube with EDTA for obtaining plasma, kept in ice until centrifugation. After removing the clot, the liquid was centrifuged at 3,000 rpm and the blood components separated into suitable microtubes, kept frozen at -20°C until being shipped to the Human Nutrition lab at the Nutrition Department (FSP/USP), where they were kept at -70°C until biochemical analyses were performed.
Serum folate and Vitamin B12 concentrations were measured using fluoroimmunoassay (Perkin Elmer, WallacOy, Turku, Finland). The cutoff for determining folate deficiency (FOLD) was 10 nmol. For Vitamin B12 deficiency (VB12D) we used a cut-off of 150 pmol/L. Serum retinol and Vitamin E were analyzed using reverse-phase High Performance Liquid Chromatography (HPLC-1100, Hewlett-Packard, United States). Children with serum retinol concentrations below 1.05 μmol/L and below 0.7 μmol/L were used for Vitamin A insufficiency and deficiency respectively (VAD)2828. World Health Organization (WHO).Indicators for assessing vitamin A deficiency and their application in monitoring and evaluating intervention programmes. Geneva: WHO; 1996.. Vitamin E insufficiency was considered for concentrations below 11.6 μmol/L. For anemia, we used the WHO44. World Health Organization (WHO), United Nations Children’s Fund (UNICEF), United Nations University (UNU). Iron deficiency anaemia. Assessment, prevention and control. A guide for programme managers. Geneva: WHO; 2001. cut-off point of Hb < 110 g/L in children six months to five years of age. Blood hemoglobin (Hb) was measured using a portable Hemocue (Hb301; HemoCue®, Angelholm, Sweden) hemoglobin-meter at the time of blood collection. Iron deficiency (ID) was determined when plasma ferritin (PF) was below 12 g/L and/or the soluble transferrin receptor (sTfR) concentration was less than 8.3 g/mL. IDA was determined when anemic children presented iron defficiency2020. Cardoso MA, Scopel KKG, Muniz PT, Villamor E, Ferreira MU. Underlying factors associated with anemia in Amazonian children: a population-based, cross-sectional study. PLoS One 2012; 7:e36341.,2929. Cook JD, Flowers CH, Skikne BS. The quantitative assessment of body iron. J Hematol 2003; 101(9):3359-3364.. These analyzes were performed by enzyme immunoassay (Ramco, Houston, Texas, USA). Knowing that PF concentration increases with inflammation and infection, and can lead to under-estimating ID3030. Thurnham DI, McCabe LD, Haldar S, Wieringa FT, Northrop-Clewes CA, McCabe GP. Adjusting plasma ferritin concentrations to remove the effects of subclinical inflammation in the assessment of iron deficiency: a meta-analysis. Am J ClinNutr 2010; 92(3):546-555., plasma C-Reactive Protein (CFP) was determined to check for infection in the study children. We used an immunochemical system - IMMAGE (Beckman Coulter, Brea, CA, USA). PCR values under 5 mg/L and/or Alfa-1 acid glycoprotein (AGP) greater than 1 g/L were considered to be associated with existing or chronic inflammation and/or infection, as described in the meta-analysis3030. Thurnham DI, McCabe LD, Haldar S, Wieringa FT, Northrop-Clewes CA, McCabe GP. Adjusting plasma ferritin concentrations to remove the effects of subclinical inflammation in the assessment of iron deficiency: a meta-analysis. Am J ClinNutr 2010; 92(3):546-555.. Samples were analyzed within 6 months of being collected. The lab ran blind analyses for internal and external quality control. Children diagnosed with anemia, ID or VAD were sent to the UBS healthcare system for treatment, as per the guidelines of the Ministry of Health. The frequency of each micronutrient deficiency was calculated. However, we know that the cut-off points for these deficiencies are not considered definitive as micronutrients, especially in children, have imperfect sensitivities and specificities3131. Pasricha S-R, Shet AS, Black JF, Sudarshan H, Prashanth NS, Biggs BA. Vitamin B-12, folate, iron, and vitamin A concentrations in rural Indian children are associated with continued breastfeeding, complementary diet, and maternal nutrition. Am J Clin Nutr 2011; 94(5):1358-1370.,3232. World Health Organization (WHO). Conclusions of a WHO Technical Consultation on folate and vitamin B-12 deficiencies. Food Nutr Bull 2008b; 29(Supl. 2):S238-S244..
Data analysis
The main outcome of interest was anemia. The frequency of micronutrient deficiencies (ID, VAD, VB12D and FOLD) were used to describe the study population.
Data was doubly inputted using Epiinfo 6.04 (Center for Disease Control and Prevention, Atlanta, Georgia) software, and exported to Stata v. 10.0 (StataCorp., College Station, Texas) for analysis. The distribution of the continuous variables was identified using the Shapiro Wilk test. To characterize the samples, absolute and relative frequencies were calculated for the categorical variables, the mean and standard deviation for continuous variables with normal distribution, and the median and interquartile intervals for variables without a normal distribution. Pearson’s Chi-squared was used to compare the amounts of anemia, ID and IDA using the dichotomous variables, with a level of significance p < 0.05.
In the first step, gross analysis using a Poisson regression looked at the effect of each of the explanatory variables on anemia individually. The following independent variables were investigated: demographic and socioeconomic characteristics, access to public services, maternal and birth characteristics, morbidities, infant nutrition practices, the use of nutritional supplements by the infant and biochemical markers. The cut-off point to select variables for inclusion in multiple models was p < 0.20. Adjusted prevalence rations and 95% confidence intervals controlled by gender were obtained using multiple Poisson regressions with robust variance, as per the existing conceptual model adapted from earlier studies77. Castro TG, Silva-Nunes M, Conde WL, Muniz PT, Cardoso MA. Anemia e deficiência de ferro em pré-escolares da Amazônia Ocidental brasileira: prevalência e fatores associados. Cad Saude Publica 2011; 27(1):131-142.,2121. Oliveira CSM, Cardoso MA, Araujo TS, Muniz PT. Anemia em crianças de 6 a 59 meses e fatores associados no Município de Jordão, Estado do Acre, Brasil. Cad Saude Publica 2011; 27(5):1008-1020.. Considering the characteristics of our study population, late inclusion of complementary nutrition was categorized as before or after 240 days, and weight at birth as less than or more than 3,500 grams.
