Abstract
Rapidly proliferating tissue may require enhanced DNA repair capacity in order to avoid fixation of promutagenic DNA lesions to mutations. Partial hepatectomy (PH) triggers cell proliferation during liver regeneration (LR). However, little is known on how DNA repair genes change and how they are regulated at the transcriptional level during LR. In the present study, the Rat Genome 230 2.0 array was used to detect the expression profiles of DNA repair genes during LR, and differential expression of selected genes was confirmed by real-time RT-PCR. 69 DNA repair genes were found to be associated with LR, more than half of which distributed in a cluster characterized by a gradual increase at 24-72h and then returning to normal. The expression of base excision repair- and transcription-coupled repair-related genes was enhanced in the early and intermediate phases of LR, whereas the expression of genes related to HR, NHEJ and DNA cross-link repair, as well as DNA polymerases and related accessory factors, and editing or processing nucleases, were mainly enhanced in the intermediate phase. The expression changes of genes in DNA damage response were complicated throughout the whole LR. Our data also suggest that the expression of most DNA repair genes may be regulated by the cell cycle during LR.
partial hepatectomy; rat genome array; DNA repair genes; liver regeneration
SHORT COMMUNICATION
Alterations in DNA repair gene expression and their possible regulation in rat-liver regeneration
Gai-Ping WangI, II; Cun-Shuan XuI, II
ICollege of Life Science, Henan Normal University, Xinxiang, Henan Province, China
IIKey Laboratory for Cell Differentiation Regulation, Xinxiang, Henan Province, China
Send correspondence to Send correspondence to: Cunshuan Xu. College of Life Science, Henan Normal University, No. 46, Construction East Road, Xinxiang 453007 Henan, China. E-mail: cellkeylab@126.com; xucs@x263.net.
ABSTRACT
Rapidly proliferating tissue may require enhanced DNA repair capacity in order to avoid fixation of promutagenic DNA lesions to mutations. Partial hepatectomy (PH) triggers cell proliferation during liver regeneration (LR). However, little is known on how DNA repair genes change and how they are regulated at the transcriptional level during LR. In the present study, the Rat Genome 230 2.0 array was used to detect the expression profiles of DNA repair genes during LR, and differential expression of selected genes was confirmed by real-time RT-PCR. 69 DNA repair genes were found to be associated with LR, more than half of which distributed in a cluster characterized by a gradual increase at 24-72h and then returning to normal. The expression of base excision repair- and transcription-coupled repair-related genes was enhanced in the early and intermediate phases of LR, whereas the expression of genes related to HR, NHEJ and DNA cross-link repair, as well as DNA polymerases and related accessory factors, and editing or processing nucleases, were mainly enhanced in the intermediate phase. The expression changes of genes in DNA damage response were complicated throughout the whole LR. Our data also suggest that the expression of most DNA repair genes may be regulated by the cell cycle during LR.
Key words: partial hepatectomy, rat genome array, DNA repair genes, liver regeneration.
The liver has an outstanding capacity for regeneration (Taub, 2004). The process of hepatic cells initiating the cell cycle and proliferating rapidly, in order to compensate for lost liver tissues after rat partial hepatectomy (PH), is called liver regeneration (LR) (Lai et al., 2005; Suzuki and Tsukamoto, 2004). Injured liver cells and cell remnants caused by PH are harmful to the organism, while injured areas therefrom are susceptible to infection by antigens and xenobiotics, all possibly leading to inflammatory and immune responses (Shao et al., 2007; Zhang et al., 2006). Furthermore, carbohydrate, lipid, and protein and amino acid metabolisms are highly active, thereby providing nutrients or energy, especially for active DNA replication in LR (Fausto et al., 2006). As a result, a wider variety of endogenous damage produced by inflammation, normal metabolic byproducts (i.e. ROS) or replication errors, may constantly occur in LR.
