Abstract
Adaptation or acclimation to hypoxia occurs via the modulation of physiologically relevant genes, such as erythropoietin, transferrin, vascular endothelial growth factor, phosphofructokinase and lactate dehydrogenase A. In the present study, we have cloned, sequenced and examined the modulation of the LDH-A gene after an Amazonian fish species, Astronotus crassipinis (the Oscar), was exposed to hypoxia and anoxia. In earlier studies, we have discovered that adults of this species are extremely tolerant to hypoxia and anoxia, while the juveniles are less tolerant. Exposure of juveniles to acute hypoxia and anoxia resulted in increased LDH-A gene expression in skeletal and cardiac muscles. When exposed to graded hypoxia juveniles show decreased LDH-A expression. In adults, the levels of LDH-A mRNA did not increase in hypoxic or anoxic conditions. Our results demonstrate that, when given time for acclimation, fish at different life-stages are able to respond differently to survive hypoxic episodes.
Amazon; fishes; gene expression; RT-PCR; LDH-A; hypoxia; anoxia
RESEARCH ARTICLE
Anoxia- and hypoxia-induced expression of LDH-A* in the Amazon Oscar, Astronotus crassipinis
Vera Maria Fonseca Almeida-ValI; Alice Reis OliveiraI; Maria de Nazaré Paula da SilvaI; Monica Stropa Ferreira-NozawaII; Roziete Mendes AraújoI; Adalberto Luis ValI; Sérgio Ricardo NozawaI, II
ILaboratório de Ecofisiologia e Evolução Molecular, Instituto Nacional de Pesquisas da Amazônia, Manaus, AM, Brazil
IILaboratório de Expressão Gênica, Centro Universitário Nilton Lins, Manaus, AM, Brazil
Send correspondence to Send correspondence to: Sergio Ricardo Nozawa Laboratório de Ecofisiologia e Evolução Molecular Instituto Nacional de Pesquisas da Amazônia Av. André Araújo 2936 69060-001 Manaus, AM, Brazil E-mail: srnozawa@gmail.com
ABSTRACT
Adaptation or acclimation to hypoxia occurs via the modulation of physiologically relevant genes, such as erythropoietin, transferrin, vascular endothelial growth factor, phosphofructokinase and lactate dehydrogenase A. In the present study, we have cloned, sequenced and examined the modulation of the LDH-A gene after an Amazonian fish species, Astronotus crassipinis (the Oscar), was exposed to hypoxia and anoxia. In earlier studies, we have discovered that adults of this species are extremely tolerant to hypoxia and anoxia, while the juveniles are less tolerant. Exposure of juveniles to acute hypoxia and anoxia resulted in increased LDH-A gene expression in skeletal and cardiac muscles. When exposed to graded hypoxia juveniles show decreased LDH-A expression. In adults, the levels of LDH-A mRNA did not increase in hypoxic or anoxic conditions. Our results demonstrate that, when given time for acclimation, fish at different life-stages are able to respond differently to survive hypoxic episodes.
Key words: Amazon, fishes, gene expression, RT-PCR, LDH-A, hypoxia, anoxia.
Introduction
Lactate dehydrogenase (LDH, E.C. 1.1.1.37) is a key enzyme in the control of energy metabolism, catalyzing the interconversion of pyruvate to lactate and regulating the levels of these metabolites in accordance with oxygen availability. In vertebrates, the active form of the enzyme is a tetramer composed of four polypeptide subunits encoded by two genes: LDH-A and LDH-B. Under anaerobic conditions, the isoform LDH-A4 (isozyme A4) preferentially converts pyruvate to lactate. This isoform is found predominantly in poorly vascularized tissues with low partial pressure of oxygen (pO2), such as skeletal muscle. On the other hand, the isoform LDH-B4 (isozyme B4) is more active in aerobic conditions, converting lactate to pyruvate in well-oxygenated tissues, such as cardiac muscle. A third gene, LDH-C, is present in vertebrates, and its expression is restricted to a few tissues, such as primary spermatocytes in mammals or in the brain and retina of advanced teleosts (Markert, 1984).
The isoform (or isozyme) systems, especially LDH, have been the subject of several studies in Amazon fishes (reviewed by Almeida-Val et al., 1995. 1999a,b, 2000). Based upon the data from more than 50 species, two different models for LDH distribution in Amazon fish species have been proposed: (i) predominance of isozyme B4 in the heart, indicating the maintenance of low rates of aerobic metabolism during episodes of hypoxia; and (ii) low expression of the LDH-B gene combined with a strong expression of the LDH-A gene in all tissues, suggesting activation of anaerobic metabolism during hypoxia (reviewed by Almeida-Val and Val, 1993). These models are mutually exclusive, and do not occur simultaneously in the analyzed groups. Similar distribution, i.e., reduction in LDH-B gene expression in the heart, was first observed in wild flatfish by Markert and Holmes (1969) and in stickleback by Rooney and Ferguson (1985) and was explained as a strategy to increase hypoxia tolerance.