For each level of determination, from the distal to the proximal, variables remained in the model if conceptually relevant and associated with the outcome in the adjusted analysis (p < 0.10, Wald test), or if including each of them in the model improved the determination coefficient or changed the RP by 10% or more of the variables in the block tested. Missing observations were included in the multiple models by created the missing category. Variables that, after adjustment for the factors of the same or higher hierarchical level, showed a p value < 0.05 were considered associated with anemia.
Results
The median age was 13.3 months, 55% of the children were male, and most of them mulatto (89.9%). Median per capita household income was a bit above the minimum wage in 2012 (R$ 622,00) Regarding their environments, about 30% of the children lived in homes with inadequately treated water, and 43% in homes with no connection to public sewage. Regarding data on the mothers, the median age and years of schooling were 25 and 10 years respectively. The median number of pre-natal visits was 7.
One-half of the children had fever and 27% had diarrhea in the 15 days before the study. More than 24% had been hospitalized in the preceding 12 months. The share of children who had used iron or Vitamin A and D supplements sometime in their lives was relatively low - 13.5% and 2.7% respectively (Table 1).
Descriptive values of the indicators of nutritional status regarding iron, frequency of anemia and other conditions are shown in Table 2. The frequencies for anemia, iron deficiency and iron deficiency anemia were 22.7%, 75.8% and 19.5% respectively. We found no case of severe anemia (Hb < 70 g/L). 27.5% of the anemic children had iron deficiency. 85.3% of the children with ID were anemic. The frequencies of Vitamin A (VAD), Vitamin B12 (VB12D), folate (FOLD) deficiency and malnutrition were 17.8%, 20.0%, 0.7% and 7.7% respectively. Of the 150 children evaluated, 82% had multiple deficiencies (ID, VAD, VB12D. Acute (CRP > 5 mg/L) or chronic (AGP > 1 g/L) inflammation or infection were found in 21.2% and 38% of the children respectively.
Regarding infant nutrition practices, only 86.7% of the children nursed in the first hour after birth. The median period during which babies nursed exclusively was 120 days, and the average age at weaning was 150 days. Complementary feeding was added late (after 8 months) to 13% of the nursing infants.
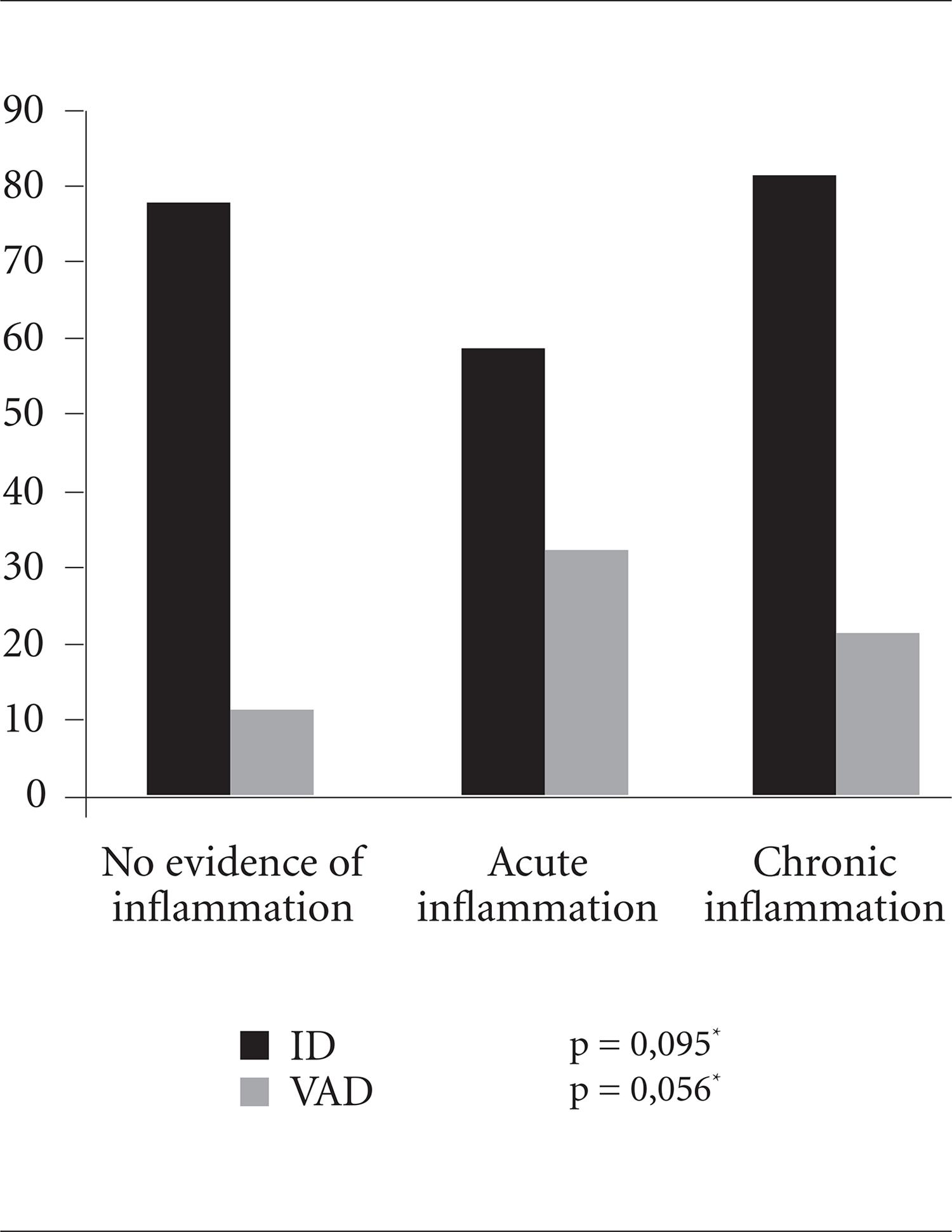
Figure 1 shows the frequency of ID and VAD using acute (PCR) and chronic (AGP) inflammation markers. The prevalence of ID was higher where there was chronic inflammation than in the presence of any other conditions, although the difference was not statistically significant. For VAD the difference between the two groups was limitrophe.
