Abstract
This work describes an alternative method for the preparation of ferroelectric thin films based on pre-calcination of oxides, to be used as precursor material for a solution preparation. In order to show the viability of the proposed method, PbZr0.53Ti0.47O3 and Bi4Ti3O12 thin films were prepared on fused quartz and Si substrates. The results were analyzed by X-ray Diffraction (XRD), Scanning Electron Microscopy (SEM), Infrared Spectroscopy (IR) and Rutherford Backscattering Spectroscopy (RBS). The films obtained show good quality, homogeneity and the desired stoichiometry. The estimated thickness for one layer deposition was approximately 1000 Å and 1500 Å for Bi4Ti3O12 and PbZr0.53Ti0.47O3 films, respectively.
ferroelectric films; PZT; bismuth titanate
Ferroelectric Thin Films Using Oxides as Raw Materials
E.B. Araújo, J.A. Eiras
Universidade Federal de São Carlos
Departamento de Física
C.P. 676, 13565-670 São Carlos - SP, Brazil
e-mail: gcferr@power.ufscar.br
Received: April 13, 1998; Revised: November 5, 1998
This work describes an alternative method for the preparation of ferroelectric thin films based on pre-calcination of oxides, to be used as precursor material for a solution preparation. In order to show the viability of the proposed method, PbZr0.53Ti0.47O3 and Bi4Ti3O12 thin films were prepared on fused quartz and Si substrates. The results were analyzed by X-ray Diffraction (XRD), Scanning Electron Microscopy (SEM), Infrared Spectroscopy (IR) and Rutherford Backscattering Spectroscopy (RBS). The films obtained show good quality, homogeneity and the desired stoichiometry. The estimated thickness for one layer deposition was approximately 1000 Å and 1500 Å for Bi4Ti3O12 and PbZr0.53Ti0.47O3 films, respectively.
Keywords: ferroelectric films, PZT, bismuth titanate
1. Introduction
Several researchers have used solid or solution deposition techniques to produce thin films. Solution depositions are normally less expensive and enable better stoichiometric control of complex mixed oxides than other physical techniques such as sputter deposition1 and laser ablation2 or chemical vapor deposition (CVD)3. One of the difficulties of the physical methods, RF sputter or laser ablation for example, is that the film composition is not the same as of that the target for complex compositions. The most frequently used solution-preparation approaches may be grouped into three categories: sol-gel process that uses alkoxide compounds as starting precursors4, hybrid processes that use chelating agents such as acetic acid5 and metalorganic decomposition (MOD) that uses large water insensitive carboxylate compounds6. Metalorganic starting reagents such as alkoxide (M(OR)n where M is a metal and R is an alkyl group) and carboxylate (M(OOCR)n) are employed to prepare the solution for film deposition. As a limitation of these can be pointed out the difficulty to obtain the precursor reagents and sometimes the low stability of complex solution.
In recent years, considerable attention has been devoted to the development of thin film technology of ferroelectric materials7,8, with a view toward the increase of application possibilities in many electronic and optical devices. The most studied ceramic compositions for film preparation are the same as those well established for bulk ferroelectric ceramics, which normally have a complex composition. Large-scale processing of high-quality thin films requires low-temperature synthesis, high reproducibility, and simplicity in all processing steps with low cost.
Due to this fact, the search for new routes for film preparation remains as an interesting and open subject in order to improve the stability of complex solutions, the control of the stoichiometry of the film composition or to reduce the cost of the process.
This work describes an alternative solution method for preparation of ferroelectric thin films starting on a pre-calcination of oxides or carbonates. The proposed method is applied in the processing of lead zirconate titanate (PbZr0.53Ti0.47O3 - PZT) and bismuth titanate (Bi4Ti3O12 - BIT) thin films. The method is based on the fact that not all precursor oxides (PbO, TiO2, ZrO2 or Bi2O3) are soluble in acid media, but a reacted oxide may be soluble. X-ray Diffraction (XRD), Scanning Electron Microscopy (SEM), Infrared Spectroscopy (IR) and Rutherford Backscattering Spectroscopy (RBS) characterization were used to evaluate the efficiency of the method and to compare the film quality to those of films obtained through other methods.
2. Experimental
It is well know that some oxides and carbonates may be not soluble in acid or alkali media9. However, a complex oxide, formed by the reaction of these oxides or carbonates, could be soluble. For example, while lead oxide (PbO) is soluble in HNO3, titanium oxide (TiO2) and zirconium oxide (ZrO2) are soluble H2SO49. On the other hand, PZT is only soluble in aqueous solution of HNO310. This fact was used in this work to produce the ion solution, instead of the conventional metalorganic or alkoxide precursors, as starting materials for production of thin films.