It is common knowledge that DNA repair processes counteract genetic damage and maintain genome integrity (Wood et al., 2001). Many researchers have discovered that inherited mutations affecting DNA repair genes are strongly associated with high cancer risks (Jass, 2006). Decreased DNA repair capacity may be an important factor predisposing to the development of preneoplastic lesions, neoplastic nodules and malignant tumors (Vielhauer et al., 2001). It is generally believed that rapidly proliferating tissue undergoing DNA synthesis may require enhanced DNA repair capacity, so as to avoid fixation of promutagenic DNA lesions to mutations (Kaufmann et al., 1991; Riis et al., 2002). While hepatic cell proliferation is activated in regenerating liver, diminished rates of DNA repair may contribute to reducing LR capacity (Schmucker, 2005). Therefore, research on how DNA repair operates to prevent the accumulation of damage or mutations, and how to retain the rate of LR, has become a hot topic (Arai et al., 2003). Some researchers have used an LR model to assess expression changes in certain DNA repair enzymes, such as UNG and ATM, as well as their corresponding repair activities. As a result, they found hepatocytes are endowed with increased DNA-repair capacity during the period of highest transformation sensitivity in the cell cycle (Gombar et al., 1981; Lu et al., 2005).
Notwithstanding, there has been surprisingly little research on the global changes of mRNA expression in DNA repair-related genes during LR (Riis et al., 2002). In this study, a total of about 180 genes involved in various DNA repair pathways, such as the mismatch, direct, excision, homologous recombination (HR), DNA nonhomologous end-joining (NHEJ), DNA cross-link and translesion repair, were obtained by searching the biological pathway maps at databases, such as RGD, GenMAPP, KEGG, BIOCARTA and Biocompare. They were then reconfirmed through pertinent article retrieval (Arias-Lopez et al., 2006; Wood et al., 2005). Gene expression profiles of the above DNA repair genes were detected using the Rat Genome 230 2.0 array, whereupon their expression changes and possible regulation patterns during LR were primarily analyzed.
A total of 76 healthy male Sprague-Dawley rats, each weighing 200 ± 10 g, were supplied by the Experimental Animal Center of Henan Normal University. They were randomly divided into 19 groups (4 rats in each), viz., 9 PH, 9 sham-operated (SO) and one normal control (NC). The rats in the PH groups underwent 2/3 PH as described by Higgins and Anderson(1931). Four rats a time were ether anesthetized at 0, 2, 6, 12, 24, 30, 36, 72, 120 and 168 h after PH, whereupon their livers were immediately removed and stored at -80 °C for use.
After isolation from the frozen livers, as indicated by the manual of Trizol reagent (Invitrogen Corporation, Carlsbad, California, USA), total RNA was purified, acccording to RNeasy mini protocol (Qiagen, Inc, Valencia, CA, USA). The quality of the final product was assessed by optical density measurement at 260/280 nm, as well as through agarose electrophoresis (180 V, 0.5 h). T7-oligo dT(24) (Keck Foundation, New Haven, CT), SuperScript II RT (Invitrogen Corporation, Carlsbad, CA) and 5 μg of total RNA were used to synthesize the first strand of cDNA, and the Affymetrix cDNA single-stranded cDNA synthesis kit for synthesis of the second. The resultant cDNA products were purified according to manufacturer's cDNA purify protocol. 12 μL of purified cDNA and the reagents in the GeneChip In Vitro Transcript Labeling Kit (ENZO Biochemical, New York, USA) were employed for synthesizing biotin-labeled cRNA, which was purified by means of RNeasy Mini Kit columns (Qiagen, Valencia, CA). 15 mL of cRNA (1 μg/μL) were incubated with 6 μL of 5 x fragmentation buffer and 9 μL of RNase free water for 35 min at 94 °C, and then digested into 35-200 bp cRNA fragments. The prehybridized Rat Genome 230 2.0 microarray was placed into a hybridization buffer, and hybridization was allowed to occur in a hybridization oven (Affymetrix) at 45 °C at 60 rpm for 16 h. The hybridized arrays were washed in a wash-buffer, and stained in a GeneChip® Fluidics Station 450 (Affymetrix Inc., Santa Clara, CA, USA). The arrays were then scanned and images captured with a GeneChip® Scanner 3000 (Affymetrix Inc., Santa Clara, CA, USA) (Guo and Xu, 2008).