Relative frequency (%) of iron (ID) and vitamin A deficiencies (VAD) according to the presence of inflammation in infants in Rio Branco (n= 150).
No infection, CRP ≤ 5mg/L and AGP ≤ 1g/L, n=69/87 (ID) and n = 10/87 (VAD); Acute inflammation, CRP > 5mg/L and AGP > 1g/L, n = 17/29 (ID) e n = 9/29 (VAD); Chronic inflammation, CRP ≤ 5mg/L and AGP > 1g/L, n = 22/28 (ID) e n = 6/28 (VAD). Only 2 children showed CRP > 5 mg/L and AGP ≤ 1g/L; Differences in absolute frequencies correspond to missing values to biochemical indicators. AGP: α-1-acid glicoprotein; CRP: C-reactive protein. * p value of the categories of inflammation in relation to each outcome.
The frequency of anemia according to the independent variables, and the relative contribution of each risk factor to this condition, were estimated using χ2. The adjusted prevalence ratios (APR) are shown in Table 3. Anemia was uniformly distributed between the two genders, with no statistically significant difference. Higher prevalence of anemia and increased risk of this outcome were related to the following variables: not being an only child [30.1%; APR: 2,11 (CI95%:1.06; 4.19), no cable TV or Internet in the home [APR: 4.57 (1.13; 18.47), malnutrition, late introduction of iron-rich foods or those that promote iron absorption, Vitamin A and B12 deficiency and CRP > 5 mg/L. Children weighing more than 3,500 grams at birth were at lower risk for anemia.
ID was 17% lower among children of mothers who had 9 or more years of schooling than among the children of mothers with fewer years of schooling. Late introduction of iron-rich foods or those that promote iron absorption increased the risk of ID by 26% compared to those children introduced to these foods at the right time. VAD was associated with ID [1.37 (1.17; 1.61)]. The only factors associated with IDA were late introduction of iron-rich foods or those that promote iron absorption [2.16 (1.14;4.09)], malnutrition [2.28 (1.12; 4.61)] and Vitamin A [1.96 (1.04; 3.70)] and B12 [2.82 (1.48; 5.37)] deficiencies.
Discussion
In this study, socioeconomic factors and infant feeding practices were associated with the presence of anemia. Other factors strongly associated with anemia were weight at birth, malnutrition, presence of infection and deficiency of other micronutrients (Vitamins A and B12).
The largest proportion of anemia was found in children where iron rich foods such as meat and other iron absorption promoters were introduced late. The strong association between this variable and the outcome reinforces the importance of nutritional education measures that stress the timely and suitable introduction of complementary nutrition, showing the nutritional damages caused by maintaining a dairy-based diet, as shown in other studies55. Hadler MC, Juliano Y, Sigulem DM. Anemia do lactente: etiologia e prevalência. J Pediatr 2002; 78(4):321-326.,1414. Spinelli MGN, Marchioni DML, Souza JMP, Souza SB, Szarfac SC. Fatores de risco para anemia em crianças de 6 a 12 meses no Brasil. Rev Panam Salud Pública 2005; 17(2):84-91.,3333. Yang W, Li X, Li Y, Zhang S, Liu L, Wang X, Li W. Anemia, malnutrition and their correlations with socio-demographic characteristics and feeding practices among infants aged 0–18 months in rural areas of Shaanxi province in northwestern China: a cross-sectional study. BMC Public Health 2012; 12:1127.,3434. Schulze KJ, Christian P, Wu LS, Arguello M, Hongjie C, Nanayakkara-Bind A, Stewart CP, Khatry SK, LeClerq S, West Junior KP. Micronutrient Deficiencies Are Common in 6- to 8-Year-Old Children of Rural Nepal, with Prevalence Estimates Modestly Affected by Inflammation. J Nutr 2014; 144(6):979-987.. Surveys by Garcia et al.1717. Garcia MT, Granado FS, Cardoso MA. Alimentação complementar e estado nutricional de crianças menores de dois anos atendidas no Programa Saúde da Família em Acrelândia, Acre, Amazônia Ocidental Brasileira. Cad Saude Publica 2011; 27(2):305-316. and Castro et al.1818. Castro TG, Baraldi L, Muniz PT, Cardoso MA. Dietary practices and nutritional status of 0-24 month-old children from Brazilian Amazonia. Publ Health 2009; 12(12):2335-2342. in a town close to the area of this study also found an inadequate pattern of nutrition in children under the age of two, with limited consumption of foods that facilitate iron absorption (meats, fruits and vegetables), and high use of iron inhibitors (cow’s milk and thickeners). This type of nutritional pattern results in insufficient ingestion of iron heme and low mineral utilization. It also can lead to microgastrointestinal bleeding due to the early introduction of cow’s milk. Studies reinforce the importance of eating meats during the weaning period, which should start as soon as complementary feeding is introduced, and always before the 27th week3535. VitoloMR, BortoliniGA. Biodisponibilidade do ferro como fator de proteção contra anemia entre crianças de 12 a 16 meses. J Pediatr 2007; 83(1):33-38..
The median duration of exclusive breastfeeding was low (120 days), and weaning early (median of 150 days). Although the importance of exclusively breast feeding for preventing anemia is known, our data did not reveal any association between breast milk and anemia or ID. This is likely due to the difficulty to obtain accurate information about this variable. The importance of breast feeding for reducing the risk of anemia and infant survival is known, and is considered the single highest-impact intervention for reducing preventable deaths of children under the age of five (90% of infant deaths in the world)3636. Jones G, Steketee Rw, Black Re, Bhutta ZA, Morris SS, Bellagio Child Survival Study Group. How many child deaths can we prevent this year? Lancet 2003; 362(9377):65-71..