To produce the solution containing the desired ions, PZT and BIT powder were previously prepared by conventional mixed oxide method. For preparation of PZT powder, lead oxide (PbO), titanium oxide (TiO2) and zirconium oxide (ZrO2) were mixed at Zr/Ti = 53/47 ratio using a ball mill during 4 hours. The mixed powder was calcined at 850 °C for 3.5 h in an electric furnace using oxidant atmosphere, obtaining finally the PbZr0.53Ti0.47O3 powder. For preparation of BIT powder, bismuth oxide (Bi2O3) and titanium oxide were mixed in appropriate proportion and mixed in a ball mill during 4 h. The mixture was then calcined at the same conditions as the PZT powder to obtain Bi4Ti3O12 powder. Under these conditions full reacted PZT and BIT powders were obtained. It is interesting to observe that this procedure is the same used to obtain powders to prepare ceramic bodies or thin films through the proposed method, described below.
PZT powder (2 g) was dissolved in 60 mL of diluted acid solution (10% of HNO3 and 90% of distilled water) during 1 h at 80 °C. After cooling, the solution was diluted with distilled water at room temperature to complete a stock solution with final concentration of 12.5 g/L. BIT powder (1 g) was dissolved in 4 mL of concentrated chloridric acid during 20 min at 80 °C. At room temperature the clear solution was diluted with ethanol to complete 125 mL of stock solution with final concentration of 8 g/L. Both solutions of ions were clear and transparent, showing that all elements were dissolved. These ions solution was the starting solution of the proposed process.
The general idea to deposit a film on a substrate is to distribute the ions homogeneously throughout the polymeric resin, prepared according to the Pechini method11,12,. The process calls for forming a chelate between dissolved ions with a hydroxycarboxylic acid (citric acid). Heating of the resin in air causes a breakdown of the polymer. Subsequently, the ions are oxidized to form the desired crystalline phases.
For preparation of the resin by the Pechini method, citric acid and ethylene glycol (citric acid/ethylene glycol = 44/56 in mol%) were mixed with 50 mL from the stock solution. The mixture was heated at 90 °C for 1 h, transparent resins being obtained. Films of the resins (PZT or BIT) were deposited at room temperature on fused quartz and Si substrates by dip coating. The as deposited films were previously heated on a hot plate before they were heat treated for crystallization. Deposited films were crack-free, uniform and well adhered on the substrates.
The structure of the annealed film was examined by X-ray diffraction (XRD), using a Rigaku X-ray diffractometer with CuKa radiation. Scanning electron microscopy (SEM JEOL 5800LV) was used to study the surface morphology of the films. Infrared spectra (IR) of coating solutions and films were measured in the wavenumber range 400 to 1400 cm-1 using a Fourier transform IR spectrometer (Bomem MB-102). The stoichoimetry and thickness of the film were investigated using Rutherford Backscattering (RBS) at 2 MeV and a normal angle of incidence of the He+ beam. All characterizations were done at room temperature.
3. Results and Discussion
The as deposited amorphous films were annealed at temperatures between 500 °C and 700 °C for crystallization. Figure 1 shows the XRD pattern of PZT film deposited on fused quartz and heat-treated for 2.5 h at 600 °C. In this figure we can identify the co-existence of a tetragonal and a rhombohedral phase. The lattice constants a and c of PZT were calculated for tetragonal phase using the (101), (111) and (200) peaks and were found to be 4.02 and 4.16 Å (c/a = 1.03), respectively. For the rhombohedral phase we used the (100), (101) and (111) peaks and obtained a = 4.13 Å and 90-a = 0.03°. This result agrees with result for PZT thin films, prepared by sol-gel technique5, with the same nominal composition.
Figure 2-A, B and C shows the XRD patterns of BIT films on Si substrate after heat treatments for 2.5 h at 500 °C, 600 °C and 700 °C, respectively.
Some structural fluctuation can be seen in Fig. 2-A for films annealed at 500 °C, when compared with the well defined (111), (008) and (006) peaks, that appear in Figs. 2-B and 2-C. As the annealing temperature was increased, from 500 °C to 700 °C, these peaks in XRD patterns became sharper and the full width at half maximum decreased, indicating better crystallinity. Figure 2-C shows that full crystallized Bi4Ti3O12 films with no preferential orientation, on the as crystallized surface, were obtained after firing at 700 °C. The calculated lattice constants a, b and c of Bi4Ti3O12 considering an orthorhombic symmetry and using the (008), (111), (117) and (020) peaks were found to be 5.58 Å, 5.53 Å and 33.67 Å, respectively. These results are very close to Bi4Ti3O12 thin films obtained by others chemical methods, like metallo-organic solution deposition13 and sol-gel14.
Figure 3 shows IR spectra for as deposited (Fig. 3-A) and crystallized at 700 °C PZT film (Fig. 3-B) on Si substrate. The IR spectrum in Fig. 3-A shows several absorption bands. The band centered at 529 cm-1, extending from about 759 cm-1 to 414 cm-1, has been associated to M-O bands in organic systems for PZT thin films prepared by sol-gel process15. The other bands in Fig. 3-A are attributed to the polymeric resin, formed by citric acid and ethylene glycol16, which disappear with the heat treatment (Fig. 3-B). The band at about 494 cm-1 (Fig. 3-B) has been assigned to the vibrations of TiO6 octahedra by Last and Spitzer17,18. Infrared reference spectra of PZT show that ZrO6 or TiO6 octahedra have also a band around the same frequency15. In Fig. 3-B the absorption bands around 1277 and 1098 cm-1 are associated to traces of carbonate (C3-2) present in the PZT film15.