Previous studies have shown that LDH tissue distribution in Amazon cichlids, including Astronotus, is related to their ability to tolerate hypoxic environments and to express some degree of phenotypic plasticity in the heart, thereby revealing their choice of habitats (Almeida-Val et al., 1995). One good example is the regulation of LDH distribution in the heart of Cichlassoma amazonarum. Depending on the availability of oxygen in their habitat, these animals' hearts exhibit the predominance of one of two isoforms: B4 or A4. When exposed to severe hypoxia (± 30 mmHg or ± 4 kPa) for 51 days, LDH-A4 expression increases in the heart and brain, while isozyme B4 increases in the liver and no longer appears in skeletal muscle (see Almeida-Val et al., 1995, for details). The most significant changes, however, were detected in the brain, which adopted muscle-type kinetics due to the new LDH distribution (Almeida-Val et al., 1995; Val et al., 1998).
The plasticity in regulating the expression of LDH-A and LDH-B in Amazon cichlids indicates the ability of these animals to base their metabolism on anaerobic glycolysis through the increase of LDH-A expression when oxygen availability is low. Therefore, such plasticity allows these animals to visit environments with low oxygen concentrations for feeding, breeding, etc. Cichlids are territorial fish with very aggressive behavior and strong parental care (Chellapa et al., 1999). Consequently, burst swimming is a common trait in their lives that depends upon anaerobic metabolism. The relative amount of red muscle fibers in Cichlids is reduced compared to other teleosts (Almeida-Val et al., 2006), and so are their heart somatic indexes (unpublished data Almeida-Val). Both characteristics support the idea that anaerobic glycolysis takes place during periods of limited oxygen availability and that heart work is sustained for short periods of time at the expense of glucose. The LDH-A4 isozyme will be more useful than LDH-B4 in such situations, since lactate accumulation can be avoided by a quick washout from the relatively small heart (unpublished results Maciel).
Oxygen concentrations in water bodies of the Amazon vary spatially, diurnally, and seasonally (Almeida-Val et al., 1999b). Due to low water oxygen capacitance, respiration of aquatic organisms and organic material decomposition, oxygen tension can reach very low levels, especially during the night when no photosynthesis occurs. This characteristic of Amazon aquatic ecosystems has been important for the appearance of several adaptive characteristics in the evolution of the Amazonian fishes, particularly some cichlid species (Almeida-Val et al., 1999a). The cichlid Astronotus ocellatus is extremely tolerant to low oxygen concentrations and anoxia, as is its sibling species Astronotus crassipinis (Muusze et al., 1998; Almeida-Val et al., 2000; Chippari-Gomes et al., 2005). Increased hypoxia tolerance was found in growing Astronotus ocellatus. Based on enzyme measurements, including LDH, we have described a size-dependent hypoxia tolerance and anaerobic/aerobic ratio in this species (Almeida-Val et al., 1999b). Both species (Astronotus ocellatus and A. crassipinis) show a decrease in metabolic rate when exposed to hypoxia (Almeida-Val et al., 2000; Chippari-Gomes et al., 2005). Based on experiments in the laboratory we also showed that these species increase their tolerance to hypoxia if dissolved oxygen is gradually decreased, compared to acute hypoxia exposure, when the animal is suddenly transferred from normoxic to hypoxic environments.
Adaptation to hypoxia occurs through the modulation of physiologically relevant genes. According to the literature (reviewed by Hochachka, 1997), low O2 concentrations induce the stabilization of the hypoxia-inducible factor (HIF-1), which induces the transcription of hypoxia-inducible genes like erythropoietin, transferrin, vascular endothelial growth factor, phosphofructokinase and lactate dehydrogenase A, among many others (Wenger, 2000).
Considering the different responses to hypoxia between juvenile and adult Amazonian Oscar, and considering that graded and acute hypoxia induce different acclimation responses in these species, the present work was planned to study the contributions of LDH regulation to these adjustments, particularly LDH-A, which is one of the genes induced by HIF-1 under hypoxia. Thus, we have cloned, sequenced and tested the modulation of LDH-A gene expression during exposure of the juvenile and adult Amazonian cichlid Astronotus crassipinis to acute and graded hypoxia and anoxia.
Material and Methods
Experimental animals and exposure to hypoxia
Different-sized specimens of Astronotus crassipinis were purchased from a fish farm (Amazonfish) located at 230 km from Manaus, on the highway AM-010 that links the cities of Manaus (59°59'14" N, 3°5'54" S) and Itacoatiara, and used for acute hypoxia experiments. Additionally, specimens (most of them larger adults) were collected from Catalão Lake (59°54'29" N, 3°9'47" S) at the Negro River, near Manaus, Amazon, Brazil, and used for graded hypoxia experiments. All animals were first acclimated to the laboratory in aerated water in 500-L indoor tanks at room temperature (26.0 ± 2.0 °C) and normoxic water for 30 days at the Laboratory of Ecophysiology and Molecular Evolution, INPA, Manaus. The animals were fed ad libitum fish pellets every other day. The animals were exposed to two experimental conditions: (i) acute hypoxia and anoxia; and (ii) graded hypoxia and anoxia.