Consistent with the literature22. World Health Organization (WHO). Center for Disease Control and Prevention. de Benoist B, McLean E, Egli I, Cogswellm M, editors. Worldwide prevalence of anaemia 1993-2005. WHO Global Database on Anaemia. Geneva: WHO; 2008., we found a strong association between anemia and socioeconomic conditions, reflected here as owning assets such as cable TV and Internet. The absence of such assets in the home was associated with a four-fold higher risk of having anemia. The North has the smallest Internet penetration in Brazil (20.2% of the population has Internet access, compared to the national average of 36.5%)3737. Brasil. Ministério do Desenvolvimento Social e Combate à Fome (MDS). Indicadores de Desenvolvimento Brasileiro. 2013. [acessado 2014 jun 23]. Disponível em: http://189.28.128.178/sage/sistemas/apresentacoes/arquivos/indicadores_de_desenvolvimento_2013.pdf
http://189.28.128.178/sage/sistemas/apre...
. In this context, this variable is a good indicator of socioeconomic level. In a study of nursing infants in Pernambuco, Lima et al.3838. Lima ACVMS, Lira PIC, Romani SAM, Eickmann SH, Piscoya MD, Lima MC. Fatores determinantes dos níveis de hemoglobina em crianças aos 12 meses de vida na Zona da Mata Meridional de Pernambuco. Rev Bras Saúde Matern Infant 2004; 4(1):35-43. found that the absence of a TV in the home was significantly associated with a lower mean blood hemoglobin level.
It is believed that the role of the mother’s years of schooling is related to an increase in the ability to understand the instructions received and with making healthier food choices, providing their children with a better diet. They tend to be more aware of healthcare in general, which results in more efficient care practices3939. Choi HJ, Lee HJ, Jang HB, Park JY, Kang JH, Park KH, Jihyun Song J. Effects of maternal education on diet, anemia, and iron deficiency in Korean school-aged children. BMC Public Health 2011; 11:870.. However, unlike some studies3333. Yang W, Li X, Li Y, Zhang S, Liu L, Wang X, Li W. Anemia, malnutrition and their correlations with socio-demographic characteristics and feeding practices among infants aged 0–18 months in rural areas of Shaanxi province in northwestern China: a cross-sectional study. BMC Public Health 2012; 12:1127. but agreeing with others55. Hadler MC, Juliano Y, Sigulem DM. Anemia do lactente: etiologia e prevalência. J Pediatr 2002; 78(4):321-326.,4040. Soh P, Ferguson EL, McKenzie JE, Homs MYV, Gibson RS. Iron deficiency and risk factors for lower iron stores in 6-24 month-old New Zealanders. Eur J Clin Nutr 2004; 58(1):71-79., this study found no association between maternal education and anemia. A possible explanation may be the homogeneous distribution of education in the study population, without enough contrast for proper comparisons.
The low prevalence of the use of supplements as prophylaxis for the main micronutrient deficiencies in young children (Vitamins A, D and iron) shows limited adherence of this population to this type of public health measure. This reduced rate of adherence is common in a number of countries other than Brazil, and is a topic of much discussion in the literature. In the case of iron supplementation, this is generally attributed to gastrointestinal effects, sensory aspects, the long period of administration, limited prescription by healthcare professionals and lack of informaiton4141. Mora J. Iron supplementation: overcoming technical and practical barriers. J Nutr 2002; 132(4):S853-S855..
Vitamin B12 deficiency is associated with Anemia and IDA, [this study found] higher numbers (20%) than the numbers found in children 6-24 months old in Acre (12%)2020. Cardoso MA, Scopel KKG, Muniz PT, Villamor E, Ferreira MU. Underlying factors associated with anemia in Amazonian children: a population-based, cross-sectional study. PLoS One 2012; 7:e36341., and lower than those found in children under the age of 2 in Colombia (50%)4242. Herrán OF, Ward JB, Villamor E. Vitamin B12 sero status in Colombian children and adult women: results from a nationally representative survey. Public Health Nutr 2014; 18(5):836-843.. Inadequate levels of Vitamin B12 have been found in numerous developing nations. The few studies available on the prevalence of Vitamin B12 deficiency used small samples and non-standard methods of nutritional assessment, making comparisons difficult. In global terms, this association has been considered uncommon, possibly due to diagnostic limitations, as ID tends to reduce mean corpuscular volume (MCV) more easily than Vitamin B12 deficiency3232. World Health Organization (WHO). Conclusions of a WHO Technical Consultation on folate and vitamin B-12 deficiencies. Food Nutr Bull 2008b; 29(Supl. 2):S238-S244.. It is likely that this finding is due to late introduction of meats to infant nutrition, found in this study.
Half of the children studied had inadequate levels of Vitamin A, and about one in five had Vitamin A deficiency, shown to be associated with anemia, thus corroborating the findings of Pedraza et al.4343. Pedraza DF, Queiroz D, Paiva AA, Cunha MAL da, Lima ZN. Food security, growth and vitamin A, hemoglobin and zinc levels of preschool children in the northeast of Brazil. Cien Saude Colet 2014; 19(2):641-650. in a study conducted in the Brazilian Northeast. In Rio Branco, 40% of the anemic children also presented VAD, but ID was not found, reinforcing the involvement of other micronutrients in anemia.