Figure 4 shows a SEM micrograph for BIT film on Si substrate, heat-treated at 700 °C for 2.5 h. The average grain size was estimated to be 76 nm. Figure 4 shows that the film presents good homogeneity, whose morphology is similar with those arising from films prepared by sol-gel technique14.
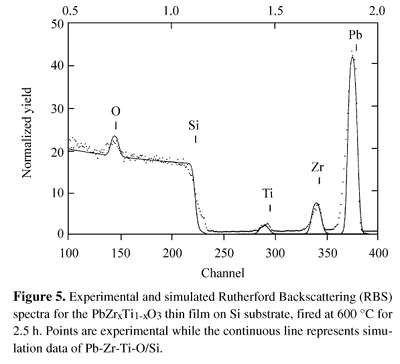
Figure 5 shows a typical RBS spectrum for one layer PZT thin film. In this figure, dots represent experimental points while the continuous line represents the simulation for PZT on Si using the RUMP program19. The values obtained from the simulation were around 1500 Å for the film thickness and a zirconium to titanium ratio of 53.8/46.2. This Zr/Ti ratio confirms that the deposited film had the same nominal composition of the PZT powder, dissolved in acid solution (see Table 1).
Figure 6 shows a RBS spectra for one layer BIT thin film. Through the simulation an estimated thickness of 1000 Å and titanium to bismuth ratio of 0.36/0.48 were obtained. Also in this case, the Ti/Bi ratio agrees very well with the desired nominal composition (see Table 1).
The above presented results show that through the proposed method thin films well crystallized have been prepared with the composition of the powders obtained by conventional ceramic method.
4. Conclusion
An alternative chemical method to prepare thin films is presented. The method is based on the dissolution of a ceramic powder in acid media, with the same composition desired for the film, to obtain a solution containing the metallic ions. The method was successfully applied to produce BIT (Bi4Ti3O12) and PZT (PbZr0.53Ti0.47O3) thin films. The obtained films showed good microstructural homogeneity, the desired composition and a quality similar to those films prepared by sol gel processing. Low cost and high stability of the solution in the preparation process are the main advantages compared with other chemical methods. The presented method can then be particularly useful to produce doped films.
Acknowledgments
The authors thank Dr. N.I. Morimoto and MSc. A.G. Pedrine, LSI-USP, Dr. M.H. Tabacniks and A.F. dos Reis, LAMFI-USP, for facilities and RBS analysis, Dr. Y.P. Mascarenhas, IFSC-USP, for XRD analysis, Mr. Paulo (DQ-UFSCar) for IR measurements and CNPq (Brazilian agency) for financial support.
- 1. Auciello, O.; Kingon, A.I.; Krupanidhi, S.B.; MRS Bulletin, p. 25, June 1996.
- 2. Auciello, O.; Ramesh, R. MRS Bulletin, p. 31, June 1996.
- 3. Keijser. M. de;. Dormans; G.J.M MRS Bulletin, p. 37, June 1996.
- 4. Johnson Jr., D.W. Am. Ceram. Soc. Bull. v. 64 n. 12, p. 1597, 1985.
- 5. Yi, G.; Wu, Z.; Sayer, M. J. Appl. Phys. v. 64, n. 5, p. 2717, 1988.
- 6. Haertling, G.H. Ferroelectrics v. 119, n. 51 1991.
- 7. Sheppard, L.M. Ceramic Bulletin v. 71 n. 1, p. 85, 1992.
- 8. Sayer, M.; Sreenivas, K. Science 247, 1056, 1990.
- 9. Weast, R.C. Handbook of Chemistry and Physics, 68th edition, CRC Press, 1988.
- 10. Araújo, E.B.; Eiras, J.A. Proceeding of 41ş Congresso Brasileiro de Cerâmica v. 1, p. 413, 1997.
- 11. Pechini, M. U.S. Pat. N° 3 330 697, July 11, 1967.
- 12. Lessing, P.A. Ceramic Bulletin v. 68, n. 5, p. 1002, 1989.
- 13. Joshi, P.C.; Krupanidhi, S.B. J. Appl. Phys. v. 72, n. 12, p. 5827, 1992.
- 14. Sedlar, M.; Sayer, M. Ceramics International v. 22, n. 241, 1996.
- 15. Lipeles, R.A. Ceramic Transactions, Ferroelectric Films v. 25, p. 147-153, 1992.
- 16. Ponchert, C.J. The Aldrich Library of Infrared Spectra, second edition, 1975.
- 17. Last, J.T. Phys. Rev v. 105, p. 1740, 1957.
- 18. Spitzer, W.G.; Miller, R.C.; Kleinman, D.A.; Howarth, L.E. Phys. Rev. v. 126, p. 1710, 1962.
- 19. Doolittle, L.R. Phys. Rev. v. B9, p. 344, 1985.
Publication Dates
-
Publication in this collection
27 Jan 2000 -
Date of issue
Jan 1999
History
-
Received
13 Apr 1998 -
Accepted
05 Nov 1998