Abstracts
S-100 dendritic cells immunolabeling were investigated in uninfested skin biopsies from five Nelores, five Holstein-Friesian and five crossbreed (Holstein-Friesian x Nelores) calves. The group of crossbreed animal was experimentally infested with 100 first-instar larvae of Dermatobia hominis and biopsies were obtained 24, 48, 72 and 168 hours after infestation. Samples obtained prior to infestation from these animals were used as control. Rabbit anti-protein S-100 antibody and the avidin-biotin-peroxidase method were used for immunolabeling. Melanocytes, nerves and endothelial cells and dermal dendritic cells (DC) were positive to protein S-100. DCs were exclusively detected in the superficial dermis close to the basal layer of both normal and parasitized animals. There was no significant difference in DC numbers that might be attributed to breed. In parasitized animals the DC had thicker and deeply stained dendritic processes compared to normal animals. Statistically significant decreases in the DC number occurred after infestation.
Dendritic cell; Protein; Cattle; Skin; Dermatobia hominis
Foram investigadas as células dendríticas (CD) na pele normal de cinco bezerros das raças Nelore, cinco da raça Holandesa Preta e Branca e cinco animais mestiços por meio da imunomarcação pela proteína S-100. Os animais mestiços foram infestados experimentalmente com 100 larvas de primeiro estádio de Dermatobia hominis e deles foram colhidas biópsias de pele parasitada às 24, 48, 72 e 168 horas após a infestação. Biópsias de pele destes animais, colhidas antes da infestação, foram utilizadas como controle. A imunomarcação das CDs foi feita empregando-se anticorpos de coelhos antiproteína S-100 e a técnica da avidina-biotina-peroxidase. Além das CDs, melanócitos, nervos e células endoteliais apresentaram imunomarcação pela proteína S-100. As DCs foram observadas exclusivamente na derme superficial, próximas à camada basal, tanto nos animais infestados como nos não-infestados. Não se detectou diferença significativa no número de CDs que pudesse ser atribuída à raça dos animais. Nos animais parasitados por D. hominis, as CDs apresentavam-se mais intensamente coradas e com os prolongamentos mais espessos do que nos controles não-parasitados. Além disso, nos animais parasitados observou-se um decréscimo significativo no número de CDs a partir de 24 horas após a infestação.
Células dendríticas; Proteínas; Bovinos; Pele; Dermatobia hominis
S-100 dendritic cells in normal and Dermatobia hominis infested cattle skin
Células dentríticas S-100 positivas em pele de bovinos normais e infestados por Dermatobia hominis
Teresa Cristina Goulart de OLIVEIRA-SEQUEIRA11 Departamento de Parasitologia do Instituto de Biociências da UNESP, Botucatu SP ; Carlos Eduardo BACHI21 Departamento de Parasitologia do Instituto de Biociências da UNESP, Botucatu SP ; Edy de LELLO31 Departamento de Parasitologia do Instituto de Biociências da UNESP, Botucatu SP
CORRESPONDENCE TO:
Teresa Cristina Goulart de Oliveira-Sequeira
Departamento de ParasitologiaInstituto de Biociências da UNESP
Campus de Botucatu
18618-000 Botucatu SP
e-mail: sequeira@ibb.unesp.br
SUMMARY
S-100 dendritic cells immunolabeling were investigated in uninfested skin biopsies from five Nelores, five Holstein-Friesian and five crossbreed (Holstein-Friesian x Nelores) calves. The group of crossbreed animal was experimentally infested with 100 first-instar larvae of Dermatobia hominis and biopsies were obtained 24, 48, 72 and 168 hours after infestation. Samples obtained prior to infestation from these animals were used as control. Rabbit anti-protein S-100 antibody and the avidin-biotin-peroxidase method were used for immunolabeling. Melanocytes, nerves and endothelial cells and dermal dendritic cells (DC) were positive to protein S-100. DCs were exclusively detected in the superficial dermis close to the basal layer of both normal and parasitized animals. There was no significant difference in DC numbers that might be attributed to breed. In parasitized animals the DC had thicker and deeply stained dendritic processes compared to normal animals. Statistically significant decreases in the DC number occurred after infestation.
UNITERMS: Dendritic cell; Protein; Cattle; Skin; Dermatobia hominis.