Acute hypoxia and anoxia exposure
For this experiment, two groups of animals were investigated: juveniles weighing 38.25 ± 6.71 g and measuring 10.33 ± 0.73 cm of fork length, and adults weighing 148.00 ± 44.25 g and measuring 16.10 ± 1.55 of fork length.
Five Astronotus crassipinis specimens of each life stage (juveniles and adults) were individually transferred to a respirometer chamber (flow cells) and exposed to hypoxia and anoxia. Fish were starved for 48 h before being placed in the flow cells of the respirometer, where they were kept in air-saturated water (~17 kPa). One group of animals was exposed to each of the following situations: normoxia, hypoxia, and anoxia.
To expose the fish to acute hypoxia, the dissolved oxygen in the water (pO2) was reduced quickly to 8.5 kPa (5 animals), 4.3 kPa (5 animals) and 0 kPa (5 animals) air saturation by reducing the aeration of the cell water over 1 h and 15 min. One group of animals was exposed to anoxia for 0 min, one for 30 min and one for 2 h. When the desired water pO2 or time under anoxia was reached, fish were bled and sacrificed with a sharp blow to their heads. Then, tissues were excised, immediately homogenized and pooled for RNA extraction. Samples from animals exposed to air-saturation water were used as a control group (normoxia). An Oxymeter YSI model 85 was used to monitor dissolved oxygen in water during the experiments.
In a second experiment mRNA from 18 muscle samples of juveniles were extracted individually and amplified with specific LDH-A primers. This experiment was planned to check homogeneity of responses among individual fishes, and only two experimental situations were tested: hypoxia (pO2 8.5 kPa), and anoxia (pO2 0.0 kPa) during one hour. The control group was exposed to normoxia (pO2 16.9 kPa) (see Figure S3 for details).
Graded hypoxia and anoxia exposure
For this experiment, juveniles of Astronotus crassipinis weighing between 43 and 67 g were acquired from a local hatchery or collected in the field and transported to the Laboratory of Ecophysiology and Molecular Evolution (INPA) where they were held indoors in 500-L fiberglass static holding tanks for at least 20 days prior to the experiments (35-45 fish/tank). Water temperature and dissolved oxygen were monitored regularly and were 29 ± 0.5 °C and 16 kPa, respectively, and 12L:12D cycle. Tanks were also equipped with glass wool filters to remove particulate material from the water. 50% of the water was replaced every other day. Fish were fed commercial dry food pellets ad libitum daily.
To assess the tolerance to graded hypoxia and to evaluate LDH-A gene expression, 70 specimens of A. crassipinis were individually placed into a 15-L metabolic chamber (flow cell) without access to the surface. The chamber was fitted with water inlet and outlet ports. Each chamber was immersed in a 100-L polystyrene box filled with water to maintain temperature and oxygen partial pressure levels. Water was re-circulated from the outer polystyrene chamber through the metabolic chamber with a submersible pump, providing a closed flow-through system in order to control the chamber and surrounding water properties (see Chippari-Gomes et al., 2005, for details). Fish were acclimated in normoxia inside the metabolic chamber (~16 kPa) for 12 h prior to experimentation. During any of the experiments, the oxygen concentration was slowly decreased to 8.8 kPa, 4.3 kPa, 2.2 kPa, 1.7 kPa, 1.0 kPa, and 0.2 kPa (n = 10 for each oxygen level). Final oxygen levels were obtained by bubbling N2 into the polystyrene container (20% decrease per hour), and the temperature was kept constant at 29 °C ± 0.5 °C. Fish were held at each oxygen level for 8 h, and the loss of equilibrium indicated the lowest survivable oxygen level. Dissolved oxygen levels were continuously monitored using an oxygen meter, YSI model 85. Again, when the desired water pO2 or time under anoxia was reached, fish were bled and sacrificed with a sharp blow to their heads. Then, tissues were excised, immediately homogenized and pooled for RNA extraction.
Design of PCR primers, cloning and sequencing LDH-A gene
Total RNA was extracted from white muscle with Trizol (Invitrogen, Life Technologies) according to the manufacturer's instructions. Genomic DNA was eliminated from the samples using DNase according to the manufacturer's instructions (Invitrogen, Life Technologies). The quality of the RNA was assessed with a UV/Vis Spectrophotometer, with a 260/280 nm absorbance ratio of 1.8-2.0. Agarose gel 1% (w/v) electrophoresis with ethidium bromide staining was performed to verify the integrity of the rRNAs. Both methods were used to check RNA purity.
LDH-A sequences were obtained from GenBank (NCBI) and were aligned using Bioedit bioinformatic tools (Hall, 1999). The degenerate primers used were: LDH-A13 (5'- AAI TTA AAG GGC GAG GCI ATG G - 3') and LDH-A14 (5'- TTI GGT TGA GGI TTT GAA GAG - 3'), which correspond to a conserved region of all the LDH-A genes. Amplified PCR products of all actual cDNAs were ligated into pGEM-T easy vector (Promega) and amplified in DH5'-alpha E. coli competent cells, after which the plasmids were extracted. Plasmids containing a LDH-A gene insert were sequenced to ensure that the correct mRNA sequences were semi-quantified. The fragments were sequenced with BigDye version 3.1 fluorescent dye (Applied Biosystems) and run on an ABI 310 automatic DNA sequencer (Applied Biosystems). The obtained sequences were analyzed using a BLAST (Megablast) program. The obtained sequence for LDH-A (A. crassipinis - GenBank accession no. 154940275) was used as a probe in a northern blot.