The prevalence of VAD was higher among children with acute inflammation, corroborating the findings of Rohner et al.4444. Rohner F, Woodruff BA, Aaron GJ, Yakes EA, Lebanan MA, Rayco-Solon P, Saniel OP. Infant and young child feeding practices in urban Philippines and their associations with stunting, anemia, and deficiencies of iron and vitamin A. Food Nutr Bull 2013; 34(Supl. 2):S17-S34.. The magnitude of the Vitamin A deficiency found was larger than the national estimate (16.3%)1111. Brasil. Ministério da Saúde (MS). Pesquisa Nacional de Demografia e Saúde da Criança e da Mulher – PNDS 2006: dimensões do processo reprodutivo e da saúde da criança. Brasília: MS; 2009., and higher than the numbers found in a study of children in the Amazon (14.2%)2020. Cardoso MA, Scopel KKG, Muniz PT, Villamor E, Ferreira MU. Underlying factors associated with anemia in Amazonian children: a population-based, cross-sectional study. PLoS One 2012; 7:e36341. and the Philippines (3%)4444. Rohner F, Woodruff BA, Aaron GJ, Yakes EA, Lebanan MA, Rayco-Solon P, Saniel OP. Infant and young child feeding practices in urban Philippines and their associations with stunting, anemia, and deficiencies of iron and vitamin A. Food Nutr Bull 2013; 34(Supl. 2):S17-S34.. This situation probably reflects insufficient consumption of foods rich in pro-vitamin A (e.g. yellow fruits and vegetables), as well as the high morbidity among the study population. Numerous studies have demonstrated the influence of Vitamin A on the iron status3434. Schulze KJ, Christian P, Wu LS, Arguello M, Hongjie C, Nanayakkara-Bind A, Stewart CP, Khatry SK, LeClerq S, West Junior KP. Micronutrient Deficiencies Are Common in 6- to 8-Year-Old Children of Rural Nepal, with Prevalence Estimates Modestly Affected by Inflammation. J Nutr 2014; 144(6):979-987.,4545. Saraiva BC, Soares MC, Santos LC, Pereira SC, Horta PM. Iron deficiency and anemia are associated with lowretinol levels in children aged 1 to 5 years. J Pediatr 2014; 90(6):593-599. and hemoglobin levels4646. Jafari SM, Heidari G, Nabipoura I, Amirinejad R, Assadi M, Bargahi A, Akbarzadehd S, Tahmasebia R, Sanjdideh Z. Serum retinol levels are positively correlated with hemoglobin concentrations, independent of iron homeostasis: a population-based study. Nutrition Research 2013; 33(4):279-285.. Because of this, some studies have suggested that combined supplementation with Vitamin A and iron would have a greater impact on reducing anemia than the administration of either one of these in isolation99. Villalpando S, Pérez-Expósito A, Shamah-Levy T, Rivera JA. Distribution of anemia associated with micronutrient deficiencies other than iron: a probabilistic sample of Mexican children. Ann Nutr Metab 2006; 50(6):506-511.,1010. Arsenault JE, Mora-Plazas M, ForeroY, Lopez-Arana S, Baylin A, Villamor E. Hemoglobin concentration is inversely associated with erythrocyte folate concentrations in Colombian school-age children, especially among children with low vitamin B12 status. Eur J Clin Nutr 2009; 63(7):842-849., as more often than not, micronutrient deficiencies are associated47.
Weight at birth has been associated with the prevalence of anemia, with higher birth weight considered protection from this outcome. The literature shows that children who weigh less at birth have lower iron reserves, and many are weaned early, accentuating iron depletion3838. Lima ACVMS, Lira PIC, Romani SAM, Eickmann SH, Piscoya MD, Lima MC. Fatores determinantes dos níveis de hemoglobina em crianças aos 12 meses de vida na Zona da Mata Meridional de Pernambuco. Rev Bras Saúde Matern Infant 2004; 4(1):35-43.. However, as this is a variable obtained from the child’s file, care must be taken in its interpretation.
Regarding morbidity, it is known that children living in unsuitable environments are more prone to infectious diseases3434. Schulze KJ, Christian P, Wu LS, Arguello M, Hongjie C, Nanayakkara-Bind A, Stewart CP, Khatry SK, LeClerq S, West Junior KP. Micronutrient Deficiencies Are Common in 6- to 8-Year-Old Children of Rural Nepal, with Prevalence Estimates Modestly Affected by Inflammation. J Nutr 2014; 144(6):979-987.. In this study, ~27% of the children between 11 and 14 months had diarrhea, and 70% had a cough in the two weeks preceding the study. The variable indicating recent inflammation or infection (CRP) was strongly associated with anemia and showed a borderline association with VAD. The prevalence of anemia in children with inflammation was almost three times higher than among those with no inflammation. The literature has ample references to a predisposition for anemia following acute infection, as in general this is accompanied by a worsening appetite, and greater loss or utilization of micronutrients, which varies depending on the duration and severity of disease3030. Thurnham DI, McCabe LD, Haldar S, Wieringa FT, Northrop-Clewes CA, McCabe GP. Adjusting plasma ferritin concentrations to remove the effects of subclinical inflammation in the assessment of iron deficiency: a meta-analysis. Am J ClinNutr 2010; 92(3):546-555.,3434. Schulze KJ, Christian P, Wu LS, Arguello M, Hongjie C, Nanayakkara-Bind A, Stewart CP, Khatry SK, LeClerq S, West Junior KP. Micronutrient Deficiencies Are Common in 6- to 8-Year-Old Children of Rural Nepal, with Prevalence Estimates Modestly Affected by Inflammation. J Nutr 2014; 144(6):979-987.,3838. Lima ACVMS, Lira PIC, Romani SAM, Eickmann SH, Piscoya MD, Lima MC. Fatores determinantes dos níveis de hemoglobina em crianças aos 12 meses de vida na Zona da Mata Meridional de Pernambuco. Rev Bras Saúde Matern Infant 2004; 4(1):35-43..