INTRODUCTION
The best known dendritic cells (DC) are epidermal Langerhans cells (LC) which in many situations are related to the development of a primary immune response. Nithiuthai; Allen20 found morphologic and quantitative changes in Langerhans cell populations in the skin of guinea pigs infested with ticks. According to these investigators, the LC may incorporate salivary antigens and present them to T-cells; in subsequent infestations they may represent the initial focal point for the triggering of a more rapid and extensive cellular and humoral response. As a consequence of these events, chemotactic factors for basophils and eosinophils would be released and would promote the accumulation of these effector cells at the sites of tick fixation.
Besides LC, other cutaneous dendritic cells are now recognized as part of a system of antigen-presenting cells (APCs) and represent a group of "accessory" cells necessary to generate a primary antibody response19. Three pools of DC from skin are recognized: epidermal dendritic cells (Langerhans cells), dermal dendritic cells (DDC), and "veiled cells" from afferent lymph. All of them are antigen-presenting cells important in mediating T-cell activation and T-cell tolerance. Infectious agents and inflammatory products can induce their activities making them mobile sentinels that bring antigens to T-cells. DCs are now subject for several studies that involve immune response as transplantation, allergy, autoimmune disease, resistance to infection and to tumor, immunodeficiency and vaccines2.
The major attributes studied in an attempt to correlate resistance to ectoparasites with the phenotypic traits of animals have been hair length, skin thickness, density of sebaceous gland and mast cells, superficiality of hair follicles and number of circulating lymphocytes3.
The only criterion used to evaluate animal resistance or susceptibility to Dermatobia hominis (Linnaeus, Jr.), (Diptera: Cuterebridae) has been the quantification of typical subcutaneous nodules provoked by the presence of larvae6,12. Cattle innate resistance to parasitism has been related to breed and coat color. So, animals of European breeds and animals with dark coats have been usually considered more susceptible17.
D. hominis is a major cause of myiasis in the Neotropical region occurring in a wide range of vertebrates host, but the most devastating impact relates to cattle in which it causes loss of weight, reduced milk production and, especially, perforation of hides13. Despite its economic and public health importance there are few reports on the host-parasite relationship, especially immune response of cattle.
We previously identified eosinophils as the major effector cells present in the exudate that surrounds first-instar larvae of D. hominis and reactive immunoglobulins at the site of parasitized cattle skin suggesting that parasite damage could be a consequence of antibody-dependent cell-mediated cytotoxicity21. According to Grogan et al.7, in human infested by D. hominis eosinophils appear attracted to larvae by eosinophil chemotactic factor (ECF) secreted by T-cells.
As DC is the key cell implicated in afferent antigen processing and presentation resulting in T-cell homing and clonal expansion, we compared the DC population in the skin of three non-infested cattle breeds (Nelore, Holstein-Friesian and their crossbreed). A second purpose was to investigate the DC responses of cattle to infestation with D. hominis.
MATERIAL AND METHOD
Animals and biopsies
Skin biopsies were obtained from Holstein-Friesian, Nelore and crossbreed animals (Holstein-Friesian x Nelore) aging 11 to 19 months (n = five animals each group). All animals had early natural exposure to D. hominis, but at time of experiment no developing larvae were found in subcutaneous tissues. The 5 crossbreed calves were infested with approximately 100 first-instar larvae of D. hominis obtained in the laboratory according to Lello; Peraçoli11. Biopsies of parasitized skin were taken 24, 48, 72 and 168 hours after infestation as previously described21.
Immunocytochemical processing
The formalin fixed biopsies were embedded in Histosec wax (Merck) and 4-5 µm sections were obtained with a steel blade microtome. The avidin-biotin-peroxidase technique9 was used for DC immunolabeling. Anti-protein S-100 was used as primary antibody (rabbit anti-S100, Dako) and goat anti-rabbit biotinylated antibody (Vector) was used as secondary antibody.
A human melanoma biopsy was processed as a positive control and omission of the primary antibody was used as a negative control.
The criteria used for the identification of dendritic cells were: (1) presence of a fully visible cell body; (2) presence of at least one dendritic process.
Morphometric analysis
Dendritic cells were counted in 10-20 fields delimited by a 100-point grid in each skin sample. Counts were performed with a Zeiss microscope with a 40x objective in a kpl 8x eyepiece. The population of dendritic cells is reported as number of cells/mm2 of skin20 for each time point.
Statistical analysis
Numbers of dendritic cells in the skin of Nelore, Holstein-Friesian and crossbreed calves were analyzed on Minitab version 11 by using a one-way analysis of variance.