Northern blot analysis
Total RNA (10-15 μg) was fractionated by agarose gel electrophoresis (1.2% (w/v), 2.2 M formaldehyde)/, transferred to Gene Screen Plus filters (New England Nuclear) using 10x SSC (1.5 M NaCl, 0.15 M sodium citrate) and hybridized to Astronotus crassipinis LDH-A and human rRNA gene probes. The obtained fragment with the above-mentioned primer was used as a probe and 32P-labelled by the random priming method with the Random Primer DNA labeling kit (Life Technologies). Hybridization to 32P-labelled LDH-A clones was performed at 42 °C for 18 h and the final wash was performed in 0.1x SSC and 0.2% SDS at 55 °C. Quantification of the hybridized RNA was performed using a densitometer (GS-700 model, BIORAD) and human 45S precursor rRNA was used to normalize the amount of RNA loaded in each sample.
To amplify a 480 bp DNA fragment from A. cassiprinis, we used degenerate primers based on the sequence of known LDH-A genes from a variety of fishes. Cloning and DNA sequence analysis revealed that the amplified fragment encoded a highly conserved nucleotide sequence that was characteristic of LDH-A genes (Figure S1). BLAST results revealed an E-value of e = 0.0 with Oreochromis niloticus (GenBank accession no. EU313200.1) and an E-value of 2e-175 with Chromis xanthochira (GenBank accession no. AY289562.1). Analysis of the deduced amino acid sequence (Supplementary material, Figure S2) in the NCBI databank (CDART: Conserved Domain Architecture Retrieval Tool) showed an E-value of 2e-63 for the encoded protein, suggesting that this gene is a member of the family of NAD-dependent 2-hydroxycarboxylate dehydrogenases.
Statistical analysis
The results obtained through RT-PCR were scanned and relative concentration of each band was measured and presented as means ± SEM (means and standard error of the means) in bar graphs as shown in Figure S3. One-way ANOVA was used for comparison of results (normoxia, hypoxia and anoxia). Different letters mean significant differences among them at 5% level.
Results
Acute hypoxia exposure
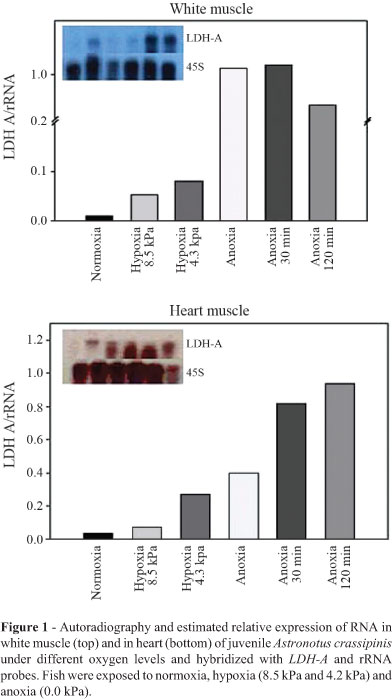
LDH-A mRNA expression was detected in skeletal and cardiac muscle in all studied groups. No expression of LDH-A mRNA was found in liver. LDH-A mRNA levels increased in white and heart muscles of juveniles exposed to acute hypoxia (Figure 1). A 10-fold increase in LDH-A expression was observed in the white muscle after 30 min of anoxia followed by a decrease after two hours. A threefold increase in LDH-A transcription was observed in cardiac muscle after deep hypoxia exposure (4.3 kPa). These higher levels of expression were retained for two hours in anoxic conditions, reaching a 10-fold increase at this time (Figure 1). No relationship between hypoxia and increasing levels of LDH-A mRNA expression was observed in the adults, which expressed high LDH-A levels in normoxia, keeping the same LDH-A expression in anoxic animals (Figure 2).
The semi-quantitative RT-PCR method was used with LDH-A specific primers for A.crassipinis and showed an increase in gene expression after anoxia (Figure S3), corroborating the results of pooled samples at the individual level.
Graded hypoxia exposure
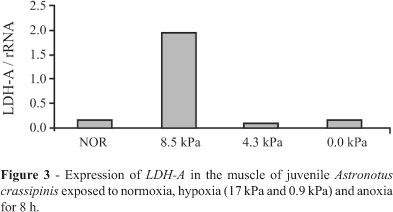
When juveniles were steadily exposed to hypoxia or anoxia and kept in this condition for eight hours, LDH-A expression increased in the skeletal muscle of the group submitted to 1.7 kPa pO2. However, animals exposed to 0.9 kPa and 0.0 kPa pO2 showed a decrease in LDH-A message levels, that were similar to normoxic levels, indicating metabolic suppression (Figure 3). We observed no loss of equilibrium (behavior that precedes death) during the whole experiment (see details in Chippari-Gomes et al., 2005).