Images showing gene expression abundance were converted into signal, signal detection (P, A, M) and experiment/control (Ri) values through Affymetrix GCOS 1.2 software (Affymetrix, USA). The data of each array were initially normalized by scaling all signals to a target intensity of 200. P values < 0.05 meant that gene expression is present (P), p < 0.065 indicated marginal expression (M), and p > 0.065 absence of expression (A). Furthermore, signal values of PH normalized to those of control were used to calculate the relative or ratio values of gene expression abundance. A ratio = 2 meant up-regulated gene expression, = 0.5, significantly down-regulated, and 0.5-2, biologically insignificant expression. To minimize technical errors inherent in microarray analysis, each sample was analyzed at least three times, and the average value was considered reliable.
As a result, 139 of the above-mentioned 180 DNA repair genes were assessed in the Rat Genome 230 2.0 array. Sixty-nine of these yielded meaningful expression changes, at least at one time-point after PH, thereby indicating significant (0.01 < p < 0.05) or extremely significant (p < 0.01) differences between PH and SO groups, thus indicating their involvement in LR. During LR, 55 genes were found to be up-regulated, 8 down-regulated and 6 up/down-regulated. Fold changes for the up-regulated ranged from 2-fold to 34-fold, and in the down-regulated from 2-fold to 5-fold (Supplementary Material, Table S1).
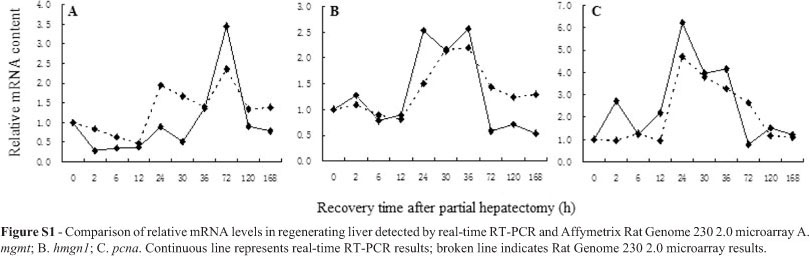
To confirm the results of the microarray analysis, some significantly changed genes were chosen for in-depth analysis by real-time quantitative RT-PCR. MGMT is an important direct repair protein which suicidally transfers the methyl moiety from O6-methylguanine to itself (Pegg et al., 1995), whereas PCNA plays a vital role in BER and the initiation of recombination-associated DNA synthesis (Li et al., 2009), and HMGN1 participates in promoting NER (Birger et al., 2003). Therefore, the above three genes involved in different DNA repair pathways were chosen. Primer sequences were designed by rimer express 2.0 software according to the mRNA sequences of mgmt, hmgn, pcna and that of the internal control gene β-actin (GenBank numbersNM_012861, NM_001013184, NM_022381 and NM_031144) (Supplementary Material, Table S2). First-strand cDNA samples underwent quantitative PCR amplification, using SYBR® Green I on a Rotor-Gene 3000A thermocycler (Corbett Robotics, San Francisco, CA). Each was analyzed in triplicate, and standard curves were generated from five repeated ten-fold serial dilutions of cDNA (Wang and Xu, 2010). The absolute values and corresponding relative values of their temporal transcriptional levels in RT-PCR assays appear in Table 1. On a whole, expression trends of the three genes detected by RT-PCR and microarray were generally consistent, thereby indicating that array-check results were reliable (Supplementary Material, Figure S1).
It is common knowledge that hepatocyte DNA replication starts at approximately 12 h after PH and normally reaches a peak at 24 h. In rats, however, there is a second peak at 36-66 h. In this study, the 10 time-points during LR could be allocated to 3 phases, viz., 0-6 h (early phase), in which hepatocytes are activated and G0/G1 transition occurs, 12-72 h (intermediate phase) when cell proliferation takes place, and 120-168 h (late phase) when regeneration terminates. There was considerable variation in different genes at the time points, as to initial expression and expression persistence during the whole process. As a result, the numbers of initially up and down-regulated DNA repair-related genes were 19 and 4, respectively, in the early phase, 49 and 8 in the intermediate, and 1 and 1 in the late. Total expression frequencies of up- and down-regulated genes in the three phases, were 26 and 5, 159 and 15, 41 and 8, respectively (Figure 1), thereby illustrating that DNA repair-related genes, mainly induced during the early and intermediate stages, played important roles in the different stages.