The frequency of anemia found in children in this part of the Amazon was similar to the findings of the 2006 PNDS1111. Brasil. Ministério da Saúde (MS). Pesquisa Nacional de Demografia e Saúde da Criança e da Mulher – PNDS 2006: dimensões do processo reprodutivo e da saúde da criança. Brasília: MS; 2009., and of a study in New Zealand4040. Soh P, Ferguson EL, McKenzie JE, Homs MYV, Gibson RS. Iron deficiency and risk factors for lower iron stores in 6-24 month-old New Zealanders. Eur J Clin Nutr 2004; 58(1):71-79.. However, they were lower than the frequency found in other Brazilian studies conducted with children of similar age included in the healthcare system. It is convenient to point out here to point out that a number of factors limit the possibility of comparing prevalence across studies, as some are derived from surveys of capillary blood samples1111. Brasil. Ministério da Saúde (MS). Pesquisa Nacional de Demografia e Saúde da Criança e da Mulher – PNDS 2006: dimensões do processo reprodutivo e da saúde da criança. Brasília: MS; 2009.,1414. Spinelli MGN, Marchioni DML, Souza JMP, Souza SB, Szarfac SC. Fatores de risco para anemia em crianças de 6 a 12 meses no Brasil. Rev Panam Salud Pública 2005; 17(2):84-91.,4444. Rohner F, Woodruff BA, Aaron GJ, Yakes EA, Lebanan MA, Rayco-Solon P, Saniel OP. Infant and young child feeding practices in urban Philippines and their associations with stunting, anemia, and deficiencies of iron and vitamin A. Food Nutr Bull 2013; 34(Supl. 2):S17-S34., which can cause hemodilution and introduce a systematic error in the hemoglobin assays. Other studies in Acre show a higher prevalence of anemia in children under the age of 277. Castro TG, Silva-Nunes M, Conde WL, Muniz PT, Cardoso MA. Anemia e deficiência de ferro em pré-escolares da Amazônia Ocidental brasileira: prevalência e fatores associados. Cad Saude Publica 2011; 27(1):131-142.,1717. Garcia MT, Granado FS, Cardoso MA. Alimentação complementar e estado nutricional de crianças menores de dois anos atendidas no Programa Saúde da Família em Acrelândia, Acre, Amazônia Ocidental Brasileira. Cad Saude Publica 2011; 27(2):305-316., which may be explained by the fact that people living in the state capital have better infrastructure and access to higher quality assets and services. In general, these children come from households of a higher socioeconomic level and education when compared to those in cities in the interior of the Amazon.
Iron deficiency has been shown to be a serious public health problem; our study found numbers higher than the results of by Pasricha et al.3131. Pasricha S-R, Shet AS, Black JF, Sudarshan H, Prashanth NS, Biggs BA. Vitamin B-12, folate, iron, and vitamin A concentrations in rural Indian children are associated with continued breastfeeding, complementary diet, and maternal nutrition. Am J Clin Nutr 2011; 94(5):1358-1370. in Indian children aged 12 to 23 months, and lower than the results found by Cardoso et al.2020. Cardoso MA, Scopel KKG, Muniz PT, Villamor E, Ferreira MU. Underlying factors associated with anemia in Amazonian children: a population-based, cross-sectional study. PLoS One 2012; 7:e36341. in children 6 to 23 months in the Amazon interior.
The prevalence of malnutrition in children under the age of 11 months (7.7%) was higher than would be estimated using PNDS1111. Brasil. Ministério da Saúde (MS). Pesquisa Nacional de Demografia e Saúde da Criança e da Mulher – PNDS 2006: dimensões do processo reprodutivo e da saúde da criança. Brasília: MS; 2009. data (4.8%), and was strongly associated with anemia, as per the findings of Yang et al.3333. Yang W, Li X, Li Y, Zhang S, Liu L, Wang X, Li W. Anemia, malnutrition and their correlations with socio-demographic characteristics and feeding practices among infants aged 0–18 months in rural areas of Shaanxi province in northwestern China: a cross-sectional study. BMC Public Health 2012; 12:1127. and Lima et al.3838. Lima ACVMS, Lira PIC, Romani SAM, Eickmann SH, Piscoya MD, Lima MC. Fatores determinantes dos níveis de hemoglobina em crianças aos 12 meses de vida na Zona da Mata Meridional de Pernambuco. Rev Bras Saúde Matern Infant 2004; 4(1):35-43.. Anemia and macro and micronutrient deficiency are related to infant growth and development, leading to short stature4747. Pedraza DF, Rocha ACD, Sales MC. Deficiência de micronutrientes e crescimento linear: revisão sistemática de estudos observacionais. Cien Saude Colet 2013; 18(11):3333-3347.. They can also interfere in Hb levels due to dietary insufficiencies.
The results of this study must be considered while bearing in mind its limitation. First, as it is a study of children seen by a basic care service, thus results may not be extrapolated to the general population of children under the age of two in Rio Branco. Second, a number of variables that are important causes of anemia such as gestational age and the normal eating habits of the study children were not analyzed due to inconsistencies in the former, and no data for the latter. Lastly, this is a cross-sectional study where data on individuals was collected only once, making it impossible to determine the timing of the outcomes and markers of recent infection and micronutrient deficiency, so care must be taken in its interpretation. Despite the absence of more accurate data on food consumption in our study, the combination of different indicators of iron status and biochemical micronutrient assays allowed us to identify other factors associated with anemia, in addition to iron deficiency. There is a single, population-based study conducted in a town in the interior of the Amazon that looked at the same range of biochemical indicators2020. Cardoso MA, Scopel KKG, Muniz PT, Villamor E, Ferreira MU. Underlying factors associated with anemia in Amazonian children: a population-based, cross-sectional study. PLoS One 2012; 7:e36341..
Our findings show that not only iron, but also other micronutrient deficiencies and the presence of infection affect the prevalence of anemia, reinforcing the need to supplement with other micronutrients and improve how morbidities are handled within the child-care services of the Rio Branco primary healthcare services. Interventions are required to improve infant health and nutrition bearing in mind the distal determinants of deficiencies such as social inequalities in terms of access to goods and services, and the importance of the nutritional advice provided by healthcare professionals and adequate nutritional practices in infancy.
Acknowledgements
To the mothers and infants who participated in the study. To the healthcare professionals and managers of the Basic Healthcare Units who participated in the study. To Universidade Federal do Acre for logistics support. To the interviewers for their contributions during fieldwork.
To the Ministry of Health/Overall Food and Nutrition Coordination for funding.
Referências
-
1Stevens GA, Finucane MM, De-Regil LM, Paciorek CJ, Flaxman SR, Branca F, Peña-Rosas JP, Bhutta ZA, Ezzati M. Global, regional, and national trends in haemoglobin concentration and prevalence of total and severe anaemia in children and pregnant and non-pregnant women for 1995-2011: a systematic analysis of population-representative data. Lancet Glob Health 2013; 1(1):e16-e25.