Discussion
The fish group has successfully radiated and is one of the most numerous groups among vertebrates, inhabiting all kinds of aquatic environments (Nelson, 2006). The evolution of the fish group occurred during the last 500 million years, under low and unstable atmospheric O2 (Webster, 2003). Hypoxia-responsive elements date from 0.6 to 0.4 billion years ago. Aquatic hypoxia was, probably, a strong regulator of gene expression and, as a consequence, of anaerobic metabolism (D'Angio and Finkelstein, 2000, Prabhakar, 2001; Semenza, 2001).
Fishes acquired the first coordinated hypoxia regulation system during the Silurian. The appearance of HIF-1α (hypoxia induction factor) coincides with the development of multicellular systems and metabolic compartmentalization (Webster, 2003). LDH-A is one of the glycolytic enzymes with a HIF-1α site. The HIF-1α pathway is activated at oxygen saturation lower than 5% (1 kPa), and although pO2 in the atmosphere is around 20 kPa, the normal pO2 in most tissues ranges between 6.7 and 9.3 kPa (Iyer et al., 1998; Hochachka, 1999; Semenza, 2001).
The sequence found for A. crassipinis LDH-A gene in the present work is extremely conservative, with very little difference from other fish species, particularly species belonging to order Perciformes. The deduced amino acid sequence for the protein suggests that this gene is a member of the NAD-dependent 2-hydroxycarboxylate dehydrogenase family, of which LDH's enzyme class is a member. Further studies of LDH-A gene sequences in fish species belonging to this family will be necessary to confirm the conservative character of LDH-A.
Studies carried out in our laboratory indicate that LDH enzyme activities and hypoxia survivorship are directly related to Astronotus ocellatus size (Almeida-Val et al., 2000). This increased ability to survive severe hypoxia in fishes with bigger sizes can be explained as an increase in their anaerobic power (represented by LDH absolute activities in their tissues and organs) rather than a decrease in specific metabolic rates (Almeida-Val et al., 1999b, 2000). Furthermore, cichlid species of the genus Astronotus show high tolerance to hypoxic and anoxic environments (Muusze et al., 1998; Chippari-Gomes et al., 2002). Both species of Astronotus (A. crassipinis and A. ocellatus) have similar responses to hypoxia and present scaling metabolism accordingly (Sloman et al. 2008). In fact, in the present study, the results showed an increase in the amount of mRNA for LDH-A in adult fishes compared to juveniles in normoxia (Figures 1 and 2).
As mentioned above, increased expression of LDH-A along with a simultaneous reduction in the expression of LDH-B occurs in the heart of several species of cichlids, and these characteristics are related to the degree of hypoxia tolerance of each species (Almeida-Val et al., 1995). The regulation of LDH-A in the heart is required to compensate for lactate accumulation during hypoxic and anoxic episodes. The presence of the LDH-A4 isoform in a highly aerobic tissue such as the heart was observed in hypoxia-tolerant species and represents the anaerobic "preference" of the cardiac muscle (Almeida-Val et al., 1993, 1995).
The current analysis of LDH-A mRNA expression in A. crassipinis exposed to experimental hypoxia revealed higher transcription rates of this gene in white and heart muscles of juveniles. We observed a maximum increase in LDH-A expression after 30 min of anoxia in their white muscle, although the induction was more effective in cardiac muscle. After exposure to 20% air saturation, we observed an increase in LDH-A transcription, and this higher level of expression was kept during the following 2 h of anoxia. The different induction patterns observed in these two tissues are possibly due to differences in the ability of these tissues to cope with hypoxia. While white muscle endures hypoxia throughout metabolic suppression, cardiac muscle maintains lower but constant ATP generation at the expenses of anaerobic glycolysis. With regard to both LDH tissue distribution and the ability to deal with hypoxia, these responses are also observed in mammalian cells (Firth et al., 1994).
In adults, however, hypoxia did not affect the expression of LDH-A mRNA. Therefore, we can infer that adult animals have developed other mechanisms of tolerance and do not depend exclusively on gene regulation to survive hypoxic episodes. Previous studies have shown that hypoxia tolerance in the sibling species A. ocellatus is directly related to the increase in size correlating to the increase in the routine metabolic rate, to a decrease in the mass-specific metabolic rate and to an increase in anaerobic power in its tissues (Almeida-Val et al., 1999b, 2000). Based upon experiments with the rainbow trout gonad epithelial cell line, a further decrease in oxygen levels (down to 0.2 kPa) results in reduced accumulation of HIF-1α. Therefore, it is possible that a similar process occurs in other fish cell systems and even in other species, given that these responses are very similar between mammalian and fish cells (Soitamo et al., 2001). As mentioned above, LDH-A, along with 100 other genes, is controlled by HIF-1α during hypoxia. Thus, we can conclude that the regulation of LDH-A gene expression in the present work is probably due to HIF-1α induction.