To facilitate the visualization and interpretation of gene expression profiles, 69 DNA repair genes, with 2-fold-plus variation in intensity, at least at one time-point after PH, were hierarchically clustered, according to expression similarities (Figure 2A). The result, in compact graphical format, showed their arrangement into three groups (Figure. 2B). Cluster C1 contained 22 genes which were rapidly up-regulated, 2-30 h after PH and persisted so, whereas the expression of the 38 genes in cluster C2, gradually increased at 24-72 h and then returned to normal, and the 9 genes in Cluster C3, were rapidly down-regulated at 6-12 h and continued to be so. Furthermore, more than half of the DNA repair genes in cluster C2 were up-regulated, mainly at 24-72 h after PH.
Four base excision repair-related genes were mainly present in the C1 cluster, this including mbd4, tdg, and apex1. However, Gombar et al. (1981) found that the specific enzyme activities of UNG reached maximal levels between 18-24 h after PH, and then rebounded by 48 h. Furthermore, Riis et al. (2002) found that ogg1 expression increased 5-fold by 24 h after PH. In the present study, the mRNA levels of UNG and OGG1 were not found to be significantly enhanced after PH, although the expression of two other glycosylases, MBD4 and TDG, was dramatically so.
The genes related to HR, NHEJ, DNA cross-link repair, DNA polymerases and related accessory factors, as well as editing and processing nucleases, were mainly contained in cluster C2. Amongst those associated with HR, brca1, mus81, blm, mre11a and brca2 were enhanced, with two expression peaks, one between 24-30 h and the other 36-72 h. Since most DNA synthesis in hepatocytes occurs between 12-24 h, with non-parenchyma cells proliferating later (Koniaris et al., 2003; Khan and Mudan, 2007), it was supposed they may play a key role in DNA replication and repair during LR. In this study, the genes involved in HR were all extremely enhanced, with 34-fold peak expression above control at 24 h, thus quite consistent with the results of Thyagarajan et al., (1996) that homologous DNA recombination activity in regenerating liver closely mirrors the first wave of DNA synthesis, reaching a peak 24h after regenerative stimulus. The genes specifically associated with NHEJ, DNA cross-link repair, editing and processing nucleases, and DNA polymerases and related accessory factors which operate in distinct DNA repair pathways or bypass specific classes of adducts in DNA (Wood et al., 2005), were enhanced between 24-36 h with one peak at 24 h after PH. The above results give us to understand that expression of most DNA repair genes is closely related to progression through the cell division cycle during liver regeneration.
The genes involved in both mismatch repair and nucleotide excision repair (NER), and DNA damage response, were distributed among all the three clusters, with most in cluster C1. Among the DNA lesion recognition genes of interest in NER, cetn2 and xpc, both involved in global genome repair (GGR), were down-regulated between 6-24 h and 36-72 h, respectively, whereas ercc8, hmgn1 and polr2g,, involved in transcription coupled repair (TCR), were up-regulated after PH, and GTF2H1, GTF2H2, and GTF2H3, the three main subunits of the general transcription factor TFIIH (Tian et al., 2004), were simultaneously enhanced at the mRNA level, after 12 h. Due to cell proliferation triggered by PH, there was an increase in RNA polymerase II-dependent transcription (Dong and Xu, 2008), and the above-mentioned NER factors or TFIIH were more essential to transcription than to their normal roles in DNA repair. Thus, during LR, their expression changes could lead to changes in transcriptional activity rather than in DNA repair. The true relationship between NER factors or TFIIH and DNA repair activity requires further study. As a component of the trimeric Cdk7-cyclin H-Mat1 complex, which functions as a cyclin-dependent kinase-activating kinase (Rossi et al., 2001), cdk7 was up-regulated at 6 h, while the expression of cyclin h did not change significantly, as alike in a previous report (Albrecht et al., 1999). LIG1, which catalyzes DNA joining in the final step of NER (Gariboldi et al., 1995), was expressed in the liver undergoing active cell proliferation. In this study, the expression of lig1 was increased at 24-36 h, thus correlated with enhanced cell proliferation activity in this phase.