The relative difference between the number of dendritic cells before infestation and at each moment after infestation was analyzed by t test22. Statistical significance was taken as p < 0.05.
RESULTS
Localization of protein S-100 in cattle skin
Immunolabeling for protein S-100 was observed in the cytoplasm and nucleus of dermal dendritic cells, nervous cells and in the endothelium of blood vessels. In addition, it was observed in rough granules of melanocytes located exclusively in the basal layer of the epithelium of dark coated animals. These melanocytes were the only epidermal cells or structures positive to protein S-100.
Morphology of dermal dendritic cells in normal cattle skin
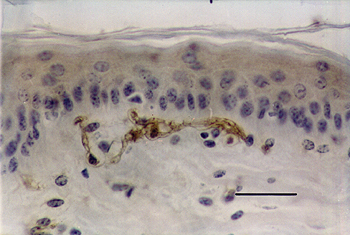
Dendritic cells identified by the presence of protein S-100 were located in the superficial dermis, often close to the basal layer of the epithelium. The cell body was well defined, oval or polygonal, and presented cytoplasmic prolongations. These dendritic processes showed lighter staining and were quite tenuous and delicate (Fig. 1).
Morphology of dendritic cells in cattle skin infested with Dermatobia hominis
In the skin of parasitized calves, dendritic cells presented the same location as in normal skin. However, the dendritic processes were thickened, rough and deeply stained. On several occasions it was possible to observe the presence of lymphocytes in close contact with dendritic cells, which seemed to envelop them through their dendritic processes (Fig. 2).
Quantitative analysis of the dermal dendritic cells number in the normal skin
The mean number (± standard error) of dendritic cells in the normal skin of Holstein-Friesian, Nelore and crossbreed calves were 16.8 ± 2.6, 17.6 ± 1.0 and 20.8 ± 2.5, respectively. No significant difference in number of cells was observed between breeds.
Quantitative analysis of the dermal dendritic cells numbers in cattle skin parasitized with Dermatobia hominis
The mean numbers of dendritic cells/mm2 counted in the skin of 5 parasitized calves, before and after infestation, are showed in Fig. 3.
The number of DC decreased after 24 hours of infestation remaining lower than original levels (p < 0.05) until the seventh day (168 hours).
DISCUSSION
In the skin of humans, protein S-100 was detected in structures not related to nervous tissues such as melanocytes and Langerhans cells4,8,10,18. The results obtained here for the skin of cattle partially agree with those reported by the above authors. We already detect S-100 protein in melanocytes, but not in epidermal Langerhans cells. Besides melanocytes, other structures not related to nervous tissues positive to S-100 protein were endothelial cells and dermal dendritic cells.
The occurrence of protein S-100 in the mammary arterioles of dogs15 and in the endothelial cells of blood vessels and dendritic cells of cattle granulomas16 suggests that differences in the distribution of S-100 protein may vary in different mammalian species.
Up to now all the immunological phenotipical traits investigated comparing breed resistance to ectoparasites focused in the intensity of infiltration of effector cells such eosinophils, basophils or mast cells3. As the infiltration of these cells occurs after antigen presentation20, we investigated DC as a possible phenotypical marker for breed resistance. We obtained no evidence that the number of dendritic cells varied as a function of this variable, but we cannot rule out this possibility because other APC, not investigated in the present work, could be important in cattle responses to D. hominis.
The major morphologic change of dendritic cells observed in the skin of cattle parasitized with D. hominis were thickening of the dendritic processes and deeper labeling by protein S-100. This same pattern of alteration was observed in Langerhans cells of human patients with allergic reactions to drugs5 and with cholesteatoma24. This kind of morphological change could be related to a manifestation of an activation process and must be better investigated in future experiments.
Significant differences in dendritic cells numbers were observed before and after calf infestation. DC number decreases 24 hours after infestation, remaining lower than the original levels until seventh day. This kinetics seems to be quite compatible with the role of these cells whose function is to incorporate antigens in the skin and migrate to regional lymph nodes where they present these antigens to T lymphocytes and trigger the cellular and humoral immune response1 These events have been described for viral infections23 and for Leishmania infection14. Thus, the reduction observed after infestation suggests that, after the antigenic stimulus originating from larval penetration, the dendritic cells may incorporate secreted and/or excreted soluble antigens and migrate to the lymph nodes were they present them to T lymphocytes. Effector cells including eosinophils and basophils observed at the site of larval establishment in cattle21, could be attracted to the larvae by chemotatic factors secreted by these sensitized T-cells.