Prolonging anoxia exposure for 120 min induced both an increase and a decrease in LDH-A gene expression in the heart and muscle of juveniles, respectively. A decrease in LDH-A expression in the muscles of juveniles allowed fish to acclimate for more than three hours in anoxia. Also, the responses of the adults seem to reflect another physiological status of both heart and skeletal muscles. In an extensive review, Nikinmaa (2002) suggested that the possible oxygen tensions leading to the stabilization of HIF-1α protein are poorly studied, and that they may vary between species and cell types. If this is true, the stabilization or decrease in LDH-A expression can be explained as a result of this process. Furthermore, these properties also vary along animal development, as suggested by Gu et al. (2000), who studied the different responses of genes of the "Per Arnt Sim" (PAS) superfamily in animals of different sizes and their roles in detecting and adapting to environmental changes.
In contrast with acute exposure, graded hypoxia exposure (animals exposed for 8 h to 8.5 kPa air saturation) resulted in the induction of LDH-A expression. Further decreases in dissolved oxygen concentrations resulted in a decrease of LDH-A expression and can be explained as the result of a generalized metabolic suppression induced by the low levels of oxygen. Chippari-Gomes et al. (2005) measured the metabolic responses in the same experiment and described an increase in blood glucose and lactate in these animals, concomitant with a decrease in glycolytic function (showed by PK, LDH and MDH activities) in the muscle of acclimated animals. Muscle LDH enzyme activities dropped at low levels of hypoxia (approximately 5 kPa) and returned to control levels at higher hypoxic levels (1.7 kPa), accounting for the high levels of blood lactate detected. Due to LDH kinetic properties and its stability in the cell milieu, the first increase in mRNA levels can account for maintaining a continuous rate of anaerobic glycolysis in these animals, since this is a regular response in animals exposed to deep and prolonged hypoxia (Hochachka and Somero, 2002).
In vitro studies of hypoxia modulation of LDH expression using cultures of hepatocytes, myocytes and tumour cells as models have shown that the expression of LDH-A mRNA and/or protein is also induced by hypoxia (Robin et al., 1984; Webster, 1987; Marti et al., 1994; Firth et al,. 1994). As an example of in vivo study, the elegant experiment done by Gracey et al. (2001) has shown the profile of the hypoxia-induced genes by microarray analysis in the teleost fish Gillichthys mirabilis. Among the many genes identified, LDH-A was induced in liver and white muscle, but not in heart of that species. The induction of LDH-A in the heart observed in Astronotus crassipinis could be an indication of its greater tolerance to hypoxia.
Studies with Astronotus spp. in our laboratory have shown that hypoxia tolerance in juveniles is accounted for by activation of anaerobic metabolism (Muusze et al., 1998; Chippari-Gomes et al., 2005), reduction of ATP turnover, and suppression of the overall metabolism (Sloman et al., 2006; Richard et al., 2007) including decreases in protein synthesis (Lewis et al., 2007) and partial arrest of gill ion fluxes (Wood et al., 2007) to avoid passive ion leak across the gills. Respiratory responses to progressive hypoxia in Oscar showed that the oxygen transfer factor increases transiently in moderate hypoxia and decreases in deep hypoxia. Based on all these measurements, we concluded that the high tolerance of acidic anaerobic by-products is more important to survive hypoxic conditions than a superior oxygen supply capacity (Scott et al., 2008).
In conclusion, the results presented in this study demonstrate that gene transcription responds immediately after hypoxia exposure, and that the intensity of this response is tissue-specific and varies according to animal size, period of acclimation and degree of hypoxia. Post-transcriptional facts, which change enzyme activities in the cells, is certainly one of the regulatory mechanisms that occur in this species, since we could not find a direct relationship between gene expression (present study) and absolute enzyme activities (Chippari-Gomes et al., 2005). As already stated in the literature, the kinetics of vertebrate LDH can be modulated by substrate, enzyme, and co-factor concentration, as well as by temperature, pH and pressure (Hochachka et al., 2002). Our results confirm previous observations on scaling hypoxia tolerance and on the higher anaerobic capacity that adults of Astronotus crassipinis have, compared to their juveniles. We also show that the activation of anaerobic metabolism is regulated in different ways in different tissues according to the physiological characteristics of different-sized animals. As expected, hypoxia tolerance in Astronotus crassipinis is based, at least in part, on gene regulation that results from a very refined oxygen-sensing system developed and conserved throughout the evolution of vertebrates.
Acknowledgments
This work was funded by the National Research Council (CNPq) of Brazil (PNOPG to VMFAV- processes # 400030/11-33) and a CNPq/FAPEAM (Amazon State Research Foundation) PRONEX grant to ALV. ARO was the recipient of a PCI/INPA/CNPq research fellowship (#380521/00-8); ALV and VMFAV are the recipients of a Research fellowship from CNPq. The authors acknowledge Collin Brauner and Jeff Richards for useful comments on the first draft of this work. The results presented here are part of the data bank I of INCT - ADAPTA, funded by CNPq/MCT and FAPEAM.