The protein kinases ATM and ATR are emerging as core sensors of DNA damage, capable of activating the downstream effector kinases CHK2/CHK1, as well as many other protein factors, through phosphorylation. The efficient transduction of DNA damage signals initiated by ATM/ATR, is not only CHK2/CHK1-dependent, but also requires a class of checkpoint mediators (Liu et al., 2006). In the ATM signalling pathway, MDC1 assists other mediators in accumulating at sites of damaged DNA (Lukas et al., 2004), whereas in the ATR, the RAD17-RFC2-5 and HUS1-RAD9-RAD1 complexes are possibly capable of recognizing and binding to DNA damage sites in substitution of RFC and PCNA (Ellison and Stillman, 2001). TELO2, essential for the mammalian S-phase checkpoint, impacts on CHK1 stability (Collis et al., 2007), and p53, one of the targets of ATM, ATR and Chk1/Chk2, contributes to G1/s arrest (Canman et al., 1998; Yang et al., 2004). GADD45, dependent on p53, also participates in activating G2/m checkpoints, DNA repair and apoptosis (Vairapandi et al., 2002). The interactions among these factors, and their expression changes during LR, are shown in Figure 3. The general view was that abundance of ATM proteins was invariable in the different cell-cycle phases. However, Lu et al. (2005) have shown that their expression levels, besides being increased, were correlated with the onset of DNA replication during LR. Nonetheless, in the present study, there appeared to be no significant change, suggesting that their role in LR may be played through their downstream targets. Among the genes of interest shown in Figure 3, most of the other mediators and effectors, apart from weakly down-regulated hus1 and telo2, were dramatically enhanced between 6-36 h. Furthermore, gadd45a and gadd45g in cluster C1, which immediately reached their peak levels at 2 h, were up-regulated almost throughout LR, this indicating their possibly significant role in signal transduction of DNA damage during the process.
In conclusion, the expression of BER- and TCR-related genes was enhanced at the transcriptional level in the early and intermediate phases of LR, whereas the expression of genes related to HR, NHEJ and DNA cross-link repair, as well as DNA polymerases and related accessory factors, and editing or processing nucleases, were mainly enhanced in the intermediate phase. Furthermore, gene expression in response to DNA damage was rather complicated throughout the whole LR process. It was also proposed that the expression of most DNA repair genes may be regulated by, or play a role in, progression through the cell division cycle during LR. However, the whole process (genem → RNA → protein) is influenced by many factors, including protein interactions. More important, it remains to be demonstrated that these changes at mRNA levels result in changes both at protein levels and in DNA repair capacity. Therefore, western blot, protein chip and RNA interference assays are required for further analysis of DNA repair genes and their regulation in regenerating liver.
Acknowledgments
Financial support by the National Basic Research 973 Pre-research Program of China, No. 2010CB534905, is acknowledged.
Supplementary Material
The following online material is available for this article:
Table S1 - Expression abundance of 69 DNA repair genes during rat liver regeneration.
Table S2 - Primer sequences used in real-time quantitative RT-PCR.
Figure S1 - Comparison of relative mRNA levels in regenerating liver detected by real-time RT-PCR and Affymetrix Rat Genome 230 2.0 microarray.
This material is made available as part of the online article from http://www.scielo.br.gmb.
Received: August 14, 2010; Accepted: December 14, 2010.
Associate Editor: Carlos F.M. Menck
License information: This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
- Albrecht JH, Rieland BM, Nelsen CJ and Ahonen CL (1999) Regulation of G(1) cyclin-dependent kinases in the liver: role of nuclear localization and p27 sequestration. Am J Physiol 277:G1207-G1216.