Another piece of evidence of the role of DCs in cattle response to Dermatobia hominis was their close association with lymphocyte as seen in Fig. 2. Lymphocytes that traffic through the skin from the vasculature interact with DCs25, suggesting that these latter cells may also play a role in antigen presentation outside of lymph nodes, especially in a secondary response, as described here.
ACKNOWLEDGEMENTS
We thank Dr. G.J.M. Rosa and Luzia A. Trinca for statistical analysis, to Mr. A.V. Salvador for technical assistance and to Dr. Alessandro F.T. Amarante for suggestions. Grants for this work were from Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP-93/2049-1) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq-300736/88-6).
RESUMO
Foram investigadas as células dendríticas (CD) na pele normal de cinco bezerros das raças Nelore, cinco da raça Holandesa Preta e Branca e cinco animais mestiços por meio da imunomarcação pela proteína S-100. Os animais mestiços foram infestados experimentalmente com 100 larvas de primeiro estádio de Dermatobia hominis e deles foram colhidas biópsias de pele parasitada às 24, 48, 72 e 168 horas após a infestação. Biópsias de pele destes animais, colhidas antes da infestação, foram utilizadas como controle. A imunomarcação das CDs foi feita empregando-se anticorpos de coelhos antiproteína S-100 e a técnica da avidina-biotina-peroxidase. Além das CDs, melanócitos, nervos e células endoteliais apresentaram imunomarcação pela proteína S-100. As DCs foram observadas exclusivamente na derme superficial, próximas à camada basal, tanto nos animais infestados como nos não-infestados. Não se detectou diferença significativa no número de CDs que pudesse ser atribuída à raça dos animais. Nos animais parasitados por D. hominis, as CDs apresentavam-se mais intensamente coradas e com os prolongamentos mais espessos do que nos controles não-parasitados. Além disso, nos animais parasitados observou-se um decréscimo significativo no número de CDs a partir de 24 horas após a infestação.
UNITERMOS: Células dendríticas; Proteínas; Bovinos; Pele; Dermatobia hominis.
Received: 13/10/1999
Accepted: 20/03/2000
2 Departamento de Patologia da Faculdade de Medicina da UNESP, Botucatu SP
3 Departamento de Morfologia do Instituto de Biociências da UNESP, Botucatu SP
-
1- AUSTYN, J.M. New insights into mobilization and phagocytic activity of dendritic cells. J. Exp. Med., v.183, n.4, p.1287-92, 1996.
-
2- BANCHEREAU, J.; STEINMAN, R.M. Dendritic cells and the control of immunity. Nature, v.392, n.6673, p.245-51, 1998.
-
3- BROWN, S.J. Highlights of contemporary research on host immune response to ticks. Vet. Parasitol., v.28, n.4, p.321-34, 1988.
-
4- COCCHIA, D.; MICHETTI, F.; DONATO, R. Immunocytochemical localization of S-100 antigen in normal human skin. Nature, v.294, n.5836, p.85-7, 1981.
-
5- DASCALU, D.I.; KLETTER, Y.; BARATZ, M.; BRENNER, S. Langerhans’ cell distribution in drug eruption. Acta Derm. Venereol., v.72, n.3, p.175-7, 1992.
-
6- GOMES, A.G.; MAIA, A.A.M. Berne: distribuição sazonal, localização no hospedeiro e susceptibilidade de bovinos mestiços na região de Uberaba, MG. Rev. Fac. Med. Vet. Zootec. Univ. São Paulo, v.25, n.1, p.109-15, 1988.
-
7- GROGAN, T.M.; PAYNE, C.M.; PAYNE, T.B.; SPIER, C.; CROMEY, D.W.; RANGEL, C.; RICHTER, L. Cutaneous myiasis: immunohistologic and ultrastructural morphometric features of human botfly lesion. Am. J. Dermatol., v.9, n.3, p.232-9, 1987.
-
8- HALLIDAY, G.M.; MCARDLE, J.P.; KNIGHT, B.A.; MULLER, H.K. New methodology for assessment of the Langerhans cell network. J. Pathol., v.148, n.2, p.127-34, 1986.
-
9- HSU, S.M.; RAINE, L.; FANGER, H. A comparative study of the peroxidase-antiperoxidase method and an avidin-biotin complex method for studying polypeptide hormones with radioimmunoassay antibodies. Am. J. Clin. Pathol., v.75, n.5, p.734-8, 1981.