-
2World Health Organization (WHO). Center for Disease Control and Prevention. de Benoist B, McLean E, Egli I, Cogswellm M, editors. Worldwide prevalence of anaemia 1993-2005. WHO Global Database on Anaemia. Geneva: WHO; 2008.
-
3Lozoff B. Iron deficiency and child development. Food Nutr Bull 2007; 28(Supl. 4):S560-S571.
-
4World Health Organization (WHO), United Nations Children’s Fund (UNICEF), United Nations University (UNU). Iron deficiency anaemia. Assessment, prevention and control. A guide for programme managers. Geneva: WHO; 2001.
-
5Hadler MC, Juliano Y, Sigulem DM. Anemia do lactente: etiologia e prevalência. J Pediatr 2002; 78(4):321-326.
-
6Carvalho AGC, Lira PIC, Barro MFA, Aléssio MLM, Lima MC, Carbonneau MA, Berger J, Léger CL. Diagnóstico por deficiência de ferro em crianças do Nordeste do Brasil. Rev Saude Publica 2010; 44(3):513-519.
-
7Castro TG, Silva-Nunes M, Conde WL, Muniz PT, Cardoso MA. Anemia e deficiência de ferro em pré-escolares da Amazônia Ocidental brasileira: prevalência e fatores associados. Cad Saude Publica 2011; 27(1):131-142.
-
8Bortolini GA, Vitolo MR.Relação entre deficiência de ferro e anemia em crianças de até 4 anos de idade. J Pediatr 2010; 86(6):488-492.
-
9Villalpando S, Pérez-Expósito A, Shamah-Levy T, Rivera JA. Distribution of anemia associated with micronutrient deficiencies other than iron: a probabilistic sample of Mexican children. Ann Nutr Metab 2006; 50(6):506-511.
-
10Arsenault JE, Mora-Plazas M, ForeroY, Lopez-Arana S, Baylin A, Villamor E. Hemoglobin concentration is inversely associated with erythrocyte folate concentrations in Colombian school-age children, especially among children with low vitamin B12 status. Eur J Clin Nutr 2009; 63(7):842-849.
-
11Brasil. Ministério da Saúde (MS). Pesquisa Nacional de Demografia e Saúde da Criança e da Mulher – PNDS 2006: dimensões do processo reprodutivo e da saúde da criança. Brasília: MS; 2009.
-
12World Health Organization (WHO). Use of multiple micronutrient powders for home fortification of foods consumed by infants and children 6-23 months of age Geneva: WHO; 2011.
-
13Instituto Brasileiro de Geografia e Estatística (IBGE). Estudos e Pesquisas. Informação Demográfica e Socioeconômica, n. 32. Síntese de Indicadores Sociais: uma análise das condições de vida da população brasileira 2013. [acessado 2014 jun 23]. Disponível em: ftp://ftp.ibge.gov.br/Indicadores_Sociais/Sintese_de_Indicadores_Sociais_2013/SIS_2013.pdf
-
14Spinelli MGN, Marchioni DML, Souza JMP, Souza SB, Szarfac SC. Fatores de risco para anemia em crianças de 6 a 12 meses no Brasil. Rev Panam Salud Pública 2005; 17(2):84-91.
-
15Jordão RE, Bernardi JLD, Barros Filho A. Prevalência de anemia ferropriva no Brasil: uma revisão sistemática. Rev Paul Pediatr 2009; 27(1):90-98.
-
16Vieira RCS, Ferreira HS. Prevalência de anemia em crianças brasileiras, segundo diferentes cenários epidemiológicos. Rev Nutrição 2010; 23(3):433-444.
-
17Garcia MT, Granado FS, Cardoso MA. Alimentação complementar e estado nutricional de crianças menores de dois anos atendidas no Programa Saúde da Família em Acrelândia, Acre, Amazônia Ocidental Brasileira. Cad Saude Publica 2011; 27(2):305-316.
-
18Castro TG, Baraldi L, Muniz PT, Cardoso MA. Dietary practices and nutritional status of 0-24 month-old children from Brazilian Amazonia. Publ Health 2009; 12(12):2335-2342.
-
19De Souza OF, Macedo, LFM, Oliveira CSM, Araújo TS, Muniz PT. Prevalence and associated factors to anaemia in children. J Hum Growth Dev 2012; 22(3):307-313.
-
20Cardoso MA, Scopel KKG, Muniz PT, Villamor E, Ferreira MU. Underlying factors associated with anemia in Amazonian children: a population-based, cross-sectional study. PLoS One 2012; 7:e36341.
-
21Oliveira CSM, Cardoso MA, Araujo TS, Muniz PT. Anemia em crianças de 6 a 59 meses e fatores associados no Município de Jordão, Estado do Acre, Brasil. Cad Saude Publica 2011; 27(5):1008-1020.
-
22Instituto Brasileiro de Geografia e Estatística (IBGE). Cidades. Resultados da amostra do Censo Demográfico 2010. [acessado 2014 maio 23]. Disponível em: http://cidades.ibge.gov.br/xtras/perfil.php?codmun=120040
» http://cidades.ibge.gov.br/xtras/perfil.php?codmun=120040 -
23Programa das nações unidas para o desenvolvimento (PNUD). Atlas do desenvolvimento humano no Brasil: atlas brasil 2013. [acessado 2014 jun 23]. Disponível em:http://www.pnud.org.br/idh/atlas2013.aspx?indiceaccordion=1&li=li_atlas2013
» http://www.pnud.org.br/idh/atlas2013.aspx?indiceaccordion=1&li=li_atlas2013 -
24Brasil. Ministério da Saúde (MS). Caderno de Informações em Saúde. [acessado 2014 maio 23]. Disponível em: http://tabnet.datasus.gov.br/tabdata/cadernos/ac.htm
» http://tabnet.datasus.gov.br/tabdata/cadernos/ac.htm -
25Secretaria Municipal de Saúde de Rio Branco. Departamento de Atenção Básica (DAB). DATASUS. Fonte: SIAB - Sistema de Informação de Atenção Básica. Relatório do Consolidado da Cobertura do Programa Saúde da Família. Rio Branco: Secretaria Municipal de Saúde de Rio Branco; 2013
-
26World Health Organization (WHO). Expert Committee on Physical Status. Physical status: the use and interpretation of anthropometry: report of a WHO Expert Committee. Geneva: WHO; 1995.