- Arai T, Kelly VP, Komoro K, Minowa O, Noda T and Nishimura S (2003) Cell proliferation in liver of Mmh/Ogg1-deficient mice enhances mutation frequency because of the presence of 8-hydroxyguanine in DNA. Cancer Res 63:4287-4292.
- Arias-Lopez C, Lazaro-Trueba I, Kerr P, Lord CJ, Dexter T, Iravani M, Ashworth A and Silva A (2006) p53 modulates homologous recombination by transcriptional regulation of the RAD51 gene. EMBO Rep 7:219-224.
- Birger Y, West KL, Postnikov YV, Lim JH, Furusawa T, Wagner JP, Laufer CS, Kraemer KH and Bustin M (2003) Chromosomal protein HMGN1 enhances the rate of DNA repair in chromatin. EMBO J 22:1665-1675.
- Canman CE, Lim DS, Cimprich KA, Taya Y, Tamai K, Sakaguchi K, Appella E, Kastan MB and Siliciano JD (1998) Activation of the ATM kinase by ionizing radiation and phosphorylation of p53. Science 281:1677-1679.
- Collis SJ, Barber LJ, Clark AJ, Martin JS, Ward JD and Boulton SJ (2007) HCLK2 is essential for the mammalian S-phase checkpoint and impacts on Chk1 stability. Nat Cell Biol 9:391-401.
- Dong HM and Xu CS (2008) Analysis of the relevance of e2fs and their target genes with rat liver regeneration. Ind J Gastroenterol 27:31-32.
- Ellison V and Stillman B (2001) Opening of the clamp: An intimate view of an ATP-driven biological machine. Cell 106:655-660.
- Fausto N, Campbell JS and Riehle KJ (2006) Liver regeneration. Hepatology 43:S45-S53.
- Gariboldi M, Montecucco A, Columbano A, Ledda-Columbano GM, Savini E, Manenti G, Pierotti MA and Dragani TA (1995) Genetic mapping and expression analysis of the murine DNA ligase I gene. Mol Carcinog 14:71-74.
- Gombar CT, Katz EJ, Magee PN and Sirover MA (1981) Induction of the DNA repair enzymes uracil DNA glycosylase and 3-methyladenine DNA glycosylase in regenerating rat liver. Carcinogenesis 2:595-599.
- Guo GB and Xu CS (2008) Expression profiles of the organic acid metabolism-associated genes during rat liver regeneration. Amino Acids 34:597-604.
- Higgins GM and Anderson RM (1931) Experimental pathology of the liver: Restoration of the liver of the white rat following partial surgical removal. Arch Pathol Lab Med:186-202.
- Jass JR (2006) Hereditary Non-Polyposis Colorectal Cancer: The rise and fall of a confusing term. World J Gastroenterol 12:4943-4950.
- Kaufmann WK, Rice JM, MacKenzie SA, Smith GJ, Wenk ML, Devor D, Qaqish BF and Kaufman DG (1991) Proliferation of carcinogen-damaged hepatocytes during cell-cycle-dependent initiation of hepatocarcinogenesis in the rat. Carcinogenesis 12:1587-1593.
- Khan AZ and Mudan SS (2007) Liver regeneration: mechanisms, mysteries and more. ANZ J Surg 77:9-14.
- Koniaris LG, McKillop IH, Schwartz SI and Zimmers TA (2003) Liver regeneration. J Am Coll Surg 197:634-659.
- Lai HS, Chen Y, Lin WH, Chen CN, Wu HC, Chang CJ, Lee PH, Chang KJ and Chen WJ (2005) Quantitative gene expression analysis by cDNA microarray during liver regeneration after partial hepatectomy in rats. Surg Today 35:396-403.
- Li X, Stith CM, Burgers PM and Heyer WD (2009) PCNA is required for initiation of recombination-associated DNA synthesis by DNA polymerase delta. Mol Cell 36:704-713.