-
10- KAHN, H.D.; MARKS, A.; THOM, H.; BAUMAL, R. Role of antibody to S-100 protein in diagnostic pathology. Am. J. Clin. Pathol., v.79, n.3, p.341-7, 1983.
-
11- LELLO, E.; PERAÇOLI, M.T.S. Cell-mediated and humoral immune response in immunized and/or Dermatobia hominis infested rabbits. Vet. Parasitol., v.47, n.1-2, p.129-38, 1993.
-
12- MAIA, A.A.M.; GUIMARÃES, M.P. Berne: susceptibilidade de bovinos, distribuição no hospedeiro, associação com outras miíases e abcessos. Arq. Bras. Med. Vet. Zootec., v.37, n.5, p.461-7, 1985.
-
13- McMULLIN, P.F.; CRAMER, L.G.; BENZ, G.; JEROMEL, P.C.; GROSS, S.J. Control of Dermatobia hominis infestation using an ivermectin slow-release bolus. Vet. Rec, v.29, n.17, p.465, 1989.
-
14- MOLL, H.; FUCHS, H.; BLANK, C.; ROELLINGHOFF, M. Langerhans cell transport Leishmania major from the infected skin to the draining lymph node for presentation to antigen-specific T cells. Eur. J. Immunol., v.23, n.7, p.1595-601, 1993.
-
15- MÖLLER, A.C.; HELLMÉN, E. S-100 protein is not specific for myoepithelial cells in the canine mammary gland. J. Comp. Pathol., v.110, n.1, p.49-55, 1994.
-
16- MOMOTANI, E.; KUBO, M.; ISHIKAWA, Y.; MATSUBARA, Y.; NAKAJIMA, Y.; YOSHINO, T. Immunohistochemical distribution of S-100 a -positive cells in bovine mycobacterial and non-mycobacterial granulomas. J. Comp. Pathol., v.108, n.3, p.291-301, 1993.
-
17- MORAES, F.R.; COSTA, A.J.; VASCONCELOS, O.T.; ROCHA, U.F. Ensaio comparativo de susceptibilidade natural de zebuínos e taurinos à larva de Dermatobia hominis (Lineu Jr., 1781). Ars. Vet., v.2, n.2, p.247-53, 1986.
-
18- NAKAJIMA, T.; WATANABE, S.; SATO, Y. An immunoperoxidase study of S-100 protein distribution in normal and neoplastic tissues. Am. J. Surg. Pathol., v.6, n.8, p.715-27, 1982.
-
19- NESTLE, F.O.; FILGUEIRA, L.; NICKOLOFF, B.J.; BURG, G. Human dermal dendritic cells process and present soluble protein antigens. J. Invest. Dermatol., v.110, n.5, p.762-6, 1998.
-
20- NITHIUTHAI, S.; ALLEN, J.R. Significant changes in epidermal Langerhans cells of guinea pigs infested with ticks (Dermacentor andersoni). Immunology, v.51, n.1, p.133-41, 1984.
-
21- OLIVEIRA-SEQUEIRA, T.C.G.; SEQUEIRA, J.L.; SCHMITT, F.L.; LELLO, E. Histological and immunological reaction of cattle skin infested with Dermatobia hominis Med. Vet. Entomol., v.10, n.4, p.323-30, 1996.
-
22- SIEGEL, S. Estatística não paramétrica para as ciências do comportamento São Paulo : Makro Books do Brasil, 1975. p.350.
-
23- SPRECHER, E.; BECKER, Y. Langerhans cell density and activity in mouse skin and lymph nodes affect herpes simplex type I (HSV I) pathogenicity. Arch. Virol., v.107, n.3-4, p.191-205, 1989.
-
24- TAKAHASHI, S.; NAKANO, Y. Immunohistochemical demonstration of Langerhans’ cell in cholesteatoma using an antiserum against S-100 protein. Arch. Otorhinolaryngol., v.246, n.1, p.48-52, 1989.
-
25- TSE, Y.; COOPER, K.D. Cutaneous dermal Ia+ cells are capable of initiating delayed type hypersensitivity responses. J. Invest. Dermatol., v.94, n.3, p.267-72, 1990.
Publication Dates
-
Publication in this collection
11 May 2001 -
Date of issue
2000
History
-
Received
13 Oct 1999 -
Accepted
20 Mar 2000