Internet Resources
Chippari-Gomes AR, Val AL and Almeida-Val VMF (2002) Comparative responses of closely related cichlids to graded hypoxia. In: International Congress on the Biology of Fish, Vancouver. Responses of Fish to Aquatic Hypoxia, pp 9-13 (http://www-heb.pac.dfo-mpo.gc.ca/congress/2002/Hypoxia/Chippari-Gomes.pdf).
Supplementary Material
The following online material is available for this article:
- Figure S1 - DNA sequence: Alignment of LDH-A nucleotide sequences from Epinephelus coioides, Lycodichthys dearborni, Notothenia angustata, Trematomus bernacchii , Pagothenia borchgrevinki, Dissostichus eleginoides, Coryphopterus nicholsi, Gillichthys seta, Gillichthys mirabilis and Astronotus crassipinnis.
- Figure S2 - LDH-A sequence from A. cassiprinis used as a probe in Northern blotting experiments
- Figure S3 - RT-PCR expression of LDH-A in Astronotus crassipinis juveniles exposed to normoxia, hypoxia and anoxia.
Received: December 23, 2009; Accepted: August 24, 2010.
Associate Editor: Louis Bernard Klaczko
License information: This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
- Almeida-Val VMF and Val AL (1993) Evolutionary trends of LDH isozymes in fishes. Comp Biochem Physiol 105:21-28.
- Almeida-Val VMF, Val AL and Hochachka PW (1993) Hypoxia tolerance in Amazon fishes: Status of an under-explored biological "goldmine". In: Hochachka PW, Lutz PL, Sick T, Rosenthal M and Van den Thillart G (eds) Surviving Hypoxia: Mechanisms of Control and Adaptation. CRC Press, Boca Raton, pp 435-445.
- Almeida-Val VMF, Farias IP, Silva MNP and Duncan WP (1995) Biochemical adjustments to hypoxia in Amazon Cichlids. Braz J Med Biol Res 28:1257-1263.
- Almeida-Val VMF, Val AL and Walker I (1999a) Long- and short-term adaptation of amazon fishes to varying O2 levels: Intra-specific phenotypic plasticity and interspecific variation. In: Val AL and Almeida-Val VMF (eds) Biology of Tropical Fishes. Editora do INPA, Manaus, pp 185-206.
- Almeida-Val VMF, Paula-Silva MN, Duncan WP, Lopes NP and Val AL (1999b) Increase of anaerobic potential during growth of an Amazonian cichlid, Astronotus ocellatus Survivorship and LDH regulation after hypoxia exposure. In: Val AL and Almeida-Val VMF (eds) Biology of Tropical Fishes. Editora do INPA, Manaus, pp 437-448.
- Almeida-Val VMF, Val AL, Duncan WP, Souza FCA, Paula-Silva MN and Land S (2000) Scaling effects on hypoxia tolerance in the Amazon fish Astronotus ocellatus (Perciformes, Cichlidae): Contribution of tissue enzyme levels. Comp Biochem Physiol 125B:219-126.
- Almeida-Val VMF, Chipari-Gomes AR and Lopes NP (2006) Metabolic and physiological adjustments to low oxygen and high temperature in fishes of the Amazon. In: Val AL, Almeida-Val VMF and Randall DJ (eds) The Physiology of Tropical Fishes, v. 21. Series Fish Physiology. Elsevier, London, pp 443-500.
- Chellapa S, Yamamoto ME and Cacho MSRF (1999) Reproductive behavior and ecology of two species of cichlid fishes. In: Val AL and Almeida-Val VMF (eds) Biology of Tropical Fishes. Editora do INPA, Manaus, pp 113-126.
- Chippari-Gomes AR, Gomes LC, Lopes NP, Val AL and Almeida-Val VMF (2005) Metabolic adjustments in two Amazonian cichlids exposed to hypoxia and anoxia. Comp Biochem Physiol B Biochem Mol Biol 141:347-355.
- D'Angio CT and Finkelstein JN (2000) Oxygen regulation of gene expression: A study in opposites. Mol Genet Metab 71:371-380.
- Firth JD, Ebert BL, Pugh CW and Ratcliffe PJ (1994) Oxygen-regulated control elements in the phosphoglycerate kinase 1 and lactate dehydrogenase A genes: Similarities with the erythropoietin enhancer. Proc Natl Acad Sci USA 91:6496-6500.
- Gracey AY, Troll JV and Somero GN (2001) Hypoxia-induced gene expression profiling in the euryoxic fish Gallichthys mirabilis Proc Natl Acad Sci USA 88:1993-1998.
- Gu YZ, Hogenesch JB and Bradfield CA (2000) The PAS superfamily: Sensors of environmental and developmental signals. Annu Rev Pharmacol Toxicol 40:519-561.
- Hall TA (1999) BioEdit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp Ser 41:95-98.
- Hochachka PW (1997) Oxygen - A key regulatory metabolite in metabolic defense against hypoxia. Am Zool 37:595-603.
- Hochachka PW (1999) Two research paths for probing the roles of oxygen in metabolic regulation. Braz J Med Biol Res 32:661-672.
- Hochachka PW and Somero GN (2002) Biochemical Adaptation: Mechanism and Process in Physiological Evolution. Oxford University Press, New York, 466 pp.