- Liu WF, Yu SS, Chen GJ and Li YZ (2006) DNA damage checkpoint, damage repair, and genome stability. Yi Chuan Xue Bao 33:381-390.
- Lu S, Shen KC, Wang Y, Brooks SC and Wang YA (2005) Impaired hepatocyte survival and liver regeneration in Atm-deficient mice. Hum Mol Genet 14:3019-3025.
- Lukas C, Melander F, Stucki M, Falck J, Bekker-Jensen S, Goldberg M, Lerenthal Y, Jackson SP, Bartek J and Lukas JU (2004) Mdc1 couples DNA double-strand break recognition by Nbs1 with its H2AX-dependent chromatin retention. EMBO J 23:2674-2683.
- Pegg AE, Dolan ME and Moschel RC (1995) Structure, function, and inhibition of O6-alkylguanine-DNA alkyltransferase. Prog Nucleic Acid Res Mol Biol 51:167-223.
- Riis B, Risom L, Loft S and Poulsen HE (2002) Increased rOGG1 expression in regenerating rat liver tissue without a corresponding increase in incision activity. DNA Repair 1:419-424.
- Rossi DJ, Londesborough A, Korsisaari N, Pihlak A, Lehtonen E, Henkemeyer M and Makela TP (2001) Inability to enter S phase and defective RNA polymerase II CTD phosphorylation in mice lacking Mat1. EMBO J 20:2844-2856.
- Schmucker DL (2005) Age-related changes in liver structure and function: Implications for disease? Exp Gerontol 40:650-659.
- Shao HY, Zhao LF and Xu CS (2007) Expression patterns and action analysis of genes associated with inflammatory responses during rat liver regeneration. World J Gastroenterol 13:369-377.
- Suzuki T and Tsukamoto I (2004) Apoptosis induced by 5-(N,N-hexamethylene)-amiloride in regenerating liver after partial hepatectomy. Eur J Pharmacol 503:1-7.
- Taub R (2004) Liver regeneration: From myth to mechanism. Nat Rev Mol Cell Biol 5:836-847.
- Thyagarajan B, Cruise JL and Campbell C (1996) Elevated levels of homologous DNA recombination activity in the regenerating rat liver. Somat Cell Mol Genet 22:31-39.
- Tian M, Jones DA, Smith M, Shinkura R and Alt FW (2004) Deficiency in the nuclease activity of xeroderma pigmentosum G in mice leads to hypersensitivity to UV irradiation. Mol Cell Biol 24:2237-2242.
- Vairapandi M, Balliet AG, Hoffman B and Liebermann DA (2002) GADD45b and GADD45g are cdc2/cyclinB1 kinase inhibitors with a role in S and G2/M cell cycle checkpoints induced by genotoxic stress. J Cell Physiol 192:327-338.
- Vielhauer V, Sarafoff M, Gais P and Rabes HM (2001) Cell type-specific induction of O6-alkylguanine-DNA alkyltransferase mRNA expression in rat liver during regeneration, inflammation and preneoplasia. J Cancer Res Clin Oncol 127:591-602.
- Wang GP and Xu CS (2010) Reference gene selection for real-time RT-PCR in eight kinds of rat regenerating hepatic cells. Mol Biotechnol 46:49-57.
- Wood RD, Mitchell M, Sgouros J and Lindahl T (2001) Human DNA repair genes. Science 291:1284-1289.
- Wood RD, Mitchell M and Lindahl T (2005) Human DNA repair genes, 2005. Mutat Res 577:275-283.
- Yang J, Xu ZP, Huang Y, Hamrick HE, Duerksen-Hughes PJ and Yu YN (2004) ATM and ATR: Sensing DNA damage. World J Gastroenterol 10:155-160.
- Zhang LX, Zhao LF, Zhang AS, Chen XG and Xu CS (2006) Expression patterns and action analysis of genes associated with physiological responses during rat liver regeneration: cellular immune response. World J Gastroenterol 12:75147521.
Publication Dates
-
Publication in this collection
08 Apr 2011 -
Date of issue
2011
History
-
Accepted
14 Dec 2010 -
Received
14 Aug 2010