-
27World Health Organization (WHO).WHO Anthro (version 3.2.2, January 2011b) and macros. [acessado 2014 jun 23]. Disponível em: http://www.who.int/childgrowth/software/es/index.html
» http://www.who.int/childgrowth/software/es/index.html -
28World Health Organization (WHO).Indicators for assessing vitamin A deficiency and their application in monitoring and evaluating intervention programmes. Geneva: WHO; 1996.
-
29Cook JD, Flowers CH, Skikne BS. The quantitative assessment of body iron. J Hematol 2003; 101(9):3359-3364.
-
30Thurnham DI, McCabe LD, Haldar S, Wieringa FT, Northrop-Clewes CA, McCabe GP. Adjusting plasma ferritin concentrations to remove the effects of subclinical inflammation in the assessment of iron deficiency: a meta-analysis. Am J ClinNutr 2010; 92(3):546-555.
-
31Pasricha S-R, Shet AS, Black JF, Sudarshan H, Prashanth NS, Biggs BA. Vitamin B-12, folate, iron, and vitamin A concentrations in rural Indian children are associated with continued breastfeeding, complementary diet, and maternal nutrition. Am J Clin Nutr 2011; 94(5):1358-1370.
-
32World Health Organization (WHO). Conclusions of a WHO Technical Consultation on folate and vitamin B-12 deficiencies. Food Nutr Bull 2008b; 29(Supl. 2):S238-S244.
-
33Yang W, Li X, Li Y, Zhang S, Liu L, Wang X, Li W. Anemia, malnutrition and their correlations with socio-demographic characteristics and feeding practices among infants aged 0–18 months in rural areas of Shaanxi province in northwestern China: a cross-sectional study. BMC Public Health 2012; 12:1127.
-
34Schulze KJ, Christian P, Wu LS, Arguello M, Hongjie C, Nanayakkara-Bind A, Stewart CP, Khatry SK, LeClerq S, West Junior KP. Micronutrient Deficiencies Are Common in 6- to 8-Year-Old Children of Rural Nepal, with Prevalence Estimates Modestly Affected by Inflammation. J Nutr 2014; 144(6):979-987.
-
35VitoloMR, BortoliniGA. Biodisponibilidade do ferro como fator de proteção contra anemia entre crianças de 12 a 16 meses. J Pediatr 2007; 83(1):33-38.
-
36Jones G, Steketee Rw, Black Re, Bhutta ZA, Morris SS, Bellagio Child Survival Study Group. How many child deaths can we prevent this year? Lancet 2003; 362(9377):65-71.
-
37Brasil. Ministério do Desenvolvimento Social e Combate à Fome (MDS). Indicadores de Desenvolvimento Brasileiro. 2013. [acessado 2014 jun 23]. Disponível em: http://189.28.128.178/sage/sistemas/apresentacoes/arquivos/indicadores_de_desenvolvimento_2013.pdf
» http://189.28.128.178/sage/sistemas/apresentacoes/arquivos/indicadores_de_desenvolvimento_2013.pdf -
38Lima ACVMS, Lira PIC, Romani SAM, Eickmann SH, Piscoya MD, Lima MC. Fatores determinantes dos níveis de hemoglobina em crianças aos 12 meses de vida na Zona da Mata Meridional de Pernambuco. Rev Bras Saúde Matern Infant 2004; 4(1):35-43.
-
39Choi HJ, Lee HJ, Jang HB, Park JY, Kang JH, Park KH, Jihyun Song J. Effects of maternal education on diet, anemia, and iron deficiency in Korean school-aged children. BMC Public Health 2011; 11:870.
-
40Soh P, Ferguson EL, McKenzie JE, Homs MYV, Gibson RS. Iron deficiency and risk factors for lower iron stores in 6-24 month-old New Zealanders. Eur J Clin Nutr 2004; 58(1):71-79.
-
41Mora J. Iron supplementation: overcoming technical and practical barriers. J Nutr 2002; 132(4):S853-S855.
-
42Herrán OF, Ward JB, Villamor E. Vitamin B12 sero status in Colombian children and adult women: results from a nationally representative survey. Public Health Nutr 2014; 18(5):836-843.
-
43Pedraza DF, Queiroz D, Paiva AA, Cunha MAL da, Lima ZN. Food security, growth and vitamin A, hemoglobin and zinc levels of preschool children in the northeast of Brazil. Cien Saude Colet 2014; 19(2):641-650.
-
44Rohner F, Woodruff BA, Aaron GJ, Yakes EA, Lebanan MA, Rayco-Solon P, Saniel OP. Infant and young child feeding practices in urban Philippines and their associations with stunting, anemia, and deficiencies of iron and vitamin A. Food Nutr Bull 2013; 34(Supl. 2):S17-S34.
-
45Saraiva BC, Soares MC, Santos LC, Pereira SC, Horta PM. Iron deficiency and anemia are associated with lowretinol levels in children aged 1 to 5 years. J Pediatr 2014; 90(6):593-599.
-
46Jafari SM, Heidari G, Nabipoura I, Amirinejad R, Assadi M, Bargahi A, Akbarzadehd S, Tahmasebia R, Sanjdideh Z. Serum retinol levels are positively correlated with hemoglobin concentrations, independent of iron homeostasis: a population-based study. Nutrition Research 2013; 33(4):279-285.
-
47Pedraza DF, Rocha ACD, Sales MC. Deficiência de micronutrientes e crescimento linear: revisão sistemática de estudos observacionais. Cien Saude Colet 2013; 18(11):3333-3347.
Publication Dates
-
Publication in this collection
Feb 2016
History
-
Received
26 Nov 2014 -
Reviewed
16 June 2015 -
Accepted
18 June 2015