- Hochachka PW, Beatty CL, Burelle Y, Trump ME, McKenzie DC and Matheson GO (2002) The lactate paradox in human high-altitude physiological performance. News Physiol Sci 17:122-126.
- Iyer NV, Leung SW and Semenza GL (1998) The human hypoxia-inducible factor 1alpha gene: HIF1A structure and evolutionary conservation. Genomics 52:159-165.
- Lewis JM, Costa I, Val AL, Almeida-Val VMF, Gamperl AK and Driedzic WR (2007) Responses to hypoxia and recovery: Repayment of oxygen debt is not associated with compensatory protein synthesis in the Amazonian cichlid, Astronotus ocellatus J Exp Biol 210:1935-1943.
- Markert CL (1984) Lactate dehydrogenase. Biochemistry and function of lactate dehydrogenase. Cell Biochem Funct 2:131-134.
- Markert CL and Holmes RS (1969) Lactate dehydrogenase isozymes of the flatfish, pleuronectiformes: Kinetic, molecular and immunochemical analysis. J Exp Zool 171:85-104.
- Marti HH, Jung HH, Pfeilschifter J and Bauer C (1994) Hypoxia and cobalt stimulate lactate dehydrogenase (LDH) activity in vascular smooth muscle cells. Pflugers Arch 429:216-222.
- Muusze B, Marcon J, Van den Thillart G and Almeida-Val VMF (1998) Hypoxia tolerance of Amazon fish: Respirometry and energy metabolism of the cichlid Astronotus ocellatus Comp Biochem Physiol 120A:151-156.
- Nelson JS (2006) Fishes of the World. 4th edition. John Wiley, New York, 601 pp.
- Nikinmaa M (2002) Oxygen-dependent cellular functions: Why fishes and their aquatic environment are a prime choice of study. Comp Biochem Physiol A Mol Integr Physiol 133:1-16.
- Prabhakar NR (2001) Oxygen sensing during intermittent hypoxia: Cellular and molecular mechanisms. J Appl Physiol 90:1986-1994.
- Richard JG, Wang YS, Brauner CJ, Gonzalez RJ, Patrick ML, Schulte PM, Chippari-Gomes AR, Almeida-Val VMF and Val AL (2007) Metabolic and ionoregulatory responses of the Amazonian cichlid, Astronotus ocellatus, to severe hypoxia. Comp Biochem Physiol B Biochem Mol Biol 177:361-374.
- Robin ED, Murphy BJ and Theodore J (1984) Coordinate regulation of glycolysis by hypoxia in mammalian cells. J Cell Physiol 118:287-289.
- Rooney CH and Ferguson A (1985) Lactate dehydrogenase isozymes and allozymes of the nine-spined stickleback Pungitius pungitius (L.) (Osteichthyes, Gasterosteidae). Comp Biochem Physiol B 81:711-715.
- Scott GR, Wood CM, Sloman KA, Iftikar FI, De Boeck G, Almeida-Val VMF and Val AL (2008) Respiratory responses to progressive hypoxia in the Amazonian Oscar, Astronotus ocellatus Resp Physiol Neurobiol 162:109-116.
- Semenza GL (2001) HIF-1, O(2), and the 3 PHDs: How animal cells signal hypoxia to the nucleus. Cell 107:1-3.
- Sloman KA, Wood CM, Scott GR, Wood S, Kajimura M, Johannsson OE, Almeida-Val VMF and Val AL (2006) Tribute to R. G. Boutilier: The effect of size on the physiological and behavioural responses of Oscar, Astronotus ocellatus, to hypoxia. J Exp Biol 209:1197-1205.
- Soitamo AJ, Rabergh CM, Gassmann M, Sistonen L and Nikinmaa M (2001) Characterization of a hypoxia-inducible factor (HIF-1alpha) from rainbow trout. Accumulation of protein occurs at normal venous oxygen tension. J Biol Chem 276:19699-19705.
- Val AL, Almeida-Val VMF and Paula-Silva MN (1998) Hypoxia adaptation in fish of the Amazon: A never-ending task. S Afr J Zool 33:107-114.
- Webster KA (1987) Regulation of glycolytic enzyme RNA transcriptional rates by oxygen availability in skeletal muscle cells. Mol Cell Biochem 77:19-28.
- Webster KA (2003) Evolution of the coordinate regulation of glycolytic enzyme genes by hypoxia. J Exp Biol 206:2911-2922.
- Wenger RH (2000) Mammalian oxygen sensing, signalling and gene regulation. J Exp Biol 203:1253-1263.
- Wood CM, Kajimura M, Sloman KA, Scott GR, Walsh PJ, Almeida-Val VMF and Val AL (2007) Rapid regulation of Na+ fluxes and ammonia excretion in response to acute environmental hypoxia in the Amazonian Oscar, Astronotus ocellatus Am J Physiol Regul Integr Comp Physiol 292:2048-2058.
Publication Dates
-
Publication in this collection
02 June 2011 -
Date of issue
2011
History
-
Received
23 Dec 2009 -
Accepted
24 Aug 2010